the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Reviews and syntheses: Review of causes and sources of N2O emissions and NO3 leaching from organic arable crop rotations
Sissel Hansen
Randi Berland Frøseth
Maria Stenberg
Jarosław Stalenga
Jørgen E. Olesen
Maike Krauss
Paweł Radzikowski
Jordi Doltra
Shahid Nadeem
Torfinn Torp
Valentini Pappa
Christine A. Watson
The emissions of nitrous oxide (N2O) and leaching of nitrate (NO3) from agricultural cropping systems have considerable negative impacts on climate and the environment. Although these environmental burdens are less per unit area in organic than in non-organic production on average, they are roughly similar per unit of product. If organic farming is to maintain its goal of being environmentally friendly, these loadings must be addressed. We discuss the impact of possible drivers of N2O emissions and NO3 leaching within organic arable farming practice under European climatic conditions, and potential strategies to reduce these. Organic arable crop rotations are generally diverse with the frequent use of legumes, intercropping and organic fertilisers. The soil organic matter content and the share of active organic matter, soil structure, microbial and faunal activity are higher in such diverse rotations, and the yields are lower, than in non-organic arable cropping systems based on less diverse systems and inorganic fertilisers. Soil mineral nitrogen (SMN), N2O emissions and NO3 leaching are low under growing crops, but there is the potential for SMN accumulation and losses after crop termination, harvest or senescence. The risk of high N2O fluxes increases when large amounts of herbage or organic fertilisers with readily available nitrogen (N) and degradable carbon are incorporated into the soil or left on the surface. Freezing/thawing, drying/rewetting, compacted and/or wet soil and mechanical mixing of crop residues into the soil further enhance the risk of high N2O fluxes. N derived from soil organic matter (background emissions) does, however, seem to be the most important driver for N2O emission from organic arable crop rotations, and the correlation between yearly total N-input and N2O emissions is weak. Incorporation of N-rich plant residues or mechanical weeding followed by bare fallow conditions increases the risk of NO3 leaching. In contrast, strategic use of deep-rooted crops with long growing seasons or effective cover crops in the rotation reduces NO3 leaching risk. Enhanced recycling of herbage from green manures, crop residues and cover crops through biogas or composting may increase N efficiency and reduce N2O emissions and NO3 leaching. Mixtures of legumes (e.g. clover or vetch) and non-legumes (e.g. grasses or Brassica species) are as efficient cover crops for reducing NO3 leaching as monocultures of non-legume species. Continued regular use of cover crops has the potential to reduce NO3 leaching and enhance soil organic matter but may enhance N2O emissions. There is a need to optimise the use of crops and cover crops to enhance the synchrony of mineralisation with crop N uptake to enhance crop productivity, and this will concurrently reduce the long-term risks of NO3 leaching and N2O emissions.
- Article
(1882 KB) - Full-text XML
-
Supplement
(181 KB) - BibTeX
- EndNote
Biologically available nitrogen (N) or reactive N is limited in most natural terrestrial ecosystems. In modern crop production, the addition of N fertiliser has become crucial to achieve high crop yields. This has resulted in cropping systems where a substantial proportion of the N added is lost to the environment, and where the excess reactive N threatens the quality of air, water and ecosystems (Robertson and Vitousek, 2009). The emissions of N2O have considerable environmental impacts through the contribution to global warming and ozone depletion (Ravishankara et al., 2009), and about 16 to 20 Tg N2O–N is emitted annually to the atmosphere; of this, close to 40 % is anthropogenic, and agriculture accounts for 67 %–80 % of the anthropogenic N2O emissions (Ussiri and Lal, 2013). About half of the anthropogenic N2O emissions originate from cultivated soils (Stehfest and Bouwman, 2006). In addition, agricultural soils are sources of indirect N2O emissions resulting from downstream microbial turnover of N from NO3 leaching or ammonia volatilisation (IPCC, 2006). NO3 lost by leaching may also contaminate drinking water and lead to eutrophication of freshwater and marine ecosystems (Dalgaard et al., 2014).
The area under organic production is increasing worldwide (Willer and Lernoud, 2018). In Europe, 2.7 % of the agricultural land is under organic farming, and in nine countries, 10 % or more of the agricultural land is managed organically (Willer et al., 2018). In 2016, 43 % (6×106 ha) of the organic farmed area in Europe was under arable crops.
Organic agriculture aims to be an environmentally friendly production system that sustains the health of soils, ecosystems and people. It should rely on ecological processes, biodiversity and nutrient cycles adapted to local conditions, rather than the use of inputs (IFOAM, 2019). Because of the serious consequences of N2O emissions and NO3 leaching, these environmental burdens are also important issues for organic farming, and there is a continued debate regarding whether an organic mode of crop production enhances or reduces greenhouse gas (GHG) emissions and NO3 leaching from agriculture (McGee, 2015; Lorenz and Lal, 2016). We have chosen to focus on arable systems because crop and soil management vary more between organic and non-organic production of arable crops than for grassland (Barbieri et al., 2017), and the yield gap is larger (De Ponti et al., 2012). These conditions will affect N2O emissions and NO3 from the systems. Previous reviews have compared the net N2O emissions and NO3 leaching from these systems. Lower area-scaled, but roughly similar yield-scaled emissions (slightly higher, similar or slightly lower) are commonly observed for N2O emissions (Tuomisto et al., 2012; Skinner et al., 2014, 2019) and NO3 leaching (Kirchmann and Bergström, 2001; Stopes et al., 2002; Aronsson et al., 2007; Tuomisto et al., 2012; Benoit et al., 2014) from organic versus non-organic arable crop production.
In this review we focus on the drivers for N2O emissions and NO3 leaching under organic arable crop rotations in European climatic conditions. There are insufficient robust field data on N2O emissions and NO3 leaching within organic arable crop rotations to allow for a meta-analysis to quantify the impact of key causes. Thus, here we use the available data to identify sources and causes of N2O emission and NO3 leaching in these rotations, as a basis for suggesting targeted mitigation strategies. We define “organic arable crop rotations” as cropping systems with associated crop and soil management commonly used on European farms dominated by arable cropping and following the European Council Regulation (EC) no. 834/2007s on organic farming (Council of the European Union, 2007). Among others, the use of synthetic N-fertilisers and N-inhibitors are prohibited, but manure and/or short-term leys may be used in these rotations. We designate “non-organic crop systems” as arable cropping systems generally based on inorganic fertilisers, the use of pesticides and often the use of narrow crop rotations, commonly called conventional farming.
Both globally and in Europe organic rotations are longer and more diverse than non-organic rotations (Barbieri et al., 2017). This is essential for nitrogen supply, and for pest and weed control (Stockdale et al., 2001). Barbieri et al. (2017) found that catch crops and undersown cover crops are 2.4 and 8.7 times more frequent in organic than in conventional systems, respectively. They further found that the share of pulses and temporary fodder crops (such as alfalfa, clover and ryegrass) were higher in organic than in non-organic crop rotations, and that the difference between organic and non-organic crop rotations was greater in this respect in Europe than in North America and globally. They further found that more legumes are included in fodder crops, and in catch crops, undersown cover crops and intercropping than in non-organic rotations. In addition to plant-derived N, organic N is applied as manure or other organic fertilisers and amendments. The great diversity of N mineralisation patterns among the organic fertilisers and crop residues is a challenge for farm management to synchronise the N release with plant N uptake. If N is released during periods with poor plant uptake, then the content of soil mineral nitrogen (SMN) and other easily available N can accumulate, creating a large risk of N losses via gaseous emissions or leaching. Because N is mainly applied through plant residues and a limited amount of organic fertilisers in arable organic systems, the N turnover from biological activity is crucial for the content and type of SMN. Plants and organic fertilisers are also important sources of soil organic carbon (SOC). We address how the supply and quality of organic matter in above- and below-ground residues and organic amendments influence the availability and type of SMN and degradable carbon.
Increasing the content of SOC enhances the risk of N2O emissions (Li et al., 2005). This is true whether the soil has a high content of SOC or the content is increased via the addition of organic matter to the soil, and is caused by the tight link between SOC and microbial N2O production (Sahrawat and Keeney, 1986). Because the impact of SOC on N2O emission is dependent on NO3 content in soil (e.g. Weier et al., 1992; Li et al., 2005), we address how the supply of N and carbon (C) via organic inputs drive N2O emissions in organic arable rotations. Based on the IPCC (2006), most inventories and farm models assume that 1 % of total N-input by fertilisers, manure and plant residues are emitted as N2O–N. Skinner et al. (2014) found no correlation between total N-input and N2O emission in organic systems. If this is a general trend, the total N-input cannot be used to estimate N2O emissions from organic crop rotations. In non-organic cropping systems, peak fluxes of N2O are commonly observed shortly after fertilisation with mineral fertilisers in moist soil (Smith et al., 2012), whereas in organic crop rotations the highest fluxes are often observed after the incorporation of plant residues (e.g. Pappa et al., 2011; Nadeem et al., 2012; Brozyna et al., 2013; Krauss et al., 2017b; Skinner et al., 2019). Because of the enhanced content of SOM and thus the larger impact of background emissions of N2O in organic versus non-organic cropping systems (Skinner et al., 2014), increased background emissions are likely to have a major impact in organic-crop rotations. In order to design good mitigation strategies, it is useful to know the relative importance of these two sources of N2O emissions.
The main sources for NO3 leaching are NO3 from the nitrification of plant residues and added organic matter, as NO3 fertilisers are prohibited in organic systems. The crop N requirement dependency on soil organic matter turnover may lead to an asynchrony between crop nutrient demand and the mineralisation of soil organic N, which enhances the risk of NO3 leaching (Di and Cameron, 2002; Crew et al., 2005). Furthermore, soil cultivation for weeding or the incorporation of plant residues have been shown to influence NO3 leaching (Askegaard et al., 2011). We discuss the main drivers for NO3 leaching in organic arable cropping systems and the associated preventative measures. We address the following questions for organic arable crop rotations:
-
How does the supply and quality of organic matter in above- and below-ground residues and organic amendments influence the availability of easily available N and degradable C?
-
How does the supply of easily available N and degradable C drive N2O emissions, and how can these be mitigated?
-
Is there a lack of correlation between total N-input and N2O emission in organic arable crop rotations, and are total N2O emissions primarily driven by background emissions or by episodes with high N2O fluxes following N additions?
-
What are the main drivers for NO3 leaching in organic arable systems, and how can the leaching be reduced?
Based on the authors own field trials, literature databases and searches through Google Scholar, we compiled data on agronomic management, soil properties and yield level of organic arable crop rotations, and measurements of SMN, N2O emissions and NO3 leaching from field trials relevant to organic crop rotations, climate and soil conditions in Europe. For SMN and NO3 leaching, we used the available literature and the data in Tables S1 and S3 in the Supplement to explore the importance of the determining factors for SMN and NO3 leaching, and to identify factors of importance for NO3 leaching in organic arable crop rotations. For SMN and NO3 leaching the structure of the available data did not allow for meaningful statistical analyses.
For N2O we used data presented in Table S2 to explore the impact of background emissions and episodes with high N2O fluxes.
We used the available literature to explore the impact of total N added and N and C added through organic inputs from living plants, plant residues and organic fertilisers on N2O emissions, as we did not have enough data on added N to be able to include this in the regression analyses.
We aimed to analyse the impact of high emission events of N2O fluxes on the total N2O emission. However, we lacked daily measurements, and we lacked data for yearly periods. Thus, it was not possible to identify the full impact of hot moments as undertaken by Molodovskaya et al. (2012). Because of the differences in measurement period, it was also not possible to make direct comparisons between the different field trials. To overcome this, we used a regression model based on N2O emissions in the actual period and peak N2O flux within this period, resulting in the following fitted model:
where F is the cumulated N2O flux (emission) in the measurement period (−278 to 8566 g N2O–N ha−1), t is the duration of the period (38 to 490 d), F∕t expresses the average daily N2O flux rate (−1.3 to 53.2 g N2O–N ha−1 d−1) and Fmax is the highest N2O-flux rate (0.1 to 605 g N2O–N ha−1 d−1) in the measurement period. The total analysis and the data used are given in S4 in the Supplement. One negative value for the average daily N2O flux (−1.3) in a barley/pea crop was removed from the analyses, as it would not have had any significant impact on the results.
We also calculated the percentage contribution of the highest daily N2O flux of the total N2O emissions in the measurement periods for all trials presented in Table S2 (n=97), and correspondingly the sum of the fluxes for the days with the five highest flux rates as a percentage of the total N2O emissions. The choice of five days was to represent what typically constitutes a peak emission event.
The impact of the following potential explanatory variables on the highest daily N2O flux rates was calculated by stepwise regression (α to enter =0.15; α to remove =0.15) by Minitab 18.1, ©2017 Minitab, Inc: clay (soil clay content, %), pH (soil pH), SOC (soil organic C, g kg−1 dry soil), WFPS (soil water filled pore space, %), NO3 (soil content of NO3, kg NO3–N ha−1) and NH4 (soil content of NH4, kg NH4–N ha−1) and temp (soil temperature, ∘C). The selection of these variables was based on the expected impact on N2O emissions (Sect. 4.1) and the data that we were able to obtain from the following studies: Ball et al. (2007), Chirinda et al. (2010), Nadeem et al. (2012), Brozyna et al. (2013), Li et al. (2015a), Baral et al. (2017), Krauss et al. (2017b), and Pugesgaard et al. (2017). The N2O flux data were log-transformed to achieve near normality and variance homogeneity: ln (daily N2O flux +2), where the daily N2O flux = g N2O–N ha−1d−1 is the highest N2O flux rate during the actual measurement period (Table S2, highest daily flux rate). The highest flux rates were chosen for analysis, as we wanted the extreme values to explore which factors are mostly influential for hot moments of N2O emissions. The total analysis and data used are given in S5.
Based on the stepwise regression, we achieved the following fitted regression model:
3.1 Supply and quality of soil organic matter
Return of crop residues to the soil is standard practice in both organic and non-organic arable production; however, because of the more diverse crop rotations in organic production systems, larger and more diverse inputs of herbage from legume-based green manures, leys, cover crops (CC) and intercrops are returned to soil than in the non-organic cropping systems commonly used in Europe (Gattinger et al., 2012). The N content in the crop residues of legume-based systems is typically higher than that from non-legume systems (Watson et al., 2002). Commonly used external sources of organic inputs in organic cropping systems are animal manures and slurries, composts or biogas residues, and organic fertilisers based on animal manure or municipal waste (Løes et al., 2017). The great diversity of N mineralisation patterns among the organic fertilisers and crop residues result in a large variation in how much N and C are rapidly degradable in the organic inputs.
Through the application of organic amendments and various crop residues from arable and forage crops, C and N are applied to soil, and the soil organic matter (SOM) content is often higher in organic than in non-organic arable crop rotations: Marinari et al. (2007) reported about 40 % more total organic C a short time after the application of organic matter compared with mineral fertiliser; Marriott and Wander (2006) documented concentrations of SOC that were about 14 % higher in organic than in non-organic systems; Gattinger et al. (2012) showed 3.5 Mg C more in SOC stocks in organic compared with non-organic production in a global meta-analysis; Aguilera et al. (2013) reported a SOC concentration 19 % higher in a meta-analysis from the Mediterranean; Hu et al. (2018a) showed 0.4 Mg C ha−1 yr−1 more SOC accumulated with an organic than a non-organic treatment at Foulum, but 0.4 Mg less C than in the non-organic treatment at Flakkebjerg in long-term field trials; and Pimentel et al. (2005) documented 15 % higher SOC concentrations in legume-based organic versus non-organic crop rotation in a long-term field trial. The quality of SOM differs between non-organic and the more diversified organic, arable crop rotation, with a higher share of labile SOM (Lynch, 2015) and thus easily degradable organic matter (C and N) in soils in organic crop rotations (Marriott and Wander, 2006; Marinari et al., 2007; Martyniuk et al., 2016). The higher content of degradable SOM in organic crop rotations is a valuable soil fertility asset as it provides a short-term pool for plant nutrient supply (Marriott and Wander, 2006; Martyniuk et al., 2016). SOM turnover rates vary with soil texture and climate: they are higher when organic carbon is less protected from decomposers (low clay content) and in warm climates with suitable moisture (Burke et al., 1989).
3.2 Soil biological activity
Soil microbes contribute directly to plant residue decomposition and to the mineralisation and turnover of SOM, in addition to earthworms and other soil fauna (Kuiper et al., 2013; Lubbers et al., 2013). The inflow of degradable organic matter provides substrate for soil organisms, and the application of organic matter increases the growth of microbial communities, their enzyme activities and the microbial diversity compared with an unfertilised control or soil fertilised with only mineral fertiliser (Anderson and Domsch, 1989; Marinari et al., 2007; Thangarajan et al., 2013), although such changes in the topsoil may be slow processes (Petersen et al., 2013). Accordingly, higher biological activity has commonly been found in arable soils managed organically compared with non-organically managed soils (Mäder et al., 2002; Gomiero, 2013; Hartmann et al., 2015; Lori et al., 2017). In their meta-analysis, Lori et al. (2017) found that organic systems had 32 % to 82 % greater microbial biomass C, microbial biomass N, total phospholipid fatty-acids, and dehydrogenase, urease and protease activities than conventional systems. They found that when both organic and non-organic systems included legumes, the organic system displayed a higher microbial N content than the non-organic counterpart. In cases where only the organic systems contained legumes, the difference in microbial N between the two systems was even more pronounced. The abundance of earthworms can be twice as high in organic than in non-organic systems (Pfiffner and Mäder., 1997; Filser et al., 1999; Hansen and Engelstad, 1999; Riley et al., 2008). More abundant earthworm populations are found when large amounts of animal manure or green manure are applied to soil (Hansen and Engelstad, 1999; Frøseth et al., 2014), when autumn ploughing is avoided (Pfiffner and Luka, 2007) and in the absence of tractor traffic (Hansen and Engelstad, 1999).
3.3 Soil N-dynamics within organic arable cropping systems
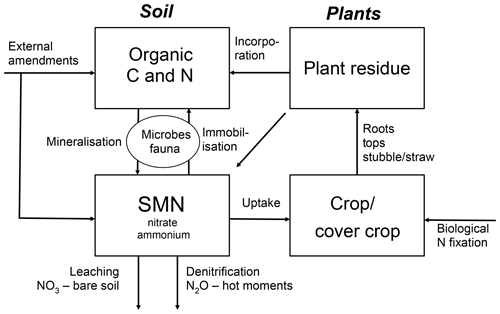
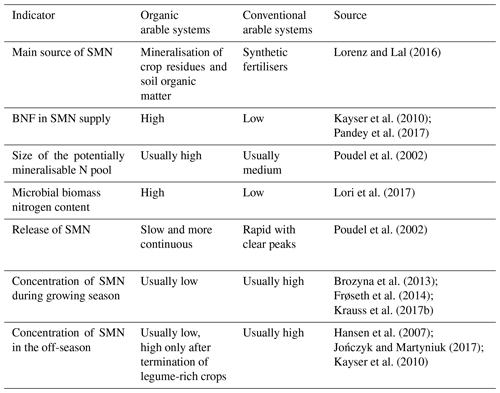
Crop N supply and SMN in organic farming relies largely on mineralisation of N in soil organic matter, N in organic amendments and crop residues and BNF of legume-based crops (Gattinger et al., 2012; Lorenz and Lal, 2016). A principal sketch for N dynamics in organic systems is given in Fig. 1 and an overview of performance of selected drivers of SMN in organic and non-organic crop rotations are given in Table 1.
Despite substantial inputs of N from biological nitrogen fixation (BNF; Kayser et al., 2010; Pandey et al., 2017) and from organic amendments, the N supply is often below optimum for plant growth in arable organic farming (Berry et al., 2002; Tuomisto et al., 2012). BNF may be a more important N source than N from organic manures and fertilisers in organic arable cropping systems (Pandey et al., 2018).
The major release of plant available N from added organic matter depends on the C:N ratio, the mineral N content and the degradability of the C of added plant residues manure, compost and other decomposed amendments. Bhogal et al. (2016) showed that for pig slurry and poultry layer manure with a C:Norg of 9–12:1, up to 70 % of the organic N was mineralised after five growing seasons, whereas in cattle slurry and straw-based farmyard manure with a C:Norg of 10–21:1, only 10 %–30 % of N was mineralised (net). For crop residues with a high C:N content there will be initial immobilisation and the net mineralisation may only start very late or after the growing season of the main crop (Li et al., 2015b). The mineralisation process of the more stable N can continue over years to decades. Simulation modelling has shown that even over a 20-year period, only 10 %–15 % of organic N in applied manure may be taken up by crops, with the rest being lost or retained in soil organic matter (Berntsen et al., 2007). The high microbial activity and high content of organic matter affect N cycling. Hu et al. (2018b) found a higher net N mineralisation of added organic matter in soils with a prehistory of CC use, indicating positive legacy effects of CC use, which was attributed to a greater microbial biomass N in systems with the use of CC.
The design of the rotation, as well as its management, influences the potentially mineralisable N (PMN) pool. Working in three different organic arable systems, Spargo et al. (2011) showed that the PMN pool amounted to 315 kg N ha−1 on average. They showed that more diversified crop rotations resulted in larger PMN pools. Poudel et al. (2002) reported a 112 % and 56 % greater PMN pool in the organic system in comparison to the conventional and low-input systems, respectively. Moreover, they observed a slower and more continuous release of mineral N in the organic systems compared with the more rapid release of mineral N from synthetic N fertilisers applied in non-organic systems. Moyo et al. (2016) reported higher PMN in soils under wheat following a cut and mulched red clover ley compared with after a ley where the residues had been removed. This indicates the importance of total N-input to the soil. The importance of total N-input in plant residues for crop N uptake was also observed by Petersen et al. (2013).
SMN was found to be very low under grass–clover leys (Table S1, Watson et al., 1993; Nadeem et al., 2012; Brozyna et al., 2013; Frøseth et al., 2014; Krauss et al., 2017b), as grasses quickly take up soil NO3 in the root zone (Brophy et al., 1987). Frøseth et al. (2014) observed low levels of SMN in a 1-year grass–clover ley, irrespective of whether the herbage was mulched or removed. After termination of a ley, the concentration of SMN usually increases (Table S1, Ball et al., 2007; Brozyna et al., 2013; Krauss et al., 2017b). Even in the year following the termination of a ley, the content of SMN can still be high (Hansen et al., 2007; Jończyk and Martyniuk, 2017). Kayser et al. (2010) pointed out that N provided by spring ploughing of both a 1-year grass–clover ley and a 3-year grassland ley resulted in high concentrations of SMN (0–90 cm, 61 kg N ha−1 and 95 kg N ha−1, respectively) in the following autumn after harvest of spring triticale. Much of this SMN may not have been available for the crops during spring and is likely to have been mineralised after the end of the growing season. In contrast, low concentrations of SMN were observed in topsoil and subsoil after spring ploughing of a 1-year grass–clover ley in four field trials in Norway, in late spring and in autumn after the harvest of a spring barley crop (Table S1) and in the following year (Frøseth et al., 2014; Frøseth, 2016).
4.1 Mechanisms for N2O emissions
Many processes contribute to N2O production in soils, but the dominant mechanisms for N2O emissions from terrestrial agricultural soils are the microbial processes of nitrification, nitrifier denitrification (as a result of incomplete nitrification) and denitrification (Firestone and Davidson, 1989; Butterbach-Bahl et al., 2013). Nitrification and denitrification are both biological processes; thus, the same mechanisms will cause N2O emissions in organic and non-organic farming systems. However, fertilisation, and crop and soil management practices differ substantially between these two systems (Sect. 3), and the relative importance of the relevant triggers consequently differ (Sect. 6).
Nitrification is the microbial oxidation of NH3 to NO2 and ultimately NO3, where N2O is produced as a by-product via some partially understood biotic and abiotic reactions of hydroxylamine (Anderson, 1964; Liu et al., 2017). Nitrifier denitrification occurs when NO2 produced during nitrification is reduced to N2O (by denitrifying organisms), instead of being oxidised to NO3, under fluctuating oxic–anoxic conditions (Firestone and Davidson, 1989). Denitrification is the microbial anaerobic reduction of NO3 via NO2 to gaseous NO, N2O and N2, which are ultimately transported through the soil to the atmosphere. Denitrification is the main source of N2O production in soils, as the N2O yield potential of denitrification is much higher (1 %–100 %) than that of nitrification (0.1 %–1 %) (e.g. Andersson et al., 1993; Butterbach-Bahl et al., 2013).
The ratio between the gaseous products of denitrification depends on NO3 availability, oxygen availability in the soil, the amount of easily decomposable carbon as an energy source, soil pH and the microbial community structure (Bakken et al., 2012). Oxygen availability depends on soil microbial activity and gas diffusivity, which, in turn, depends on soil moisture content, texture and density. Gas diffusivity is a promising predictor for N2O fluxes from soils with varying bulk density as observed by Balaine et al. (2013), who found that the production of N2O increased when the relative gas diffusivity was between 0.006 and 0.020 and the soil became anaerobic.
As explained above, the risk of N2O emission increases as the soil carbon content increases (Li et al., 2005). N2O and N2 production correlates with total organic C, water soluble C and mineralisable C in soil, but the increased availability of C also decreases the ratio of N2O:N2 (Sahrawat and Keeney, 1986). Emissions from soil organic matter (background emission) will vary between years because of variations in temperature and precipitation (Brozyna et al., 2013; Hansen et al., 2014) and between different categories of crops due to different time windows with high SMN (Dobbie and Smith, 2003).
Organic amendments and plant residues that provide carbon that is easily decomposable by microbes may enhance microbial activity and deplete soil oxygen via enhanced soil respiration. In addition, degradable carbon is an energy source for denitrifying bacteria. In accordance with this, Köster et al. (2011) concluded that bacterial denitrification was the main process for producing N2O during the first 3 weeks following the application of biogas residues, and high carbon availability was an important cause of this. Li et al. (2016) concluded that denitrification was the main cause of N2O emission after the addition of legume-based residues. Several studies have shown higher rates of N loss through denitrification from soils treated with organic amendments such as manure, composts and plant residues when compared with unamended or mineral N treated soils (Thangarajan et al., 2013). In line with this, the incorporation of residues by tillage increases soil respiration and N2O fluxes due to microbial stimulation (Krauss et al., 2017a).
Low soil pH inhibits the activity of the N2O reductase enzyme and, thus, the N2O:N2 ratio increases (Liu et al., 2010). At higher soil pH, the denitrification rate is higher, but the N2O:N2 ratio is lower as a greater part is completely denitrified to N2. At low temperatures, nitrous oxide reductase is hampered (Holtan-Hartwig et al., 2002), but, conversely, denitrification rates are also reduced (Butterbach-Bahl et al., 2013).
4.2 Legumes during active plant growth
In general, unfertilised legumes have small N2O emissions during their growing period, particularly when grown in mixtures with non-legumes. Low N2O emission are found during growth of grain legumes (Rochette and Janzen, 2005; Dusenbury et al., 2008; Pappa et al., 2011; Jensen et al., 2012; Jeuffroy et al., 2013), green manure crops and CC (Baggs et al., 2000b; Brozyna et al., 2013; Li et al., 2015a; Peyrard et al., 2016; Shelton et al., 2018) as well as for grass–clover leys (Baggs et al., 2000b; Ball et al., 2002; Nadeem et al., 2012; Brozyna et al., 2013; Krauss et al., 2017b; Skinner et al., 2019). This is consistent with low SMN concentrations during growth (Sect. 3) and negligible N2O emissions associated with BNF by the legume–rhizobium symbioses (Rochette and Janzen, 2005; Carter and Ambus, 2006). However, following termination or senescence of the legume crops, reactive N released from dying roots and nodules may lead to enhanced N2O emission (Rochette and Janzen, 2005).
Surface mulching of harvested herbage may theoretically enhance N2O emissions due to mineral N released from the herbage. However, several studies have shown that mulching of grass–clover herbage on the growing ley only causes a slight increase in N2O emissions (Möller and Stinner, 2009; Nadeem et al., 2012; Brozyna et al., 2013). None of these studies measured ammonia volatilisation from mulched herbage, which could have been a major loss of mineralised N corresponding to the findings of Larsson et al. (1998). Volatilised NH3 will be redeposited elsewhere and may result in increased N2O formation downstream (IPCC, 2006).
4.3 Crop residues
As outlined in Sect. 4.1, there is an enhanced risk of N2O emissions from agricultural soils when easily degradable carbon and N are simultaneously available, and denitrification is probably the main source for this. Because legume-based crop residues also increase SMN (Sect. 3.3), increased N2O emissions have been reported in field trials whether the residues are from grain legumes (Pappa et al., 2011; Jeuffroy et al., 2013), grass–clover (Baggs et al., 2000b; Ball et al., 2007; Nadeem et al., 2012; Brozyna et al., 2013; Skinner et al., 2019), intercropped clover (Pappa et al., 2011) or CCs (Baggs et al., 2000b; Peyrard et al., 2016; Pugesgaard et al., 2017). However, the increase of N2O fluxes after the incorporation of crop residues and other plant material might be small and a negligible part of the total N2O emissions (Peyrard et al., 2016; Pugesgaard et al., 2017; Shelton et al., 2018).
The C:N ratio of incorporated herbage does affect N2O emissions, with higher emissions expected from herbage with a low C:N ratio (Chen et al., 2013). From this, one should expect higher N2O emissions from legume residues than from cereals or grasses (e.g. Rochette and Janzen, 2005). However, Larsson et al. (1998) observed the same N2O–N emission factor (EF; 1 % of applied N) from mulched alfalfa (C:N ratio of 11) as from mulched grass with a C:N ratio of 21, but a higher EF than from a mulched grass with a low N-content (C:N ratio of 36, EF =0.1 %). The N2O fluxes might be high despite a high C:N ratio when the carbon source is easily degradable as observed with fodder radish by Li et al. (2015a) (Table S2).
N2O emissions may also be associated with the previous incorporation of plant residues. In accordance with this, Skinner et al. (2019) observed enhanced N2O fluxes after a maize crop succeeding a grass–clover ley. Measurements of N2O fluxes shortly after the incorporation of plant material, or measurements in the following year, only tell part of the story. The enhanced content of various fractions of SOM derived from crop residues, ley and CC (Sect. 3.2) are likely to increase the long-term background emissions of N2O (Sect. 4.1). In a 10-year-old field experiment with and without legume-rich CC in the crop rotation, Pugesgaard et al. (2017) concluded that crop residues were an important source of N2O, and that mineralisable C, rather than N input, was the main driver for N2O emission. Contrary to this, Peyrard et al. (2016) observed that although N2O fluxes increased for a few days after incorporation of CC, the contribution of such events to cumulative N2O emissions were negligible in a 3-year low-input field trial. In their study, however, the CC treatments started when the N2O measurements started. More studies in long-term experiments with the continuous use of CC are needed to verify the actual impact of crop residues in a long-term perspective in various field situations, as the addition of plant material to soil also affects the soil structure, the soil biological activity and N turnover.
4.3.1 Freeze/thaw and dry/wet cycles
The mechanisms behind freeze/thaw have been comprehensively reviewed by Congreves et al. (2018), showing that the causes of N2O emissions are different for these two mechanisms and that freeze/thaw cycles have a larger impact on N2O emissions in temperate agroecosystems than drying/rewetting. Wagner-Riddle et al. (2017) estimated that by neglecting freeze/thaw N2O emissions, global agricultural N2O emissions are underestimated by 17 % to 28 %. Freezing/thawing of soil rich in organic matter and soil biota, or soil covered with plant residues, may result in a N2O boost as easily degradable C and N is released from cells through lysis after frost. As summarised in the introduction and in Sect. 3 these conditions are particularly relevant for organic crop rotations. Flessa et al. (1995) observed that 46 % of total annual N2O emissions from a sunflower crop that was solely fertilised with farmyard manure (12 Mg ha−1) occurred during December and January, mainly due to high N2O peak fluxes (650 g N2O–N ha−1d−1) after the thawing of the first freezing period during winter. Correspondingly, Westphal et al. (2018) did not observe any enhanced N2O fluxes after the late summer incorporation of a ley dominated by alfalfa (0–10 g N2O–N ha−1 d−1), but fluxes were greatly enhanced during the spring thaw in the following year (60 g N2O–N ha−1 d−1).
When CCs are killed by frost, N2O fluxes will increase during thawing of the soil due to the release of easily degradable C and N in the plant material. Li et al. (2015a) observed significantly higher N2O emissions from the frost-sensitive fodder radish that is rich in readily degradable carbon than from other less frost sensitive CCs. Winter emissions were even greater when fodder radish was harvested in late autumn (30 October), leaving only roots and stubble (Table S2). This suggests that N and C in roots of frost sensitive CCs can be an important source of N2O emissions after thawing. Also, frost may enhance N2O emissions in leys. Sturite et al. (2014) observed that enhanced N2O emission during thawing of a frozen grass–clover ley correlated with clover content in the ley.
Under drought conditions, the nitrification process prevails and N2O is produced at very low rates. However, with rewetting easily degradable N and C are mineralised, resulting in increased N2O fluxes. Hansen et al. (2014) observed that the N2O flux increased with increasing clover content during the rewetting of a grass–clover ley after drought. Hence, both freezing/thawing and rewetting may have a large impact in organic systems.
4.3.2 Soil and tillage effects
N2O emissions associated with crop residues are affected by tillage depth and soil type. Large N2O emissions have been observed when crop residues are placed near the soil surface in heavy soil (Peyrard et al., 2016; Krauss et al., 2017b), whereas the highest N2O emissions in lighter soil types have been observed either after rotary harrow (Baggs et al., 2000a) or after ploughing (Petersen et al., 2011). When the crop residues are squeezed and mixed with a rotary harrow, easily available N and degradable C become available for denitrifying bacteria in the soil and the potential for denitrification is large. In line with this, Krauss et al. (2017b) observed high N2O fluxes a few days after weeds and crop residues were superficially incorporated with a rotary harrow in a moist calcareous clay soil (WFPS 80 %) (Table S2, highest observed peak in single plot 800 g N2O–N ha−1 d−1). Similarly, Peyrard et al. (2016) observed enhanced N2O fluxes (max rates 60 g N2O–N ha−1 d−1) up to several days after crop destruction when crop residues (sunflower, wheat and faba bean) were mulched or placed near the soil surface of a calcareous-clay, but not by ploughing or mechanical weeding. Baggs et al. (2000a) observed higher N2O fluxes when lettuce residues were incorporated by rotary harrow than by ploughing (peak of 67 g N2O–N ha−1 d−1).
Restricted gas diffusivity is another possible explanation for the observed lower N2O fluxes with the deep incorporation of crop residues in dense soil. With reduced gas diffusivity more N2O is likely reduced to N2 in accordance with a general trend of a larger ratio of N2O–N ∕ (N2O–N + N2-N) close to the soil surface and smaller fluxes deeper in the soil profile (Sahrawat and Keeney, 1986). Kuntz et al. (2016) observed a decreased O2 concentration at a soil depth of 8 cm and a corresponding reduction of N2O to N2 with surface application of carbon-rich material. As another example, Petersen et al. (2011) found that the largest fluxes were observed when residues were incorporated by ploughing compared to reduced tillage in their loamy sandy soil. Possible explanations for this could be that residues came directly into contact with mineral N from the injected slurry after ploughing, fostering enhanced microbial turnover of C and N, and that, in this soil, the aeration with O2 was still available at plough depth.
4.4 Organic fertilisers
Organic fertilisers vary widely in the content and types of N and C compounds causing large variations in N2O emissions after application. Animal slurries have a higher content of NH4–N and contain more easily degradable N and C than solid manures and composts; thus, they are stronger triggers for rapid N2O emissions shortly after application (Charles et al., 2017, Sect. 3). In accordance with this, Krauss et al. (2017b) observed higher N2O emissions shortly after the application of cattle slurry than after the application of composted solid cattle manure. Correspondingly, in a field experiment with spring barley fertilised with various organic slurries, Baral et al. (2017) observed the highest N2O EF in the treatment with the highest application of organic matter, and thus the highest content of easily degradable C. Meijide et al. (2007) and Chantigny et al. (2007) found that the use of digested slurry, with a lower content of degradable C compared with untreated pig slurry, reduced soil N2O emissions by 25 % and 50 %, respectively.
The effect of organic fertilisers depends on soil type and the content of SOC. Degradable C applied with organic fertilisers will trigger microbial respiration and denitrification in a soil with a low SOC content to a greater extent than it would in a soil with a high SOC content (Chantigny et al., 2010; Pelster et al., 2012), whereas the impact of easily available N is higher in a soil with a high SOC content (Petersen et al., 2008). Due to the higher SOC content and higher share of labile SOM in organic crop rotations compared with non-organic crop rotations (Sect. 3.1), the short-term effect of organic fertilisers with a high content of degradable C on N2O emission are likely lower in organic than in non-organic crop rotations.
Also, the absence of synthetic fertilisers containing easily available N means that organic fertilisers are likely to have a smaller short-term impact on N2O emissions in organic than in non-organic crop production. In a meta-analysis, Charles et al. (2017) found that the N2O EF was higher when soils received organic amendments in combination with synthetic fertilisers. They found EFs for liquid manures + synthetic fertilisers of 2.14 % (±0.53), for composts + synthetic fertilisers they reported 0.37 % (±0.24), and the corresponding EFs for manure and compost were 1.12 % (±0.18) and 0.00 % (±0.17), respectively.
However, the long-term impact of manures is not included in these EFs. In contrast with the short-term fertiliser effect, a long-term fertilisation with organic fertilisers may enhance N2O emissions through enhanced background emissions. Chang et al. (1998) observed that annual N2O emissions increased with manure rate when different rates of solid feedlot manure and thus N application were applied for 21 years. Their manure rate was far greater than would be applied under organic farming conditions, but the possibility for enhanced background N2O emissions after long-term input of organic matter via manuring and the application of crop residues should be considered. Krauss et al. (2017b) found that fertilisation with slurry and manure compost increased annual N2O emissions during winter wheat after more than 10 years of differentiated management compared with sole slurry fertilisation (mean values in the period, 369 d, were 2.2 and 2.9 kg N2O–N ha−1, respectively). They related this to higher microbial biomass and SOC. Mean values for the upper 10 cm of soil were 28 and 30 Mg C ha−1 for fertilisation with slurry and manure compost and sole slurry fertilisation, respectively.
4.5 Contribution of total N-input and high emission events to N2O emissions
Skinner et al. (2014) concluded in a review that soil characteristics (soil N content) had a greater impact on N2O emissions from organic production than the total N-input by fertilisation. Lack of correlation between N2O emissions and N fertilisation in organic production corresponds to the more recent findings of Krauss et al. (2017b) and Pugesgaard et al. (2017). Pugesgaard et al. (2017) observed no significant correlation between N2O emissions and N input in fertiliser/manure, for either annual N2O emissions or spring emissions, but N2O emissions were correlated with N input in residues from the previous main crop and CC. This agrees with the findings of Bouwman et al. (2002), Van Groenigen et al. (2010), Shcherbak et al. (2014) and Peyrard et al. (2016), who observed low EFs when N fertilisation was below optimum as commonly found in organic production systems.
As discussed earlier, N2O emissions in organic crop rotations are driven by enhanced background emissions from the long-term input of organic matter and episodes with enhanced N2O emission after the application of crop residues or organic fertiliser. However, this poses the question as to whether N2O emissions are primarily driven by background emissions or by episodes with high N2O fluxes. We used data from mainly organic field trials (Table S2) and one non-organic trial fertilised with organic fertilisers (Baral et al., 2017) to calculate the impact of the highest N2O fluxes on the total N2O emissions. In Frick (CH), Edinburgh (UK), Aberdeen (UK) and Ås (NO), the highest daily flux rates were 605, 211, 297 and 94 g N2O–N ha−1 d−1, respectively (Table S2). Because of the high flux rates, we hypothesised that high flux events were responsible for a major part of the N2O emissions from these systems. The single days with the highest fluxes correspond to 18 % (65 d measurement period) in Frick, 2 % in Edinburgh (161 d), 17 % in Aberdeen (38 d) and 2 % in Ås (218 d) of the cumulated N2O emissions in the measurement periods. The five highest daily N2O fluxes corresponded to 22, 7, 55 and 5 % of the N2O emission in the measurement periods in these investigations, respectively. However, in field trials conducted on well-structured sandy loams at either Foulum or Flakkebjerg in Denmark (Table S2, 105 to 365 d), peak N2O fluxes from 1 or 5 days only constituted from < 1 % to 8 % and 5 % to 14 % of the total emission in the periods, respectively. The highest daily flux rate in these trials was only 78 g N2O–N ha−1 d−1. This was in a non-organic treatment heavily fertilised with cattle slurry and digested sewage sludge (476 kg total N ha−1, Table S2; Baral et al., 2017). From this we reject the hypothesis that high flux events were responsible for a major part of the N2O emissions from these systems, rather background emissions seemed to be the major N2O source. However, a simple regression model (Eq. 1) showed that the average daily N2O flux correlated positively with episodes with high N2O fluxes. From this we can conclude that when the conditions for high N2O fluxes are met for 1 or more days, there is a large chance of high total N2O emissions in the period. The small peaks in the Danish field trials reveal that well-drained sandy soils promote rapid water infiltration and good gas-diffusivity and, in turn, low N2O emissions.
We wanted to explore the impact of soil conditions on the highest N2O flux peak. In a stepwise regression (Eq. 2), the content of clay, SOC, NO3–N in soil and soil temperature had significant positive impacts on peak N2O fluxes in the selected investigations shown in Table S2 (Pclay, PSOC < 0.001, , Ptemperature < 0.01, PWFPS < 0.05). The content of NH4–N in soil did not affect peak N2O fluxes. These findings indicate that denitrification is the main cause of high N2O-flux rates in these studies. To visualise the impact of different factors on the mean and the highest N2O flux and the contribution of the five highest daily fluxes to the cumulated N2O emissions in the measurement period, we grouped the investigations according to the percentage (%) of clay in soil, N added with organic fertilisers, SOC, soil pH, experimental period, type of crop and mean daily precipitation in the period (Fig. 2). Because of the lack of information, we were neither able to include total N-input nor soil porosity in the regression analyses and the box plots.
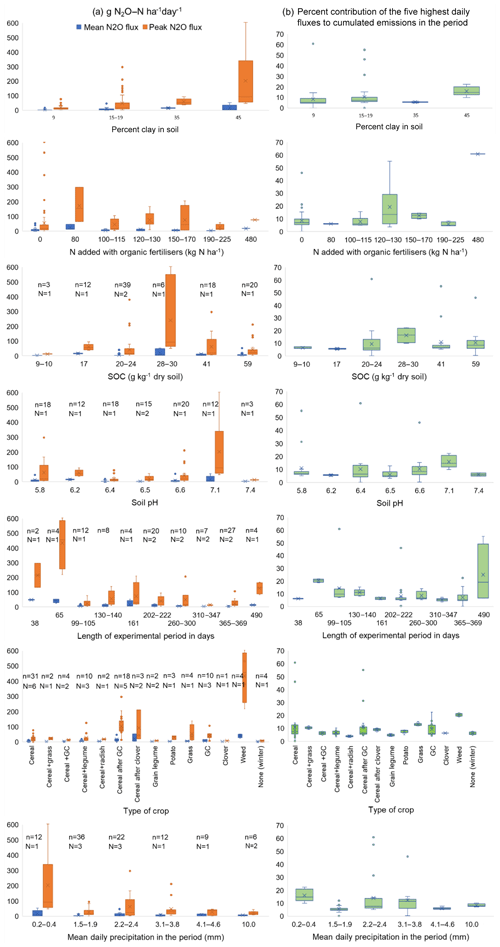
Figure 2Box plot (the lines in the boxes indicate the mean, the crosses indicate the median, and the whiskers indicate the upper and lower quartile) for (a) the mean N2O flux in the period (g N2O–N ha−1 d−1) and the highest daily flux rate in the same period (g N2O–N ha−1 d−1), and (b) the percent (%) contribution of the five highest daily fluxes to cumulated N2O emissions in the period. N denotes the number of sites, and n is the number of observations.
In line with the findings of Skinner et al. (2014), we did not find a clear impact of N added with organic fertilisers; however, the soils with the lowest SOC content showed the lowest mean and lowest peak N2O emissions. This supports the hypothesis that the SOC content of the soil, and thus the content of SOM, are important drivers for N2O emissions. At high SOC contents other factors seem to be more important. The impact of the type of crop on N2O emissions mainly follow the trends we have seen in other investigations (Sect. 5.2, 5.3): higher N2O emissions when cereals succeed a short-term ley (grass–clover or clover), and mixtures with monocotyledons and legumes do not have higher N2O emissions than cultures with only monocotyledons. The high residual effects of previous leys support the idea that background emissions are the main driving force for N2O emission in organic crop rotations. The high N2O emissions (up to 600 g N2O–N ha−1 d−1) from one occasion in Switzerland with the incorporation of weeds in wet soil have a large impact on all box plots (Fig. 2). None of the other groupings in Fig. 2 showed any clear causal effect on N2O emissions.
The low effect we observed of pH on N2O emissions (Eq. 2, Fig. 2) is contradictory to enhanced N2O emissions caused by a hampered N2O reductase enzyme often observed in acid soils (Bakken et al., 2012). A reason for this could be that in these soils and with these management methods, other factors meant more for N2O emissions than pH. Although denitrification was likely to have been the main cause of N2O fluxes at the highest N2O flux rates, the soil NO3 concentrations are often low between the episodes with high N2O flux rates, as observed by Chirinda et al. (2010), Nadeem et al. (2012), Brozyna et al. (2013), Li et al. (2015a) and Pappa et al. (2008).
4.6 Impacts of earthworms
Abundant earthworm populations in organic crop rotations (Sect. 3.3) are likely to influence N2O fluxes as they significantly affect mineralisation and reduction of N compounds to N2O and N2 (Prieto, 2011). N2O is emitted from intestinal microbes but is also released from nitrates emitted in body fluids in the earthworm gut as well as from casts, middens and burrows (Prieto, 2011). Conversely, earthworms improve soil porosity and aggregate stability (Bronick and Lal, 2005) and thus gas diffusivity and water infiltration in soils, which will reduce N2O emissions. Epigeic species (living near the surface and feeding on surface litter) and anecic species (deep burrowing) are well known to enhance N2O production, because they feed directly on decomposing herbage (Evers et al., 2010; Lubbers et al., 2011; Nebert et al., 2011). Endogeic earthworms that feed on SOM particles are most common in cultivated arable soils (Hansen and Engelstad, 1999), and they do not increase denitrification (Postma-Blaauw et al., 2006). There are too few published results to robustly predict the impact of earthworms in arable organic crop rotations on N2O emissions as this will depend on local climatic and edaphic conditions.
5.1 Mechanisms for NO3 leaching
NO3 leaching is an abiotic process driven by diffusion and convection (e.g. Johnsson et al., 1987), where NO3 is transported out of the root zone along with the downward water flow. In addition to soil water content, soil texture and structure are important in determining leaching rates. Fine textured soils have slower infiltration rates than coarse textured soils, and porous sandy soils are most vulnerable to leaching, also because these soils often have more shallow rooting depths than loamy soils (Askegaard et al., 2005). The impact of soil type was clearly demonstrated by Askegaard et al. (2011) who found that, depending on soil type (coarse sand > loamy sand > sandy loam) and precipitation, 20–100 kg N ha−1 yr−1 were leached on average for the crop rotations. In their study, the location on coarse sand had 200–300 mm more rainfall per year than the other locations. The leaching was considerably higher than for Swedish clay soils which showed a value of 20 kg N ha−1 yr−1 (Stenberg et al., 2012).
Due to its high mobility in soil, NO3 can easily be lost from the agroecosystem by leaching during periods with high drainage rates. A well-developed active root system enhances NO3 uptake, whereas a poor root system will not utilise all of the NO3 within the soil profile (Dunbabin et al., 2003). NO3 remaining in the soil after the growing season of crops, or subsequently mineralised, will greatly increase the risk of leaching loss. This could occur outside of the growing season, but also when there is poor crop establishment caused by unfavourable seedbed structure or from crop failure caused by diseases or pests (Stenberg et al., 2012). If crop failure coincides with rainy weather, the risk of severe NO3 leaching is large. This was observed by Torstensson et al. (2006) and De Notaris et al. (2018) in organic farming systems, where potato growth was restricted due to early crop termination following disease outbreaks. Annual leaching was 75 kg N ha−1 when potato was cultivated in the year after green manure compared with 98 kg N ha−1 after pea/barley (Torstensson et al., 2006; Table S3), whereas De Notaris et al. (2018) observed substantially higher leaching rates. They measured 213 kg N ha−1 yr−1 leached when the potato followed green manure and 133 kg N ha−1 yr−1 after grain legumes in a year with early occurring potato late blight. This was substantially higher than in previous years (140 and 78 kg N ha−1, respectively).
Extreme rainfall events and/or periods of drought can significantly affect leaching for a variety of reasons. A field experiment over 13 years in the UK showed that N leaching in winter from fertilised grass (non-organic) was highly correlated with the preceding summer's soil moisture deficit, with the highest losses following dry summers (Tyson et al., 1997). In this case, poor grass growth due to drought led to a build-up of NO3 from unused fertiliser present in the autumn. Prolonged mineralisation of organic fertilisers or crop residues due to drought may also lead to a similar situation in organic farming systems. Tosti et al. (2016) found, under Mediterranean rain-fed conditions, that the risk of NO3 leaching was mainly at the onset of drainage due to rainfall, i.e. at the initial stage of growth, and was typically variable among years depending on the timing of heavy rains. Thus, amendments applied at the pre-crop stage would be a risky practice for NO3 leaching. Most N leaching studies in organic farming in Mediterranean environments focused on row and vegetables crops (e.g. Campanelli and Canali, 2012), because these systems are most demanding in N inputs and thus have higher N applications and potential leaching than in common arable crops.
5.2 Legumes
In a crop rotation with a large contribution from BNF, some of the N inputs will be retained in crop residues and in particular in mulched green manures (Frøseth et al., 2014). In their review, Crews and Peoples (2005) found that when the N input was based on BNF, the proportion of the N retained in the soil was higher (58 % of legume N) than in the fertilised systems (31 % of fertiliser N). From this, it can be assumed that the risk of N release outside the growing season are high in rotations with legumes. However, in their meta-analyses of crop yield and N dynamics as influenced by CCs, Tonitto et al. (2006) concluded that NO3 leaching was reduced by 40 % on average in legume-based systems relative to conventional fertiliser-based systems. The reason for this is probably the large difference in N input between legume-based systems relative to conventional fertiliser-based systems. The response of NO3 leaching to N input in fertiliser, manure and residues may also differ between sites due to soil type and precipitation (Pandey et al., 2018).
5.2.1 Grain legumes
Nitrate leaching reported from crop residues of grain legumes vary. The highest values are found when grain legumes are grown in monoculture rather than in mixtures with e.g. cereals, and when CCs are not used (Plaza-Bonilla et al., 2015). Stenberg et al. (2012) observed higher NO3 leaching after faba bean than after non-leguminous crops. On a clay soil in Sweden, they observed an average leaching of 20 kg N ha−1 yr−1, which was twice the value seen for spring cereals. On average over 3 years on loamy sand in Denmark, De Notaris et al. (2018) reported NO3 leaching that was about twice as high following a barley–pea intercrop compared with spring wheat or spring barley. In a sandy soil in northwestern Germany, Kayser et al. (2010) observed that 83 kg N ha−1 leached in triticale following field bean. In a worst-case scenario, Askegaard et al. (2011) observed annual NO3 leaching of 270 kg N ha−1 during and after a lupin crop on a course sandy soil in a situation where the lupin crop did not ripen, leaving a large amount of N in crop residues (same experiment as De Notaris et al., 2018). Pappa et al. (2008) observed very low N leaching during and after a barley–pea intercrop, but they observed a significant effect of the pea cultivar on N leaching in the autumn and winter period.
5.2.2 Forage legumes
Many authors (Stalenga and Jończyk, 2008; Kayser et al., 2010; Neumann et al., 2011) emphasise that one of the most critical times for NO3 leaching in organic crop rotations occurs after soil incorporation of a grass–clover ley. N leaching is low during the growing period of grass–clover leys (Kayser et al., 2010), but because of the large amounts of mineralised N after the termination of the ley (Sect. 3.3), the risk of NO3 leaching is large for 1–2 years following termination of these crops (Berntsen et al., 2005). The leaching may occur shortly after ley termination, during winter, or during the succeeding seasons, depending on the time of incorporation, quality of the herbage, the weather and the crop sequence.
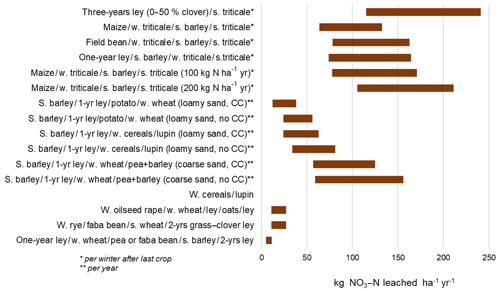
Stenberg et al. (2012) observed higher NO3 leaching following termination of a grass–clover ley than following faba bean, but values were still low (4 kg N ha−1 higher on average). They found the highest leaching when the grass–clover ley lasted for 2 years (up to 40 kg N ha−1 yr−1 leached). This corresponds to the finding of Kayser et al. (2010), who observed greater NO3 leaching during the winter after spring incorporation of a 3-year ley than after a 1-year ley (121 versus 83 kg N ha−1, Fig. 3). However, the crop yield of triticale was much better after the 3-year ley than after the 1-year ley. The percentage share of clover (0–5, 30 % and 50 %) did not influence the amount leached after ley termination, nor did the crop yield. Eriksen et al. (2008) measured NO3 leaching after 1 to 8-year-old grass–clover leys, but found that the length of the ley had no effect on NO3 leaching. Stenberg et al. (2012) observed that cereals succeeding a grass–clover ley had nearly double the yearly N leaching compared with cereals with no legume pre-crop. The highest NO3 leaching occurred after the cultivation of a winter rye (48 kg N ha−1 yr−1).
De Notaris et al. (2018) observed that NO3 leaching during the cultivation of spring wheat was about 50 kg N ha−1 higher when the spring wheat succeeded a 2-year green manure crop (alfalfa or grass–clover) than when it succeeded a grain legume (107 versus 50 kg N ha−1). Similarly, Askegaard et al. (2011) observed peaks in NO3 leaching in autumn and winter after ploughing-in a grass–clover ley. At the crop rotation level, inclusion of grass–clover or alfalfa on 25 % of the area increased the NO3 leaching rate by 6–12 kg N ha−1 (De Notaris et al., 2018). Forage legumes may also be undersown as intercrops to increase soil fertility in organic crop rotations. Pappa et al. (2008) found that clover intercropped in spring barley only increased annual NO3 leaching by 1–2 kg NO3–N ha−1.
5.3 Cover crops
CCs are grown between main crops to minimise NO3 leaching. Many field trials in non-organic systems have shown reduced leaching using CCs (e.g. Rasse et al., 2000; Torstensson and Aronsson, 2000; Constantin et al., 2010; Valkama et al., 2015). This is also the case for organic crop rotations (Tonitto et al., 2006; Askegaard et al., 2011; Tosti et al., 2014, 2016; De Notaris et al., 2018). The reduction in NO3 leaching can be substantial. Studies in Nordic countries report reductions of 50 %–60 % in N leaching (Torstensson and Aronsson, 2000, Fig. 3; Askegaard et al., 2011; De Notaris et al., 2018). If the cash crop fails, the effect of CCs on reduced leaching can be even higher. In a year when potato late blight caused crop failure in potato, the CCs reduced N leaching by 95 % when the potato succeeded a grain legume (from 133 to 6 kg N ha−1 leached), and by 92 % when the potato succeeded a green manure ley (from 213 to 17 kg N ha−1 leached; calculated from Table S3, De Notaris et al., 2018).
De Notaris et al. (2018) concluded that the use of CCs had a larger impact on leaching than a substantial variation in N surplus between alternative cropping systems. In three long-term field trials (13–17 years) in northern France, Constantin et al. (2010) observed that CCs were the most efficient management option for reducing NO3 leaching (from 36 % to 62 %). Good establishment and growth of the CC is essential to obtain sufficient uptake of SMN and thus reduce NO3 leaching. Stenberg et al. (1999) found no significant reduction in NO3 leaching during winter from a ryegrass CC that was undersown in spring. They explained this by poor CC establishment. De Notaris et al. (2018) also observed occasions with a very small effect of CCs on NO3 leaching. They related this to CC growth and identified threshold values in CC above-ground biomass determined in November, above which N leaching was reduced to a stable low level. NO3 leaching from spring wheat averaged 15 kg N ha−1 yr−1 with CC biomass above 0.9 Mg ha−1, and 41 kg N ha−1 yr−1 with CC biomass below 0.9 Mg ha−1. In potatoes, the average N leaching was 11 and 80 kg N ha−1 yr−1 with CC biomass above and below 1.5 Mg ha−1, respectively.
Including legumes in CC mixtures does not seem to reduce the ability of CCs to reduce NO3 leaching (Tonitto et al., 2006; Tosti et al., 2014; De Notaris et al., 2018; Shelton et al., 2018). In a field trial with barley, hairy vetch and a 50:50 mixture of both species as CC, Tosti et al. (2014) found that, in all years, the barley–vetch mixture decreased N leaching to the same level as pure barley, both during its own growing cycle and after CC incorporation into the soil. De Notaris et al. (2018) concluded that the same degree of reduced NO3 leaching was obtained with legume-based CCs as with non-legume CCs. The CC was either undersown in spring or after the harvest of the main crop. The undersown legumes were white clover and red clover, and winter vetch was used in the mixture sown after harvest. Shelton et al. (2018) found greater NO3 leaching from intercropped hairy vetch than from simultaneously grown wheat and wheat–hairy vetch mixture. When the CC is a pure stand of legumes, the CC does not necessarily reduce N leaching (Tosti et al., 2014; Valkama et al., 2015; Shelton et al., 2018). Tosti et al. (2014) concluded that hairy vetch sown as a pure crop in autumn showed high BNF, but no NO3 leaching mitigation effect compared with bare soil. Valkama et al. (2015) found in their meta-analysis of Nordic studies of undersown CCs that legumes (white and red clovers) in pure stand did not diminish the risk of NO3 leaching.
5.4 Tillage
Tillage stimulates soil N mineralisation, at least in the short term. Tillage is also often associated with soil incorporation of plant residues that may lead to net N immobilisation or mineralisation, depending on the quality of the residues. Therefore, timing of tillage is crucial for the fate of the mineralised N, whether SMN becomes available for crop uptake or is subject to leaching. In general, incorporation should consider soil type, climate conditions and type of herbage (C:N ratio). Thorup-Kristensen and Dresbøll (2010) suggested the late incorporation of CCs in high-rainfall areas on sandy soils, and earlier in low-rainfall areas on NO3 retentive soils. Field studies have shown rapid N mineralisation from N-rich plant material, even at low temperatures (Breland, 1994; Thorup-Kristensen and Dresbøll, 2010). Thus, spring incorporation has been recommended to increase N recovery by subsequent crops. However, under Scandinavian conditions, there may still be a deficit in crop-available N, even after spring incorporation of a green manure ley (Känkänen et al., 1998; Frøseth et al., 2014). Under such conditions, Torstensson and Aronsson (2000) suggest that late autumn incorporation of CCs, instead of spring incorporation, would be preferable with respect to N availability for the subsequent crop and would not substantially increase NO3 leaching. Under Mediterranean climatic conditions, characterised by mild, rainy winters and warm to hot, dry summers, there is a risk of NO3 leaching if residues are incorporated prior to the wet season. No studies were found that measured NO3 leaching with respect to the timing of tillage in organic arable farming under these conditions.
In organic crop production, the timing of cultivation for mechanical control of perennial weeds may conflict with the aims of high N use efficiency (Melander et al., 2016). Askegaard et al. (2011) found, on sandy soils in Denmark, that the management of crop and soil during autumn was the main determinant of N leaching. Stubble harrowing in autumn for controlling perennial weeds, followed by bare soil during winter, led to an average of 25 kg N ha−1 more leached than for soils left untouched with a cover of weeds/volunteers. NO3 leaching increased with increasing number of autumn soil cultivations.
Reducing tillage intensity may also enhance the need for weed management, and thereby the risk of NO3 leaching. In their meta-analysis comparing different reduced tillage intensities in organic farming, Cooper et al. (2016) found that the weed incidence was consistently higher, by about 50 %, when tillage intensity was reduced, although this did not always lower the yields. Compared with conventional tillage, reduced tillage may reduce NO3 leaching, but this depends on the establishment and growth of the succeeding crop (Känkänen et al., 1998).
Bare fallow has traditionally been a method to control perennial weeds by repeated tillage of superficial or deeper top soil layers. In practical organic farming, a bare fallow can sometimes be used before or after the main crop if conditions have promoted perennial weeds. However, if carried out in the growing season, soil temperature and moisture conditions favour soil microbial activity and therefore the build-up of SMN, which greatly increases the risk of NO3 leaching (Borgen et al., 2012).
Easily available N and degradable C added through organic inputs enhance the risk of high N2O emissions in the short term due to enhanced biological activity (Sect. 3.3) and increased denitrification potential (Sect. 4.1), and in the longer term because of higher soil content of N and C in labile organic matter (Sect. 3.1) contributing to N2O emissions from mineralised SOM (background emission, Sect. 4.1, 4.3, 4.4, 4.5). There is no strong correlation between total N-input and N2O emissions in organic arable crop rotations (Sect. 4.5), which is another indication of the strong impact of background emissions in these systems (Sect. 4.5). The same is also true of the relatively low impact of episodes with high N2O fluxes on the total N2O emission in various investigations (Sect. 4.5, Fig. 2).
From this, we postulate that background emissions in most organic crop rotations are a more important driver for N2O emissions than episodes with a high N2O flux rate (hot moments). Nevertheless, reducing periods with hot moments of denitrification and thus high N2O fluxes rates are important for reducing total emissions. One mitigation measure is to avoid large applications of residues from crops comprising easily available N and degradable C like clover or Brassica (Sect. 4.3). Mulching of grass–clover on top of a ley seems to only slightly increase N2O emissions compared with no mulch (Sect. 4.2). Incorporating organic material in the soil surface does not reduce the risk of high N2O fluxes, and N2O emissions are often higher after surface application than after ploughing (Sect. 4.3.2). Mixing by rotary harrow can enhance emissions, particularly in moist soil. If the crop residues are removed, composted, used in a biogas plant or treated by other methods before targeted soil application, such events can be avoided. To avoid these measures resulting in GHG emissions being simply moved to another place, this requires measures that minimise GHG emissions during and after treatment of the plant residues (see Sect. 7). The largest stimulation of denitrification via the application of plant residues or other organic material with a high content of degradable C seems to be in soils with a low content of SOC, particularly if the soil has a high NO3 content or if easily available N is applied (Sect. 4.4). If crop residues consist of less degradable C and easily available N, such as straw from cereals and grain legumes, they do not stimulate rapid denitrification and enhanced N2O flux rate (Sect. 4.3), and the N2O flux is likely reduced (Xia et al., 2018).
Background emissions are highly influenced by the content and mineralisation patterns of SOM and the release of SMN and degradable C (Sects. 3 and 4.1). Thus, weather conditions will have a large impact on the background emissions, which are likely to be higher in warm and moist years than in cold or dry years. These conditions cannot be influenced by farmers, but it is important to develop strategies to decrease the SMN content to reduce the risk of N2O emissions and NO3 leaching in periods with the risk of SMN accumulation. The choice and sequence of crops and the use of CC is a central strategy for simultaneously mitigating N2O emissions and NO3 leaching in organic systems. Poor timing of N released from crop residues from preceding crops and crop N uptake (Sects. 3.3 and 5.2) is a major challenge. There is a need to reduce periods with bare soil, although this may conflict with the need for mechanical weed control (Melander et al., 2016).
CCs take up surplus SMN and protect soil from erosion, they reduce NO3 leaching, and during growth CCs do not contribute to N2O emissions (Sect. 4.2). However, in areas with frost, frost sensitive crops like fodder radish, will release easily available N and degradable C during thawing, and thus stimulate denitrification (Sect. 4.3.1). The choice of CC must be adapted to local conditions to ensure good establishment and appropriate tolerance to frost and droughts. A mixture of legumes and non-legumes (for instance grasses or cereals) are just as efficient for reducing N leaching as sole non-legume CCs, whereas sole legumes are not as efficient (Sect. 5.3). CCs containing legumes have a lower C:N ratio than CCs without legumes, which enhances the N fertiliser value for the following crop (Li et al., 2015b).
The knowledge about the rooting pattern of different crop species can be used as a tool for designing crop rotations that achieve higher N use efficiency and thereby reduces the risk of NO3 leaching. Deep-rooted crops, especially tap-rooted crops, can recover NO3 from deeper soil layers before and after more shallow-rooted cash crops, such as leek (Thorup-Kristensen, 2006). As shown by Fan et al. (2016), the root distribution and rooting depth may differ between varieties, although plant breeders do not normally select crops based on the root system.
For the choice of crop, species and varieties should be well-adapted to the climate conditions on the farm and the soil fertility level. Crops in good conditions also compete more successfully against weeds. This decreases the need for soil management to achieve weed control, and thereby reduces the risk of NO3 leaching. The timing of the release of N from residues and amendments and crop uptake are crucial for minimising the risk of NO3 leaching. This can be achieved by timing of soil tillage and the incorporation of residues. In any case, the effect of mitigation strategy is highly dependent on soil type and precipitation.
Because of the large impact of poorly aerated soil on N2O emissions (Sect. 4.1) and plant growth, measures should be taken to improve and maintain a good soil structure. In organic production, soil fauna, microorganisms and the development and maintenance of soil structure, are supported by crop rotations that include legumes or grass–clover leys, use of CCs and the application of organic fertilisers. Even so, traffic and tillage under wet soil conditions are damaging to soil structure and should therefore be avoided.
A way to reduce N2O emissions and NO3 leaching per unit produced could be to increase yields in organic production as more land is commonly needed per unit product in organic than in non-organic production (De Ponti et al., 2012; Meier et al., 2015). However, as discussed by Röös et al. (2018) this is not straightforward as many of the available measures have negative side effects. More targeted, and thus efficient, use of N applied through crop residues and organic fertilisers seems to have co-benefits in terms of higher productivity as well as reduced N2O emissions and NO3 leaching. This may be achieved by the recycling of these residues through biogas and targeted application to crops according to their N demand (Brozyna et al., 2013; Knudsen et al., 2014).
The knowledge of N cycling and losses in arable organic farming is constrained by few investigations that have adequately quantified the magnitude and timing of N2O emissions and NO3 leaching. Many of the reported results are from a few experiments, including a long-term experiment in Denmark (Olesen et al., 2000). Such long-term experiments are important for quantifying N transformation and loss processes in organic cropping systems, where the legacy effects of N in plant residues and other organic amendments are often considerably larger than in non-organic systems. Lessons from these experiments can help to develop methods and technologies to improve synchronisation of N released from crop residues and soil organic matter and the N demand of cash crops. However, there are few long-term experiments with organic arable crop rotations, and even fewer have been systematically used for quantifying the N cycling processes. Therefore, research on existing and new long-term experiments covering relevant soil and climatic conditions should be coordinated to make data and facilities available for research on N dynamics, including N2O emissions, NO3 leaching, NH3 volatilisation and the fate of added N. In this way, useful lessons can be learned regarding the impact of long-term management on the N dynamics. The availability of historical data on N input and crop management is a prerequisite to achieve this, and such data could be made available and structured through a coordination of long-term experiments.
CCs have been shown to efficiently reduce NO3 leaching (Sect. 5.3), whereas they can reduce or increase N2O emissions depending on the conditions (Sect. 4.2, 4.3). To optimise the use of CCs for crop productivity, environment and climate, improved knowledge is needed of locally adapted crop rotations and CCs that maintain living plants that can take up available N and simultaneously reduce weed pressure.
Simulation models can be used to quantify N transformation and loss processes and how different management practices affect N storage in SOM, as well and short-term effects of crop residues and organic fertilisers on N2O emissions and NO3 leaching. However, process-based models fail to accurately simulate the impact of grass–clover leys on the turnover of N and C (Frøseth, 2016; Doltra et al., 2019). To our knowledge, no field-scale model includes all C and N turnover processes, such as loss of C and N from above ground tissues (phyllodeposition) and rhizodeposition. These processes may be of greater importance in organic than in non-organic systems, because of the greater emphasis on fertility building measures. This may lead to an underestimation of the amount of N and C returned to soil. Improving models on these aspects requires more comprehensive data on all C and N inputs and flows in the cropping systems. This can only be achieved through studies using isotopic labelling of C and N, and consideration should be given to also conducting such studies in long-term experiments. There is further uncertainty on the release of N from legumes and thus the impact of legume residues on N2O emission and NO3 leaching (see Sects. 4.2, 4.3 and 5.2). Therefore, more research on the mineralisation pattern of various legume residues would be useful.
Heterogeneity is another aspect that makes it difficult to predict the availability of N and C in organic systems and thus N2O emissions. In addition to the soil heterogeneity, variability is created from incorporated crop residues, CCs, short-term leys and organic fertiliser that are seldom evenly distributed. The uneven impact of freezing/thawing and drying/rewetting will add to this variability and make it difficult to predict hot spots and hot moments of N2O emissions. There is also a need to develop technologies to better measure this heterogeneity and to account for the heterogeneity in N dynamic models adapted to organic crop rotations.
Composting of crop residues and manures is common on organic farms and is a way to make the organic matter more resistant and useful as a soil amendment. However, composting manure without emissions of N2O and other greenhouse gases is challenging (Chadwick et al., 2011) and needs to be addressed to develop a more comprehensive picture of greenhouse gas emissions from organic arable production and how to mitigate them. Neither composting nor biogas fermentation are always feasible. Improved understanding of different pathways for microbial degradation of crop residues is also needed, to better manage crop residues and organic waste for C and N retention properties.
Organic arable production is based on diverse crop rotations, N inputs via BNF in legumes, external inputs of manure and compost, and the recovery of excess N in grassland and cover crops. This results in considerable inputs on N in organic matter from plant residues, green manures and the application of organic fertilisers and soil amendments that enhance the content of total and labile SOC in organic arable production compared with non-organic production. When the conditions for the mineralisation of SOC are met this will lead to a high availability of easily available N and degradable C, and an enhanced content of SMN, if available N is not taken up by growing plants. Conditions with a high content of SMN and degradable C provide hot moments for high N2O fluxes. Degradable C will increase microbial growth and thus O2 consumption in soil leading to anaerobic conditions; this provides suitable conditions for the denitrification of available NO3. In organic arable crop rotations background emissions are more important in most situations than episodes with high N2O fluxes with respect to the total N2O emissions from organic arable crop rotations. SMN from mineralisation of SOM and plant residues in combination with periods of bare soil or sparse plant growth and precipitation surplus provide drivers for NO3 leaching.
Main mitigation strategies are the targeted use of growing plants to take up surplus soil mineral N from the root zone, and the targeted use of crop residues and organic fertilisers to synchronise the availability of N with the crop demand. Continued use of CCs has the proven ability to reduce NO3 leaching from organic arable crop rotations but does increase N2O emissions in some situations. A CC mixture of legumes and non-legumes (for instance grasses or cereals) is just as efficient as sole non-legumes and has a better impact on soil fertility than non-legumes. More research is needed to develop locally adapted crop rotations and CC mixtures that concurrently reduce N2O emissions and NO3 leaching.
The underlying research data used in this article is given in the Supplement Tables S1–S3, and S4 and S5.
| Abbreviations | |
| BNF | Biological nitrogen fixation |
| C | Carbon |
| CC | Cover crops |
| CH4 | Methane |
| EF | Emission factor, which refers to the percentage of N applied emitted as N2O–N |
| N | Nitrogen |
| N2O | Nitrous oxide |
| NO3 | Nitrate |
| PMN | Potentially mineralisable N |
| SMN | Soil mineral nitrogen |
| SOC | Soil organic carbon |
| SOM | Soil organic matter |
| Terminology | |
| Background emissions | N2O emissions that derive from N released from SOM as opposed to N added through fertilisation, soil amendments or plant residue applied in the current year. |
| Degradable C | Easily degradable carbon compounds. |
| Hot moments of N2O flux | Brief and disproportionally high short-term N2O flux event due to the combination of multiple influencing factors (Molodovskaya et al., 2012). |
| N2O emission | The cumulative flux reported for one field treatment during the actual measurement period. |
| NO3 leached | NO3 transported below the root zone. |
| PMN | The amount of SMN released from organic matter under ideal soil environmental conditions. |
| SMN | The content of mineral N in the form of ammonium (NH4) and NO3 in soil. |
The supplement related to this article is available online at: https://doi.org/10.5194/bg-16-2795-2019-supplement.
SH was responsible for the overall development and content of the paper with contributions from all co-authors, in addition to Table S2 and Fig. S2. RBF and MS were jointly responsible for N leaching and Table S3. RBF was responsible for Fig. 3 and contributed to Table S1 with data from Norway. JS was responsible for SMN, Table S1 and Table 1. JEO contributed to the overall content, structure and language of the paper, and was responsible for Fig. 1, as well as complementary data from the Danish field trials in Table S2. MK contributed to the content of the paper and complementary data from a Swiss field trial in Table S2. PR was responsible for the section on earthworms, and the main editing of the references and reference list. JD contributed to the content of the paper, mainly from a Mediterranean perspective. SN contributed to the content of the paper, mainly N2O and complementary data from Norwegian field trial in Table S2. TT was responsible for the statistical analyses on the cause of N2O fluxes based on Table S2. VP contributed the complementary data from field trials in Edinburgh and Aberdeen in Table S2. CW contributed to the overall development and content of the paper and was responsible for language editing.
The authors declare that they have no conflict of interest.
In addition to the FertilCrop project the authors also acknowledge support from the Scottish Government RESAS Strategic Research Programme 2016-2021 and from Møre og Romsdal county council. The text in this report is the sole responsibility of the authors and does not necessarily reflect the views of the funding bodies that financed this project. The authors acknowledge Bruce Ball for complementary data from a field trial in Aberdeen used in Table S2, and an anonymous referee for valuable and comprehensive comments.
This research (FertilCrop project) has been supported by the FP7 ERA-net project, CORE Organic Plus and the co-fund from the European Commission (grant no. 563).
This paper was edited by Lutz Merbold and reviewed by two anonymous referees.
Aguilera, E., Lassaletta, L., Gattinger, A., and Gimeno, B. S.: Managing soil carbon for climate change mitigation and adaptation in Mediterranean cropping systems: A meta-analysis, Agr. Ecosyst. Environ., 168, 25–36, https://doi.org/10.1016/j.agee.2013.02.003, 2013.
Anderson, J. H.: The metabolism of hydroxylamine to nitrite by Nitrosomonas, Biochem. J., 91, 8–17, 1964.
Anderson, T.-H. and Domsch, K. H.: Ratios of microbial biomass carbon to total organic carbon in arable soils, Soil Biol. Biochem., 21, 471–479, https://doi.org/10.1016/0038-0717(89)90117-X, 1989.
Aronsson, H., Torstensson, G., and Bergstrom, L.: Leaching and crop uptake of N, P and K from organic and conventional cropping systems on a clay soil, Soil Use Manage., 23, 71–81, 2007.
Askegaard, M., Olesen, J. E., and Kristensen, K.: Nitrate leaching from organic arable crop rotations: effects of location, manure and catch crop, Soil Use Manage., 21, 181–188, 2005.
Askegaard, M., Olesen, J. E., Rasmussen, I. A., and Kristensen, K.: Nitrate leaching from organic arable crop rotations is mostly determined by autumn field management, Agr. Ecosyst. Environ., 142, 149–160, https://doi.org/10.1016/j.agee.2011.04.014, 2011.
Baggs, E. M., Rees, R. M., Smith, K. A., and Vinten, A. J. A.: Nitrous oxide emission from soils after incorporating crop residues, Soil Use Manage., 16, 82–87, 2000a.
Baggs, E. M., Watson, C. A., and Rees, R. M.: The fate of nitrogen from incorporated cover crop and green manure residues, Nutr. Cycl. Agroecosys., 56, 153–163, 2000b.
Bakken, L. R., Bergaust, L., Liu, B., and Frostegård, A.: Regulation of denitrification at the cellular level: a clue to the understanding of N2O emissions from soils., Philos. T. R. Soc. B., 367, 1226–1234, https://doi.org/10.1098/rstb.2011.0321, 2012.
Balaine, N., Clough, T. J., Beare, M. H., Thomas, S. M., Meenken, E. D., and Ross, J. G.: Changes in Relative Gas Diffusivity Explain Soil Nitrous Oxide Flux Dynamics, Soil Sci. Soc. Am. J., 77, 1496, https://doi.org/10.2136/sssaj2013.04.0141, 2013.
Ball, B. C., Mctaggart, I. P., and Watson, C. A.: Influence of organic ley-arable management and afforestation in sandy loam to clay loam soils on fluxes of N2O and CH4 in Scotland, Agr. Ecosyst. Environ., 90, 305–317, 2002.
Ball, B. C., Watson, C. A., and Crichton, I.: Nitrous oxide emissions, cereal growth, N recovery and soil nitrogen status after ploughing organically managed grass/clover swards, Soil Use Manage., 23, 145–155, https://doi.org/10.1111/j.1475-2743.2006.00072.x, 2007.
Baral, K. R., Labouriau, R., Olesen, J. E., and Petersen, S. O.: Nitrous oxide emissions and nitrogen use efficiency of manure and digestates applied to spring barley, Agr. Ecosyst. Environ., 239, 188–198, https://doi.org/10.1016/j.agee.2017.01.012, 2017.
Barbieri, P., Pellerin, S., and Nesme, T.: Comparing crop rotations between organic and conventional farming, Sci. Rep.-UK, 7, 1–10, https://doi.org/10.1038/s41598-017-14271-6, 2017.
Benoit, M., Garnier, J., Anglade, J., and Billen, G.: Nitrate leaching from organic and conventional arable crop farms in the Seine Basin (France), Nutr. Cycl. Agroecosys., 100, 285–299, 2014.
Berntsen, J., Petersen, B. M., Olesen, J. E., Eriksen, J. E., and Soegaard, K.: Simulation of residual effects and nitrate leaching after incorporation of different ley types, Eur. J. Agron., 23, 290–304, 2005.
Berntsen, J., Petersen, B. M., Sørensen, P., and Olesen, J. E.: Simulating Residual Effects of Animal Manures Using N-15 Isotopes, Plant Soil, 290, 173–187, 2007.
Berry, B. M., Sylvester-Bradley, R., Philipp, L., Hatch, D. J., Cuttle, S. P., Rayns, F. W., and Gosling, P.: Is the productivity of organic farms restricted by the supply of available nitrogen?, Soil Use Manage., 18, 248–255, https://doi.org/10.1111/j.1475-2743.2002.tb00266.x, 2002.
Bhogal, A., Williams, J. R., Nicholson, F. A., Chadwick, D. R., Chambers, K. H., and Chambers, B. J.: Mineralization of organic nitrogen from farm manure applications, Soil Use Manage., 32, 32–43, 2016.
Borgen, S. K., Lunde, H. W., Bakken, L. R., Bleken, M. A., and Breland, T. A.: Nitrogen dynamics in stockless organic clover-grass and cereal rotations, Nutr. Cycl. Agroecosys., 92, 363–378, 2012.
Bouwman, A. F., Boumans, L. J. M., and Batjes, N. H.: Modeling global annual N2O and NO emissions from fertilized fields, Global Biogeochem. Cy., 16, 1080, https://doi.org/10.1029/2001GB001812, 2002.
Breland, T. A.: Measured and predicted mineralization of clover green manure at low temperatures at different depths in two soils, Plant Soil, 166, 13–20, 1994.
Bronick, C. J. and Lal, R.: Soil structure and management: A review, Geoderma, 124, 3–22, https://doi.org/10.1016/j.geoderma.2004.03.005, 2005.
Brophy, L. S., Heichel, G. H., and Russelle, M. P.: Nitrogen transfer from forage legumes to grass in a systematic planting design, Crop Sci., 27, 753–758, 1987.
Brozyna, M. A., Petersen, S. O., Chirinda, N., and Olesen, J. E.: Effects of grass-clover management and cover crops on nitrogen cycling and nitrous oxide emissions in a stockless organic crop rotation, Agr. Ecosyst. Environ., 181, 115–126, https://doi.org/10.1016/j.agee.2013.09.013, 2013.
Burke, I. C., Yonker, C. M., Parton, W. J., Cole, C. V., Flach, K., and Schimel, D. S.: Texture, Climate, and Cultivation Effects on Soil Organic Matter Content in U.S. Grassland Soils, Soil Sci. Sos. Am. J., 53, 800–805, 1989.
Butterbach-Bahl, K., Baggs, E. M., Dannenmann, M., Kiese, R., and Zechmeister-Boltenstern, S.: Nitrous oxide emissions from soils: how well do we understand the processes and their controls?, Philos. T. R. Soc. B., 368, 20130122, https://doi.org/10.1098/rstb.2013.0122, 2013.
Campanelli, G. and Canali, S.: Crop production and environmental effects in conventional and organic vegetable farming systems: The case of a long-term experiment in Mediterranean conditions (Central Italy), J. Sustain. Agr., 36, 599–619, 2012.
Carter, M. S. and Ambus, P.: Biologically fixed N2 as a source for N2O production in a grass-clover mixture, measured by 15N2, Nutr. Cycl. Agroecosys., 74, 13–26, 2006.
Chadwick, D., Sommer, S., Thorman, R., Fangueiro, D., Cardenas, L., Amon, B., and Misselbrook, T.: Manure management: Implications for greenhouse gas emissions, Anim. Feed Sci. Tech., 166–167, 514–531, 2011.
Chang, C., Cho, C. M., and Janzen, H. H.: Nitrous oxide emission from long-term manured soils, Soil Sci. Soc. Am. J., 62, 677–682, 1998.
Chantigny, M. H., Angers, D. A., Rochette, P., Bélanger, G., Massé, D., and Côté, D.: Gaseous Nitrogen Emissions and Forage Nitrogen Uptake on Soils Fertilized with Raw and Treated Swine Manure, J. Environ. Qual., 36, 1864, https://doi.org/10.2134/jeq2007.0083, 2007.
Chantigny, M. H., Rochette, P., Angers, D. A., Bittman, S., Buckley, K., Massé, D., Bélanger, G., Eriksen-Hamel, N., and Gasser, M.-O.: Soil Nitrous Oxide Emissions Following Band-Incorporation of Fertilizer Nitrogen and Swine Manure, J. Environ. Qual., 39, 1545–1553, https://doi.org/10.2134/jeq2009.0482, 2010.
Charles, A., Rochette, P., Whalen, J. K., Angers, D. A., Chantigny, M. H., and Bertrand, N.: Global nitrous oxide emission factors from agricultural soils after addition of organic amendments: A meta-analysis, Agr. Ecosyst. Environ., 236, 88–98, https://doi.org/10.1016/j.agee.2016.11.021, 2017.
Chen, H. H., Li, X. C., Hu, F., and Shi, W.: Soil nitrous oxide emissions following crop residue addition: a meta-analysis, Global Change Biol., 19, 2956–2964, https://doi.org/10.1111/gcb.12274, 2013.
Chirinda, N., Carter, M. S., Albert, K. R., Ambus, P., Olesen, J. E., Porter, J. R., and Petersen, S. O.: Emissions of nitrous oxide from arable organic and conventional cropping systems on two soil types, Agr. Ecosyst. Environ., 136, 199–208, 2010.
Congreves, K. A., Wagner-Riddle, C., Si, B. C., and Clough, T. J.: Nitrous oxide emissions and biogeochemical responses to soil freezing-thawing and drying-wetting, Soil Biol. Biochem., 117, 5–15, https://doi.org/10.1016/j.soilbio.2017.10.040, 2018.
Constantin, J., Mary, B., Laurent, F., Aubrion, G., Fontaine, A., Kerveillant, P., and Beaudoin, N.: Effects of catch crops, no till and reduced nitrogen fertilization on nitrogen leaching and balance in three long-term experiments, Agr. Ecosyst. Environ., 135, 268–278, https://doi.org/10.1016/j.agee.2009.10.005, 2010.
Cooper, J., Baranski, M., Stewart, G., Nobel-de Lange, M., Bàrberi, P., Fließbach, A., Peigné, J., Berner, A., Brock, C., Casagrande, M., Crowley, O., David, C., De Vliegher, A., Döring, T. F., Dupont, A., Entz, M., Grosse, M., Haase, T., Halde, C., Hammerl, V., Huiting, H., Leithold, G., Messmer, M., Schloter, M., Sukkel, W., van der Heijden, M. G. A., Willekens, K., Wittwer, R., and Mäder, P.: Shallow non-inversion tillage in organic farming maintains crop yields and increases soil C stocks: a meta-analysis, Agron. Sustain. Dev., 36, https://doi.org/10.1007/s13593-016-0354-1, 2016.
Council of the European Union: Council Regulation (EC) No 834/2007 of 28 June 2007 on organic production and labelling of organic products and repealing Regulation (EEC) No 2092/91, Official Journal of the European Union, L 189/1, available at: http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32007R0834&from=EN (last access: 12 July 2019) 2007.
Crews, T. E. and Peoples, M. B.: Can the synchrony of nitrogen supply and crop demand be improved in legume and fertilizer-based agroecosystems? A review, Nutr. Cycl. Agroecosys., 72, 101–120, https://doi.org/10.1007/s10705-004-6480-1, 2005.
Dalgaard, T., Hansen, B., Hasler, B., Hertel, O., Hutchings, N. J., Jacobsen, B., Kronvang, B., Olesen, J. E., Schjørring, J., Termansen, M., and Vejre, H.: Policies for agricultural nitrogen management – trends, challenges and prospects for improved efficiency in Denmark, Environ. Res. Lett., 9, 115002, https://doi.org/10.1088/1748-9326/9/11/115002, 2014.
De Notaris, C., Rasmussen, J., Sørensen, P., and Olesen, J. E.: Nitrogen leaching: A crop rotation perspective on the effect of N surplus, field management and use of catch crops, Agr. Ecosyst. Environ., 255, 1–11, https://doi.org/10.1016/J.AGEE.2017.12.009, 2018.
De Ponti, T., Rijk, B., and Van Ittersum, M. K.: The crop yield gap between organic and conventional agriculture, Agr. Syst., 108, 1–9, https://doi.org/10.1016/j.agsy.2011.12.004, 2012.
Di, H. J. and Cameron, K. C.: Nitrate leaching in temperate agroecosystems: sources, factors and mitigating strategies, Nutr. Cycl. Agroecosys., 64, 237–256, 2002.
Dobbie, K. E. and Smith, K. A.: Nitrous oxide emission factors for agricultural soils in Great Britain: the impact of soil water filled pore space and other controlling variables, Global Change Biol., 99, 204–218, https://doi.org/10.1046/j.1365-2486.2003.00563.x, 2003.
Doltra, J., Gallejones, P., Olesen, J. E., Hansen, S., Frøseth, R. B., Krauss, M., Stalenga, J., Jończyk, K., Martínez-Fernández, A., and Pacini, G. C.: Simulating soil fertility management effects on crop yield and soil nitrogen dynamics in field trials under organic farming in Europe, Field Crop. Res. 233, 1–11, https://doi.org/10.1016/j.fcr.2018.12.008, 2019.
Dunbabin, V., Diggle, A., and Rengel, Z.: Is there an optimal root architecture for nitrate capture in leaching environments?, Plant Cell Environ., 26, 835–844, 2003.
Dusenbury, M. P., Engel, R. E., Miller, P. R., Lemke, R. L., and Wallander, R.: Nitrous Oxide Emissions from a Northern Great Plains Soil as Influenced by Nitrogen Management and Cropping Systems, J. Environ. Qual., 37, 542–550, https://doi.org/10.2134/jeq2006.0395, 2008.
Eriksen, J., Askegaard, M., and Søegaard, K.: Residual effect and nitrate leaching in grass-arable rotations: effect of grassland proportion, sward type and fertilizer history, Soil Use Manage., 24, 373–382, 2008.
Evers, A. K., Demers, T. A., Gordon, A. M., and Thevathasan, N. V.: The effect of earthworm (Lumbricus terrestris L.) population density and soil water content interactions on nitrous oxide emissions from agricultural soils, Appl. Environ. Soil Sci., 2010, 737096, https://doi.org/10.1155/2010/737096, 2010.
Fan, J., McConkey, B., Wang, H., and Janzen, H.: Root distribution by depth for temperate agricultural crops, Field Crop. Res., 189, 68–74, https://doi.org/10.1016/j.fcr.2016.02.013, 2016.
Filser, J., Dette, A., Fromm, H., Lang, A., Munch, J. C., Winter, K., and Beese, F.: Reactions of soil organisms in site-specific management. The first long-term study at the landscape scale, Ecosyst. Suppl., 28, 139–147, 1999.
Firestone, M. and Davidson, E.: Microbiological basis of NO and N2O production and consumption in soil, in: Exchange of trace gases between terrestrial ecosystems and the atmosphere, edited by: Andreac, M. O. and Schimel, D. S., John Wiley & Sons Ltd., Hoboken, NJ, USA, 7–21, 1989.
Flessa, H., Dörsch, P., and Beese, F.: Seasonal variation of N2O and CH4 fluxes in differently managed arable soils in southern Germany, J. Geophys. Res., 100, 115–124, 1995.
Frøseth, R. B.: Nitrogen dynamics in an organic green manure – cereal rotation and mineralization of clover leaves at low temperature, Philosophiae Doctor (PhD) Thesis 2016:25, Norwegian University of Life Sciences, Ås, Norway, 58 pp., 2016.
Frøseth, R. B., Bakken, A. K., Bleken, M. A., Riley, H., Pommeresche, R., Thorup-Kristensen, K., and Hansen, S.: Effects of green manure herbage management and its digestate from biogas production on barley yield, N recovery, soil structure and earthworm populations, Eur. J. Agron., 52, 90–102, https://doi.org/10.1016/j.eja.2013.10.006, 2014.
Gattinger, A., Muller, A., Haeni, M., Skinner, C., Fliessbach, A., Buchmann, N., Mäder, P., Stolze, M., Smith, P., Scialabba, N. E.-H., and Niggli, U.: Enhanced top soil carbon stocks under organic farming, P. Natl. Acad. Sci. USA, 109, 18226–18231, https://doi.org/10.1073/pnas.1209429109, 2012.
Gomiero, T.: Alternative Land Management Strategies and Their Impact on Soil Conservation, Agriculture, 3, 464–483, https://doi.org/10.3390/agriculture3030464, 2013.
Hansen, E. M., Eriksen, J., and Vinther, F. P.: Catch crop strategy and nitrate leaching following grazed grass-clover. Soil Use Manage., 23, 348–358, 2007.
Hansen, S. and Engelstad, F.: Earthworm populations in a cool and wet district as affected by tractor traffic and fertilisation, Appl. Soil Ecol., 13, 237–250, https://doi.org/10.1016/S0929-1393(99)00037-2, 1999.
Hansen, S., Bernard, M.-E., Rochette, P., Whalen, J. K., and Dörsch, P.: Nitrous oxide emissions from a fertile grassland in Western Norway following the application of inorganic and organic fertilizers, Nutr. Cycl. Agroecosys., 98, 71–85, https://doi.org/10.1007/s10705-014-9597-x, 2014.
Hartmann, M., Frey, B., Mayer, J., Mäder, P., and Widmer, F.: Distinct soil microbial diversity under long-term organic and conventional farming, ISME J., 9, 1177–1194, https://doi.org/10.1038/ismej.2014.210, 2015.
Holtan-Hartwig, L., Dorsch, P., and Bakken, L. R.: Low temperature control of soil denitrifying communities: kinetics of N2O production and reduction, Soil Biol. Biochem., 34, 1797–1806, 2002.
Hu, T., Sørensen, P., and Olesen, J .E.: Soil carbon varies between different organic and conventional management schemes in arable agriculture, Eur. J. Agron., 94, 79–88, 2018a.
Hu, T., Olesen, J. E., Christensen, B. T., and Sørensen, P.: Release of C and N from fodder radish (Raphanus sativus) shoots and roots incubated in soils of different management history, Acta Agr. Scand. B-S. P., 68, 749–756, 2018b.
IFOAM: The Principles of Organic Agriculture, available at: https://www.ifoam.bio/sites/default/files/poa_english_web.pdf, last access: April 2019.
IPCC: Guidelines for National Greenhouse Gas Inventories. Agriculture, Forestry and Other Land Use, International Panel oif Climate Change, available at: https://www.ipcc-nggip.iges.or.jp/public/2006gl/index (last accessL July 2019), 2006.
Jensen, E. S., Peoples, M. B., Boddey, R. M., Gresshoff, P. M., Hauggaard-Nielsen, H., Alves, B. J. R., and Morrison, M. J.: Legumes for mitigation of climate change and the provision of feedstock for biofuels and biorefineries. A review, Agron. Sustain. Dev., 32, 329–364, https://doi.org/10.1007/s13593-011-0056-7, 2012.
Jeuffroy, M. H., Baranger, E., Carrouée, B., de Chezelles, E., Gosme, M., Hénault, C., Schneider, A., and Cellier, P.: Nitrous oxide emissions from crop rotations including wheat, oilseed rape and dry peas, Biogeosciences, 10, 1787–1797, https://doi.org/10.5194/bg-10-1787-2013, 2013.
Johnsson, H., Bergström, L., Jansson, P.-E., and Paustian, K.: Simulated nitrogen dynamics and losses in a layered agricultural soil, Agr. Ecosyst. Environ., 18, 333–356, 1987.
Jończyk, K. and Martyniuk, S.: Productivity of crop rotations and selected indicators of soil fertility in different types of ecological farms, J. Res. Appl. Agric. Eng., 62, 153–157, 2017.
Känkänen, H., Kangas, A., Mela, T., Nikunen, U., Tuuri, H., and Vuorinen, M.: Timing incorporation of different green manure crops to minimize the risk of nitrogen leaching, Agric. Food Sci. Finland., 7, 553–567, 1998.
Kayser, M., Müller, J., and Isselstein, J.: Nitrogen management in organic farming: Comparison of crop rotation residual effects on yields, N leaching and soil conditions, Nutr. Cycl. Agroecosys., 87, 21–31, https://doi.org/10.1007/s10705-009-9309-0, 2010.
Kirchmann, H. and Bergström, L.: Do organic farming practices reduce nitrate leaching?, Commun. Soil Sci. Plan., 32, 997–1028, 2001.
Knudsen, M. T., Meyer-Aurich, A., Olesen, J. E., Chirinda, N., and Hermansen, J. E.: Carbon footprints of crops from organic and conventional arable crop rotations using a life cycle assessment approach, J. Clean. Prod., 64, 609–618, 2014.
Köster, J. R., Cárdenas, L., Senbayram, M., Bol, R., Well, R., Butler, M., Mühling, K. H., and Dittert, K.: Rapid shift from denitrification to nitrification in soil after biogas residue application as indicated by nitrous oxide isotopomers, Soil Biol. Biochem., 43, 1671–1677, https://doi.org/10.1016/j.soilbio.2011.04.004, 2011.
Krauss, M., Krause, H. M., Spangler, S., Kandeler, E., Behrens, S., Kappler, A., Mäder, P., and Gattinger, A.: Tillage system affects fertilizer-induced nitrous oxide emissions, Biol. Fert. Soils, 53, 49–59, https://doi.org/10.1007/s00374-016-1152-2, 2017a.
Krauss, M., Ruser, R., Müller, T., Hansen, S., Mäder, P., and Gattinger, A.: Impact of reduced tillage on greenhouse gas emissions and soil carbon stocks in an organic grass-clover ley – winter wheat cropping sequence, Agr. Ecosyst. Environ., 239, 324–333, https://doi.org/10.1016/j.agee.2017.01.029, 2017b.
Kuiper, I., Deyn G. B., Thakur M. P., and Groenigen J. W.: Soil invertebrate fauna affect N2O emissions from soil, Global Change Biol., 19, 2814–2825, 2013.
Kuntz, M., Morely, N. J., Hallett, P. D., Watson, C., and Baggs, E. M.: Residue-C effects on dinitrification vary with soil depth, Soil Biol. Biochem., 103, 365–375, 2016.
Larsson, L., Ferm, M., Kasimir-Klemedtsson, A., and Klemedtsson, L.: Ammonia and nitrous oxide emissions from grass and alfalfa mulches, Nutr. Cycl. Agroecosys., 51, 41–46, 1998.
Li, C. S., Frolking, S., and Butterbach-Bahl, K.: Carbon Sequestration in Arable Soils Is Likely to Increase Nitrous Oxide Emissions, Offsetting Reductions in Climate Radiative Forcing, Climatic Change, 72, 321–338, 2005.
Li, X., Petersen, S. O., Sørensen, P., and Olesen, J. E.: Effects of contrasting catch crops on nitrogen availability and nitrous oxide emissions in an organic cropping system, Agr. Ecosyst. Environ., 199, 382–393, https://doi.org/10.1016/j.agee.2014.10.016, 2015a.
Li, X., Sørensen, P., Li, F., Petersen, S. O. and Olesen, J. E.: Quantifying biological nitrogen fixation of different catch crops, and the residual effects of roots and tops on nitrogen uptake in barley using in situ 15N labelling, Plant Soil, 395, 273–287, 2015b.
Li, X., Sørensen, P., Olesen, J. E., and Petersen, S. O.: Evidence for denitrification as main source of N2O emission from residue-amended soil, Soil Biol. Biochem., 92, 153–160, https://doi.org/10.1016/j.soilbio.2015.10.008, 2016.
Liu, B., Mørkved, P. T., Frostegård, Å., and Bakken, L. R.: Denitrification gene pools, transcription and kinetics of NO, N2O and N2 production as affected by soil pH, FEMS Microbiol. Ecol., 72, 407–417, https://doi.org/10.1111/j.1574-6941.2010.00856.x, 2010.
Liu, S., Han, P., Hink, L., Prosser, J., Wagner M., and Brüggemann N.: Abiotic conversion of extracellular NH2OH contributes to N2O emission during ammonia oxidation, Environ. Sci. Technol., 51, 13122–13132, https://doi.org/10.1021/acs.est.7b02360, 2017.
Løes, A.-K., Bünemann, E. K., Cooper, J., Hörtenhuber, S., Magid, J., Oberson, A., and Möller, K.: Nutrient supply to organic agriculture as governed by EU regulations and standards in six European countries, Org. Agric., 7, 395–418, https://doi.org/10.1007/s13165-016-0165-3, 2017.
Lorenz, K. and Lal, R.: Environmental Impact of Organic Agriculture, Adv. Agron., 139, 99–152, 2016.
Lori, M., Symnaczik, S., Mäder, P., De Deyn, G., Gattinger, A., and Dentener, F.: Organic farming enhances soil microbial abundance and activity – A meta-analysis and meta-regression, PLoS One, 12, 1–25, https://doi.org/10.1371/journal.pone.0180442, 2017.
Lubbers, I. M., Brussaard, L., Otten, W., and Van Groenigen, J. W.: Earthworm induced N mineralization in fertilized grassland increases both N2O emission and crop N uptake, Eur. J. Soil Sci., 62, 152–161, 2011.
Lubbers I. M., Van Groenigen K. J., Fonte S. J., Six J., Brussaard L., and Van Groenigen J. W.: Greenhouse-gas emissions from soils increased by earthworms, Nat. Clim. Change, 3, 187–194, 2013.
Lynch, D. H.: Nutrient Cycling and Soil Health in Organic Cropping Systems – Importance of Management Strategies and Soil Resilience, Sustain. Agric. Res., 4, 80, https://doi.org/10.5539/sar.v4n3p80, 2015.
Mäder, P., Fliessbach, A., Dubois, D., Gunst, L., Fried, P., and Niggli, U.: Soil fertility and biodiversity in organic farming, Science, 296, 1694–1697, https://doi.org/10.1126/science.1071148, 2002.
Marinari, S., Masciandaro, G., Ceccanti, B., and Grego, S.: Evolution of soil organic matter changes using pyrolysis and metabolic indices: A comparison between organic and mineral fertilization, Bioresource Technol., 98, 2495–2502, https://doi.org/10.1016/j.biortech.2006.09.001, 2007.
Marriott, E. E. and Wander, M. M.: Total and Labile Soil Organic Matter in Organic and Conventional Farming Systems, Soil Sci. Soc. Am. J., 70, 950–959, https://doi.org/10.2136/sssaj2005.0241, 2006.
Martyniuk, S., Koziel, M., Stalenga, J., and Jonczyk, K.: Labile factions of soil organic matter and microbial characteristics of soil under organic and conventional crop management systems, Biol. Agric. Hortic., 32, 1–6, https://doi.org/10.1080/01448765.2015.1015054, 2016.
McGee, J. A.: Does certified organic farming reduce greenhouse gas emissions from agricultural production?, Agric. Hum. Values, 32, 255–263, https://doi.org/10.1007/s10460-014-9543-1, 2015.
Meier, M. S., Stoessel, F., Jungbluth, N., Juraske, R., Schader, C., and Stolze, M.: Environmental impacts of organic and conventional agricultural products – Are the differences captured by life cycle assessment?, J. Environ. Manage., 149, 193–208, https://doi.org/10.1016/j.jenvman.2014.10.006, 2015.
Meijide, A., Díez, J. A., Sánchez-Martín, L., López-Fernández, S., and Vallejo, A.: Nitrogen oxide emissions from an irrigated maize crop amended with treated pig slurries and composts in a Mediterranean climate, Agr. Ecosyst. Environ., 121, 383–394, https://doi.org/10.1016/j.agee.2006.11.020, 2007.
Melander, B., Rasmussen, I. A., and Olesen, J. E.: Incompatibility between fertility building methods and the management of perennial weeds in organic cropping systems, Agr. Ecosyst. Environ., 220, 184–192, 2016.
Molodovskaya, M., Singurindy, O., Richards, B. K., Warland, J., Johnson, M. S., and Steenhuis, T. S.: Temporal Variability of Nitrous Oxide from Fertilized Croplands: Hot Moment Analysis, Soil Sci. Soc. Am. J., 76, 1728, https://doi.org/10.2136/sssaj2012.0039, 2012.
Moyo, H., Davies, W. P., Cannon, N. D., and Conway, J. S.: Influences of two year red clover-grass ley management on nitrogen economy and following wheat performance, Biol. Agric. Hortic., 32, 98–109, https://doi.org/10.1080/01448765.2015.1057865, 2016.
Möller, K. and Stinner, W.: Effects of different manuring systems with and without biogas digestion on soil mineral nitrogen content and on gaseous nitrogen losses (ammonia, nitrous oxides), Eur. J. Agron., 30, 1–16, https://doi.org/10.1016/j.eja.2008.06.003, 2009.
Nadeem, S., Hansen, S., Azzaroli Bleken, M., and Dörsch, P.: N2O emission from organic barley cultivation as affected by green manure management, Biogeosciences, 9, 2747–2759, https://doi.org/10.5194/bg-9-2747-2012, 2012.
Nebert, L. D., Bloem, J., Lubbers, I. M., and van Groenigen, J. W.: Association of earthworm-denitrifier interactions with increased emission of nitrous oxide from soil mesocosms amended with crop residue, Appl. Environ. Microb., 77, 4097–4104, 2011.
Neumann, A., Torstensson, G., and Aronsson, H.: Losses of nitrogen and phosphorus via the drainage system from organic crop rotations with and without livestock on a clay soil in southwest Sweden, Org. Agric., 1, 217–229, https://doi.org/10.1007/s13165-011-0017-0, 2011.
Olesen, J. E., Askegaard, M., and Rasmussen, I. A.: Design of an organic farming crop rotation experiment, Acta Agr. Scand. B-S. P., 50, 13–21, 2000.
Pandey, A., Li, F., Askegaard, M., and Olesen, J. E.: Biological nitrogen fixation in organic and conventional arable crop rotations in Denmark, Eur. J. Agron, 90, 87–95, 2017.
Pandey, A., Li, F., Askegaard, M., Rasmussen, I. A., and Olesen, J. E.: Nitrogen balances in organic and conventional arable crop rotations and their relations to nitrogen yield and leaching losses, Agr. Ecosyst. Environ., 265, 350–362, 2018.
Pappa, V. A., Rees, R. M., Walker, W. A., and Baddely, J. A.: Intercropping can reduce environmental impacts, Agric. Environ. VII L. Manag. a Chang. Environ. Proc. SAC SEPA Bienn. Conf., 26–27 March 2008, Edinburgh, UK, 287–290, 2008.
Pappa, V. A., Rees, R. M., Walker, R. L., Baddeley, J. A., and Watson, C. A.: Nitrous oxide emissions and nitrate leaching in an arable rotation resulting from the presence of an intercrop, Agr. Ecosyst. Environ., 141, 153–161, https://doi.org/10.1016/j.agee.2011.02.025, 2011.
Pelster, D. E., Chantigny, M. H., Rochette, P., Angers, D. A., Rieux, C., and Vanasse, A.: Nitrous Oxide Emissions Respond Differently to Mineral and Organic Nitrogen Sources in Contrasting Soil Type, J. Environ. Qual., 41, 427–435, https://doi.org/10.2134/jeq2011.0261, 2012.
Petersen, S. O., Schjonning, P., Thomsen, I. K., and Christensen, B. T.: Nitrous oxide evolution from structurally intact soil as influenced by tillage and soil water content, Soil Biol. Biochem., 40, 967–977, https://doi.org/10.1016/j.soilbio.2007.11.017, 2008.
Petersen, S. O., Mutegi, J. K., Hansen, E. M., and Munkholm, L. J.: Tillage effects on N2O emissions as influenced by a winter cover crop, Soil Biol. Biochem., 43, 1509–1517, https://doi.org/10.1016/j.soilbio.2011.03.028, 2011.
Petersen, S. O., Schjonning, P., Olesen, J. E., Christensen, S., and Christensen, B. T.: Sources of Nitrogen for Winter Wheat in Organic Cropping Systems, Soil Sci. Soc. Am. J., 77, 155–165, 2013.
Peyrard, C., Mary, B., Perrin, P., Vericel, G., Grehan, E., Justes, E., and Leonard, J.: N2O emissions of low input cropping systems as affected by legume and cover crops use, Agr. Ecosyst. Environ., 224, 145–156, https://doi.org/10.1016/j.agee.2016.03.028, 2016.
Pfiffner L. and Mäder, P.: Effects of biodynamic, organic and conventional production systems on earthworm populations, Biol. Agric. Hortic., 15, 1–4, https://doi.org/10.1080/01448765.1997.9755177, 1997.
Pfiffner, L. and Luka, H.: Earthworm Populations in Two Low-Input Cereal Farming Systems, Appl. Soil Ecol., 37, 184–91, https://doi.org/10.1016/j.apsoil.2007.06.005, 2007.
Pimentel, D., Hepperly, P., Hanson, J., Douds, D., and Seidel, R: Environmental, energetic, and economic comparisons of organic and conventional farming systems, Bioscience, 55, 573–582, 2005.
Plaza-Bonilla, D., Nolot, J. M., Raffaillac, D., and Justes, E.: Cover crops mitigate nitrate leaching in cropping systems including grain legumes: field evidence and model simulations, Agr. Ecosyst. Environ., 212, 1–12, 2015.
Postma-Blaauw, M. B., Bloem, J., Faber, J. H., Van Groenigen, J. W., De Goede, R. G., and Brussaard, L.: Earthworm species composition affects the soil bacterial community and net nitrogen mineralization, Pedobiologia, 50, 243–256, 2006.
Poudel, D. D., Horwath, W. R., Lanini, W. T., Temple, S. R., and Van Bruggen, A. H. C.: Comparison of soil N availability and leaching potential, crop yields and weeds in organic, low-input and conventional farming systems in northern California, Agr. Ecosyst. Environ., 90, 125–137, 2002.
Prieto, R. A.: The role of earthworms in nitrous oxide emissions from forage agroecosystems, in: Masters Abstracts International, (Vol. 50, No. 02), 2011.
Pugesgaard, S., Petersen, S. O., Chirinda, N., and Olesen, J. E.: Crop residues as driver for N2O emissions from a sandy loam soil, Agr. Forest. Meteorol., 233, 45–54, https://doi.org/10.1016/j.agrformet.2016.11.007, 2017.
Rasse, D., Ritchie, J., Peterson, W., Wei, J., and Smucker, A.: Rye cover crop and nitrogen fertilization effects on nitrate leaching in inbred maize fields, J. Environ. Qual., 29, 298–304, https://doi.org/10.2134/jeq2000.00472425002900010037x, 2000.
Ravishankara, A. R., Daniel, J. S., and Portmann, R. W.: Nitrous Oxide (N2O): The Dominant Ozone-Depleting Substance Emitted in the 21st Century, Science 326, 123–125, https://doi.org/10.1126/science.1176985, 2009.
Riley, H., Pommeresche, R., Eltun, R., Hansen, S., and Korsaeth, A.: Soil structure, organic matter and earthworm activity in a comparison of cropping systems with contrasting tillage, rotations, fertilizer levels and manure use, Agr. Ecosyst. Environ., 124, 275–284, 2008.
Robertson, G. P. and Vitousek, P. M.: Nitrogen in Agriculture: Balancing the Cost of an Essential Resource, Annu. Rev. Env. Resour., 34, 97–125, https://doi.org/10.1146/annurev.environ.032108.105046, 2009.
Rochette, P. and Janzen, H. H.: Towards a revised coefficient for estimating N2O emissions from legumes, Nutr. Cycl. Agroecosys., 73, 171–179, 2005.
Röös, E., Mie, A., Wivstad, M., Salomon, E., Johansson, B., Gunnarsson, S., Wallenbeck, A., Hoffmann, R., Nilsson, U., Sundberg, S., and Watson, C. A.: Risks and opportunities of increasing yields in organic farming, A review, Agron. Sustain. Dev., 38, 14, https://doi.org/10.1007/s13593-018-0489-3, 2018.
Sahrawat, K. L. and Keeney, D. R.: Nitrous Oxide Emissions from Soils, Adv. Soil S., 4, 103–147, 1986.
Shcherbak, I., Millar, N., and Robertson, G. P.: Global metaanalysis of the nonlinear response of soil nitrous oxide (N2O) emissions to fertilizer nitrogen, P. Natl. Acad. Sci. USA, 111, 9199–9204, https://doi.org/10.1073/pnas.1322434111, 2014.
Shelton, R. E., Jacobsen, K. L., and McCulley, R. L.: Cover Crops and Fertilization Alter Nitrogen Loss in Organic and Conventional Conservation Agriculture Systems, Front. Plant Sci., 8, 1–14, https://doi.org/10.3389/fpls.2017.02260, 2018.
Skinner, C., Gattinger, A., Muller, A., Mäder, P., Flieβbach, A., Stolze, M., Ruser, R., and Niggli, U.: Greenhouse gas fluxes from agricultural soils under organic and non-organic management – A global meta-analysis, Sci. Total Environ., 468–469, 553–563, https://doi.org/10.1016/j.scitotenv.2013.08.098, 2014.
Skinner, C., Gattinger, A., Krauss, M., Krause, H.-M., Mayer, J., van der Heijden, M. G. A., and Mäder, P.: The impact of long-term organic farming on soil-derived greenhouse gas emissions, Sci. Rep., 9, 1702. https://doi.org/10.1038/s41598-018-38207-w, 2019.
Smith, K. A., Dobbie, K. E., Thorman, R., Watson, C. J., Chadwick, D. R., Yamulki, S., and Ball, B. C.: The effect of N fertilizer forms on nitrous oxide emissions from UK arable land and grassland, Nutr. Cycl. Agroecosys., 93, 127–149, 2012.
Spargo, J. T., Cavigelli, M. A., Mirsky, S. B., Maul, J. E., and Meisinger, J. J.: Mineralizable soil nitrogen and labile soil organic matter in diverse long-term cropping systems, Nutr. Cycl. Agroecosys., 90, 253–266, 2011.
Stalenga, J. and Jończyk, K.: Nutrient management and soil organic matter balance in the organic crop production system evaluated by NDICEA model, J. Res. Appl. Agric. Eng., 53, 78–84, 2008.
Stehfest, E. and Bouwman, L.: N2O and NO emission from agricultural fields and soils under natural vegetation: summarizing available measurement data and modeling of global annual emissions, Nutr. Cycl. Agroecosys., 74, 207–228, 2006.
Stenberg, M., Aronsson, H., Linden, B., Rydberg, T., and Gustafson, A.: Soil mineral nitrogen and nitrate leaching losses in soil tillage systems combined with a catch crop, Soil Till. Res., 50, 115–125, https://doi.org/10.1016/S0167-1987(98)00197-4, 1999.
Stenberg, M., Ulén, B., Söderström, M., Roland, B., Delin, K., and Helander, C. A.: Tile drain losses of nitrogen and phosphorus from fields under integrated and organic crop rotations, A four-year study on a clay soil in southwest Sweden, Sci. Total Environ., 434, 79–89, https://doi.org/10.1016/j.scitotenv.2011.12.039, 2012.
Stockdale, E. A., Lampkin, N. H., Hovi, M., Keatinge, R., Lennartsson, E. K. M., MacDonald, D. W., Padel, S., Tattersall, F. H., Wolfe, M. S., and Watson, C. A.: Agronomic and Environmental Implications of Organic Farming Systems, Adv. Agron., 70, 261–327, 2001.
Stopes, C., Lord, E. I., Philipps, L., and Woodward, L.: Nitrate leaching from organic farms and conventional farms following best practice, Soil Use Manage., 18, 256–263, 2002.
Sturite, I., Rivedal, S., and Dörsch, P.: Nitrous oxide emissions from clover rich leys during the long Northern winter, edited by: Cordovil, C. M. D. S., in: Proceedings of the 18th Nitrogen Workshop – The nitrogen challenge: building a blueprint for nitrogen use efficiency and food security, 30 June–3 July 2014, Lisbon, Portugal, 517–518, 2014.
Thangarajan, R., Bolan, N. S., Tian, G. L., Naidu, R., and Kunhikrishnan, A.: Role of organic amendment application on greenhouse gas emission from soil, Sci. Total Environ., 465, 72–96, https://doi.org/10.1016/j.scitotenv.2013.01.031, 2013.
Thorup-Kristensen, K.: Effect of deep and shallow root systems on the dynamics of soil inorganic N during 3-year crop rotations, Plant Soil, 288, 233–248, 2006.
Thorup-Kristensen, K. and Dresbøll, D. B.: Incorporation time of nitrogen catch crops influences the N effect for the succeeding crop, Soil Use Manage., 26, 27–35, 2010.
Tonitto, C., David, M. B., and Drinkwater, L.: Replacing bare fallows with cover crops in fertilizer-intensive cropping systems: A meta-analysis of crop yield and N dynamics, Agr. Ecosyst. Environ., 112, 58–72, https://doi.org/10.1007/s10705-010-9354-8, 2006.
Torstensson, G. and Aronsson, H.: Nitrogen leaching and crop availability in manured catch crop systems in Sweden, Nutr. Cycl. Agroecosys., 56, 139–152, https://doi.org/10.1023/A:1009821519042, 2000.
Torstensson, G., Aronsson, H., and Bergstrom, L.: Nutrient use efficiencies and leaching of organic and conventional cropping systems in Sweden, Agron. J., 98, 603–615, 2006.
Tosti, G., Benincasa, P., Farneselli, M., Tei, F., and Guiducci, M.: Barley–hairy vetch mixture as cover crop for green manuring and the mitigation of N leaching risk, Eur. J. Agron., 54, 34–39, https://doi.org/10.1016/J.EJA.2013.11.012, 2014.
Tosti, G., Farneselli, M., Benincasa, P., and Guiducci, M.: Nitrogen fertilization strategies for organic wheat production: Crop yield and nitrate leaching, Agron. J., 108, 770–781, https://doi.org/10.2134/agronj2015.0464, 2016.
Tuomisto, H. L., Hodge, I. D., Riordan, P., and Macdonald, D. W.: Does organic farming reduce environmental impacts? – A meta-analysis of European research, J. Environ. Manage., 112, 309–320, https://doi.org/10.1016/j.jenvman.2012.08.018, 2012.
Tyson, K. C., Scholefield, D., Jarvis, S. C., and Stone, A. C.: A comparison of animal output and nitrogen leaching losses recorded from drained fertilized grass and grass/clover pasture, J. Agric. Sci., 129, 315–323, 1997.
Ussiri, D. and Lal, R.: Global Sources of Nitrous Oxide, in Soil Emission of Nitrous Oxide and its Mitigation, Springer Netherlands, Dordrecht, the Netherlands, 131–175, 2013.
Valkama, E., Lemola, R., Känkänen, H., and Turtola, E.: Meta-analysis of the effects of undersown catch crops on nitrogen leaching loss and grain yields in the Nordic countries, Agr. Ecosyst. Environ., 203, 93–101, https://doi.org/10.1016/j.agee.2015.01.023, 2015.
Van Groenigen, J. W., Velthof, G. L., Oenema, O., Van Groenigen, K. J., and Van Kessel, C.: Towards an agronomic assessment of N2O emissions: A case study for arable crops, Eur. J. Soil Sci., 61, 903–913, https://doi.org/10.1111/j.1365-2389.2009.01217.x, 2010.
Wagner-Riddle, C., Congreves, K. A., Abalos, D., Berg, A. A., Brown, S. E., Ambadan, J. T., Gao, X., and Tenuta, M.: Globally important nitrous oxide emissions from croplands induced by freeze-thaw cycles, Nat. Geosci., 10, 279–283, https://doi.org/10.1038/ngeo2907, 2017.
Watson, C. A., Fowler, S. M., and Wilman, D.: Soil inorganic-N and nitrate leaching on organic farms, J. Agric. Sci., 120, 361–369, 1993.
Watson, C. A., Atkinson, D., Gosling, P., Jackson, L. R., and Rayns, F. W.: Managing soil fertility in organic farming systems, Soil Use Manage., 18, 239–247, 2002.
Weier, K. L., Doran, W. L., Power, J. F., and Walters, D. T: Denitrification and the dinitrogen/nitrous oxide ratio as affected by soil water, available carbon, and nitrate, Soil Sci. Soc. Am. J., 57, 66–72, 1992.
Westphal, M., Tenuta, M., and Entz, M. H.: Nitrous oxide emissions with organic crop production depends on fall soil moisture, Agr. Ecosyst. Environ., 254, 41–49, https://doi.org/10.1016/j.agee.2017.11.005, 2018.
Willer, H. and Lernoud, J.: The World of Organic Agriculture. Statistics and Emerging Trends 2018, Research Institute of Organic Agriculture (FiBL), Frick, and IFOAM – Organics International, Bonn, Germany, 2018.
Willer, H., Schaack, D., and Lernoud, J.: Organic farming and market development in Europe and the European Union, edited by: Willer, H. and Lernoud, J., in: The World of Organic Agriculture, Statistics and Emerging Trends 2018, Research Institute of Organic Agriculture (FiBL), Frick, and IFOAM – Organics International, Bonn, Germany, 218–255, 2018.
Xia, L., Lam, S. K., Wolf, B., Kiese, R., Chen, D., and Butterbach-Bahl, K.: Trade-offs between soil carbon sequestration and reactive nitrogen losses under straw return in global agroecosystems, Global Change Biol., 24, 5919–5932, https://doi.org/10.1111/gcb.14466, 2018.
- Abstract
- Introduction
- Methodology
- Drivers of SMN and degradable C in organic arable crop rotations
- Drivers of N2O emissions in organic arable crop rotations
- Drivers of NO3 leaching in organic arable crop rotations
- Key drivers of N2O emissions and NO3 leaching and suggested mitigation strategies
- Research and innovation needs
- Conclusions
- Data availability
- Appendix A
- Author contributions
- Competing interests
- Acknowledgements
- Financial support
- Review statement
- References
- Supplement
- Abstract
- Introduction
- Methodology
- Drivers of SMN and degradable C in organic arable crop rotations
- Drivers of N2O emissions in organic arable crop rotations
- Drivers of NO3 leaching in organic arable crop rotations
- Key drivers of N2O emissions and NO3 leaching and suggested mitigation strategies
- Research and innovation needs
- Conclusions
- Data availability
- Appendix A
- Author contributions
- Competing interests
- Acknowledgements
- Financial support
- Review statement
- References
- Supplement