the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Methane production by three widespread marine phytoplankton species: release rates, precursor compounds, and potential relevance for the environment
Thomas Klintzsch
Gerald Langer
Gernot Nehrke
Anna Wieland
Katharina Lenhart
Frank Keppler
Methane (CH4) production within the oceanic mixed layer is a widespread phenomenon, but the underlying mechanisms are still under debate. Marine algae might contribute to the observed CH4 oversaturation in oxic waters, but so far direct evidence for CH4 production by marine algae has only been provided for the coccolithophore Emiliania huxleyi.
In the present study we investigated, next to E. huxleyi, other widespread haptophytes, i.e., Phaeocystis globosa and Chrysochromulina sp. We performed CH4 production and stable carbon isotope measurements and provide unambiguous evidence that all three investigated marine algae are involved in the production of CH4 under oxic conditions. Rates ranged from 1.9±0.6 to 3.1±0.4 µg of CH4 per gram of POC (particulate organic carbon) per day, with Chrysochromulina sp. and E. huxleyi showing the lowest and highest rates, respectively. Cellular CH4 production rates ranged from 16.8±6.5 (P. globosa) to 62.3±6.4 ag CH4 cell−1 d−1 (E. huxleyi; ag = 10−18 g). In cultures that were treated with 13C-labeled hydrogen carbonate, δ13CH4 values increased with incubation time, resulting from the conversion of 13C–hydrogen carbonate to 13CH4. The addition of 13C-labeled dimethyl sulfide, dimethyl sulfoxide, and methionine sulfoxide – known algal metabolites that are ubiquitous in marine surface layers – resulted in the occurrence of 13C-enriched CH4 in cultures of E. huxleyi, clearly indicating that methylated sulfur compounds are also precursors of CH4. By comparing the algal CH4 production rates from our laboratory experiments with results previously reported in two field studies of the Pacific Ocean and the Baltic Sea, we might conclude that algae-mediated CH4 release is contributing to CH4 oversaturation in oxic waters. Therefore, we propose that haptophyte mediated CH4 production could be a common and important process in marine surface waters.
- Article
(1836 KB) - Full-text XML
-
Supplement
(312 KB) - BibTeX
- EndNote
Methane (CH4), the second most important anthropogenic greenhouse gas after CO2, is the most abundant reduced organic compound in the atmosphere and plays a central role in atmospheric chemistry (Denman et al., 2007; Kirschke et al., 2013; Lelieveld et al., 1998). The mixing ratio of CH4 in the atmosphere has been increasing dramatically from preindustrial values of about 715 parts per billion by volume (ppbv) to about 1868 ppbv (October 2018, NOAA). The global atmospheric CH4 budget is determined by the total emission (540–568 Tg CH4 yr−1) of various sources from terrestrial and aquatic surface areas that are balanced primarily by one major sink (hydroxyl radicals) in the atmosphere. The world's oceans are considered to be a minor source of CH4 to the atmosphere (1 %–3 %, Saunois et al., 2016). However, in recent years the widespread occurrence of in situ CH4 production in the ocean mixed layer has received much attention, since CH4 formation in the oxygenated ocean mixed layer challenges the paradigm that biological methanogenesis is a strictly anaerobic process.
Methane is primarily formed by the degradation of buried organic matter under heat and pressure (thermogenic) inside the Earth's crust or produced by the incomplete combustion of biomass (pyrogenic). However, CH4 resulting from microbial processes, carried out by methanogenic archaea under anoxic conditions in soils and sediments or the digestion system of ruminants, is categorized as biogenic or microbial (Kirschke et al., 2013). In contrast to these well-known sources, recent studies have confirmed direct CH4 release from eukaryotes, including plants, animals, fungi, lichens, and the marine alga Emiliania huxleyi even in the absence of methanogenic archaea and in the presence of oxygen or other oxidants (Keppler et al., 2006; Ghyczy et al., 2008; Lenhart et al., 2012, 2015b, 2016). A very recent study also confirmed Cyanobacteria as CH4 producers, suggesting that CH4 production occurs in all three domains of life (Bizic-Ionescu et al., 2018a). These novel sources, from the domains Eucarya and Bacteria, might be classified as biotic non-archaeal CH4 (Boros and Keppler, 2018).
In situ CH4 production in oxygenated surface waters in the marine environment was first reported by Scranton and Farrington (1977) and Scranton and Brewer (1977) and some decades later also for lakes (Grossart et al., 2011). Significant quantities of CH4, produced in upper oxic waters near the air–water interface, might overcome oxidation and thus significantly contribute to CH4 fluxes from aquatic environments to the atmosphere (Bogard et al., 2014). It turned out that in situ CH4 production in the upper oxic waters is a common feature of both oceans and lakes (Forster et al., 2009; Reeburgh, 2007; Tang et al., 2014; Donis et al., 2017; Bižić-Ionescu et al., 2018b; Bange et al., 1994). These results have stimulated the scientific community to study in more detail the phenomenon of CH4 occurrence in oxygenated surface waters. In this context, emissions from Cyanobacteria or algae might help to explain the phenomenon of dissolved CH4 oversaturation. In addition, it has been suggested that CH4 might be produced under phosphorus limitation by the bacterial cleavage of methylphosphonate (MPn) in oligotrophic Pacific regions (Karl et al., 2008; Metcalf et al., 2012; Repeta et al., 2016; Valle and Karl, 2014).
In contrast to this apparently non-oxygen-sensitive pathway, many other studies have identified the “traditionally” archaeal methanogenesis in anoxic microenvironments as a CH4 source. Floating particles (Karl and Tilbrook, 1994), as well as the digestive tracts of zooplankton (de Angelis and Lee, 1994; Stawiarski et al., 2019; Schmale et al., 2018) or fishes (Oremland, 1979), have been found as anoxic micro-niches for methanogens. It has been suggested that some methanogens might be active under oxic conditions by being equipped with enzymes to counteract the effects of molecular oxygen during methanogenesis (Angel et al., 2011). Potential substrates for methylotrophic methanogens in such micro-niches are the algae metabolites dimethylsulfoniopropionate (DMSP) and their degradation products dimethyl sulfide (DMS) or dimethyl sulfoxide (DMSO) (Zindler et al., 2013; Damm et al., 2008; Florez-Leiva et al., 2013). Furthermore, DMSP might also be converted to CH4 by nitrogen-limited bacteria (Damm et al., 2010, 2015). However, in coastal waters where DMS and DMSP production is enhanced, CH4 was found to be mainly related to sedimentary sources (Borges et al., 2018).
In contrast to microbial processes, which are considered to be driven by enzymes, CH4 might also be derived by the chemical reaction of chromophoric dissolved organic matter (CDOM) and DMS induced by UV or visible light under both oxic and anoxic conditions (Zhang et al., 2015). A similar photochemical CH4 formation was described earlier for acetone by Bange and Uher (2005), but the production of CH4 from acetone was considered negligible under oxic conditions.
Under highly oxidative conditions nonheme iron-oxo (IV) species catalyze CH4 formation from methyl thioethers and their sulfoxides (Althoff et al., 2014; Benzing et al., 2017). Iron-oxo species are intermediates in a number of biological enzymatic systems (Hohenberger et al., 2012). Thus, marine algae containing elevated concentrations of methyl thiolethers and their sulfoxides such as DMSP, DMSO, methionine (MET), or methionine sulfoxide (MSO) might be biochemical reactors for non-archaeal CH4 production as was already proposed by Lenhart et al. (2016) and Keppler et al. (2009). Marine phytoplankton plays a central role in the global carbon cycle: approximately half of Earth's primary production is carried out by marine phytoplankton (Field et al., 1998). In this context it is important to mention that almost 40 years ago researchers (Scranton and Brewer, 1977; Scranton and Farrington, 1977; Scranton, 1977) already mentioned the possibility of the in situ formation of CH4 by marine algae, since CH4 production was examined in cultures of E. huxleyi and Thalassiosira pseudonana. Furthermore, direct isotopic evidence for CH4 production by marine algae in the absence of methanogenic archaea has only been provided for E. huxleyi (Lenhart et al., 2016). Based on the application of stable carbon isotope techniques, it could be clearly shown that both hydrogen carbonate and a position-specific 13C-labeled MET were carbon precursors of the observed CH4 production. However, it remains unclear whether CH4 production also occurs among other marine algae and if there are also other carbon precursors involved in the formation process.
In the present study we investigated, next to the coccolithophore E. huxleyi, two other marine, non-calcifying haptophytes, namely Phaeocystis globosa and Chrysochromulina sp., for CH4 formation. The investigated species are all bloom-forming and often found as dominant members in marine phytoplankton communities worldwide (Schoemann et al., 2005; Thomsen, 1994; Brown and Yoder, 1994). Furthermore, they are well-known for their high DMSP, DMS, and DMSO productivity (Liss et al., 1994; Keller, 1989; Holligan et al., 1993; Stefels et al., 2007; Matrai and Keller, 1993). We therefore conducted stable isotope experiments using 13C-labeled DMS, MSO, and DMSO to identify potential methyl group precursor compounds that eventually lead to CH4 production. Finally, we discuss the laboratory CH4 production rates in relation to their potential significance in marine environments.
2.1 Cultures and culture conditions
Three algal species, E. huxleyi RCC1216 obtained from the Roscoff Culture Collection (http://roscoff-culture-collection.org/, last access: 21 October 2019), P. globosa PLY 575, and Chrysochromulina sp. PLY 307 obtained from the Marine Biological Association of the United Kingdom (https://www.mba.ac.uk/facilities/culture-collection, last access: 21 October 2019), were studied. In order to keep non-axenic algae cultures largely free of bacteria, the cultures were diluted regularly, resulting in quasi-constant exponential algal growth while minimizing bacterial cell density. All incubation experiments were carried out in controlled and sterile laboratory conditions under a 16 ∕ 8 h light–dark cycle at a light intensity of 350 µmol photons m−2 s−1 and a temperature of 20 ∘C. All samples were taken at the end of the light cycle. Monoclonal cultures were grown in full-batch mode (Langer et al., 2013) in sterile filtered (0.2 µm ∅ pore size) natural North Sea seawater (sampled off Helgoland, Germany) enriched in nutrients according to F/2 medium (Guillard and Ryther, 1962). The initial dissolved inorganic carbon (DIC) of the F/2 medium was 2152±6 µmol L−1 (measured by Shimadzu TOC-V CPH). The DIC value falls within the range of typical DIC concentrations of North Sea seawater.
2.2 Determination of cell densities
Cell densities were determined from four aliquots of each culture sample using either a Fuschs–Rosenthal or Neubauer counting chamber, depending on cell density.
2.3 Incubation with 13C-labeled hydrogen carbonate
To investigate CH4 production by algal cultures, borosilicate glass bottles (Schott, Germany) filled with 2.0 L of 0.2 µm filtered F/2 medium and with 0.35 L headspace volume were used in our investigations of Chrysochromulina sp. and P. globosa. For the investigations of E. huxleyi 0.85 L medium and 0.4 L headspace volume were used (Schott, Germany). The vials were sealed airtight with lids (GL 45, PP, two-port, Duran Group) equipped with one three-way port for liquid and a second port fitted with a septum for gas sampling. For measurements of the mixing ratio and stable carbon isotope value of CH4 (δ13C−CH4) samples of headspace (20 mL) were taken from each vial. Afterwards, samples (2 mL) for determining cell densities were taken. In order to maintain atmospheric pressure within the vial, the surrounding air was allowed to enter via the three-way port and through a sterile filter to avoid biological contamination. The inflow of surrounding air was taken into consideration when CH4 production was calculated. Cultures that were studied during the incubation were inoculated from a pre-culture grown in dilute-batch mode (Langer et al., 2009). To investigate algal-derived CH4 formation six vials were inoculated with algae and another six vials contained medium only. In addition, three vials of each group were treated with 13C–hydrogen carbonate () to investigate CH4 formation by measuring stable carbon isotope values of CH4. Four different treatments were used: medium either with (medium + ) or without (medium, data not available) and cultures supplemented either with (medium + culture + ) or without (medium + culture). The different treatments and the number of replicates for the experiments with Chrysochromulina sp. and P. globosa are provided in Fig. 1. Please note that stable isotope measurements using were not performed for E. huxleyi as evidence for the isotope labeling of CH4 was already provided by Lenhart et al. (2016). To study the CH4 formation of E. huxleyi by measuring headspace concentration, three replicates (culture and medium group, n=3) were used.
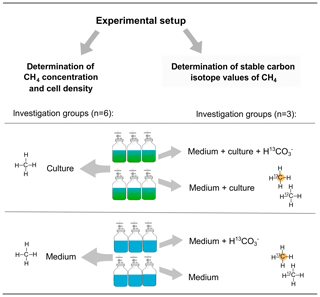
Figure 1Experimental setup for measuring CH4 formation by Chrysochromulina sp. and P. globosa. Methane formation was investigated by concentration measurements within six vials containing either algae or medium only (left column). For stable isotope measurements of CH4, 13C-labeled hydrogen carbonate () was added to three vials of both groups (right column).
The overall incubation time was 9, 11, and 6 d for Chrysochromulina sp., P. globosa, and E. huxleyi, respectively. Headspace and liquid samples were collected on a daily basis for E. huxleyi and in 2–3 d intervals from cultures of Chrysochromulina sp. and P. globosa. The incubation time and sampling intervals varied between species because of variations in the growth rate and the cell density in the stationary phase. Cell densities were plotted versus time, and the exponential growth rate (μ) was calculated from exponential regression using the natural logarithm (Langer et al., 2013). The exponential growth phase (from which μ was calculated) was defined by the cell densities which corresponded to the best fit (r2>0.99) of the exponential regression. This was done by using the first three (Chrysochromulina sp. and E. huxleyi) or four data points (P. globosa) of the growth curve. For stable carbon isotope experiments 48.7 µmol L−1 NaH13CO3 in final concentration was added to the F/2 medium. The added amount of NaH13CO3 corresponds to 2 % of the DIC of the North Sea seawater (2152±6 µmol L−1), resulting in a theoretically calculated δ13C value of DIC of ‰ . To determine the δ13C−CH4 values of the source, the Keeling plot method was applied (Keeling, 1958). For a detailed discussion of the Keeling plot method for determination of the isotope ratio of CH4 in environmental applications, please refer to Keppler et al. (2016). Oxygen concentration was monitored daily (using inline oxygen sensor probes; PreSens, Regensburg) at the end of the light cycle (Fig. S1 in the Supplement).
2.4 Determination of CH4 production rates
Since the experiment in the Sect. 2.3 was not designed to obtain POC quotas (POC: particulate organic carbon), we conducted an additional experiment. To best compare the CH4 formation rates of the three algae species it is necessary to obtain exponential growth to ensure constant growth rates and constant (at a given time of day) cellular POC quotas over the course of the experiment. Exponential growth is a prerequisite for calculating production on the basis of growth rate and quota (here CH4 quota). The point is a general, technical one and is not confined to CH4 production. The studies by Langer et al. (2012, 2013) discuss this point in the context of batch culture experiments. Briefly, production on this account is the product of growth rate and quota (e.g., CH4, calcite, organic carbon). Production here is an integrated value, typically over many cell divisions. For this calculation of production to be meaningful a constant growth rate is required. The exponential growth phase fulfills this criterion, whereas the transition phase and the stationary phase do not. Therefore, production cannot be calculated meaningfully in the non-exponential phases. The problem can, however, be minimized by using small increments (1 d) because growth rate can be regarded as quasi-constant (see also Lenhart et al., 2016). The CH4 production rates can be calculated by multiplying the growth rate μ with the corresponding cellular or POC–CH4 quota that was measured at the end of the experiment. For this additional experiment the cultures were grown in 160 mL crimped serum bottles filled with 140 mL medium and 20 mL headspace (n=4). Oxygen concentration was monitored (using inline oxygen sensor probes; PreSens, Regensburg) at the end of each light and dark cycle (Fig. S2). The growth rate (μ) was calculated from cell densities of the beginning and end of the experiment according to Eq. (1):
where N0 and N1 are the cell densities at the beginning (t0) and end of the experiment (t1). The daily cellular CH4 production rates (CH4Pcell, ag CH4 cell−1 d−1, ag = 10−18 g) were calculated according to Eq. (2):
where m(CH4) is the amount of CH4 that was produced at the end of the experiment.
To calculate POC-based CH4 production rates the cellular organic carbon content (POCcell) was derived from cell volume (Vcell) by using the Eq. (3) according to Menden-Deuer and Lessard (2000):
The cell volume was determined by measuring the cell diameter in light micrographs using the program ImageJ (Schindelin et al., 2012). According to Olenina (2006) a ball shape can be assumed for calculating the cell volume for the three species investigated here. The daily cellular CH4 production rates (CH4PPOC, µg CH4 g−1 POC d−1) were calculated from growth rate and CH4–POC quotas at the end of the experiment according to Eq. (4).
The CH4 production potential (CH4–PP) was used to translate differences in cellular production rates to the community level. According to Gafar et al. (2018), the CH4–PP can be calculated for different periods of growth by calculating a cellular standing stock for each time period from a known starting cell density (N0) (whereby constant exponential growth is assumed). The corresponding amount of produced CH4 (CH4PP) for each period of growth and standing stock is the product of the cellular standing stock and CH4 quota (Eq. 5).
In the present study the CH4–PP was calculated for a standing stock that is obtained after 7 d of growth starting with a single cell.
2.5 Incubation with 13C labeled DMS, DMSO, and MSO
The sulfur-bonded methyl group(s) in DMS, DMSO, and MSO were investigated as precursors for algal-derived CH4 in an incubation experiment with E. huxleyi. For all tested compounds only the C atom of the sulfur-bonded methyl group(s) was labeled with 13C (R–S–13CH3, 99 %). A final concentration of 10 µM was used for each compound. The different treatments to investigate potential CH4 formation by 13C2–DMS, 13C2–DMSO, 13C–MSO are provided in Fig. 2. Three independent replicates and repeated measurements over time were used. Headspace and vial size were analogous to the experiment described in Sect. 2.3 for E. huxleyi. Samples were taken daily during an overall incubation time of 6 d.
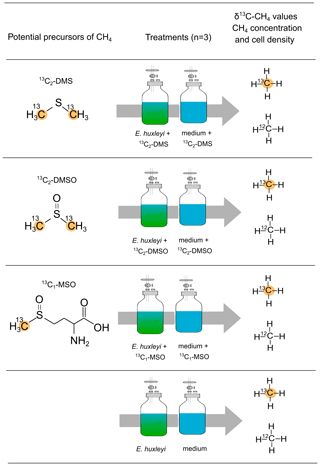
Figure 2Experimental setup to investigate potential precursor compounds of CH4. Dimethyl sulfide (13C2–DMS), dimethyl sulfoxide (13C2–DMSO), and methionine sulfoxide (13C–MSO) were added to the vials containing either a culture of E. huxleyi or medium only. For all tested compounds only the carbon atom of the sulfur-bonded methyl group(s) was labeled with 13C.
2.6 Determination of CH4 mass
A 5 mL gas sample was collected from the headspace of the vials using a gastight Hamilton gas syringe. The sample was analyzed by a gas chromatographer (GC-14B, Shimadzu, Japan; column: 2 m, ∅=3.175 mm inner diameter, high-grade steel tube packed with molecular sieve 5A 60/80 mesh from Supelco) equipped with a flame ionization detector (FID). Quantification of CH4 was carried out by comparison of the integrals of the peaks eluting at the same retention time as that of the CH4 authentic standard using two reference standards containing 9837 and 2192 ppbv. Mixing ratios were corrected for headspace pressure, which was monitored using a pressure-measuring device (GMSD 1, 3 BA, Greisinger). The CH4 mass () was determined by its mixing ratio () and the ideal gas law (Eq. 6):
where is molar mass, p is pressure, T is temperature, R is the ideal gas constant, and V is volume.
The dissolved CH4 concentration was calculated by using the equation of Wiesenburg and Guinasso (1979).
2.7 GC-C-IRMS measurements
Stable carbon isotope values of the CH4 of headspace samples were analyzed by gas chromatography–stable isotope ratio mass spectrometry (GC-C-IRMS; Deltaplus XL, Thermo Finnigan, Bremen, Germany). All δ13C–CH4 values were corrected using two CH4 working standards (Isometric Instruments, Victoria, Canada) with values of ‰ and ‰. The results were normalized by two-scale anchor calibration according to Paul et al. (2007). The average standard deviation of the analytical measurements was in the range of 0.1 ‰ to 0.3 ‰ (based on three repeated measurements of CH4 working standards). All δ13C–CH4 values are expressed in the conventional δ notation, in per mille (‰) vs. Vienna Pee Dee Belemnite (VPDB), using Eq. (7).
For a detailed description of the δ13C–CH4 measurements by GC-IRMS and technical details of the pre-concentration system, we refer the reader to previous studies by Comba et al. (2018) and Laukenmann et al. (2010).
2.8 Statistics
To test for significant differences in cell density, CH4 formation, and CH4 content between the treatments, two-way analysis of variance (ANOVA) (considering repeated measurements) and a post hoc test (Fisher least significant difference (LSD) test; alpha 5 %) were used.
3.1 Algal growth and CH4 formation
To investigate CH4 production by algal cultures, incubations with 13C-labeled hydrogen carbon were applied as described in Sect. 2.3. The growth curves during incubation of the three algal species are presented in Fig. 3 (panels a, b, c). The initial cell densities were cells mL−1 for Chrysochromulina sp., cells mL−1 for P. globosa, and cells mL−1 for E. huxleyi. The exponential growth rate µ was highest for E. huxleyi (1.71±0.04 d−1), i.e., 3 or 5 times higher than for P. globosa and Chrysochromulina sp. (with 0.33±0.08 d−1 and 0.52±0.07 d−1, respectively). The dotted lines in Fig. 1a, b, and c mark the time points of exponential growth. Maximum cell densities were lowest for Chrysochromulina sp. with cells mL−1, followed by E. huxleyi with cells mL−1, and highest for P. globosa with cells mL−1. Significant CH4 formation was observed in all three cultures over the whole incubation period of 5 to 11 d (Fig. 3d, e, f), whereas no increase in CH4 over time was observed in the control groups. For all species the increase in headspace CH4 was significant (p≤0.05) at second time point of measurement and at all following time points (p≤0.001). At the end of the incubation period the amounts of produced CH4 were 34.9±7.3, 99.3±8.2, and 45.0±3.1 ng for Chrysochromulina sp., P. globosa, and E. huxleyi, respectively. A linear correlation was found between the absolute number of cells and the amount of produced CH4 of Chrysochromulina sp., P. globosa, and E. huxleyi (Fig. 3g, h, i).
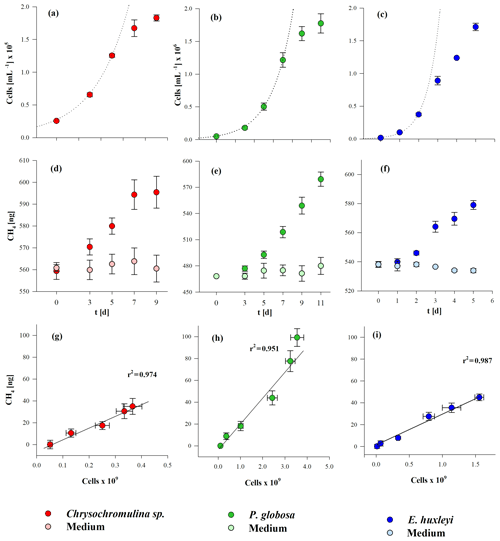
Figure 3Cell growth (a, b, c) and CH4 production (d, e, f) in the course of time and the correlation between the total number of cells and produced CH4 (g, h, i) from three algae species. Chrysochromulina sp. (a, d, g), P. globosa (b, e, h), and E. huxleyi (c, f, i). Please note that the cell numbers of Chrysochromulina sp. are presented in 105 and those of P. globosa and E. huxleyi are in 106. Mean values of six (Chrysochromulina sp., P. globosa) and three (E. huxleyi) replicated culture experiments are shown, and error bars mark the SD.
3.2 Stable carbon isotope values of CH4 during incubation with 13C–hydrogen carbonate
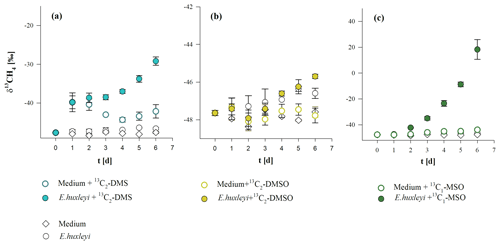
Stable carbon isotope values of CH4 (δ13CH4 values) for Chrysochromulina sp. and P. globosa are presented in Fig. 4a, c. We observed the conversion of 13C carbon (provided by 13C–hydrogen carbonate) to 13CH4 in cultures of both species, indicated by increasing δ13CH4 values over time. Stable isotope values increased from initial atmospheric (laboratory air) levels of ‰ and ‰ up to ‰ and ‰ for Chrysochromulina sp. and P. globosa, respectively, whilst the δ13CH4 values of the control groups (algae without 13C–hydrogen carbonate or 13C–hydrogen carbonate in medium without culture) did not change over time. The increase in δ13CH4 values in the headspace CH4 depended on the amount of released CH4 that was added to the initial (atmospheric) background level. To calculate the δ13CH4 values of the CH4 source that raised the CH4 quantity above background level, the Keeling plot method (Keeling, 1958; Pataki et al., 2003) was used (Fig. 4b, d). The calculated δ13CH4 values of the CH4 source were ‰ (Chrysochromulina sp.) and ‰ (P. globosa) and thus close to the theoretical calculated 13C value of the DIC (2014±331 ‰ ) resulting from the addition of 13C–hydrogen carbonate. Please note that 13C–hydrogen carbonate stable isotope labeling experiments with E. huxleyi were already performed by Lenhart et al. (2016) and were not repeated in this study. This is why δ13CH4 values and the respective Keeling plot of E. huxleyi are not shown in Fig. 4.
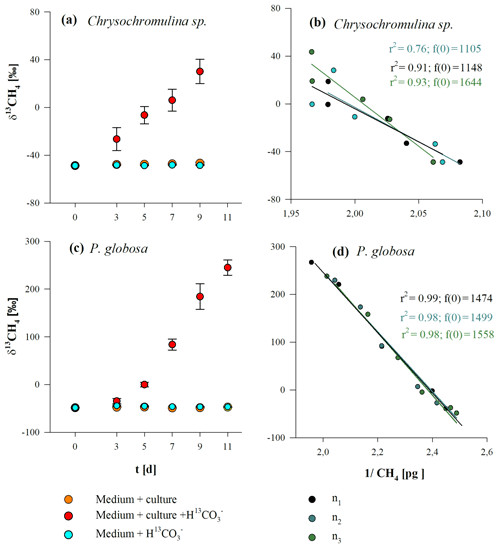
Figure 4δ13CH4 values (a, c) and respective Keeling plots (b, d) from Chrysochromulina sp. (a, b) and P. globosa (c, d) after the addition of . Panels (a, c) show the δ13CH4 values of three investigation groups (“culture + ”, “culture”, and “”), whereas each data point presented is the mean value of three replicated culture experiments with error bars showing SD. Panels (b, d) show the Keeling plots for the treatments “culture + ” from each replicated culture experiment (n1, n2, n1), where f(0) refers to the 13C value of the CH4 source.
3.3 CH4 production and production potential
Since the experiment in the section above (isotope measurements) was not designed to obtain POC quotas, we conducted an additional experiment to estimate the CH4 production rates of the three algal species. From an initial cell density of , , and cells mL−1 cultures were grown up to , , and cells mL−1 for Chrysochromulina sp., P. globosa, and E. huxleyi, respectively. These cell densities corresponded to the cell densities of the exponential growth phase obtained from the experiment in Sect. 3.1. The POC-normalized daily CH4 production rate was highest in E. huxleyi, followed by P. globosa and Chrysochromulina sp. However, the cellular or POC-normalized daily production rates of the three algal species were of the same order of magnitude (Table 1). We calculated the CH4 production potential (CH4PP), which is the amount of CH4 produced within a week of growth (Gafar et al., 2018), to translate the cellular production rates (µ × CH4 cell−1) of each species to the community level. The CH4–PP was 2 orders of magnitude higher for E. huxleyi than the other two species. This is a consequence of the higher growth rate of E. huxleyi. We furthermore observed the oxygen concentrations during the light and dark periods to ensure oxic conditions. The measured oxygen concentrations were always saturated or supersaturated relative to equilibration with ambient air (Fig. S2).
3.4 CH4 formation from 13C-labeled methyl thiol ethers
The three methylated sulfur compounds MSO, DMSO, and DMS were tested for potential CH4 formation in incubation experiments with E. huxleyi. The treatments were initiated in parallel from a batch culture by inoculating cells mL−1, and cultures were grown to final cell densities of cells mL−1 (Fig. 5a). Cell densities and CH4 formation correlated in all treatments, while no difference in cell growth pattern or CH4 formation was observed when isotope-labeled methyl thioether and sulfoxides were added to the culture (Fig. 5a, b, c). Differences between treatments were found in δ13CH4 values of headspace CH4. The initial δ13CH4 value of headspace ( ‰, laboratory air) increased slightly over time in untreated cultures (without isotope treatment) to ‰ (Fig. 6b). In contrast, experiments in which 13C2–DMS, 13C2–DMSO, and 13C–MSO were applied to cultures of E. huxleyi, δ13CH4 values increased to ‰, ‰, and ‰, respectively, over a time period of 6 d (Fig. 6a, b, c) and differed significantly from control groups (p<0.05). The results unambiguously show that a fraction of the 13C-labeled methyl groups of the added substances was converted to 13C–CH4 in cultures of E. huxleyi. Much smaller changes in δ13CH4 values were observed for controls of sterile filtered media to which only 13C2–DMS and 13C–MSO were added ( ‰ and ‰, respectively; Fig. 6a, c; day 6), whereas δ13CH4 values did not change over time in the seawater controls (no addition of isotopic-labeled compounds) or in the seawater controls treated with 13C2–DMSO (Fig. 6b). Based on the initial amount of 13C-labeled substance that was added to the cultures and the total amount of 13CH4 at the end of the incubation period, 9.5±0.2 pmol (13C2–DMS), 3.0±3.2 pmol (13C2–DMSO), and 30.1±3.6 pmol (13C–MSO) of 8.5 µmol were converted to CH4.
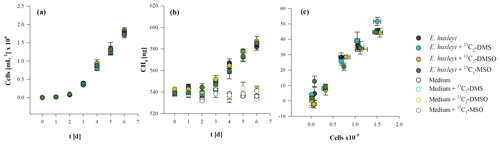
Figure 5Cell growth (a), CH4 production (b), and the relation between the total number of cells and produced CH4 (c) from E. huxleyi treated with 13C2–DMS, 13C2–DMSO, and 13C–MSO or without any treatment. Mean values of three replicated culture experiments are shown, and error bars mark the SD.
Our results of CH4 production and stable carbon isotope measurements provide unambiguous evidence that next to E. huxleyi (Lenhart et al., 2016) other widespread marine algal species, namely Chrysochromulina sp. and P. globosa, are involved in the production of CH4 under oxic conditions at rates of 1.9±0.6 to 3.1±0.4 µg CH4 g−1 POC d−1. The three investigated genera of marine phytoplankton have a worldwide distribution and they are representatives of the most widespread marine haptophytes (Schoemann et al., 2005; Thomsen, 1994; Brown and Yoder, 1994). The results indicate that CH4 production could be a common process across marine haptophytes. We first discuss the stable isotopic evidence of CH4 formation, the role of precursor compounds, and the likely mechanisms involved. Finally, we discuss the laboratory CH4 production rates in relation to their potential significance in marine environments and provide a first rough estimation of how these production rates might contribute to CH4 concentration in oxic surface waters previously reported in open-ocean algal blooms.
In cultures of Chrysochromulina sp. and P. globosa that were treated with 13C-labeled hydrogen carbonate, δ13CH4 values increased with incubation time, clearly resulting from the conversion of 13C–hydrogen carbonate to 13CH4. These results demonstrate that all three investigated algal species are instrumental in the production of CH4 under oxic conditions (Fig. S1) and that hydrogen carbonate serves as a carbon source for 13CH4. Our findings are in agreement with the stable isotope evidence of CH4 production by E. huxleyi (Lenhart et al., 2016). However, we do not consider hydrogen carbonate to be the direct carbon precursor of CH4. In a first step hydrogen carbonate and its isotope label are converted to CO2 and subsequently fixed by algal primary production, forming POC. Therefore, we would expect a large fraction of the 13C label of the hydrogen carbonate ( ‰) to be transferred to the POC towards the end of the experiment (when the volume-normalized POC content is highest). The experiments were started by the inoculation of cells from pre-cultures that were grown on DIC with natural 13C∕12C abundance (δ13C values ∼0 ‰). This means that during ongoing incubation the δ13C–POC value should get close to δ13C–DIC values, resulting from the addition of 13C–hydrogen carbonate when cultures grow in the new 13C-enriched medium. Consequently, the δ13C–POC values are considered to be somewhat lower than the theoretically calculated δ13C–DIC values ( ‰) of the medium. Our assumptions are in line with the δ13CH4 source signature values (averaged over 9 or 11 d, respectively) obtained via the Keeling plot method, which were ‰ and ‰ for Chrysochromulina sp. and P. globosa, respectively, and were thus somewhat lower than for the theoretical calculated 13C value of the DIC ( ‰ ) resulting from the addition of 13C–hydrogen carbonate. Unfortunately, δ13C–DIC and δ13C–POC values could not be determined in our set of experiments to allow for more detailed calculations. However, our results clearly indicate that hydrogen carbonate is the principal inorganic carbon precursor of 13CH4 produced in algae, but intermediate metabolites are likely to be formed from which CH4 is released, possibly by cleavage of sulfur-bonded methyl groups of methyl thioethers and sulfoxides (Althoff et al., 2014; Lenhart et al., 2016; Benzing et al., 2017).
4.1 CH4 formation from 13C-labeled methyl thioethers
4.1.1 Methyl thioethers are precursors of CH4
Methyl thioethers and their sulfoxides are ubiquitous in marine environments as they are often produced by algae at substantial rates. It is also known that these compounds are metabolized in the three investigated algal species (Liss et al., 1994; Keller, 1989). Based on the addition of 13C2–DMSO, 13C2–DMS, and 13C–MSO, whereby only the sulfur-bonded methyl groups (–S–CH3) were 99 % labeled with 13C, it was possible to clearly monitor 13CH4 formation by stable carbon isotope measurements in cultures of E. huxleyi. The δ13CH4 values increased over time significantly in 13C2–DMS, 13C2–DMSO, and 13C–MSO treated cultures, above the δ13CH4 values of the control groups (Fig. 6a–c). The 13C labeling experiment showed that DMS, DMSO, and MSO are potentially important methyl precursors for CH4, but the contribution of these compounds to the overall CH4 production in cultures of E. huxleyi could not be determined in our experiments due to the complexity of the formation of these compounds in the algal cells. This can be illustrated by the following. The contribution of a substance to the total CH4 released is the product of both the added 13C-labeled fraction (added to the water sample and uptake by the cells) and the internally formed fraction of these compounds (DMS, DMSO, and MSO), which will roughly show natural 13C abundance. Therefore, the stable isotope value of CH4 will be diluted by the fraction of naturally formed methyl sulfur compounds in the algal cells, and thus the contribution of DMS, DMSO, and MSO to CH4 formation cannot be estimated on the basis of their added amount alone. The 13CH4 quantity from the conversion of added 13C-labeled substance contributed 0.03 % (13C2–DMSO) up to 0.84 % (13C–MSO) to the overall released CH4. However, even if the added 13C-labeled compounds might only explain ≤1 % of CH4 formed by the algae, their overall contribution (including non-labeled sulfur compounds, which we are not able to measure) might provide a much larger fraction of the released CH4. The intracellular DMS concentration can reach 1 mM (Sunda et al., 2002) in cells of E. huxleyi, while the concentration of added 13C2–DMS was 0.01 mM in medium (final concentration). If intracellular 13C2–DMS was in equilibrium with bulk seawater 13C2–DMS and all CH4 was produced from intracellular DMS, then the contribution of the 13C-labeled compound would be about 1 %. However, even if the biggest fraction of CH4 in algae cultures was not released by the 13C-labeled substances, the significant increase in delta notation in 13C2–DMS, 13C2–DMSO, and 13C–MSO treated cultures above the δ13CH4 values of the control groups demonstrates that 13C-labeled precursor substances were converted to CH4 by algal cultures (Fig. 6a–c). This is also indicated when the absolute conversion quantities of 13C-labeled substance in algal cultures are considered: these were ca. 9 (13C2–DMS), 3 (13C2–DMSO), and 30 (13C–MSO) times higher than in seawater control groups. Hence, the stable isotope labeling approach should be considered as a proof of concept that methyl groups of all tested substance serve as precursor compounds of CH4. These isotope labeling results are also in good agreement with recent results from laboratory experiments in which 13C–MET was added to cultures of E. huxleyi (Lenhart et al., 2016).
In addition, we also found an indication for a purely chemical CH4 formation pathway from control samples using sterile seawater and the addition of either 13C2–DMS or 13C–MSO. The 13C2–DMS-spiked seawater group and the 13C2–DMS-spiked algae group are very close to each other up to day 2 (see Figs. 5a and 6a). For this time period, it can be assumed that the chemical conversion has taken place in both samples to the same extent, since the samples are relatively similar because the algal cell density is only 5 % (day 2) of the final cell density. However, the following days (day 3 to day 6), when algal cell density increased drastically, the δ13CH4 values of the algae cultures also increased significantly compared with δ13CH4 values of the seawater. This clearly indicates that the conversion of 13C2–DMS to CH4 increases with increasing cell counts. However, the relatively slight increase in δ13CH4 values in the control samples (Fig. 6a, c) indicates that this is only a minor pathway. The CH4 conversion from 13C–DMS and 13C–MSO in seawater was approximately 3- and 30-fold lower than in the corresponding treatments with algae and only becomes obvious when applying very sensitive stable isotope labeling experiments. A similar observation was already made by Lenhart et al. (2016) when applying 13C–MET in seawater. However, this observation might be in agreement with previously findings by Zhang et al. (2015), who described a photochemically and CDOM-related conversion of DMS to CH4 in oxygenated natural seawater.
4.1.2 Potential mechanism of CH 4 formation from thioethers
The CH4 formation from thioethers (MET, DMS) and their corresponding sulfoxides (MSO, DMSO) might be catalyzed by nonheme iron-oxo (IV), thus forming methyl radicals (• CH3) from homolytically broken sulfur methyl bounds (R–CH3), leading to CH4 under oxidative conditions (Althoff et al., 2014; Benzing et al., 2017). The tested compounds are found in high cellular concentrations in E. huxleyi, Chrysochromulina sp., and P. globosa, and nonheme iron-oxo (IV) has been identified as an active intermediate in the catalytic cycles of a number of biological enzymatic systems (Hohenberger et al., 2012). Therefore, the postulated reaction might be a likely pathway for CH4 production in investigated algal species. Furthermore, DMS and DMSO were described to be part of an antioxidant system as these compounds can readily scavenge hydroxyl radicals in cells of E. huxleyi (Sunda et al., 2002). Furthermore, CH4 is released via a methyl radical that is subtracted from DMSO when hydroxyl radicals are being scavenged – and accordingly DMS after its sulfoxidation (Herscu-Kluska et al., 2008). Since MET and MSO have similar functional groups to DMS and DMSO, respectively, it was proposed that the reaction described above is taking place analogously for these compounds (Bruhn et al., 2012; Lenhart et al., 2015a). Consequently, the CH4 formation in investigated algal species might be a response of oxidative stress that forms hydroxyl radicals or other reactive oxygen species (ROS), which in turn might react with the applied methylated sulfur compounds, generating methyl radicals and eventually CH4.
The algal metabolites DMSP, DMS, and DMSO are ubiquitous in marine surface layers and nanomolar concentrations were found in blooms of Chrysochromulina sp., P. globosa, and E. huxleyi. Several field studies showed that these compounds are linked to CH4 formation in seawater (Zindler et al., 2013; Damm et al., 2008; Florez-Leiva et al., 2013). Various authors have proposed that DMSP and its degradation products DMSO and DMS are used by methylotrophic methanogenic archaea, inhabiting anoxic microsites, as substrates for methanogenesis. In addition it was reported that, if nitrogen is limited but phosphorus is replete, marine bacteria might also use DMSP as a carbon source, thereby releasing CH4 (Damm et al., 2010). One scenario which we cannot rule out would be the production of CH4 precursors by algae and the usage of these precursors by bacteria to produce CH4. While we think that this is less likely than CH4 production by algae alone, it would, if true, show that bacteria need algae-produced precursors to produce CH4. The latter scenario would be relevant in the field because algae coexist with bacteria in the oceans. Therefore, bacteria might be involved in the CH4 production process in our cultures, but even if they were, they would still depend on algal growth. For further discussion of a potential contribution of heterotrophs and/or methanogenic archaea, see the Supplement (S3). The correlations we describe in the Supplement clearly show that CH4 production depends on algal growth. It is therefore highly unlikely that bacteria are solely responsible for CH4 production in our cultures.
4.2 POC-normalized production
For all three algal species significant correlations between CH4 mass and cell density were found (r2>0.95 for all species; Fig. 3g, h, i), suggesting that CH4 formation occurred over the entire growth curve. However, since CH4 production can only be determined in the exponential phase (Langer et al., 2013) we additionally ran dilute batch cultures to determine CH4 production. All three species displayed similar CH4 production ranging from 1.9±0.6 to 3.1±0.4 µg CH4 g−1 POC d−1, with Chrysochromulina sp. and E. huxleyi showing the lowest and highest rates, respectively. The CH4 production for E. huxleyi was found to be 2-fold higher than rates reported for the same strain and comparable culture conditions by Lenhart et al. (2016) (0.7 µg CH4 g−1 POC d−1). The lower production reported by Lenhart et al. (2016) may be explained by the fact that CH4 production was not obtained from exponentially growing cultures. We also compared the cellular CH4 production rates of E. huxleyi reported by Scranton (1977) with those of our study. Scranton (1977) reported a production rate of nmol CH4 cell−1 h−1. This value is close to the production rate of nmol CH4 cell−1 h−1 in our study. Scranton (1977) concluded from observed CH4 production rates in laboratory experiments that natural populations might be adequate to support the widespread supersaturation of CH4 observed in the open ocean. However, we suggest that the CH4 production of various algae might differ substantially under changing environmental conditions, as already shown for terrestrial plants (Abdulmajeed and Qaderi, 2017; Martel and Qaderi, 2017). Moreover, the cellular concentrations of potential precursor compounds such as methylated sulfur compounds might vary greatly between species and cultures. The investigated algal species can reach millimolar intracellular concentrations of DMS and DMSP (Sunda et al., 2002; Liss et al., 1994; Keller, 1989), and even if the conversion rate of methylated sulfur compounds to CH4 in algal cells might be low, they could be sufficient to explain a substantial fraction of the CH4 production rates by marine algae.
4.3 Implication for the marine environment and algal blooms
In general, the distribution of chlorophyll has not shown a consistent correlation with CH4 distributions in field studies. There are studies in which no correlation was observed (e.g., Lamontagne et al., 1975; Foster et al., 2006; Watanabe et al., 1995) or a correlation was found within a few depth profiles (Burke et al., 1983; Brooks et al., 1981). Many field measurements in oxygenated surface waters in marine and limnic environments have shown examples of elevated CH4 concentrations spatially related to phytoplankton occurrence (e.g., Conrad and Seifer, 1988; Owens et al., 1991; Oudot et al., 2002; Damm et al., 2008; Grossart, et al., 2011; Weller et al., 2013; Zindler et al., 2013; Tang et al., 2014; Bogard et al., 2014; Rakowski et al., 2015). Taken together these studies suggest that phytoplankton is not the sole source of CH4 in oxygenated surface waters, but importantly, they also suggest that phytoplankton is one of the sources of CH4. We therefore compared the CH4 production rates of our cultures with two field studies for the Pacific Ocean (Weller et al., 2013) and the Baltic Sea (Schmale et al., 2018) to evaluate the potential relevance of algal CH4 production. It was estimated that the gross CH4 production in a southwest Pacific Ocean mesoscale eddy is 40–58 pmol CH4 L−1 d−1 (Weller et al., 2013). Using reported phytoplankton cell densities (1.7×108 to 2.9×108 cells L−1, Weller et al., 2013), we calculated a maximal cellular production of 5.5 ag CH4 cell−1 d−1 for this eddy. The species investigated in this study showed ca. 3–11 times higher cellular production (Table 1). Hence, each of the three haptophyte algae studied here could account for the CH4 production reported by Weller et al. (2013).
Schmale et al. (2018) reported CH4 enrichments that were observed during summer in the upper water column of the Gotland Basin, central Baltic Sea. Furthermore, they found that zooplankton is one but not the only CH4 source in the oxygenated upper waters. While the authors ruled out a major contribution of algae to the observed sub-thermocline CH4 enrichment because of the low sub-thermocline phytoplankton biomass, they considered primary-production-associated CH4 formation to be one likely source in the phytoplankton-rich mixed layer. The average phytoplankton carbon biomass of the mixed layer was approximately 600 µg L−1 (averaged from Fig. 9 in Schmale et al., 2018). For the reported average net CH4 production rate in the mixed layer (95 pmol CH4 L−1 d−1), we calculated that a production rate of 2.5 µg g−1 POC d−1 is required if the CH4 is produced by the algal biomass. This rate would be within the range of CH4 production rates observed in our study. These calculations should be considered as a first rough estimate to assess whether the CH4 production rates of laboratory-grown cultures can significantly contribute to CH4 supersaturation associated with phytoplankton. We did not distinguish between species and did not take into account environmental factors or the complexity of microbiological communities.
Judging from cellular production, the species studied here are of similar importance for oceanic CH4 production in biogeochemical terms. Regarding the highest cellular production, with that of E. huxleyi as 100 %, P. globosa produces 27 % and Chrysochromulina sp. 71 % (Table 1). However, several recent studies have emphasized that production potential (PP), as opposed to cellular production, is a biogeochemically meaningful parameter (Gafar et al., 2018; Marra, 2002; Schlüter et al., 2014; Kottmeier et al., 2016). The concept of production potential goes back at least to the first half of the 20th century (Clarke et al., 1946). Briefly, the production potential of substance X is the amount of X which a phytoplankton community or culture produces in a given time. For details, see Sect. 2 and the references above. Cellular production, by contrast, is the rate of production of X of a single cell, and therefore the cellular production is ill-qualified to express community-level production. We calculated the CH4–PP (Sect. 2) for our three species, and when the one of E. huxleyi is considered to be 100 %, P. globosa has a CH4–PP of 0.9 % and Chrysochromulina sp. 0.8 % (Table 1). In terms of CH4 production in the field, therefore, E. huxleyi outperforms the other two haptophytes by 2 orders of magnitude. It can be concluded that the CH4–PP under given environmental conditions is species-specific, and therefore community composition will have an influence on algal sea surface water CH4 production.
It can be hypothesized that changing environmental conditions might drastically affect algal CH4 production, which has to be taken into account when calculating annual averages. The effect of dominant environmental parameters such as light intensity and temperature on algal CH4 production will therefore be the subject matter of future studies.
We provide the data in heiDATA, which is an institutional repository for research data of Heidelberg University (https://doi.org/10.11588/data/9N1LSR; Klintzsch and Keppler, 2019).
The supplement related to this article is available online at: https://doi.org/10.5194/bg-16-4129-2019-supplement.
GL, GN, TK, KL, and FK conceived the study and designed the experiments; TK performed the experiments under the supervision of FK and GL; AW helped with the cultivation of algae and measurements; TK, FK, and GL analyzed the data; FK, GL, GN, KL, and TK discussed the results, and GL, TK, and FK wrote the paper.
The authors declare that they have no conflict of interest.
We thank Markus Greule, Bernd Knape, and Stefan Rheinberger for conducting analytical measurements and for the technical support that helped to produce this dataset. We acknowledge financial support by the Deutsche Forschungsgemeinschaft within the funding program Open Access Publishing, by the Baden-Württemberg Ministry of Science, Research and the Arts, and by Ruprecht-Karls-Universität Heidelberg.
This research has been supported by the Deutsche Forschungsgemeinschaft (grant nos. KE 884/8-2, KE 884/11-1, and KE 884/16-2), the Natural Environment Research Council (grant no. NE/N011708/1), the Baden-Württemberg Ministry of Science, Research and the Arts, and Ruprecht-Karls-Universität Heidelberg.
This paper was edited by Jack Middelburg and reviewed by Mary Scranton and one anonymous referee.
Abdulmajeed, A. M. and Qaderi, M. M.: Intrashoot variation in aerobic methane emissions from pea plants exposed to multiple abiotic stresses, Acta Physiol. Plant., 39, 124, https://doi.org/10.1007/s11738-017-2420-y, 2017.
Althoff, F., Benzing, K., Comba, P., McRoberts, C., Boyd, D. R., Greiner, S., and Keppler, F.: Abiotic methanogenesis from organosulphur compounds under ambient conditions, Nat. Commun., 5, 4205, https://doi.org/10.1038/ncomms5205, 2014.
Angel, R., Matthies, D., and Conrad, R.: Activation of Methanogenesis in Arid Biological Soil Crusts Despite the Presence of Oxygen, Plos One, 6, e20453, https://doi.org/10.1371/journal.pone.0020453, 2011.
Bange, H. W. and Uher, G.: Photochemical production of methane in natural waters: implications for its present and past oceanic source, Chemosphere, 58, 177–183, https://doi.org/10.1016/j.chemosphere.2004.06.022, 2005.
Bange, H. W., Bartell, U., Rapsomanikis, S., and Andreae, M. O.: Methane in the Baltic and North Seas and a reassessment of the marine emissions of methane, Global Biogeochem. Cy., 8, 465–480, 1994.
Benzing, K., Comba, P., Martin, B., Pokrandt, B., and Keppler, F.: Nonheme Iron-Oxo-Catalyzed Methane Formation from Methyl Thioethers: Scope, Mechanism, and Relevance for Natural Systems, Chemistry, A European Journal, 23, 10465–10472, https://doi.org/10.1002/chem.201701986, 2017.
Bizic-Ionescu, M., Klintzch, T., Ionescu, D., Hindiyeh, M. Y., Guenthel, M., Muro Pastor, A. M., Keppler, F., and Grossart, H.-P.: Widespread formation of methane by Cyanobacteria in aquatic and terrestrial environments, https://doi.org/10.1101/398958, 2018a.
Bižić-Ionescu, M., Ionescu, D., Günthel, M., Tang, K. W., and Grossart, H.-P.: Oxic Methane Cycling: New Evidence for Methane Formation in Oxic Lake Water, in: Biogenesis of Hydrocarbons, edited by: Stams, A. J. M and Souna, D. Z., Springer International Publishing, Basel, 1–22, 2018b.
Bogard, M. J., del Giorgio, P. A., Boutet, L., Chaves, M. C. G., Prairie, Y. T., Merante, A., and Derry, A. M.: Oxic water column methanogenesis as a major component of aquatic CH4 fluxes, Nat. Commun., 5, 5350, https://doi.org/10.1038/ncomms6350, 2014.
Borges, A. V., Speeckaert, G., Champenois, W., Scranton, M. I., and Gypens, N.: Productivity and temperature as drivers of seasonal and spatial variations of dissolved methane in the Southern Bight of the North Sea, Ecosystems, 21, 583–599, 2018.
Boros, M. and Keppler, F.: Production and Signaling of Methane, in: Gasotransmitters, edited by: Wang, R., Royal Society of Chemistry, London, 192–234, https://doi.org/10.1039/9781788013000, 2018
Brooks, J. M., Reid, D. F., and Bernard, B. B.: Methane in the upper water column of the northwestern Gulf of Mexico, J. Geophys. Res.-Ocean., 86, 11029–11040, https://doi.org/10.1029/JC086iC11p11029, 1981.
Brown, C. W. and Yoder, J. A.: Coccolithophorid blooms in the global ocean, J. Geophys. Res.-Ocean., 99, 7467–7482, 1994.
Bruhn, D., Møller, I. M., Mikkelsen, T. N., and Ambus, P.: Terrestrial plant methane production and emission, Physiol. Plantarum, 144, 201–209, 2012.
Burke Jr., R. A., Reid, D. F., Brooks, J. M., and Lavoie, D. M.: Upper water column methane geochemistry in the eastern tropical North Pacific1, Limnol. Oceanogr., 28, 19–32, https://doi.org/10.4319/lo.1983.28.1.0019, 1983.
Clarke, G. L., Edmondson, W. T., and Ricker, W. E.: Dynamics of Production in a Marine Area, Ecol. Monogr., 16, 321–337, https://doi.org/10.2307/1961639, 1946.
Comba, P., Faltermeier, D., Gieger, S., Keppler, F., Schöler, H. F., and Schroll, M.: Iron catalyzed demethylation of acetic acid, J. Coord. Chem., 71, 1704–1714, https://doi.org/10.1080/00958972.2018.1490414, 2018.
Conrad, R. and Seiler, W.: Methane and hydrogen in seawater (Atlantic Ocean), Deep-Sea Res. Pt. A, 35, 1903–1917, https://doi.org/10.1016/0198-0149(88)90116-1, 1988.
Damm, E., Kiene, R. P., Schwarz, J., Falck, E., and Dieckmann, G.: Methane cycling in Arctic shelf water and its relationship with phytoplankton biomass and DMSP, Mar. Chem., 109, 45–59, https://doi.org/10.1016/j.marchem.2007.12.003, 2008.
Damm, E., Helmke, E., Thoms, S., Schauer, U., Nöthig, E., Bakker, K., and Kiene, R. P.: Methane production in aerobic oligotrophic surface water in the central Arctic Ocean, Biogeosciences, 7, 1099–1108, https://doi.org/10.5194/bg-7-1099-2010, 2010.
Damm, E., Rudels, B., Schauer, U., Mau, S., and Dieckmann, G.: Methane excess in Arctic surface water-triggered by sea ice formation and melting, Sci. Rep., 5, 16179, https://doi.org/10.1038/srep16179, 2015.
de Angelis, M. A. and Lee, C.: Methane production during zooplankton grazing on marine phytoplankton, Limnol. Oceanogr., 39, 1298–1308, 1994.
del Valle, D. A. and Karl, D. M.: Aerobic production of methane from dissolved water-column methylphosphonate and sinking particles in the North Pacific Subtropical Gyre, Aquat. Microb. Ecol., 73, 93–105, 2014.
Denman, K. L., Brasseur, G., Chidthaisong, A., Ciais, P., Cox, P. M., Dickinson, R. E., Hauglustaine, D., Heinze, C., Holland, E., Jacob, D., Lohmann, U., Ramachandran, S., Dias, P. L. d. S., Wofsy, S. C., and Zhang, X.: Couplings between changes in the climate system and biogeochemistry., in: Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, edited by: Solomon, S., Qin, D., Manning, M., Chen, Z., Marquis, M., Averyt, K. B., Tignor, M., and Miller, H. L., Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, 500–587, 2007.
Donis, D., Flury, S., Stöckli, A., Spangenberg, J. E., Vachon, D., and McGinnis, D. F.: Full-scale evaluation of methane production under oxic conditions in a mesotrophic lake, Nat. Commun., 8, 1661, https://doi.org/10.1038/s41467-017-01648-4, 2017.
Field, C. B., Behrenfeld, M. J., Randerson, J. T., and Falkowski, P.: Primary production of the biosphere: Integrating terrestrial and oceanic components, Science, 281, 237–240, 1998.
Florez-Leiva, L., Damm, E., and Farías, L.: Methane production induced by dimethylsulfide in surface water of an upwelling ecosystem, Prog. Oceanogr., 112, 38–48, 2013.
Forster, G., Upstill-Goddard, R. C., Gist, N., Robinson, C., Uher, G., and Woodward, E. M. S.: Nitrous oxide and methane in the Atlantic Ocean between 50∘ N and 52∘ S: latitudinal distribution and sea-to-air flux, Deep-Sea Res. Pt. II, 56, 964–976, 2009.
Gafar, N. A., Eyre, B. D., and Schulz, K. G.: A conceptual model for projecting coccolithophorid growth, calcification and photosynthetic carbon fixation rates in response to global ocean change, Front. Mar. Sci., 4, 433, https://doi.org/10.3389/fmars.2017.00433, 2018.
Ghyczy, M., Torday, C., Kaszaki, J., Szabo, A., Czobel, M., and Boros, M.: Hypoxia-induced generation of methane in mitochondria and eukaryotic cells – An alternative approach to methanogenesis, Cell. Physiol. Biochem., 21, 251–258, 2008.
Grossart, H.-P., Frindte, K., Dziallas, C., Eckert, W., and Tang, K. W.: Microbial methane production in oxygenated water column of an oligotrophic lake, P. Natl. Acad. Sci. USA, 108, 19657–19661, https://doi.org/10.1073/pnas.1110716108, 2011.
Guillard, R. R. and Ryther, J. H.: Studies of marine planktonic diatoms: I. Cyclotella nana Hustedt, and Detonula confervacea (Cleve) Gran, Can. J. Microbiol., 8, 229–239, 1962.
Herscu-Kluska, R., Masarwa, A., Saphier, M., Cohen, H., and Meyerstein, D.: Mechanism of the reaction of radicals with peroxides and dimethyl sulfoxide in aqueous solution, Chemistry – A European Journal, 14, 5880–5889, 2008.
Hohenberger, J., Ray, K., and Meyer, K.: The biology and chemistry of high-valent iron–oxo and iron–nitrido complexes, Nat. Commun., 3, 720, https://doi.org/10.1038/ncomms1718, 2012.
Holligan, P. M., Fernández, E., Aiken, J., Balch, W. M., Boyd, P., Burkill, P. H., Finch, M., Groom, S. B., Malin, G., and Muller, K.: A biogeochemical study of the coccolithophore, Emiliania huxleyi, in the North Atlantic, Global Biogeochem. Cy., 7, 879–900, 1993.
Karl, D. M. and Tilbrook, B. D.: Production and transport of methane in oceanic particulate organic matter, Nature, 368, 732–734, 1994.
Karl, D. M., Beversdorf, L., Bjorkman, K. M., Church, M. J., Martinez, A., and DeLong, E. F.: Aerobic production of methane in the sea, Nat. Geosci., 1, 473–478, https://doi.org/10.1038/ngeo234, 2008.
Keeling, C. D.: The concentration and isotopic abundances of atmospheric carbon dioxide in rural areas, Geochim. Cosmochim. Ac., 13, 322–334, https://doi.org/10.1016/0016-7037(58)90033-4, 1958.
Keller, M. D.: Dimethyl sulfide production and marine phytoplankton: the importance of species composition and cell size, Biol. Oceanogr., 6, 375–382, 1989.
Keppler, F., Hamilton, J. T. G., Brass, M., and Rockmann, T.: Methane emissions from terrestrial plants under aerobic conditions, Nature, 439, 187–191, 2006.
Keppler, F., Boros, M., Frankenberg, C., Lelieveld, J., McLeod, A., Pirttila, A. M., Rockmann, T., and Schnitzler, J. P.: Methane formation in aerobic environments, Environ. Chem., 6, 459–465, https://doi.org/10.1071/en09137, 2009.
Keppler, F., Schiller, A., Ehehalt, R., Greule, M., Hartmann, J., and Polag, D.: Stable isotope and high precision concentration measurements confirm that all humans produce and exhale methane, J. Breath Res., 10, 016003, https://doi.org/10.1088/1752-7155/10/1/016003, 2016.
Kirschke, S., Bousquet, P., Ciais, P., Saunois, M., Canadell, J. G., Dlugokencky, E. J., Bergamaschi, P., Bergmann, D., Blake, D. R., Bruhwiler, L., Cameron-Smith, P., Castaldi, S., Chevallier, F., Feng, L., Fraser, A., Heimann, M., Hodson, E. L., Houweling, S., Josse, B., Fraser, P. J., Krummel, P. B., Lamarque, J. F., Langenfelds, R. L., Le Quere, C., Naik, V., O'Doherty, S., Palmer, P. I., Pison, I., Plummer, D., Poulter, B., Prinn, R. G., Rigby, M., Ringeval, B., Santini, M., Schmidt, M., Shindell, D. T., Simpson, I. J., Spahni, R., Steele, L. P., Strode, S. A., Sudo, K., Szopa, S., van der Werf, G. R., Voulgarakis, A., van Weele, M., Weiss, R. F., Williams, J. E., and Zeng, G.: Three decades of global methane sources and sinks, Nat. Geosci., 6, 813–823, https://doi.org/10.1038/ngeo1955, 2013.
Klintzsch, T. and Keppler, F.: Methane production by three widespread marine phytoplankton species: release rates, precursor compounds, and potential relevance for the environment, https://doi.org/10.11588/data/9N1LSR, heiDATA, V1, UNF:6:dxf+QSeRKu9v6uPyAUhX8Q==[fileUNF], 2019.
Kottmeier, D. M., Rokitta, S. D., and Rost, B.: H+-driven increase in CO2 uptake and decrease in uptake explain coccolithophores' acclimation responses to ocean acidification, Limnol. Oceanogr., 61, 2045–2057, https://doi.org/10.1002/lno.10352, 2016.
Lamontagne, R. A., Smith, W. D., and Swinnerton, J. W.: C1-C3 Hydrocarbons and Chlorophyll a Concentrations in the Equatorial Pacific Ocean, in: Analytical Methods in Oceanography, Adv. Chem., 147, Am. Chem. Soc., 147, 163–171, 1975.
Langer, G., Nehrke, G., Probert, I., Ly, J., and Ziveri, P.: Strain-specific responses of Emiliania huxleyi to changing seawater carbonate chemistry, Biogeosciences, 6, 2637–2646, https://doi.org/10.5194/bg-6-2637-2009, 2009.
Langer, G., Oetjen, K., and Brenneis, T.: Calcification of Calcidiscus leptoporus under nitrogen and phosphorus limitation, J. Exp. Mar. Biol. Ecol., 413, 131–137, 2012.
Langer, G., Oetjen, K., and Brenneis, T.: Coccolithophores do not increase particulate carbon production under nutrient limitation: A case study using Emiliania huxleyi (PML B92/11), J. Exp. Mar. Biol. Ecol., 443, 155–161, 2013.
Laukenmann, S., Polag, D., Heuwinkel, H., Greule, M., Gronauer, A., Lelieveld, J., and Keppler, F.: Identification of methanogenic pathways in anaerobic digesters using stable carbon isotopes, Eng. Life Sci., 10, 509–514, https://doi.org/10.1002/elsc.201000074, 2010.
Lelieveld, J., Crutzen, P. J., and Dentener, F. J.: Changing concentration, lifetime and climate forcing of atmospheric methane, Tellus B, 50, 128–150, 1998.
Lenhart, K., Bunge, M., Rathering, S., Neu, T., Schüttman, I., Greule, M., Kammann, C., Schnell, S., Müller, C., Zorn, H., and Keppler, F.: Evidence for methane production by saprotrophic fungi, Nat. Commun., 3, 1046, https://doi.org/10.1038/ncomms2049, 2012.
Lenhart, K., Althoff, F., Greule, M., and Keppler, F.: Technical Note: Methionine, a precursor of methane in living plants, Biogeosciences, 12, 1907–1914, https://doi.org/10.5194/bg-12-1907-2015, 2015a.
Lenhart, K., Weber, B., Elbert, W., Steinkamp, J., Clough, T., Crutzen, P., Pöschl, U., and Keppler, F.: Nitrous oxide and methane emissions from cryptogamic covers, Glob. Change Biol., 21, 3889–3900, https://doi.org/10.1111/gcb.12995, 2015b.
Lenhart, K., Klintzsch, T., Langer, G., Nehrke, G., Bunge, M., Schnell, S., and Keppler, F.: Evidence for methane production by the marine algae Emiliania huxleyi, Biogeosciences, 13, 3163–3174, https://doi.org/10.5194/bg-13-3163-2016, 2016.
Liss, P., Malin, G., Turner, S., and Holligan, P.: Dimethyl sulphide and Phaeocystis: a review, J. Mar. Syst., 5, 41–53, 1994.
Marra, J.: Approaches to the Measurement of Plankton Production, in: Phytoplankton Productivity and Carbon Assimilation in Marine and Freshwater Ecoystems, edited by: Williams, P. J., Thomas, D. R., and Reynolds, C. S., London, Blackwell, 222–264, 2002.
Martel, A. B. and Qaderi, M. M.: Light quality and quantity regulate aerobic methane emissions from plants, Physiol. Plantarum, 159, 313–328, 2017.
Matrai, P. A. and Keller, M. D.: Dimethylsulfide in a large-scale coccolithophore bloom in the Gulf of Maine, Cont. Shelf Res., 13, 831–843, 1993.
Menden-Deuer, S. and Lessard, E. J.: Carbon to volume relationships for dinoflagellates, diatoms, and other protist plankton, Limnol. Oceanogr., 45, 569–579, 2000.
Metcalf, W. W., Griffin, B. M., Cicchillo, R. M., Gao, J. T., Janga, S. C., Cooke, H. A., Circello, B. T., Evans, B. S., Martens-Habbena, W., Stahl, D. A., and van der Donk, W. A.: Synthesis of Methylphosphonic Acid by Marine Microbes: A Source for Methane in the Aerobic Ocean, Science, 337, 1104–1107, https://doi.org/10.1126/science.1219875, 2012.
Olenina, I., Hajdu, S., Edler, L., Andersson, A., Wasmund, N., Busch, S., Göbel, J., Gromisz, S., Huseby, S., Huttunen, M., Jaanus, A., Kokkonen, P., Ledaine, I., and Niemkiewicz, E.: Biovolumes and size-classes of phytoplankton in the Baltic Sea HELCOM Balt, Sea Environ. Proc. No. 106, 144 pp., 2006.
Oremland, R. S.: Methanogenic activity in plankton samples and fish intestines – mechanism for insitu methanogenesis in oceanic surface waters, Limnol. Oceanogr., 24, 1136–1141, 1979.
Oudot, C., Jean-Baptiste, P., Fourré, E., Mormiche, C., Guevel, M., Ternon, J.-F., and Le Corre, P.: Transatlantic equatorial distribution of nitrous oxide and methane, Deep-Sea Res. Pt. I, 49, 1175–1193, https://doi.org/10.1016/S0967-0637(02)00019-5, 2002.
Owens, N. J. P., Law, C. S., Mantoura, R. F. C., Burkill, P. H., and Llewellyn, C. A.: Methane flux to the atmosphere from the Arabian Sea, Nature, 354, 293–296, https://doi.org/10.1038/354293a0, 1991.
Pataki, D. E., Ehleringer, J. R., Flanagan, L. B., Yakir, D., Bowling, D. R., Still, C. J., Buchmann, N., Kaplan, J. O., and Berry, J. A.: The application and interpretation of Keeling plots in terrestrial carbon cycle research, Global Biogeochem. Cy., 17, 1022, https://doi.org/10.1029/2001GB001850, 2003.
Paul, D., Skrzypek, G., and Fórizs, I.: Normalization of measured stable isotopic compositions to isotope reference scales – a review, Rapid Commun. Mass Sp., 21, 3006–3014, https://doi.org/10.1002/rcm.3185, 2007.
Rakowski, C., Magen, C., Bosman, S., Gillies, L., Rogers, K., Chanton, J., and Mason, O. U.: Methane and microbial dynamics in the Gulf of Mexico water column, Front. Mar. Sci., 2, 69, https://doi.org/10.3389/fmars.2015.00069, 2015.
Reeburgh, W. S.: Oceanic methane biogeochemistry, Chem. Rev., 107, 486–513, https://doi.org/10.1021/cr050362v, 2007.
Repeta, D. J., Ferrón, S., Sosa, O. A., Johnson, C. G., Repeta, L. D., Acker, M., DeLong, E. F., and Karl, D. M.: Marine methane paradox explained by bacterial degradation of dissolved organic matter, Nat. Geosci., 9, 884–887, 2016.
Saunois, M., Bousquet, P., Poulter, B., Peregon, A., Ciais, P., Canadell, J. G., Dlugokencky, E. J., Etiope, G., Bastviken, D., and Houweling, S.: The global methane budget 2000–2012, Earth Syst. Sci. Data, 8, 697–751, 2016.
Schindelin, J., Arganda-Carreras, I., Frise, E., Kaynig, V., Longair, M., Pietzsch, T., Preibisch, S., Rueden, C., Saalfeld, S., and Schmid, B.: Fiji: an open-source platform for biological-image analysis, Nat. Methods, 9, 676, https://doi.org/10.1038/nmeth.2019, 2012.
Schlüter, L., Lohbeck, K. T., Gutowska, M. A., Gröger, J. P., Riebesell, U., and Reusch, T. B. H.: Adaptation of a globally important coccolithophore to ocean warming and acidification, Nat. Clim. Change, 4, 1024, https://doi.org/10.1038/nclimate2379, 2014.
Schmale, O., Wäge, J., Mohrholz, V., Wasmund, N., Gräwe, U., Rehder, G., Labrenz, M., and Loick-Wilde, N.: The contribution of zooplankton to methane supersaturation in the oxygenated upper waters of the central Baltic Sea, Limnol. Oceanogr., 63, 412–430, 2018.
Schoemann, V., Becquevort, S., Stefels, J., Rousseau, V., and Lancelot, C.: Phaeocystis blooms in the global ocean and their controlling mechanisms: a review, J. Sea Res., 53, 43–66, 2005.
Scranton, M. I.: The marine geochemistry of methane, Ph.D., Massachusetts Institute of Technology and Woods Hole Oceanographic Institution, Massachusetts, USA, 251 pp., 1977.
Scranton, M. I. and Brewer, P. G.: Occurrence of methane in the near-surface waters of the western subtropical North-Atlantic, Deep-Sea Res., 24, 127–138, https://doi.org/10.1016/0146-6291(77)90548-3, 1977.
Scranton, M. I. and Farrington, J. W.: Methane production in the waters off Walvis Bay, J. Geophys. Res., 82, 4947–4953, https://doi.org/10.1029/JC082i031p04947, 1977.
Stawiarski, B., Otto, S., Thiel, V., Gräwe, U., Loick-Wilde, N., Wittenborn, A. K., Schloemer, S., Wäge, J., Rehder, G., Labrenz, M., Wasmund, N., and Schmale, O.: Controls on zooplankton methane production in the central Baltic Sea, Biogeosciences, 16, 1–16, https://doi.org/10.5194/bg-16-1-2019, 2019.
Stefels, J., Steinke, M., Turner, S., Malin, G., and Belviso, S.: Environmental constraints on the production and removal of the climatically active gas dimethylsulphide (DMS) and implications for ecosystem modelling, Biogeochemistry, 83, 245–275, https://doi.org/10.1007/s10533-007-9091-5, 2007.
Sunda, W., Kieber, D. J., Kiene, R. P., and Huntsman, S.: An antioxidant function for DMSP and DMS in marine algae, Nature, 418, 317–320, https://doi.org/10.1038/nature00851, 2002.
Tang, K. W., McGinnis, D. F., Frindte, K., Brüchert, V., and Grossart, H.-P.: Paradox reconsidered: Methane oversaturation in well-oxygenated lake waters, Limnol. Oceanogr., 59, 275–284, 2014.
Thomsen, H. A. Buck, K.R. and Chavez F.P.: Haptophytes as components of marine phytoplankton, in: The Haptophyte Algae, edited by: Green, J. C. and Leadbeater, B. S. C., Clarendon Press, Oxford, 187–208, 1994
Watanabe, S., Higashitani, N., Tsurushima, N., and Tsunogai, S.: Methane in the western North Pacific, J. Oceanogr., 51, 39–60, https://doi.org/10.1007/bf02235935, 1995.
Weller, D. I., Law, C. S., Marriner, A., Nodder, S. D., Chang, F. H., Stephens, J. A., Wilhelm, S. W., Boyd, P. W., and Sutton, P. J. H.: Temporal variation of dissolved methane in a subtropical mesoscale eddy during a phytoplankton bloom in the southwest Pacific Ocean, Prog. Oceanogr., 116, 193–206, https://doi.org/10.1016/j.pocean.2013.07.008, 2013.
Wiesenburg, D. A. and Guinasso, N. L.: Equilibrium solubilities of methane, carbon monoxide, and hydrogen in water and sea water, J. Chem. Eng. Data, 24, 356–360, https://doi.org/10.1021/je60083a006, 1979.
Zhang, Y. and Xie, H.: Photomineralization and photomethanification of dissolved organic matter in Saguenay River surface water, Biogeosciences, 12, 6823–6836, https://doi.org/10.5194/bg-12-6823-2015, 2015.
Zindler, C., Bracher, A., Marandino, C. A., Taylor, B., Torrecilla, E., Kock, A., and Bange, H. W.: Sulphur compounds, methane, and phytoplankton: interactions along a north-south transit in the western Pacific Ocean, Biogeosciences, 10, 3297–3311, https://doi.org/10.5194/bg-10-3297-2013, 2013.