the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Increasing soil carbon stocks in eight permanent forest plots in China
Jianxiao Zhu
Chuankuan Wang
Zhang Zhou
Guoyi Zhou
Xueyang Hu
Lai Jiang
Yide Li
Guohua Liu
Chengjun Ji
Shuqing Zhao
Peng Li
Jiangling Zhu
Zhiyao Tang
Chengyang Zheng
Richard A. Birdsey
Yude Pan
Jingyun Fang
Forest soils represent a major stock of organic carbon (C) in the terrestrial biosphere, but the dynamics of soil organic C (SOC) stock are poorly quantified, largely due to lack of direct field measurements. In this study, we investigated the 20-year changes in SOC stocks in eight permanent forest plots, which represent boreal (1998–2014), temperate (1992–2012), subtropical (1987–2008), and tropical forest biomes (1992–2012) across China. SOC contents increased significantly from the 1990s to the 2010s, mostly in the upper 0–20 cm soil depth, and soil bulk densities do not change significantly during the same period. As a result, the averaged SOC stocks increased significantly from 125.2±85.2 Mg C ha−1 in the 1990s to 133.6±83.1 Mg C ha−1 in the 2010s across the forest plots, with a mean increase of 127.2–907.5 kg C ha−1 yr−1. This SOC accumulation resulted primarily from increasing leaf litter and fallen logs, which accounts 3.6 %–16.3 % of above-ground net primary production. Our findings provided direct evidence that China's forest soils have been acting as significant C sinks, although their strength varies in forests with different climates.
- Article
(2732 KB) - Full-text XML
-
Supplement
(815 KB) - BibTeX
- EndNote
Terrestrial ecosystems have absorbed approximately 30 % of the carbon dioxide (CO2) emitted from human activity since the beginning of the industrial era (IPCC, 2013). Forests have contributed more than half of these carbon (C) fluxes of terrestrial ecosystems (Pan et al., 2011). Since soils contain a huge C stock in forest ecosystems, even a slight change in this stock will induce a considerable feedback to atmospheric CO2 concentrations (Lal, 2004; Luo et al., 2011). Thus, accurate assessment of the changes in soil organic carbon (SOC) is critical to understanding how forest soils will respond to global climate change. However, it is difficult to capture the SOC change with short-term measurements (Smith, 2004) because the soil C pool typically has a longer turnover time and higher spatial variability compared to the vegetation C pool (Schrumpf et al., 2011; Canadell and Schulze, 2014).
Previous efforts have estimated the changes in regional SOC stocks with indirect approaches, such as regional assessments (Yang et al., 2014) and model simulations (Todd-Brown et al., 2013). These estimates often involve large uncertainties due to the inherently high spatial variability of soils and a lack of direct measurements representing large areas (Sitch et al., 2015). One reliable approach to reducing the uncertainties is to conduct long-term monitoring of forest SOC stocks at sites that represent broader landscapes (Prietzel et al., 2016). Unfortunately, such repeated, accurate field-based measurements of SOC stocks from which to generate change estimates are generally lacking and inadequate worldwide (Zhao et al., 2019).
A few soil resampling studies have explored SOC changes in different forests, but the results are often contradictory. For instance, Schrumpf et al. (2014) found that SOC in deciduous broadleaved forests in central Germany increased, with a change rate of 650.0 kg C ha−1 yr−1 from 2004–2009. In contrast, Prietzel et al. (2016) indicated that SOC stocks in German forests decreased significantly, with average change rates of 988.2 kg C ha−1 yr−1 in forests in the Alps between 1986 and 2011, and 441.1 kg C ha−1 yr−1 in the Berchtesgaden region between 1976 and 2011. Kiser et al. (2009) found that the hardwood forest soils in central Tennessee, USA, exhibited a slight C source and that the relative change rate ranged from −0.4 % yr−1 to 0.3 % yr−1 between 1976 and 2006. Chen et al. (2015) synthesized global SOC changes and found that the relative rates of change in forest SOC stocks were contradictory among long-term experiments (0.2 % yr−1), regional comparisons (0.3 % yr−1), and repeated soil samplings (−0.1 % yr−1). Such discrepancies can be partly attributed to insufficient observations and inconsistent methodologies. The different effects of changing environmental factors and nitrogen inputs on soil C dynamics may also be involved (Norby and Zak, 2011). In addition, to date these studies have primarily been conducted in the forests of Europe and the USA, but few have been carried out in China's forests.
Forests in China cover an area of 156 Mha (Guo et al., 2013) and range from boreal coniferous forests and deciduous broadleaved forests in the northeast to tropical rain forests and evergreen broadleaved forests in the south and southwest. They include almost all major forest biomes of the Northern Hemisphere (Fang et al., 2012). Such variations in climate and forest types have provided ideal opportunities to examine the spatial patterns of SOC in relation to meteorological and biological factors. At the national scale, the mean annual air temperature of China increased by more than 1 ∘C between 1982 and 2011, which is considerably higher than the global average (Fang et al., 2018). Since the 1980s, the Chinese Government has implemented several large-scale national forest protection projects. These climatic changes and conservation practices in China have significantly stimulated C uptake into forest ecosystems (Fang et al., 2014, 2018; Feng et al., 2019). Several studies have assessed the temporal dynamics of SOC stock across China's forests using model simulations (Piao et al., 2009) or regional assessments (Pan et al., 2011; Tang et al., 2018). However, these estimates revealed contrasting trends in SOC dynamics and also lacked direct measurements of SOC change.
Therefore, in this study we measured SOC density (C amount per unit area) of eight permanent forest plots from tropical, subtropical, temperate, and boreal forests in China during two periods in the 1990s and 2010s to quantify their SOC changes. We then analyzed the potential biotic and climatic drivers in the SOC dynamics across these forests. Finally, we assessed the changes in SOC stocks in China's forests using the site data obtained from this study.
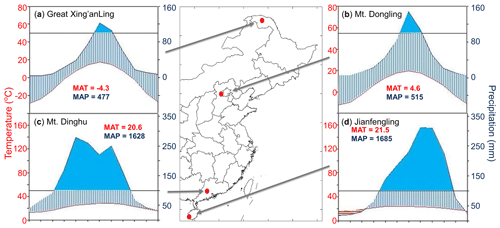
Figure 1Locations and climatic conditions of the sites. (a) Great Xing'anling, the boreal site; (b) Mt. Dongling, the temperate site; (c) Mt. Dinghu, the subtropical site; and (d) Jianfengling, the tropical site. The blue and red lines in climatic diagrams are the monthly mean values of precipitation and temperature, respectively. The blue areas indicate the period in the year when the precipitation exceeded 100 mm month−1. MAT is mean annual temperature, and MAP is mean annual precipitation. Publisher’s note: Please note that the above figure contains disputed territories.
2.1 Study sites
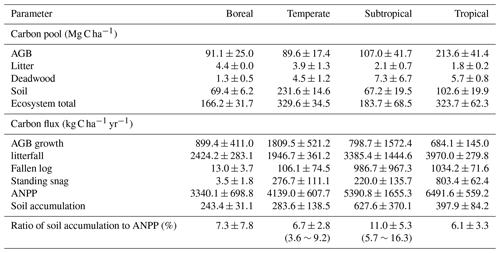
We investigated eight permanent forest plots in four forest sites (from north to south: Great Xing'anling, Mt. Dongling, Mt. Dinghu, and Jianfengling) (Fig. 1). The four sites spanned a wide range from 18.7 to 52.6∘ N in latitude, and belonged to boreal, temperate, subtropical, and tropical climate zones, respectively, with a climatic difference of approximately 26 ∘C in mean annual temperature and 1200 mm in mean annual precipitation. The eight plots are comprised of a boreal larch forest (Larix gmelinii), two temperate deciduous broadleaved forests (Betula platyphylla and Quercus wutaishanica), a temperate pine plantation (Pinus tabuliformis), a subtropical evergreen broadleaved forest, a subtropical pine plantation (P. massoniana), a subtropical pine and broadleaved mixed forest, and a tropical mountain rainforest (for details, see Table 1).
Table 1Location, forest type, mean annual temperature (MAT), and mean annual precipitation (MAP) at eight forest plots in four climate zones, together with forest origin and study periods.
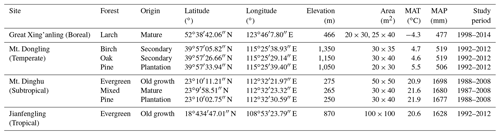
Stand characteristics of all eight plots are summarized in Table 1. The boreal larch forest was a 100-year-old mature stand at the time of the first sampling (Wang et al., 2001). Three temperate forest plots (birch, oak, and pine forests) were located along an elevation gradient on Mt. Dongling, Beijing. Both birch and oak forest plots were 55-year-old secondary forests at the time of the first sampling, dominated by B. platyphylla and Q. wutaishanica, respectively. The temperate pine plantation was 30 years old at the time of the first sampling and was dominated by P. tabuliformis (Fang et al., 2007). Three subtropical forest plots were located in Dinghu Biosphere Reserve in Guangdong Province, southern China (Zhou et al., 2006). The subtropical evergreen broadleaved forest is an old-growth stand that is more than 400 years old, co-dominated by Castanopsis chinensis, Canarium pimela, Schima superba, and Engelhardtia roxburghiana. The subtropical pine (P. massoniana) plantation was approximately 40 years old at the time of the first sampling. The mature mixed pine and broadleaved forest was approximately 110 years old at the time of the first sampling and represented the mid-successional stages of monsoon evergreen broadleaved forest in this region. The tropical mountain rainforest plot was located at the Jianfengling National Natural Reserve, southwestern Hainan (Zhou et al., 2013). It had not been disturbed for more than 300 years and was dominated by species in the families Lauraceae and Fagaceae, such as Mallotus hookerianus, Gironniera subaequalis, Cryptocarya chinensis, Cyclobalanopsis patelliformis, and Nephelium topengii. For detailed descriptions of these eight plots, see the Supplement.
2.2 Soil sampling and calculation of SOC content
The first sampling was conducted between 1987 and 1998 in each of the eight forests (Table 1). We remeasured the same sample plots in each forest between 2008 and 2014 using identical sampling protocols.
In each forest plot, two to five pits were dug to collect soil samples for analyzing the physical and chemical properties during the two sampling periods (mostly in the 1990s during the first sampling period and in the 2010s during the second sampling period). The samples were taken at depth intervals of 10 cm down to the maximum soil depth. In brief, for the boreal forest, three soil pits were established down to the 40 cm soil depth in random locations in the growing season in 1998. In August 2014, three soil pits were again randomly excavated to the same soil depth to allow sampling for SOC content and bulk density. For the three temperate forests, two soil profiles (100 cm depth) were dug in each plot to collect soil samples at 10 cm intervals during the summer of 1992. In the summer of 2012, three soil profiles were dug, and soils were sampled from the same horizons in each soil profile (Zhu et al., 2015). The first sampling in the three subtropical forests was conducted in September 1988 in the evergreen and pine plots, and in 1987 for the mixed plot, both at the end of the rainy season and at the beginning of the dry season. Five soil pits (60 cm depth) were randomly excavated to collect samples for the calculation of SOC content and bulk density. In September 2008, the soil sampling was repeated. For the tropical forest, five soil profiles (100 cm depth) were established at 10 cm intervals during summer 1992 and again in summer 2012.
We used consistent sampling and analysis approaches to determine the bulk density and SOC content between the two sampling times. Three bulk density samples were obtained for each layer using a standard container that was 100 cm3 in volume. The soil moisture was determined by weighing to the nearest 0.1 g after 48 h oven-drying at 105 ∘C. The bulk density was calculated as the ratio of the oven-dried mass to the container volume. Another three paired samples for C analysis were air-dried. Following this, fine roots were removed by hand and sieved (2 mm mesh). The SOC content was measured using the wet oxidation method (Nelson and Sommers, 1982) and was calculated according to Eq. (1):
where CCi, Bdi, and Vi are SOC content (%), bulk density (kg m−3), and volume (m3) at the ith soil horizon, respectively. HFi is calculated as and is a dimensionless factor that represents the fine soil fraction within a certain soil volume.
2.3 Calculation of above-ground biomass (AGB) and net primary production
Diameter at breast height (DBH, 1.3 m) and height of all living trees with DBH >5 cm were both measured in each plot in the 1990s and 2010s. The AGB of different components (stem, bark, branches, and foliage) was estimated for all tree species using allometric equations (Table S1 in the Supplement). A standard factor of 0.5 was used to convert biomass to C (Leith and Whittaker, 1975). The net increment of AGB (ΔStore) was calculated for each plot as the difference between the biomass in the 1990s and the 2010s. The above-ground net primary production (ANPP, kg C ha−1 yr−1) was calculated from Eq. (2):
where Litterfall and ΔStore are litter production and above-ground net biomass increment per year, respectively. Mortality (defined as above-ground deadwood production) was estimated as the summed production of fallen logs and standing snags per year.
2.4 Litter and fallen log production
Annual litterfall was collected from June 2010 to June 2013 in the tropical sites, from June 1990 to June 2008 in the subtropical sites, from April to November 2011–2014 in the temperate sites, and from May to October 2010–2014 in the boreal sites. Litter (leaves, flowers, fruits, and woody material < 2 cm diameter) was collected monthly from 10 to 15 L traps (1×1 m2, 1 m above ground) in each plot to calculate annual litter production. After collection, the samples were taken to the laboratory, oven-dried at 65 ∘C to a constant mass and weighed. The 10–15 replicates from each plot were averaged as the monthly mean value. Annual litter production (kg C ha−1 yr−1) was estimated as the sum of the monthly production in the year of collection.
Log production represents the mortality (i.e., death of entire trees) per year. Annual log production was determined from 2010 to 2013 in tropical sites, from 1989 to 1996 in subtropical sites, from 2011 to 2014 in temperate sites, and from 2010 to 2014 in boreal sites. Stocks of fallen logs were harvested and weighed during each investigated year.
2.5 Forest area and fossil fuel emission data
To calculate the amount of C sequestration in China's forest soils, we estimated the changes in the national forest SOC stocks. We used the mean SOC accumulation rates obtained from this study and the data of forest area for each forest type documented in the national forest inventory in 1989–1993, which approximates the first sampling period in the present study (Guo et al., 2013). The changes in national forest SOC stock were calculated as the product of SOC density, SOC density change rate, and forest area for major forest types during the period 1989–1993. In addition, to evaluate the relative importance of forest soil C sequestration in the national C budget, we obtained the data of fossil fuel emissions during 1991–2010 from the Carbon Dioxide Information Analysis Center (Zheng et al., 2016).
3.1 Changes in SOC
SOC stocks were investigated in eight permanent forest plots in four forest sites from northern to southern China over two periods: the 1990s and 2010s. The changes in SOC contents, bulk density, and SOC stocks in the top 20 cm soil layer between the 1990s and the 2010s are shown in Fig. 2 and Figs. S1 and S2 in the Supplement. The paired t-test analysis indicated that SOC content in the 0–20 cm depth was significantly higher in the 2010s than in the 1990s (3.2±0.7 % vs. 2.9±0.6 %; ; P<0.001) (Table 2). The average rate of increase in SOC content was 0.02 % yr−1 in the top 20 cm depth, ranging from 0.01 % yr−1 to 0.04 % yr−1 across the study sites. These rates of increase in SOC content in the 0–10 cm horizon (0.03±0.02 % yr−1) were 3 times larger than those in the 10–20 cm horizon (0.01±0.01 % yr−1) (Table S2). At the same time, the bulk density of the top 20 cm soil layer decreased in most sites (6 of 8 sites), with an average rate of decrease of 2.7±3.7 mg cm−3 yr−1 (Table S3). As a result, the SOC stock in the top 20 cm soil layer was found to have increased significantly in the past 2 decades (, P<0.001, Table 2), with an average accumulation rate of 332.4±200.2 kg C ha−1 yr−1 (0.7±0.4 % yr−1; Fig. 2; see Table S3). The temperate pine plantation experienced the largest increase in SOC stock in the top 20 cm depth (630.8±111.2 kg C ha−1 yr−1). In contrast, the smallest rate of increase was observed in the subtropical mixed forest (117.3±25.2 kg C ha−1 yr−1). It should be noted that SOC stock in the top 20 cm depth in the subtropical evergreen old-growth forest increased from 35.6±6.0 Mg C ha−1 in 1988 to 45.6±6.9 Mg C ha−1 in 2008 (increased by 498.3±78.8 kg C ha−1 yr−1), which led to the highest relative accumulation rate (1.4±0.2 % yr−1) among the study sites.
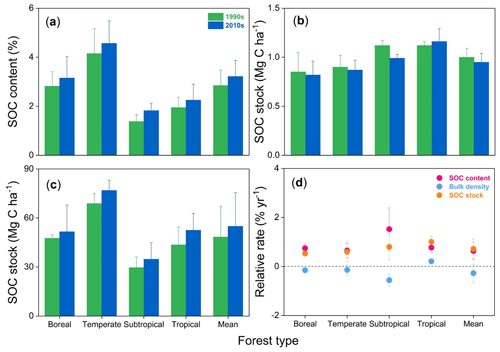
Figure 2Mean soil organic carbon (SOC) content (a), bulk density (b), SOC stock (c), and their relative change rates (d) within 0–20 cm soil depth in the 1990s and the 2010s for the four forest sites in China. For more details, see Table S2 in the Supplement.
Table 2Results of the paired-sample t tests for soil organic carbon (SOC) content, bulk density, and SOC stock at different soil depths in the eight forest plots between the 1990s and the 2010s.
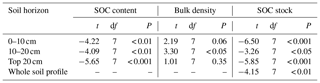
We further compared SOC stocks of the whole soil profile between 1990s and 2010s at a depth of 0–40 cm in the boreal site, 0–60 cm in the subtropical site, and 0–100 cm in the temperate and tropical sites (Fig. 3). The SOC stocks of all sampling sites in the 2010s were higher than those in the 1990s. The paired t-test analysis revealed a significant increase in SOC stocks for the whole soil profile during the sampling period (, P<0.01; Table 2). The mean SOC stocks of the whole soil profile in the eight forests increased from 125.2±85.2 Mg C ha−1 in the 1990s to 133.6±83.1 Mg C ha−1 in the 2010s, with an accumulation rate of 421.2±274.4 kg C ha−1 yr−1 and a relative increase rate of 0.6±0.5 % (Fig. 2). The SOC accumulation rates displayed large variability among different climate zones and forest types. For different climate zones, the SOC accumulation rates in the subtropical and tropical sites were relatively higher than those in the boreal and temperate sites (Fig. 3). The greatest increase in SOC stock occurred in the subtropical evergreen old-growth forest (907.5±60.1 kg C ha−1 yr−1), and the least in the temperate deciduous oak forest (127.2±25.3 kg C ha−1 yr−1; Table S3). The relative rates of increase in the subtropical evergreen old-growth forest (1.3±0.1 % yr−1) and the subtropical mixed forest (1.5±0.2 % yr−1) were higher than those in the temperate forests (0.1±0.0 % yr−1 in the oak forest, 0.1±0.0 % yr−1 in the pine forest, and 0.2±0.0 % yr−1 in the birch forest; Table S3).
In addition, the rates of SOC increase (127.2–907.5 kg C ha−1 yr−1) was equivalent to 3.6 %–16.3 % of ANPP (3340.1–6944.7 kg C ha−1 yr−1), with the highest rate in the subtropical evergreen forest (16.3±4.2 %) and the lowest in the temperate oak forest (3.6±3.4 %) (Tables 3 and S4).
3.2 Relationships between SOC change rates and biotic and climatic variables
To understand the possible mechanisms for the rates of SOC increase as described above, we analyzed the driving forces for this significantly increased SOC stock using measurements of AGB growth rate, above-ground litter and fallen log production, and ANPP (Table 3). The linear regression analysis showed that there was no significant correlation between SOC change rates and AGB growth rate (P>0.05; Fig. 4a). The SOC accumulation rates were positively and significantly associated with annual litter (R2=0.66; P=0.01; Fig. 4b) and fallen log production (R2=0.69; P=0.01; Fig. 4c). The SOC accumulation rates across these forests were closely associated with the observed ANPP (R2=0.55; P=0.03; Fig. 4d) and also showed an increasing trend with increasing mean annual temperature and precipitation, despite being insignificant (both P>0.1; Fig. 4e and f). The multiple regression analysis indicated the relative effects of biotic factors (AGB growth rate, litter, and fallen log production) and climatic factors (mean annual temperature and precipitation) on the rates of SOC increase (Fig. 4g). When the effects of climatic factors were under control, the biotic factors independently explained 56.4 % of the variations. By comparison, when the effects of biotic factors were under control, only 7.5 % of the variations were explained by the climatic factors.
4.1 SOC accumulation
Previous evidence of forest SOC changes comes mainly from individual experiments (Prietzel et al., 2006; Kiser et al., 2009; Häkkinen et al., 2011) or regional comparisons (Lettens et al., 2005; Pan et al., 2011; Ortiz et al., 2013) in European and American forests. In this study, we performed a broadscale forest soil resampling to evaluate changes in SOC stock across eight permanent forest plots in China. Our measurements suggest that SOC stocks exhibited a significant accumulation in these forests from the 1990s to the 2010s, at the accumulation rate of 127.2–907.5 kg C ha−1 yr−1. These accumulation rates are comparable to those of other studies that were primarily conducted in boreal and temperate forests in other regions (−11.0–812.0 kg C ha−1 yr−1, Fig. 5). In detail, the rate of SOC accumulation of the boreal forest in the present study was estimated as 243.4 kg C ha−1 yr−1, which was within the range of boreal forests in European and American forests (115.6–740.0 kg C ha−1 yr−1) (Prietzel et al., 2006; Dölle and Schmidt, 2009; Häkkinen et al., 2011; Wang et al., 2011; Rantakari et al., 2012; Chapman et al., 2013; Schrumpf et al., 2014). The rates of SOC accumulation in the three temperate forests ranged from 127.2 to 390.8 kg C ha−1 yr−1, comparable to the regional comparison data of 200.0 kg C ha−1 yr−1 in the temperate forests of China (Yang et al., 2014). Evidence from soil inventory-based studies of SOC dynamics also demonstrated that soil of boreal and temperate forests in European countries is likely to accumulate C (Berg et al., 2009; Nielsen et al., 2012; Tefs and Gleixner, 2012; Grüneberg et al., 2014). The mean rate of SOC accumulation in the humus layers of boreal forests in Sweden was estimated to be 251.0 kg C ha−1 yr−1 during the period 1961–2002 (Berg et al., 2009). Nielsen et al. (2012) assessed the rates of SOC change in Denmark's broadleaved deciduous and coniferous forests using two soil inventories conducted during 1990 and 2005. The estimated rates of SOC change in the broadleaved and coniferous forests were 90.0 and 310.0 kg C ha−1 yr−1, respectively. Two soil inventories provided data for analysis of the mineral soils of forests in Germany, which were found to have sequestrated 410.0 kg C ha−1 yr−1 during the period of 1987–2008 (Grüneberg et al., 2014). Therefore, evidence from long-term observations and from the repeated soil sampling in individual studies and in national soil inventory reports, suggests that soils of boreal and temperate forests in the Northern Hemisphere have functioned as C sinks during past decades.
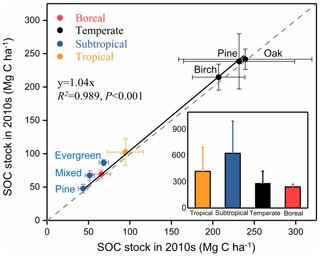
Figure 3Comparison of soil organic carbon (SOC) stocks in eight forest plots in China between the 1990s and the 2010s. The SOC stocks in all forests during the two periods are above the 1:1 line, suggesting that all these forests have increased their SOC stock during the study period. The inset graph shows the SOC sink rates by forest biome (i.e., boreal, temperate, subtropical, and tropical forests), which are categorized from the eight forest plots. SOC stocks and change rates are presented as means ±1 SD. For details, see Fig. 1 and Tables 1 and S1.
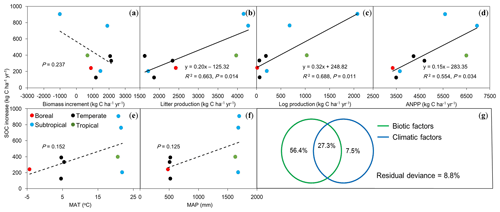
Figure 4Relationships between rates of increase in soil organic carbon (SOC) against biotic and climatic factors in eight forests in China. (a) Biomass increment, (b) litter production, (c) log production, (d) above-ground net primary production (ANPP), (e) mean annual temperature (MAT), (f) mean annual precipitation (MAP), and (g) the relative effects of biotic (a, b, c) and climatic (e, f) factors on SOC increase rates (kg C ha−1 yr−1) using partial regression analyses. Solid lines indicate significant relationships (P<0.05), and dashed lines represent insignificant trends (P>0.05) between SOC increase rates and biotic and climatic factors.
In other subtropical and tropical forest ecosystems, direct evidence of SOC dynamics is relatively scarce (Tang and Li, 2013). However, based on the estimates from regional comparisons, Pan et al. (2011) showed that global tropical forests were a source of 1.4 Pg C ha−1 yr−1 from 1990 to 2007. At the global scale, tropical land-use changes have caused a sharp drop in forest area, which also led to a large release of C from tropical forest soils. Without land-use change and deforestation, soils in subtropical and tropical forests have functioned as a considerable C sink during the past 2 decades in this study (627.6±370.1 and 397.9±84.2 kg C ha−1 yr−1, respectively; Table 3). Limited forest management (e.g., litter and deadwood harvest), as well as catastrophic land-use changes, can result in the loss of C from forest soil. Prietzel et al. (2016) reported a large loss of SOC in forests in the German Alps, where half of the woody biomass and deadwood had been harvested over recent decades. On the one hand, harvesting the forest floor can decrease litter and deadwood inputs into soils and subsequently lead to the loss of soil C (Davidson and Janssens, 2006). On the other hand, a decrease in the amount of the forest floor may lead to an increase in soil erosion, especially in mountain forests (Evans et al., 2013). Additionally, high-elevation ecosystems are expected to be more sensitive to warming than other regions, with associated changes in soil freezing and thawing events and in snow cover, which may be another reason for the SOC losses in forests in the German Alps.
4.2 Links between biotic and climatic factors and in SOC accumulation
The forest biomass of China has functioned as a significant C sink over recent decades (Pan et al., 2011; Fang et al., 2014, 2018). The increase in C accumulation by vegetation supplied more C inputs into soils, including inputs of litter, woody debris, and root exudates, and resulted in SOC accumulation (Zhu et al., 2017). However, the rate of SOC change did not increase with the rate of biomass change in this study (Table S4). We found that soil in the subtropical old-growth forest increased at the highest sink rate of 907.5±60.1 kg C ha−1 yr−1 but that vegetation functioned as a significant C source ( kg C ha−1 yr−1). This was because the relatively higher annual litterfall and fallen log production occurred in the old-growth forest, which subsequently resulted in soil C accumulation (Fig. 4). The positive (but not significant) trend between climatic factors and SOC dynamics may largely be induced by the internal correlations between climatic and biotic factors (Fig. 4).
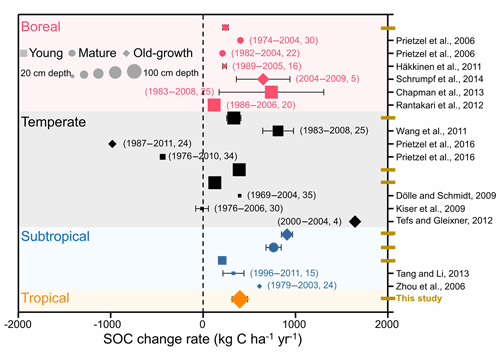
Figure 5Comparison of the changes in forest soil organic carbon (SOC) stocks according to repeated soil samplings and/or long-term observation. Different colors, shapes, and sizes represent different forest biomes, ages, and soil depths, respectively. The numbers in parentheses indicate the sampling times and intervals between the two soil samplings.
The heterotrophic respiration of global forest soil has increased significantly over past decades (Bond-Lamberty et al., 2018), suggesting that the increment in the rate of soil C input outweighs that of the rate of soil C output. The increasing heterotrophic respiration of forest soil is mainly due to ongoing climate change, and especially to increasing temperature. The increment in forest growth rate is due to increasing temperature, together with increasing CO2 and nitrogen fertilization (Norby et al., 2010; Feng et al., 2019). Thus, the sensitivity of forest net primary production to ongoing climate change should outweigh that of respiration. We also found that SOC stock increased from 68.4 to 86.6 Mg C ha−1, albeit the biomass C stock decreased significantly from 1988 to 2008 in the subtropical old-growth plot. The greatest amount of litter and deadwood production and standing crop occurred in the old-growth plot, which resulted in relatively higher soil C sequestration in the old-growth plot compared to other plots (Fig. 4, Table S4). Biotic factors explained the variation in SOC dynamics better than climatic factors. In this study, we did not, however, measure root-derived C inputs to SOC, although below-ground production also makes a significant contribution to SOC accumulation (Nadelhoffer and Raich, 1992; Majdi, 2001; Pausch and Kuzyakov, 2018). Above-ground inputs are mineralized from litter and deadwood, and below-ground inputs may benefit from interactions with soils (Rasse et al., 2005). Even if the effect of climatic factors were controlled and below-ground biotic factors were not included in the analysis, the above-ground biotic factors would explain 56.4 % of the variation in the rate of SOC accumulation.
4.3 Regional carbon budget
The rate of SOC accumulation (421.2±274.4 kg C ha−1 yr−1, Fig. 2 and Table S3) is more than one-half of the vegetation C uptake rate in China's forests (702.0 kg C ha−1 yr−1) (Guo et al., 2013; Fang et al., 2018). This result suggests that China's forest soils have contributed to a negative feedback to climate warming during the past 2 decades, rather than the positive feedback predicted by coupled C–climate models (Cox et al., 2000; He et al., 2016; Wang et al., 2018).
If we roughly use the inventory-based forest area of 138.8 Mha in China (Guo et al., 2013) and extend the current SOC sink rates obtained in this study to all the forests in the country, China's forest soils have sequestered approximately 1.1±0.5 Pg C during the past 2 decades (57.1±26.5 Tg C yr−1). This C accumulation would be equivalent to 2.4 %–6.8 % of the country's fossil CO2 emissions during the contemporary period (1991–2010) (Zheng et al., 2016). By comparing forest SOC data obtained from published literature during the 2000s and a national soil inventory during the 1980s, Yang et al. (2014) estimated significant C accumulation in the forest soils of China. Although they did not estimate the national C budget of these forest soils, we can calculate the national C sequestration rate of forest soil as 67.2 Tg C yr−1, based on the C sequestration rates and forest areas of the different forest types in their study. Our results further confirm the assessment, based on repeated measurements at eight permanent forest plots, that soils in China's forests have functioned as a C sink for atmospheric CO2 during the past 2 decades.
According to previous estimates, the C sinks of three C sectors: forest vegetation biomass (Fang et al., 2014), deadwood, and litter (Zhu et al., 2017) during the past 2 decades were 70.9, 3.9, and 2.8 Tg C yr−1, respectively (Table S5). If these previous estimates are incorporated into the soil C accumulation rate of 57.1±26.5 Tg C yr−1 in the current study, then China's forests may have sequestered a total of 134.7 Tg C per year between the 1990s and the 2010s. This is equivalent to 14.5 % of the contemporary fossil CO2 emissions in the country (Zheng et al., 2016). According to the estimate of Pan et al. (2011), the C sink rate of forests in the temperate regions of the Northern Hemisphere was 647.1 Tg C yr−1. The C sequestration of China's forests represents 20.8 % of the total temperate regions. The sequestration rate of China's forests is slightly higher than the mean value of the total temperate regions, relative to the forest area of China (i.e., 18.9 % of the forest areas in the temperate regions). This result indicates that the role of forest soils in the regional C cycle cannot be ignored, although a large uncertainty about the national C budget of forest soils remains in our estimates.
4.4 Uncertainty analysis
We investigated the SOC stocks in eight permanent plots across four forest biomes in China. These plots spanned a long-term timescale (approximately 20 years) and a broad spatial scale (approximately 34∘ of latitude). We also measured several C fluxes (i.e., biomass change rate, production of litterfall and deadwood) that were relevant to the rate of SOC change. Even so, the following three factors may introduce uncertainties related to the estimation of SOC dynamics.
First, the sampling times and intervals between SOC investigations were different across the sites. The first sampling was performed from 1987 to 1998 and the second was carried out from 2008 to 2014. As a result, the sampling interval ranged from 16 years in the boreal forest plot to 21 years in the subtropical mixed forest plot (Table 1). Nonuniform sampling times and intervals may lead to uncertainties in relation to SOC stocks across the forest plots.
Second, the depth of soil varied substantially, ranging from 40 cm in the boreal site to 100 cm in the temperate and tropical sites. In addition, different numbers (2–5) of soil profiles were dug in different plots during the first sampling period. To ensure consistency between the two sampling times, the same number of soil profiles were dug in similar locations to perform SOC stock investigations during the second sampling period. We performed continuous observation of litterfall and deadwood production, but the observation times and durations varied across the plots. Variability in these items may reduce the comparability of SOC dynamics among plots.
Finally, the rates of SOC change in our study and in inventory-based forest areas and forest types were used to estimate the C budget of forest soil in China. However, only eight permanent forest plots were observed in this study, and this will inevitably lead to uncertainty with respect to national estimations.
The SOC stocks within the top 20 cm increased by 2.4–12.6 Mg C ha−1 across the forests during the past 2 decades, with an annual accumulation rate of 332.4±200.2 kg C ha−1. If all soil horizon profiles were included, the soils may have been found to have sequestered 3.6 %–16.3 % of the annual net primary production across the investigated sites, and the averaged accumulated rate (421.2 kg C ha−1 yr−1) may have been more than one-half of the vegetation C uptake rate (702.0 kg C ha−1 yr−1) in China's forests. These results demonstrate that these forest soils have functioned as an important C sink over recent decades, although the phenomenon may not occur uniformly in forests worldwide. Forest soils store large amounts of C and accumulate it steadily (and often slowly) but will release it rapidly to the atmosphere once they are disturbed.
Allometric equations of above-ground biomass and the data for soil bulk density, SOC content, stock, and their change rates of the eight permanent plots are listed as in the Supplement. The remaining data that support the findings of this study are available from the corresponding author upon request.
The supplement related to this article is available online at: https://doi.org/10.5194/bg-17-715-2020-supplement.
JF designed the research. JZ and JF designed the data analysis. JZ, JF, ZZ, LJ, XH, HY, GL, CW, and GZ performed SOC measurements. JF, YL, CJ, and GL designed sampling and analytical programs and performed data quality control. JZ, JF, CW, SZ, PL, JZ, ZT, CZ, RAB, and YP contributed to the writing of the manuscript.
The authors declare that they have no conflict of interest.
We thank Qiong Cai and Tianli Zheng for their assistance in the preparation of the manuscript.
This research has been supported by the National Key Research and Development Program of China (grant no. 2017YFC0503906), the National Natural Science Foundation of China (grant nos. 31700374 and 31621091), and the International Programs, US Forest Service (grant no. 07-JV-11242300-117).
This paper was edited by Yakov Kuzyakov and reviewed by three anonymous referees.
Berg, B., Johansson, M. B., Nilsson, Å., Gundersen, P., and Norell, L.: Sequestration of carbon in the humus layer of Swedish forests – direct measurements, Can. J. Forest Res., 39, 962–975, https://doi.org/10.1139/X09-022, 2009.
Bond-Lamberty, B., Bailey, V. L., Chen, M., Gough, C. M., and Vargas, R.: Globally rising soil heterotrophic respiration over recent decades, Nature, 560, 80–83, https://doi.org/10.1038/s41586-018-0358-x, 2018.
Canadell, J. G. and Schulze, E. D.: Global potential of biospheric carbon management for climate mitigation, Nat. Commun., 5, 1–12, https://doi.org/10.1038/ncomms6282, 2014.
Chapman, S. J., Bell, J. S., Campbell, C. D., Hudson, G., Lilly, A., Nolan, A. J., Robertson, A. H. J., Potts, J. M., and Towers, W.: Comparison of soil carbon stocks in Scottish soils between 1978 and 2009, Eur. J. Soil Sci., 64, 455–465, https://doi.org/10.1111/ejss.12041, 2013.
Chen, L., Smith, P., and Yang, Y.: How has soil carbon stock changed over recent decades?, Glob. Change Biol., 21, 3197–3199, https://doi.org/10.1111/gcb.12992, 2015.
Cox, P. M., Betts, R. A., Jones, C. D., Spall, S. A., and Totterdell, I. J.: Acceleration of global warming due to carbon-cycle feedbacks in a coupled climate model, Nature, 408, 184–187, https://doi.org/10.1038/35041539, 2000.
Davidson, E. A. and Janssens, I. A.: Temperature sensitivity of soil carbon decomposition and feedbacks to climate change, Nature, 440, 165–173, https://doi.org/10.1038/nature04514, 2006.
Dölle, M. and Schmidt, W.: Impact of tree species on nutrient and light availability: evidence from a permanent plot study of old-field succession, Plant Ecol., 203, 273–287, https://doi.org/10.1007/s11258-008-9547-2, 2009.
Evans, A. M., Perschel, R. T., and Kittler, B. A.: Overview of forest biomass harvesting guidelines, J. Sustain. Forest., 32, 89–107, https://doi.org/10.1080/10549811.2011.651786, 2013,.
Fang, J., Shen, Z., Tang, Z., Wang, X., Wang, Z., Feng, J., Liu, Y., Qiao, X., Wu, X., and Zheng, C.: Forest community survey and the structural characteristics of forests in China, Ecography, 35, 1059–1071, https://doi.org/10.1111/j.1600-0587.2013.00161.x, 2012.
Fang, J., Guo, Z., Hu, H., Kato, T., Muraoka, H., and Son, Y.: Forest biomass carbon sinks in East Asia, with special reference to the relative contributions of forest expansion and forest growth, Glob. Change Biol., 20, 2019–2030, https://doi.org/10.1111/gcb.12512, 2014.
Fang, J., Yu, G., Liu, L., Hu, S., and Chapin III, F. S.: Climate change, human impacts, and carbon sequestration in China, P. Natl. Acad. Sci. USA, 115, 4015–4020, https://doi.org/10.1073/pnas.1700304115, 2018.
Fang, J. Y., Liu, G. H., Zhu, B., Wang, X. K., and Liu, S. B.: Carbon budgets of three temperate forest ecosystems in Dongling Mt., Beijing, China, Sci. China Earth Sci., 50, 92–101, https://doi.org/10.1007/s11430-007-2031-3, 2007.
Feng, Y., Zhu, J., Zhao, X., Tang, Z., Zhu, J., and Fang, J.: Changes in the trends of vegetation net primary productivity in China between 1982 and 2015, Environ. Res. Lett., 14, 124009, https://doi.org/10.1088/1748-9326/ab4cd8, 2019.
Grüneberg, E., Ziche, D., and Wellbrock, N.: Organic carbon stocks and sequestration rates of forest soils in Germany, Glob. Change Biol., 20, 2644–2662, https://doi.org/10.1111/gcb.12558, 2014.
Guo, Z. D., Hu, H. F., Li, P., Li, N. Y., and Fang, J. Y.: Spatio-temporal changes in biomass carbon sinks in China's forests from 1977 to 2008, Sci. China Life Sci., 56, 661–671, https://doi.org/10.1007/s11427-013-4492-2, 2013.
Häkkinen, M., Heikkinen, J., and Mäkipää, R.: Soil carbon stock increases in the organic layer of boreal middle-aged stands, Biogeosciences, 8, 1279–1289, https://doi.org/10.5194/bg-8-1279-2011, 2011.
He, Y., Trumbore, S. E., Torn, M. S., Harden, J. W., Vaughn, L. J., Allison, S. D., and Randerson, J. T.: Radiocarbon constraints imply reduced carbon uptake by soils during the 21st century, Science, 353, 1419–1424, https://doi.org/10.1126/science.aad4273, 2016.
IPCC: Summary for policymakers, in: Climate Change 2013: The Physical Science Basis, edited by: Stocker, T. F., Qin, D., Plattner, G. K., Tignor, M., Allen, S. K., Boschung, J., Nauels, Xia, Y., Bex, V., and Midgley, P. M., Contribution of Working Group I to the Fifth Assessment, Report of the Intergovernmental Panel on Climate Change, Cambridge, CUP, 1–30, 2013.
Kiser, L. C., Kelly, J. M., and Mays, P. A.: Changes in forest soil carbon and nitrogen after a thirty-year interval, Soil Sci. Soc. Am. J., 73, 647–653, https://doi.org/10.2136/sssaj2008.0102, 2009.
Lal, R.: Soil carbon sequestration impacts on global climate change and food security, Science, 304, 1623–1627, https://doi.org/10.1126/science.1097396, 2004.
Leith, H. and Whittaker, R. H.: Primary productivity of the biosphere: ecological studies, Berlin, Springer, 237–263, https://doi.org/10.1007/978-3-642-80913-2, 1975.
Lettens, S., Van Orshoven, J., van Wesemael, B., De Vosc, B., and Muysa, B.: Stocks and fluxes of soil organic carbon for landscape units in Belgium derived from heterogeneous data sets for 1990 and 2000, Geoderma, 127, 11–23, https://doi.org/10.1016/j.geoderma.2004.11.001, 2005.
Luo, Y., Melillo, J., Niu, S., Beier, C., Clark, J. S., Classen, A. T., Davidson, E., Dukes, J. S., Evans, R. D., Field, C. B., Czimczik, C. I., Keller, M., Kimball, B. A., Kueppers, L. M., Norby, R. J., Pelini, S. L., Pendall, E., Rastetter, E., Six, J., Smith, M., Tjoelker, M. G., and Torn, M. S.: Coordinated approaches to quantify long-term ecosystem dynamics in response to global change, Glob. Change Biol., 17, 843–854, https://doi.org/10.1111/j.1365-2486.2010.02265.x, 2011.
Majdi, H.: Changes in fine root production and longevity in relation to water and nutrient availability in a Norway spruce stand in northern Sweden, Tree Physiol., 21, 1057–1061, https://doi.org/10.1023/A:1011905124393, 2001.
Nadelhoffer, K. J. and Raich, J. W.: Fine root production estimates and belowground carbon allocation in forest ecosystems, Ecology, 73, 1139–1147, https://doi.org/10.2307/1940664, 1992.
Nelson, D. W. and Sommers, L. E.: Total carbon, organic carbon, and organic matter, chap. 29, in: Methods of Soil Analysis, Part 2. Chemical and Microbiological Properties, 2nd Edn., edited by: Sparks, A. L., American Society of Agronomy, Inc, Soil Science Society of Agronomy, Inc., 539–579, https://doi.org/10.2136/sssabookser5.3.c34, 1982.
Nielsen, O. K., Mikkelsen, M. H., Hoff mann, L., Gyldenkærne, S., Winther, M., Nielsen, M., Fauser, P., Thomsen, M., Plejdrup, M. S., Albrektsen, R., Hjelgaard, K., Bruun, H. G., Johannsen, V. K., Nord-Larsen, T., Bastrup-Birk, A., Vesterdal, L., Møller, I. S., Rasmussen, E., Arfaoui, K., Baunbæk, L., and Hansen, M. G.: Denmark's National Inventory Report 2012, Emission Inventories 1990–2010 – Submitted under the United Nations Framework Convention on Climate Change and the Kyoto Protocol. Scientific Report from DCE–Danish Centre for Environment and Energy, 19, http://www.risoe.dtu.dk/rispubl/NEI/NEI-DK-5700.pdf (last access: 3 August 2012); OSTI as DE01047219, 2012.
Norby, R. J. and Zak, D. R.: Ecological lessons from Free-Air CO2 Enrichment (FACE) experiments, Annu. Rev. Ecol. Evol. S., 42, 181–203, https://doi.org/10.1146/annurev-ecolsys-102209-144647, 2011.
Norby, R. J., Warren, J. M., Iversen, C. M., Medlyn, B. E., and McMurtrie, R. E.: CO2 enhancement of forest productivity constrained by limited nitrogen availability, P. Natl. Acad. Sci. USA, 107, 19368–19373, https://doi.org/10.1073/pnas.1006463107, 2010.
Ortiz, C. A., Liski, J., Gärdenäs, A. I., Lehtonen, A., Lundblad, M., Stendahl, J., Ågren, G. I., and Karltun, E.: Soil organic carbon stock changes in Swedish forest soils – a comparison of uncertainties and their sources through a national inventory and two simulation models, Ecol. Model., 251, 221–231, https://doi.org/10.1016/j.ecolmodel.2012.12.017, 2013.
Pan, Y., Birdsey, R. A., Fang, J. Houghton, R., Kauppi, P. E., Kurz, W. A., Phillips, O. L., Shvidenko, A., Lewis, S. L., Canadell, J. G., Ciais, P., Jackson, R. B., Pacala, S. W., McGuire, A. D., Piao, S., Rautiainen, A., Sitch, S., and Hayes, D.: A large and persistent carbon sink in the world's forests, Science, 333, 988–993, https://doi.org/10.1126/science.1201609, 2011.
Pausch, J. and Kuzyakov, Y.: Carbon input by roots into the soil: quantification of rhizodeposition from root to ecosystem scale, Glob. Change Biol., 24, 1–12, https://doi.org/10.1111/gcb.13850, 2018.
Piao, S., Fang, J., Ciais, P., Peylin, P., Huang, Y., Sitch, S., and Wang, T.: The carbon balance of terrestrial ecosystems in China, Nature, 458, 1009, https://doi.org/10.1038/nature07944, 2009.
Prietzel, J., Stetter, U., Klemmt, H. J., and Rehfuess, K. E.: Recent carbon and nitrogen accumulation and acidification in soils of two Scots pine ecosystems in Southern Germany, Plant Soil, 289, 153–170, https://doi.org/10.1007/s11104-006-9120-5, 2006.
Prietzel, J., Zimmermann, L., Schubert, A., and Christophel, D.: Organic matter losses in German Alps forest soils since the 1970s most likely caused by warming, Nat. Geosci., 9, 543–548, https://doi.org/10.1038/ngeo2732, 2016.
Rantakari, M., Lehtonen, A., Linkosalo, T., Tuomi, M., Tamminen, P., Heikkinen, J., Liski, J., Mäkipää, R., Ilvesniemi, H., and Sievänen, R.: The Yasso07 soil carbon model – Testing against repeated soil carbon inventory, Forest Ecol. Manag., 286, 137–147, https://doi.org/10.1016/j.foreco.2012.08.041, 2012.
Rasse, D. P., Rumpel, C., and Dignac, M. F.: Is soil carbon mostly root carbon? Mechanisms for a specific stabilisation, Plant Soil, 269, 341–356, https://doi.org/10.1007/s11104-004-0907-y, 2005.
Schrumpf, M., Schulze, E. D., Kaiser, K., and Schumacher, J.: How accurately can soil organic carbon stocks and stock changes be quantified by soil inventories?, Biogeosciences, 8, 1193–1212, https://doi.org/10.5194/bg-8-1193-2011, 2011.
Schrumpf, M., Kaiser, K., and Schulze, E. D.: Soil organic carbon and total nitrogen gains in an old growth deciduous forest in Germany, PLoS ONE, 9, e89364, https://doi.org/10.1371/journal.pone.0089364, 2014.
Sitch, S., Friedlingstein, P., Gruber, N., Jones, S. D., Murray-Tortarolo, G., Ahlström, A., Doney, S. C., Graven, H., Heinze, C., Huntingford, C., Levis, S., Levy, P. E., Lomas, M., Poulter, B., Viovy, N., Zaehle, S., Zeng, N., Arneth, A., Bonan, G., Bopp, L., Canadell, J. G., Chevallier, F., Ciais, P., Ellis, R., Gloor, M., Peylin, P., Piao, S. L., Le Quéré, C., Smith, B., Zhu, Z., and Myneni, R.: Recent trends and drivers of regional sources and sinks of carbon dioxide, Biogeosciences, 12, 653–679, https://doi.org/10.5194/bg-12-653-2015, 2015.
Smith, P.: How long before a change in soil organic carbon can be detected?, Glob. Change Biol., 10, 1878–1883, https://doi.org/10.1111/j.1365-2486.2004.00854.x, 2004.
Tang, G. and Li, K.: Tree species controls on soil carbon sequestration and carbon stability following 20 years of afforestation in a valley-type savanna, Forest Ecol. Manag., 291, 13–19, https://doi.org/10.1016/j.foreco.2012.12.001, 2013.
Tang, X., Zhao, X., Bai, Y., Tang, Z., Wang, W., Zhao, Y., Wan, H., Xie, Z., Shi, X., Wu, B., Wang, G., Yan, J., Ma, K., Du, S., Li, S., Han, S., Ma, Y., Hu, H., He, N., Yang, Y., Han, W., He, H., Yu, G., Fang, J., and Zhou, G.: Carbon pools in China's terrestrial ecosystems: New estimates based on an intensive field survey, P. Natl. Acad. Sci. USA, 115, 4021–4026, https://doi.org/10.1073/pnas.1700291115, 2018.
Tefs, C. and Gleixner, G.: Importance of root derived carbon for soil organic matter storage in a temperate old-growth beech forest – Evidence from C, N and 14C content, Forest Ecol. Manag., 263, 131–137, https://doi.org/10.1016/j.foreco.2011.09.010, 2012.
Todd-Brown, K. E., Randerson, J. T., Post, W. M., Hoffman, F. M., Tarnocai, C., Schuur, E. A. G., and Allison, S. D.: Causes of variation in soil carbon simulations from CMIP5 Earth system models and comparison with observations, Biogeosciences, 10, 1717–1736, https://doi.org/10.5194/bg-10-1717-2013, 2013.
Wang, C., Gower, S. T., Wang, Y., Zhao, H., Yan, P., and Bond-Lamberty, B. P.: The influence of fire on carbon distribution and net primary production of boreal Larix gmelinii forests in north-eastern China, Glob. Change Biol., 7, 719–730, https://doi.org/10.1046/j.1354-1013.2001.00441.x, 2001.
Wang, W., Qiu, L., Zu, Y., Su, D., An, J., Wang, H., Zheng, G., Sun, W., and Chen, X.: Changes in soil organic carbon, nitrogen, pH and bulk density with the development of larch (Larix gmelinii) plantations in China, Glob. Change Biol., 17, 2657–2676, https://doi.org/10.1111/j.1365-2486.2011.02447.x, 2011.
Wang, X., Ciais, P., Wang, Y., and Zhu, D.: Divergent response of seasonally dry tropical vegetation to climatic variations in dry and wet seasons, Glob. Change Biol., 24, 4709–4717, https://doi.org/10.1111/gcb.14335, 2018.
Yang, Y., Li, P., Ding, J., Zhao, X., Ma, W., Ji, C., and Fang, J.: Increased topsoil carbon stock across China's forests, Glob. Change Biol., 20, 2687–2696, https://doi.org/10.1111/gcb.12536, 2014.
Zhao, X., Yang, Y., Shen, H., Geng, X., and Fang, J.: Global soil–climate–biome diagram: linking surface soil properties to climate and biota, Biogeosciences, 16, 2857–2871, https://doi.org/10.5194/bg-16-2857-2019, 2019.
Zheng, T. L., Zhu, J. L., Wang, S. P., and Fang, J. Y.: When will China achieve its carbon emission peak?, Natl. Sci. Rev., 3, 8–15, https://doi.org/10.1093/nsr/nwv079, 2016.
Zhou, G., Liu, S., Li, Z., Zhang, D., Tang, X., Zhou, C., Yan, J., and Mo, J.: Old-growth forests can accumulate carbon in soils, Science, 314, 1417, https://doi.org/10.1126/science.1130168, 2006.
Zhou, Z., Jiang, L., Du, E., Hu, H., Li, Y., Chen, D., and Fang, J.: Temperature and substrate availability regulate soil respiration in the tropical mountain rainforests, Hainan Island, China. J. Plant Ecol., 6, 325–334, https://doi.org/10.1093/jpe/rtt034, 2013.
Zhu, J., Hu, H., Tao, S., Chi, X., Li, P., Jiang, L., Ji, C., Zhu, J., Tang, Z., Pan, Y., Birdsey, R. A., He, X., and Fang, J.: Carbon stocks and changes of dead organic matter in China's forests, Nat. Comm., 8, 1–10, https://doi.org/10.1038/s41467-017-00207-1, 2017.
Zhu, J. X., Hu, X. Y., Yao, H., Liu, G. H., Ji, C. J., and Fang, J. Y.: A significant carbon sink in temperate forests in Beijing: based on 20-year field measurements in three stands, Sci. China Life Sci., 58, 1135–1141, https://doi.org/10.1007/s11427-015-4935-z, 2015.