the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Late Quaternary palaeoenvironmental evolution and sea level oscillation of Santa Catarina Island (southern Brazil)
Karin A. F. Zonneveld
Paulo A. Souza
Rodrigo R. Cancelli
Sea level oscillation during the Quaternary played a major role in the geomorphology and vegetation dynamics of coastal areas in southern Brazil, encompassing ecosystems that often have a unique biodiversity. Understanding the natural evolution of these areas is essential for decision making regarding land use regulations towards sustainable development, as well as to preserve the uniqueness of the coastal ecosystems. The southern Brazil coastal plain is formed by marine, transitional and continental Quaternary deposits controlled by past variations of the sea level. These variations shaped the coastal landscape and influenced the development of different Atlantic rainforest formations, such as mangroves and restingas. In particular, the restinga formation corresponds to a specific ecosystem that covers sandy soils of marine and fluvial–marine origin formed during the Quaternary on the Brazilian coastal plain. In this contribution, we present high-resolution palynological and stable isotope data from a Holocene core retrieved from the coastal plain of Santa Catarina Island (southern Brazil). We were able to identify four different environmental zones for the last 6520 yr BP. The first zone (6520–2920 cal yr BP) is characterized by a lagoon with large marine-water influence. Notably, the observed dinoflagellate cyst association suggests that marine waters entering the region had their origin in the relatively warm and saline Brazil Current waters. During the second zone (2920–1520 cal yr BP), marine-water contribution to the lagoon decreased until it became disconnected from the sea. The third zone (1520–550 cal yr BP) was marked by the decrease of the water level until it dried out and led to the colonization of herbaceous vegetation over the palaeo-lagoon. The last zone (550 cal yr BP–recent) is characterized by the consolidation of the coastal-plain Atlantic rainforest (restinga vegetation). Our results form an example of the strong sensitivity of southern Brazilian ecosystem change caused by relative sea level variations. As such, this study might contribute to the debate about the potential effects of current climate change induced by global sea level variations.
- Article
(10225 KB) - Full-text XML
- BibTeX
- EndNote
The comprehension of past environmental changes in the Quaternary is valuable for understanding modern and future environmental dynamics. This is particularly important in coastal areas where land–ocean interactions play a major role in the geomorphology and vegetation dynamics. This highly dynamic environment is often reshaped by anthropogenic activities including the removal of vegetation, land use and hydrological changes. Such activities threaten the ecological and economical sustainability of the coastal areas that often have a unique biodiversity (Ramesh et al., 2015; Newton et al., 2016). The study of the natural evolution of these areas in terms of landscape and vegetation dynamics is essential to understand and preserve the uniqueness of these coastal ecosystems and to support land use regulations towards sustainable development of coastal areas.
Brazilian coastal areas and their ecosystems are under strong anthropogenic pressure. In such areas, the dominant biome is the Atlantic rainforest, a global biodiversity hotspot recognized as one of the most important ecosystems of the earth (UNESCO, 2011). In particular, the restinga vegetation corresponds to a specific ecosystem within the Atlantic rainforest that covers sandy soils of marine and fluvial–marine origin formed during the Quaternary on the Brazilian coastal plain (Scarano, 2002; Magnago et al., 2010). The southern Brazil coastal plain is formed by marine, transitional and continental Quaternary deposits, controlled by the relative variations of sea level that directly influenced the development of different Atlantic rainforest formations, such as restingas and mangroves (IBGE, 2012). A characteristic region for this system can be found in the south of Santa Catarina Island. This area hosts preserved fragments of the Atlantic rainforest located near the coastline. It forms a particular region that allows the study of the interaction between sea level changes and vegetation dynamics.
The study of the past dynamics of coastal areas can be achieved by means of several scientific tools, such as sedimentological (e.g. Dillenburg et al., 2006; Zazo et al., 2013), archeological (Martin et al., 1986), isotopic (e.g. Martin et al., 1986; Carr et al., 2015) and palaeontological (e.g. Angulo et al., 1999; Chemello and Silenzi, 2011; Toniolo et al., 2020), including palynological studies (e.g. Borromei and Quattrocchio, 2007; Leroy et al., 2013). Notably, the palynological records throughout sediment cores typically provide information on terrestrial and marine settings encompassing the environmental and vegetation changes within the same core (e.g. Mourelle et al., 2015; Kuhn et al., 2017). Additionally, the variations in terms of the relative abundances of marine and continental palynomorphs are commonly used to determine sea level oscillations (e.g. van Soelen et al., 2010; Candel and Borromei, 2016).
Previous palynological studies were conducted in the southernmost portion of the southern Brazil coastal plain (i.e. Rio Grande do Sul coastal plain – see summaries in Lorscheitter, 2003; Bauermann et al., 2009; Mourelle et al., 2018). However, the Santa Catarina coastal-plain sector is geomorphologically distinct, and similar studies are scarce, located only in the continental portion (Behling, 1995; Behling and Negrelle, 2001; Amaral et al., 2012; Cancelli, 2012; Kuhn et al., 2017; França et al., 2019; Val-Péon et al., 2019; Cohen et al., 2020; Silva et al., 2021) and at the continental slope in the western South Atlantic (ca. 200 km north of Santa Catarina Island; Gu et al., 2017). In general, previous studies indicate a sequence of marine-influenced environments followed or not by lagoons and succeeded by terrestrialization. Nevertheless, most of the studies focused on the pollen record, and the characterization of the marine-influenced and transitional environments were less explored.
This contribution provides the first high-resolution multi-proxy pollen, dinoflagellate cyst and isotopic study in Santa Catarina Island. We aim to reconstruct a detailed environmental evolution of the southern part of Santa Catarina Island (Fig. 1) throughout the Holocene with the following goals: (i) to understand the Atlantic rainforest dynamics and consolidation in Santa Catarina Island, (ii) to identify the effects of Holocene sea level variations in the coastal landscape and vegetation evolution, and (iii) to compare with previous palynological studies regarding the environmental evolution of the southern Brazil coastal plain.
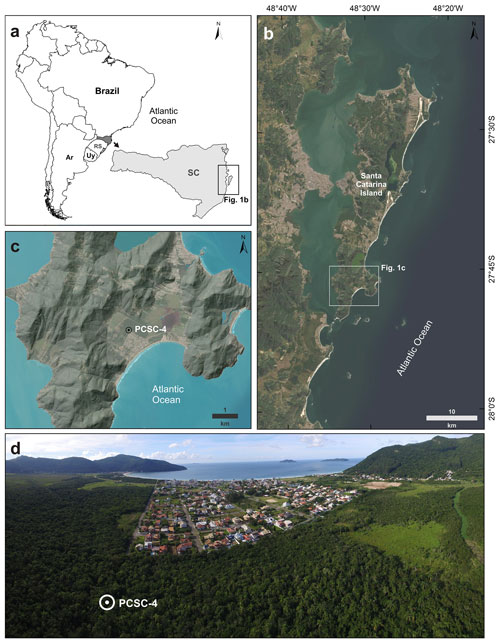
Figure 1Location and images of the study area. (a) Location of Santa Catarina Island in southern Brazil (SC: Santa Catarina state; RS: Rio Grande do Sul state; Uy: Uruguay; Ar: Argentina). (b) Santa Catarina Island and location of the Pântano do Sul beach. (c) Shaded relief model of the Pântano do Sul beach and PCSC-4 core location (basemap imagery source (b) and (c): ESRI (Esri, Maxar, Earthstar Geographics, and the GIS User Community, 2020); shaded relief basemap source (c): INPE, 2020). (d) Panoramic aerial photo of the sampling location; note the preserved Atlantic rainforest in the sampling area and the urban development.
Santa Catarina Island is located in the Santa Catarina sector of the southern Brazil coastal plain. The physiographic and structural aspects of the island are similar to the continental region, since they were united when the sea level was below the current level (Horn Filho, 2006). The topography is dominated by granitic coastal mountains with altitudes of up to 532 m and the coastal plain, which consists of Pleistocene and Holocene marine, beach, aeolian, lagoonal and paludal environment deposits (Horn Filho, 2006). The formation of the latter deposit is associated with transgressive and regressive events regarding the relative sea level that occurred during the Quaternary (Caruso Jr., 1993).
Santa Catarina Island is situated in a subtropical zone, and the climate is characterized as humid oceanic without a dry season and with hot summers (Cfa, according to Koppen's classification; Alvares et al., 2013). The Cfa climate type comprises a maximum average temperature of more than 22 ∘C and a minimum average between −3 and 18 ∘C, and rainfall is well distributed along the year, with annual accumulated precipitation of 1766 mm in Santa Catarina Island (Alvares et al., 2013; INMET, 2022). The region is influenced by the South Atlantic tropical anticyclone and the polar migratory anticyclone. The South Atlantic anticyclone produces the Atlantic tropical air mass, a warm and humid mass that is active throughout the year, while the polar migratory anticyclone generates the Atlantic polar air mass, which is characterized by low temperatures and high humidity. The migration of the polar migratory anticyclone to the region generates the polar front that is characterized by unstable weather and an increase of the precipitation (Nimer, 1990).
The Atlantic rainforest covers a large portion of southern Brazil and the entire Santa Catarina coastal plain. This biome encompasses different forest formations and associated ecosystems. In the highlands (Serra Geral plateau), it is characterized as a mosaic of Araucaria forest and grasslands, while in the coastal plain, there is dense arboreal vegetation and some pioneer formations such as the restinga, mangroves and salt marshes. These pioneer vegetations are conditioned by edaphic factors and are composed of plants that have adapted to the ecological parameters of first-occupation character (Oliveira Filho and Fontes, 2000; Scarano, 2002; Magnago et al., 2010; IBGE, 2012).
Santa Catarina Island is bordered by the South Atlantic Ocean. Surface currents in the region are dominated by the southward-flowing Brazil Current (BC; Fig. 2), which is relatively warm and saline (Peterson and Stramma, 1991). The BC originates at about 10∘ S from the bifurcation of the westward-flowing South Equatorial Current (Silveira et al., 2000; Souza and Robison, 2004). The BC contacts the northward-flowing Malvinas Current (MC), which is characterized by cold and low-salinity waters that have their origin in the Antarctic Circumpolar Current. At the contact zone, the so-called Brazil–Malvinas Confluence (BMC), MC waters dive under the BC waters. The mixed water masses are transported successively eastward as part of the South Atlantic Current (Piola and Matano, 2019). Along the coast, an additional northward-flowing water mass can be observed, the Brazilian Coastal Current (BCC). This last current consists of low-salinity water discharged from the Rio de La Plata and Patos Lagoon that, on its way north, mixes with the other water masses (Souza and Robinson, 2004). The positions of the maximal northward extension of the BCC, as well as of the BMC, vary strongly between the seasons (Piola et al., 2000; Piola, 2005). During austral summer, the BMC reaches its southernmost position. The maximal northern extension of the BCC, as well as of the colder MC waters, can be observed near Santa Catarina Island in austral winter (e.g. https://podaac-tools.jpl.nasa.gov/soto/, last access: 5 May 2023, “state of the ocean, temperature”).
Below surface waters, to a depth of approximately 600 m, southward-flowing South Atlantic Central Water can be observed, which overlies the cold and less-saline Antarctic Intermediate Water (Piola and Matano, 2001).
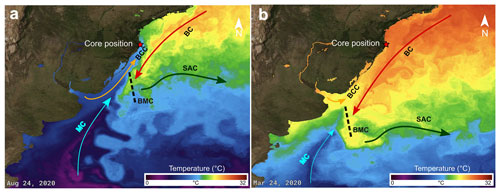
Figure 2Present configuration of sea surface currents and temperature in the western South Atlantic Ocean in the (a) winter and (b) summer (source: JPL MUR MEaSUREs Project, 2015; basemap satellite imagery source: NASA Earth Observatory Blue Marble 2005). MC: Malvinas Current; BMC: Brazil–Malvinas Confluence; BC: Brazil Current; BCC: Brazilian Coastal Current; SAC: South Atlantic Current.
3.1 Sediment core collection
This study was carried out on a sedimentary core (PCSC-4) retrieved from Pântano do Sul beach (27∘46′36.49′′ S, 48∘31′45.96′′ W; Fig. 1), located at the southernmost part of Santa Catarina Island. The core was drilled using a Russian peat corer on a peat deposit, reaching a maximum depth of 650 cm. The sampling site is located approximately 1 km from the current coastline and ca. 1 m above the present sea level. The 50 cm long sediment sections were sealed and transported to the Laboratório de Palinologia Marleni Marques Toigo at the Federal University of Rio Grande do Sul.
3.2 Radiocarbon dating
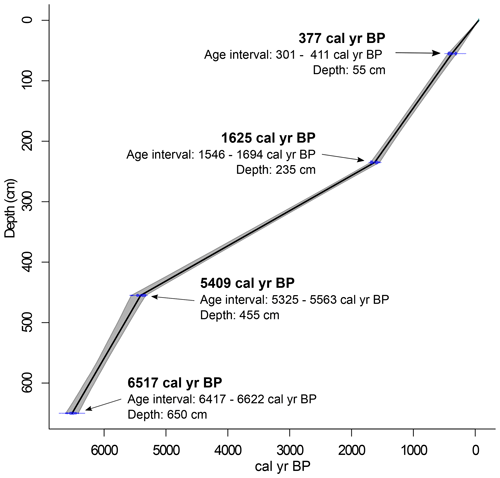
Four bulk organic-rich sediment samples were selected along the core and analysed with the accelerator mass spectrometry (AMS) at the Center for Applied Isotope Studies (CAIS) Laboratory of the University of Georgia (USA) for radiocarbon dating. Sample selection was made after the palynological analyses that aimed at obtaining ages for significant changes in the palynological record. The radiocarbon dates were calibrated using the Southern Hemisphere calibration curves (SHCal20; Hogg et al., 2020), rounded to the nearest decade and reported as calendar years before present (cal yr BP). The age–depth model was constructed with the software RStudio (RStudio Team, 2021), using the Clam 2.4.0 package (Blaauw, 2010) with linear interpolation (Fig. 3).
3.3 Granulometric analyses
Grain size analyses and calculation of organic matter content were made for 64 samples with 10 cm intervals along the core. The samples were equally separated into two sub-samples to determine the grain size analyses and the organic matter content in the sediment. These analyses, as well as the calculations of the statistical parameters, were carried out at the Center for Studies of Oceanic and Coastal Geology (CECO) at the Federal University of Rio Grande do Sul. All the samples were dried in an oven at 40 ∘C and were then weighted. The analyses were performed by a sieving–pipetting method, following the statistical parameters of Folk and Ward (1957) and the textural classification of Shepard (1954). To determine the organic matter content, the samples were calcined in muffle at 550 ∘C over 4 h and were weighed before and after the calcination. The organic matter content of the sediments was determined by the loss of ignition after this process.
3.4 Elemental C and N and 13C isotopes
A total of 33 samples with 3 cm3 of sediment were collected at 20 cm intervals from the top to the bottom of the core (0–640 cm) added to the basal sample (650 cm) for total organic carbon (TOC), total nitrogen (TN) and δ13C analyses. At first, the samples were dried in an oven at 60 ∘C and weighted. Samples were treated with 10 % HCl to eliminate carbonate and were then washed with Milli-Q water until the pH reached 5. Samples were dried in a freeze-dryer, weighted again and then homogenized to be analysed in the elemental analyser coupled to isotope ratio mass spectrometry (EA-IRMS) in the Hinrichs Laboratory at the University of Bremen. TOC and TN values are expressed as a percentage of dry weight, and δ13C is expressed in delta-per-mil notation, with an accuracy of ±0.17 ‰ with respect to the VPDB standard. The (weight weight) was calculated using the elemental results ratio.
3.5 Palynological analysis
A total of 66 samples of 3 cm3 each were obtained throughout the core for pollen and spore analyses, with 10 cm spacing between them. After a preliminary taxonomic recognition, samples where dinoflagellate cysts were recorded (650–310 cm) were resampled for more detailed analyses, totalling 35 samples with the same spacing and bulk volume of sample (3 cm3). One Lycopodium clavatum spore tablet (18.584 ± 371 spores) was added to each sample before the chemical processing of both pollen and spore samples and dinoflagellate samples to allow concentration calculations (Stockmarr, 1971).
The pollen and spore samples were processed following standard preparation techniques (Faegri et al., 1989), using HF (40 %), HCL (10 %), KOH (10 %) and acetolysis. To concentrate the material, samples were sieved using a <250 µm sieve; ZnCl2 was used for heavy-liquid separation, checking the residues to be sure that no material was lost in the separation. Slides were prepared from drops of the final residue, mounted with Entellan.
The dinoflagellate cyst samples were prepared using similar procedures. However, to avoid damage to the cysts, the samples were not prepared using hot acids, KOH and acetolysis. The dinoflagellate cyst samples were decalcified with diluted HCl (10 %) and treated with HF (40 %) to remove silicates. After chemical treatments, the samples were sieved over a 20 µm mesh screen, and residues were transferred to an Eppendorf vial, where the material was concentrated in 1 mL portions. Slides were mounted with glycerin jelly for microscopic analysis.
Pollen and spore samples were counted until reaching a minimum of 300 pollen grains monitored by saturation curves. The other palynomorphs (i.e. spores, algae, acritarchs and microforaminiferal linings) and L. clavatum spores were counted in parallel. Concentrations (palynomorphs cm−3) were calculated using the L. clavatum spores as reference values.
Dinoflagellate cyst samples were counted until reaching their saturation curves. The total dinoflagellate sum adds all counted dinoflagellate cysts, and the relative abundances of each taxa in the dinoflagellate analyses are indicated as a percentage of the total dinoflagellate sum. Concentrations (dinoflagellate cysts cm−3) were calculated using the L. clavatum spores as reference values.
To integrate the dinoflagellate cysts and pollen and spore counts, we used the ratio of L. clavatum counts from the pollen and spore sample and the corresponding dinoflagellate cyst samples as a conversion factor. The dinoflagellate cyst counts were multiplied by this ratio and added to the final integrated diagram.
The total sum represents the sum of all palynomorphs (including dinoflagellate cysts), whereas the pollen sum refers to the total amount of pollen grains. The relative abundances of pollen grains were calculated as a percentage of the pollen sum, whereas the relative abundances of the other palynomorphs were calculated in relation to the total sum.
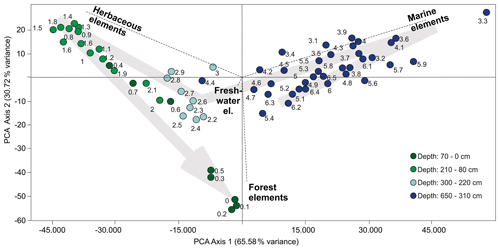
The environmental zones were established from changes in the palynomorph assemblages and from cluster analysis based on percentage values of the total sum. The depth-constrained cluster analysis (CONISS) was performed using the Edwards & Cavalli-Sforza's chord distance square-root transformation. Cluster analyses, percentage and concentration diagrams were constructed using the Tilia version 1.7.16 (Grimm, 2011). For the principal component analyses (PCA), we used the software Canoco (Šmilauer and Lepš, 2014) and PAST 4.03 (Hammer et al., 2001). Multivariate analyses were performed on palynological relative-abundance data.
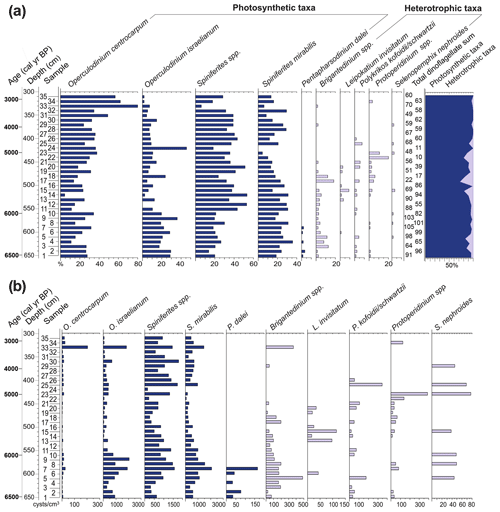
The taxonomic determinations of the pollen and spores were based on comparison with modern equivalents in palynological reference collections (MP-Pr slides of the LPMMT/IGeo/UFRGS) and from the literature (e.g. Hooghiemstra, 1984; Neves and Lorscheitter, 1992; Herrera and Urrego, 1996; Lorscheitter et al., 1998; Colinvaux et al., 1999; Macedo et al., 2009; Cancelli et al., 2012). Dinoflagellate cysts were identified following the online key for dinoflagellate cyst determinations (Zonneveld and Pospelova, 2015, and references therein). Dinoflagellate cysts were grouped according to their life strategies, photosynthetic taxa (Operculodinium centrocarpum, O. israelianum, Spiniferites spp., Spiniferites mirabilis and Pentapharsodinium dalei) and heterotrophic taxa (Brigantedinium spp., Leipokatium invisitatum, Polykrikos kofoidii, P. schwartzii, Protoperidinium spp. and Selenopemphix nephroides).
4.1 Radiocarbon dating
The radiocarbon dating results are presented in Table 1, including uncalibrated and calibrated ages obtained from four selected samples. Calibrated ages indicate that the deposition of the studied core occurred entirely during the middle-to-late-Holocene interval, where the lowermost level (650 cm) has an age of 6520 cal yr BP, whereas the uppermost level (55 cm depth) revealed an age of 380 cal yr BP. The remaining samples presented intermediate ages.
4.2 Granulometric analysis
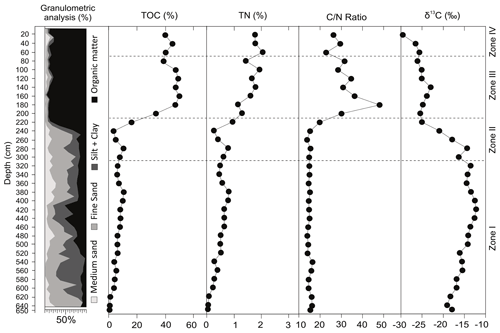
The core consists of unconsolidated sediments composed of medium sand, fine sand, silt and clay added to variable amount of organic matter (Fig. 4). The organic matter is dominant (>80 %) from 220 cm to the top of the core. Regarding the distribution of clastic sediments, in general, there is a mixture of silt, clay and fine to medium sand from the base up to 220 cm (see distribution in Fig. 3). Localized calcareous shells in living position and shell fragments occur, scattered from the base until 400 cm of depth.
4.3 Elemental C and N and 13C isotopes
The geochemical data are presented as individual profiles along the studied core (Fig. 4) and as the binary plot of δ13C × (Fig. 5). The total organic carbon (TOC) concentration varies from 0.5 % to 49.6 % and shows two main intervals separated by a gradual transition between them. The interval from the base of the core up to 240 cm shows an average value of 5.8 %, whereas between 220–200 cm, the average is 24.5 %. Samples from 180 cm to the top present an average TOC value of 44.6 %. The total nitrogen (TN) ranges from a minimum of 0.04 % at 650 cm depth to 2.1 % at 60 cm depth. ratios (weight weight) show nearly constant values of ca. 15 from the base up to 240 cm, followed by an abrupt increase from 240 to 180 cm (15.2–47.8) and a subsequent subtle decrease from 180 cm to the top (47.8–26.2; Fig. 4). The δ13C results are in the range of −12.2 ‰ to −29.6 ‰. The δ13C values are higher at the base of the sediment core (650–240 cm depth), with a range of −12.2 ‰ to −20.9 ‰, followed by a downward trend towards the top of the core.
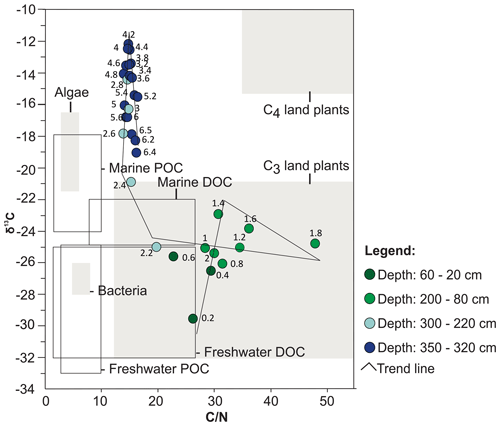
Figure 5Binary diagram illustrating the relationship between δ13C and (according to Meyers, 1994, and Wilson et al., 2005) and the results obtained from each interval of depth of the PCSC-4 core, south of Santa Catarina Island, southern Brazil. DOC: dissolved organic carbon; POC: particulate organic carbon.
4.4 Palynological record
A total of 114 distinct palynomorphs were identified along the 650 cm of the studied core, including pollen grains (59 taxa), spores (16), freshwater algae (4), marine algae (1), acritarchs (3), dinoflagellate cysts (10), indeterminate spores (8) and indeterminate pollen grains (13), as well as, fungi, microforaminiferal linings, scolecodonts and copepod eggs.
The palynological diagrams show the distribution of palynomorphs in the samples, grouped according to their ecological affinities (habit or habitat; Figs. 6–9). Both visual examination and the cluster and PCA analyses (Fig. 10) show that the samples can be grouped into four zones with characteristic species associations. For simplification purposes, the results will be presented according to the four zones discussed in Sect. 5 in ascending stratigraphic order.
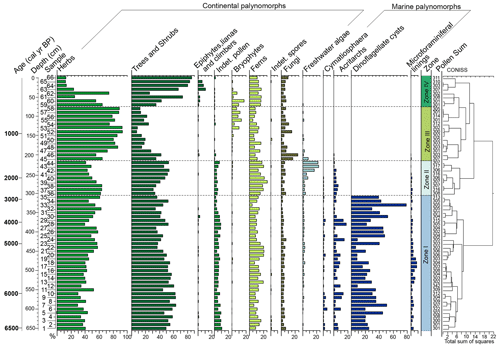
Figure 6Percentage diagram of the palynomorphs grouped according to their ecological affinities (habit or habitat) from the PCSC-4 core, south of Santa Catarina Island, southern Brazil, as well as the identified zones and cluster analyses.
4.4.1 Zone I (650–310 cm, samples 1–35)
This zone was recognized in the basal part of the core. It is characterized by high contents of marine palynomorphs (12 %–79 %), represented mainly by dinoflagellate cysts (8 %–78 %). Additionally, the other marine palynomorphs, i.e. microforaminiferal linings (<8 %), acritarchs (<17 %) and the prasinophyte Cymatiosphaera (<5 %), have the highest concentrations at this zone. The dinoflagellate cysts include both photosynthetic (Operculodinium centrocarpum, O. israelianum, Spiniferites spp., S. mirabilis and P. dalei) and heterotrophic taxa (Brigantedinium spp. Leipokatium invisitatum, Polykrikos kofoidii, P. schwarzii, Protoperidinium spp. and Selopemphix nephoides; Fig. 9).
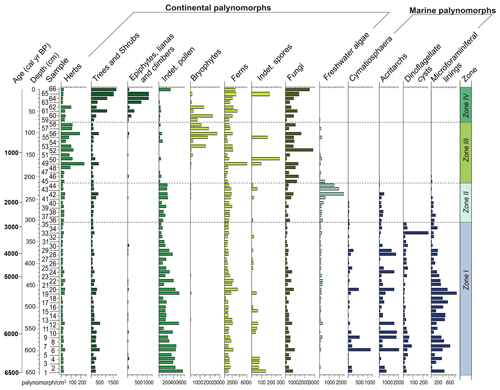
Figure 7Concentration diagram of the palynomorphs grouped according to their ecological affinities (habit or habitat) from the PCSC-4 core, south of Santa Catarina Island, southern Brazil, as well as the identified zones.
The acritarch association is composed of species of the genus Micrhystridium, specifically Acritarch sp. 1 and Acritarch sp. 2. Freshwater algae are observed in low percentages through this zone and are mainly formed by Botryococcus spp., followed by Spirogyra, Pseudoschizaea rubina and Zygnema.
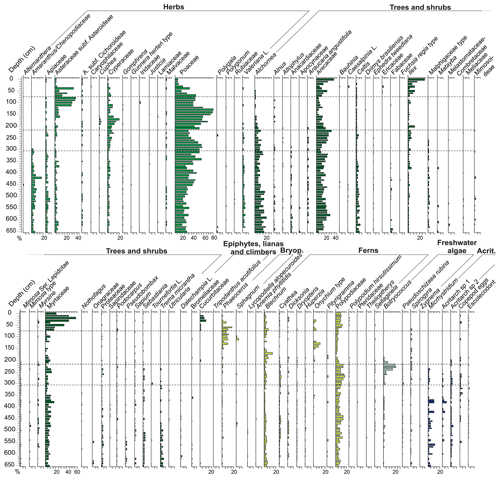
Figure 8Relative-abundance diagram of the palynomorph taxa according to their ecological affinities (habit or habitat) from the PCSC-4 core, south of Santa Catarina Island, southern Brazil, as well as the identified zones.
Terrestrial palynomorphs are dominated by pollen of trees and shrubs (34 %–63 %), followed by herbs (28 %–60 %), ferns (4 %–21 %) and indeterminate pollen grains (2 %–11 %). Arecaceae (4 %–26 %), Alchornea (4 %–20 %) and Myrtaceae (2 %–16 %) dominated the tree and shrub pollen, whereas Poaceae (15 %–55%), Amaranthus and Chenopodiaceae (1 %–17 %), Asteraceae (1 %–9 %) and Apiaceae (1 %–7 %) dominated the herb pollen. Ferns are most represented by Polypodiaceae (3 %–13 %) and Blechnum (1 %–5 %). Epiphytes, lianas and climbers, bryophytes, indeterminate spores, and fungi are scarce.
4.4.2 Zone II (300 to 220 cm, samples 36–44)
This zone is characterized by a drastic reduction of marine palynomorphs with respect to the previous zone (e.g. microforaminiferal linings, acritarchs and Cymatiosphaera) and the disappearance of dinoflagellate cysts. Freshwater algae assemblage remains nearly unchanged; however, Botryococcus increases its percentages throughout this zone (1 %–22 %). The terrestrial palynomorphs are dominated by herbs (41 %–65 %), followed by trees and shrubs (31 %–54 %). Ferns have a relative increase (13 %–25 %), and indeterminate pollen grains have a relative decrease (2 %–5 %) with respect to Zone I. Epiphytes, lianas and climbers, bryophytes, indeterminate spores, fungi, and algae maintain relatively lower abundances.
4.4.3 Zone III (210 to 80 cm, samples 45–58)
This zone is marked by the reduction of freshwater algae (<8 %) and the disappearance of Botryococcus, Pseudoschizaea rubina and Zygnema. The marine palynomorphs were not recorded. Fungi increase their percentages (2 %–24 %), as do herb pollen grains (55 %–92 %). The latter are represented mainly by Poaceae, Asteraceae subf. Asteroideae and Cyperaceae. Additionally, bryophyte spores are mainly represented by species of Phaeoceros, which occur significantly for the first time in the core, reaching up to 13 %. Trees and shrubs decrease their relative abundances (8 %–42 %), as do the indeterminate pollen grains (<2 %). Similarly to the previous zone, epiphytes, lianas and climbers, bryophytes, and indeterminate spores have low relative abundances.
4.4.4 Zone IV (70 to 0 cm, samples 59–66)
This zone comprises the upper portion of the core. It is characterized by an increase in the pollen sum of arboreal taxa (34 %–86 %) and of epiphytes, lianas and climbers (0.3 %–10 %). Herb pollen taxa decrease their relative abundances (12 %–73 %), as do bryophyte taxa (<17 %). The arboreal assemblage is mainly represented by Myrtaceae, Arecaceae and Ilex; epiphytes, lianas and climbers are represented by Cucurbitaceae. Pteridophytes show an increase of their percentages and are represented mainly by Blechnum and Polypodiaceae taxa. Indeterminate pollen grains and indeterminate spores maintain low relative abundances.
5.1 Zone I: lagoonal stage (6520–2920 cal yr BP)
High percentages of marine palynomorphs, including dinoflagellate cysts (8 %–78 %), microforaminiferal linings (<8 %), acritarchs (<17 %) and Cymatiosphaera (<5 %), are evidence of sea waters reaching the sampling site. Microforaminiferal linings are abundant in estuarine marshes, with variable salinity water influence (Batten, 1996). Species of the acritarch genus Micrhystridium are characteristic of shallow coastal water associations (Montenari and Leppig, 2003; Félix and Souza, 2012). The prasinophyte Cymatiosphaera, on the other hand, is known to be typically associated with marine water (Mudie et al., 2021).
The dinoflagellate cysts include photosynthetic (Operculodinium centrocarpum, O. israelianum, Spiniferites spp., S. mirabilis and P. dalei) and heterotrophic taxa (Brigantedinium spp., Leipokatium invisitatum, Polykrikos kofoidii, P. schwarzii, Protoperidinium spp. and Selenopemphix nephroides; Fig. 9). All the above-mentioned dinoflagellate cysts are commonly registered in the South Atlantic Ocean, as well as in coastal sites (Zonneveld et al., 2013). The presence of heterotrophic taxa in the association suggests high nutrient inputs in the waters. The photosynthetic association is dominated by Operculodinium centrocarpum and Spiniferites spp. On a global scale, the species Operculodinium centrocarpum has a cosmopolitan distribution (e.g. Zonneveld et al., 2013). However, in the western South Atlantic, this species is typically present in the relatively warm waters of the BC (Gu et al., 2019). Throughout this zone, specimens of Operculodinium israelianum and Spiniferites mirabilis are registered, which are typical for warm, temperate waters (Zonneveld et al., 2013). The dinoflagellate cyst association therefore indicates that marine waters entering into the study site had their origin in the relatively warm, saline BC waters.
Even though, nowadays, waters of the BCC and MC can seasonally reach the coast at the same latitude as that of the core position, we did not observe any evidence that this has been the case in Zone I (Fig. 2). Species that are characteristically abundant in MC and BCC waters, such as Selenopemphix antarctica and/or Impagidinium variaseptum (Zonneveld et al., 2013; Gu et al., 2019), were not observed in the studied material. The presence of a warm, temperate dinoflagellate cyst association, characteristic for high nutrient concentrations, implies that the lagoon waters were relatively warm and nutrient rich in this zone.
The presence of freshwater algae indicates freshwater influence despite the significant marine contribution. In addition, Botryococcus is a euryhaline freshwater algae that may have its photosynthetic activity inhibited, directly or indirectly, by the water salinity (Tyson, 1995). Also, Batten and Grenfell (1996) cite Botryococcus occurrence in calm or stagnant waters, such as lakes, swamps or marshes, although it can withstand relatively higher salinity from other environments such as mangroves and estuaries. Therefore, the low concentrations of this freshwater algae in this zone might be caused by the presence of marine influence. The observed palynomorph association suggests that calm conditions and mixing of marine and freshwater prevailed, which is typical for a lagoon environment.
Pollen grains (herbs, trees and shrubs) were transported by streams and/or wind and were deposited in the lagoon in low concentrations (Fig. 7). The herb taxa are mostly represented by Poaceae, Amaranthus and Chenopodiaceae, Apiaceae, Asteraceae subf. Asteroideae, and Cyperaceae (Fig. 8). These herbs are better adapted to sandy soils and include many halophytes (Lorscheitter, 2003). This suggests that saline soil conditions prevailed in the margins of the lagoon.
In this zone, the pollen of the tree and shrub taxa are mainly composed of Alchornea, Arecaceae, Myrtaceae and Celtis. The presence of Celtis and Trema, pioneer taxa, suggests an unstable environment subject to ecological succession. These pioneer taxa, together with the tree and shrub taxa (Alchornea, Myrtaceae and Arecaceae) and the halophytes herbs taxa (e.g. Amaranthus and Chenopodiaceae), indicate seasonally flooded open restinga in the surrounding area, with sandy soil subject to salinity (Freitas and Carvalho, 2012). Indeterminate pollen grains occur at higher concentrations in this zone and the subsequent Zone II in comparison to the two upper zones. This can be explained by the input of pollen grains transported by streams and wind into the lagoon partly damaging some of the grains. Alternatively, the presence of indeterminate pollen in this phase may be related to the transport of organic particles and the granulometry of sediments (fine sands and silts) in the very redeposition of lacustrine–lagoonal sediments configuring a high-energy and oxygenated environment that is more greatly influenced by marine currents than by the continental (terrestrial biomass) contribution. However, the predominance of fine sand, silt and clay sediment, as well as the presence of preserved calcareous shells in living position, could indicate deposition in a predominantly calm water body. Therefore, we interpret that the water body was likely calm with sporadic higher-energy events (e.g. storms). The pollen grains that originated from the Andes (Nothofagus and Alnus) are expected to be transported over long distances by air dispersion to the deposition site. These palynomorphs have been previously found in many Quaternary sedimentary profiles from the southern Brazil coastal plain (e.g. Cancelli et al., 2012; Diniz and Medeanic, 2012; Masetto and Lorscheitter, 2016; Kuhn et al., 2017; Silva et al., 2021).
The combination of high δ13C values (−19 to −12) and low (14–16) ratios indicates mixtures of terrestrial and marine palynomorphs and shows little variation throughout this zone (Figs. 4, 5). Low ratios are attributed to the presence of nitrogen-enriched freshwater algae organic matter (Meyers, 1994, 1997; Wilson et al., 2005). Among terrestrial plants, higher δ13C values indicate the predominance of C4 plants over C3 plants. This can be explained by the deposition of herb remnants from the borders of the lagoonal body. TOC values do not exceed 10 %. Such low TOC values are typical of lagoonal and estuarine environment (Tyson, 1995; Lorente et al., 2014).
5.2 Zone II: regressive stage and sea disconnection (2920–1520 cal yr BP)
In this zone, the predominance of fine sediments and the occurrence of freshwater algae and marine palynomorphs still indicates the presence of a calm brackish water body (Fig. 11; Zone II). However, the significant reduction of the marine indicators (e.g. microforaminiferal linings, acritarchs and Cymatiosphaera) and the disappearance of dinoflagellate cysts point to a progressive reduction of sea water input into the water body. Towards the end of this zone (ca. 1520 cal yr BP), the lagoon was probably disconnected from the sea (Fig. 11; Zone II). This is supported by the observation of the increase in the abundance of Botryococcus towards the top of the zone, suggesting that freshwater inputs into the lagoon progressively decreased its salinity, transferring the lagoon into a freshwater lake. Previous studies suggest that an increase of Botryococcus concentration might be related to a decrease of the water level in a lake (Tyson, 1995). It is therefore likely that, during the process of the closing of the lagoon and the freshening of the waters, the water table dropped consistently.
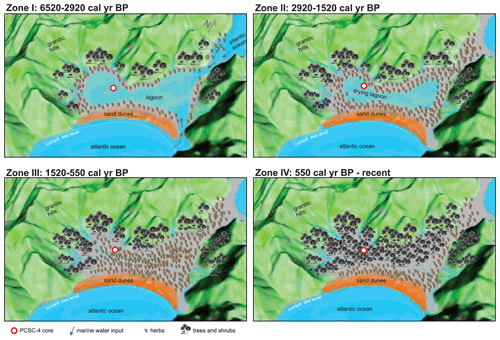
Figure 11Schematic model of the environmental evolution of the southernmost portion of the south of Santa Catarina Island (Pântano do Sul), southern Brazil.
No significant changes were observed in the spore–pollen assemblage. However, the decrease of Amaranthus and Chenopodiaceae throughout this zone suggests a progressive desalination of the soil of the adjacent areas of the lagoonal body. The ratios remain at low values of ca. 15 % throughout this zone, indicating that freshwater algae are still major contributors to the total organic matter. The binary plot of δ13C × shows a trend towards lower δ13C values, likely related to the increase of freshwater phytoplankton input.
5.3 Zone III: early development of the restinga forest (coastal plain Atlantic rainforest; 1520–550 cal yr BP)
The transition from Zone II to III is marked by a drastic decline of freshwater algae concentrations and the disappearance of Botryococcus, Pseudoschizaea rubina and Zygnema. This reduction suggests a reduced freshwater input into the area. Moreover, marine palynomorphs are no longer recorded in this zone because of the disconnection with the sea that occurred in the previous zone.
The development of soils rich in organic matter at the site is evidenced by the increase of fungi. The abundance of fungal fragments is indicative of aerobic biodegradation of plant remains (Sebag et al., 2006). Additionally, the increase of the organic matter in the sediment and the high values of TOC (average of ca. 44 %) indicate the development of soils rich in organic matter in this zone.
High percentages of Poaceae and Cyperaceae taxa and δ13C enrichment from the beginning of this zone up to 140 cm of depth can be observed in this zone (Figs. 4, 8). In particular, Cyperaceae is an emergent aquatic plant common in restinga vegetation in lagoon environments, marshes and swampy lowlands (Falkenberg, 1999). This suggests that the herbs that previously occupied the margins of the lagoon advanced and colonized the palaeo-lagoon area and the environment of ongoing humid soil conditions. The concentration values for trees and shrubs remain constant throughout this zone, suggesting that the input of arboreal pollens that were transported from adjacent areas covered by Atlantic rainforest did not change (Fig. 11; Zone III). Consequently, the observed decrease of the relative abundances of trees and shrubs associated with increasing herbs supports our previous suggestion of a significant increase in the development of herbs in the palaeo-lagoon. The strong increase in the ratios (Fig. 4) in this zone indicates an input of carbon-enriched material and suggests the increase of dense vegetation in the adjacent areas. The ratios greater than 20 originated from vascular land plants (Meyers, 1994), and δ13C values around −25 ‰ indicate an influence of C3 plants (Meyers, 1997). The binary plot of δ13C × also shows that the organic matter was influenced by C3 plants (Fig. 5). In the upper part of the section related to this zone, Poaceae and Cyperaceae (herbs) decreased their relative abundances, whereas Myrtaceae and Arecaceae (trees and shrubs) increase, showing a succession from herbal to arboreal taxa of the restinga forest.
5.4 Zone IV: restinga forest (coastal plain Atlantic rainforest; 550 cal yr BP–present)
This zone is characterized by a decrease in the relative abundances of herbs in favour of trees and shrubs. This suggests that forests developed in the palaeo-lagoon. The high relative abundances of arboreal taxa typical of the Atlantic rainforest (Arecaceae, Ilex and Myrtaceae) marks the consolidation of this arboreal forest in the area during this zone. In addition, the increase of epiphytes, lianas and climbers (mainly Cucurbitaceae taxa) suggest the advance of arboreal components in the vegetational succession of the forest as well (IBGE, 2012; Sevegnani and Schroeder, 2013).
Pteridophytes that are typically associated with the arboreal restinga forest (Blechnum and Polypodiaceae; Falkenberg, 1999) increase their concentration and support the consolidation of the arboreal restinga forest in the region. The isotope data indicate a depletion in the δ13C values during this zone, reaching the lowest value in the uppermost sample (−30). These δ13C values reveal the dominance of C3 plants, which is also indicated in the pollen record. In addition, the high TOC values and abundant organic matter contents in the sediment (∼ 40 and ∼ 90 %, respectively) indicate the occurrence of a dense forest similar to the one that covers the area nowadays.
5.5 Environmental evolution of southern Brazil coastal plain and regional implications
The observed transition from a lagoonal environment at ca. 6520 cal yr BP to the actual restinga forest in the Pântano do Sul area is interpreted to be directly related to sea level changes throughout the Holocene. In general, there is an observed sequence of decreasing marine water contribution that indicates that the relative sea level was higher than the current sea level in the first zone, successively decreasing during the second zone. Because of the disconnection of the sea and the lagoonal body evidenced by the absence of marine palynomorphs in Zone III and Zone IV, we interpret that the sea level reached the current level at some point during one of these zones, most likely in Zone III. Previous studies on the Brazilian coastal plain suggested the existence of high-frequency oscillations in the relative sea level with two regressive zones during the late Holocene (Suguio et al., 1985; Martin et al., 2003). The authors suggested that the sea level was slightly below its present elevation at ca. 4200–3700 and 2700–2100 yr BP. However, recent studies suggest a regular decline in the relative sea level without significant oscillations during the late Holocene (Angulo et al., 1999, 2006, 2022; Ybert et al., 2003). In particular, Angulo et al. (2006, 2022) suggested that the highstand in the Holocene occurred between 5000 and 5800 yr BP without a distinct peak. The phase succession observed in our study can be better explained by a regular decline without significant oscillations, as proposed by Angulo et al. (2006, 2022; Fig. 12). Moreover, other palynological studies in the southern Brazil coastal plain in the Rio Grande do Sul (e.g. Cordeiro and Lorscheitter, 1994; Lorscheitter and Dillenburg, 1998; Meyer et al., 2005; Masetto and Lorscheitter, 2019) and Santa Catarina states (Behling and Negrelle, 2001; Amaral et al., 2012; Cancelli, 2012; Kuhn et al., 2017; Val-Peón et al., 2019; Cohen et al., 2020; Silva et al., 2021) also identified the marine influence ca. 6000–5000 yr BP at their sites and showed a similar sea level dynamic. In addition, palynological studies of palaeoenvironmental reconstitutions performed in the coastal areas of Uruguay and Argentina also indicated a highstand sea level between ca. 6000–5000 yr BP, followed by a regressive event (e.g. Borel and Gómez, 2006; García-Rodríguez et al., 2010; Mourelle et al., 2015; Vilanova and Prieto, 2012).
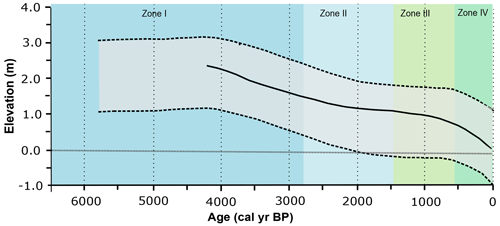
Figure 12Palaeo-sea-level reconstructions for the southern Brazilian coast and palaeoenvironmental zones. Grey area: relative palaeo-sea-level envelope (Angulo et al., 2006) to the south of 28∘ S latitude; solid line: sea level model curve for Ponta do Papagaio (Angulo et al., 2022; modified from Angulo et al., 2022).
In general, the environmental zone succession identified in this study is similar to those presented in previous palaeoenvironmental studies developed on the southern Brazil coastal plain, particularly in the Santa Catarina sector (e.g. Amaral et al., 2012; Cancelli, 2012; Kuhn et al., 2017; Val-Peón et al., 2019; Silva et al., 2021). Most of these studies indicate a succession of three zones from a lagoonal and/or estuarine environment (I) to a transitional and/or swampy regime (II) and an arboreal forest environment (III). However, in this contribution, we were able to define the transition from a herbaceous restinga (Zone III) to the arboreal restinga forest (Zone IV). Some of the above-mentioned studies indicate a maximum relative sea level (RSL) ranging from ca. 5200 to 4500 yr BP (Fig. 13).
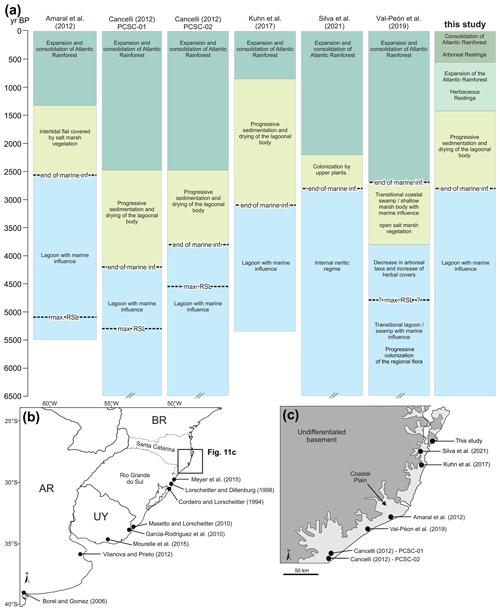
Figure 13Palynological studies and their location. (a) Summarized environmental changes of the palynological studies in similar settings of the Santa Catarina coastal plain. (b) Sites from the Rio Grande do Sul state, Uruguay (UY) and Argentina (AR). (c) Sites from the Santa Catarina state (a). Max RSL: maximum relative sea level. Inf.: influence. Cross bars at the lower limits indicate that the core was older than 6500 cal yr BP.
The distinct ages for the end of marine influence and the development of the Atlantic rainforest described in the previous studies is probably related to the different distances and/or altitudes of the depositional sites in relation to the current coastline and sea level. Localities nearer to the sea (e.g. Kuhn et al., 2017, and this study) were more affected by the sea level rise and show late development of the Atlantic rainforest in comparison to those located further from the shoreline (e.g. Cancelli, 2012). This supports that the restinga forest development was mainly controlled by edaphic factors and was less sensitive to climate factors during the Holocene (Scheel-Ybert, 2000; Amaral et al., 2012; Melo and Boeger, 2015).
Palynological, stable isotopic and sedimentological analyses at the southernmost part of Santa Catarina Island allowed us to recognize four environmental zones from 6520 cal yr BP to present, namely Zone I (lagoon with sea influence), Zone II (lagoon without sea influence), Zone III (early development of the restinga forest) and Zone IV (restinga forest). The dinoflagellate cyst association suggests that marine waters entering the region had their origin in the relatively warm, saline BC waters. Even though, nowadays, waters of the Brazil Coastal Current and MC can seasonally reach the coast at the same latitude as that of the core position, we did not observe any evidence that this has been the case in Zone I. Pollen records indicate that the Atlantic rainforest was already present in the Santa Catarina coastal plain before 6520 cal yr BP; however, this was likely restricted to areas which were not affected by the maximum transgressive sea level in the Holocene. Furthermore, we observed that the development of the restinga forest in the area occurred subsequently to the drying up of a lagoon. This study enhances our knowledge of the evolution of the southern Brazil coastal plain through information regarding the geomorphological and vegetational changes during the Holocene. Furthermore, this study represents an example of the strong sensitivity of the southern Brazilian ecosystem change caused by relative sea level variations. As such, it contributes to the debate about the potential effects of current climate change induced by global sea level changes.
All data required to reproduce the analytical results for this study are available at https://data.neotomadb.org/55958 (Kuhn, 2023).
LAK: conceptualization, methodology, formal analysis, investigation, writing – original draft, writing – review and editing, funding acquisition. KAFZ: conceptualization, methodology, formal analysis, resources, writing – review and editing, supervision, funding acquisition. PAS: conceptualization, methodology, resources, writing – review and editing, supervision, project administration. RRC: conceptualization, formal analysis, investigation, writing – review and editing.
The contact author has declared that none of the authors has any competing interests.
Publisher’s note: Copernicus Publications remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The authors thank Pedro H. Simas for the fieldwork assistance and guidance at the study area, Guilherme S. Hoerlle for the fieldwork and writing assistance, and Beatriz Fontana for the English revision. We thank Hermann Behling and Aline Freitas for the constructive reviews. This paper is part of the LAK PhD Thesis developed at the Programa de Pós-graduação em Geociências, Universidade Federal do Rio Grande do Sul.
This research has been supported by the CNPq-National Council for Scientific
and Technological Development of Brazil (grant no. 141324/2017) and
CAPES-Brazilian Coordination of Higher Education Staff Improvement (grant
no. 88887.467306/2019-00).
The article processing charges for this open-access publication were covered by the University of Bremen.
This paper was edited by Petr Kuneš and reviewed by Hermann Behling and Aline Freitas.
Alvares, C. A., Stape, J. L., Sentelhas, P. C., de Moraes Gonçalves, J. L., and Sparovek, G.: Köppen's climate classification map for Brazil, Meteorol. Z., 22, 711–728, https://doi.org/10.1127/0941-2948/2013/0507, 2013.
Amaral, P. G. C., Fonseca, P. C. G., Sylvestre, F., and Pessenda, L. C. R.: Paleoenvironmental reconstruction of a Late Quaternary lagoon system in southern Brazil (Jaguaruna region, Santa Catarina state) based on multi-proxy analysis, J. Quaternary Sci., 27, 181–191, https://doi.org/10.1002/jqs.1531, 2012.
Angulo, R., Lessa, G., and Souza, M.: A critical review of mid- to late-Holocene sea-level fluctuations on the eastern Brazilian coastline, Quaternary Sci. Rev., 25, 486–506, https://doi.org/10.1016/j.quascirev.2005.03.008, 2006.
Angulo, R. J., Giannini, P. C. F., Suguio, K., and Pessenda, L. C. R.: Relative sea-level changes in the last 5500 years in southern Brazil (Laguna–Imbituba region, Santa Catarina State) based on vermetid 14C ages, Mar. Geol., 159, 323–339, https://doi.org/10.1016/S0025-3227(98)00204-7, 1999.
Angulo, R. J., de Souza, M. C., Giannini, P. C. F., Dillenburg, S. R., Barboza, E. G., da Camara Rosa, M. L. C., Hesp, P. A., and Pessenda, L. C. R.: Late-Holocene sea levels from vermetids and barnacles at Ponta do Papagaio, 27∘50′ S latitude and a comparison with other sectors of southern Brazil, Quaternary Sci. Rev., 286, 107536, https://doi.org/10.1016/j.quascirev.2022.107536, 2022.
Batten, D. J.: Palynofacies, in: Palynology: Principles and Applications, edited by: Jansonius, J., and McGregor, D. J., American Association of Stratigraphic Palynologists Foundation, Dallas, 1011–1084, ISBN: 9-931871-03-4, 1996.
Batten D. J. and Grenfell H. R.: Botryococcus, in: Palynology: principles and applications, edited by: Jansonius, J. and McGregor, D. C., American Association of Stratigraphic Palynologists Foundation, Dallas, 1, 205–214, 1996.
Bauermann, S. G., Behling, H., and Macedo, R. B.: Biomas regionais e evolução da paisagem no Rio Grande do Sul com base em paleopalinologia, in: Quaternário Do Rio Grande Do Sul: Integrando Conhecimentos, edited by: Ribeiro, A. M., Bauermann, S. G., and Scherer, C. S., SBP, Porto Alegre, 81–93, ISBN: 978-85-63122-01-8, 2009.
Behling, H.: Investigations into the Late Pleistocene and Holocene history of vegetation and climate in Santa Catarina (S Brazil), Veg. Hist. Archaeobot., 4, 127–152, 1995.
Behling, H. and Negrelle, R. R. B.: Tropical Rain Forest and Climate Dynamics of the Atlantic Lowland, Southern Brazil, during the Late Quaternary, Quaternary Res., 56, 383–389, https://doi.org/10.1006/qres.2001.2264, 2001.
Blaauw, M.: Methods and code for `classical'age-modelling of radiocarbon sequences, Quat. Geochronology, 5, 512–518, https://doi.org/10.1016/j.quageo.2010.01.002, 2010.
Borel, C. M. and Gómez, E. A.: Palynology from the Holocene of Canal del Medio, Bahía Blanca estuary, Buenos Aires, Argentina, Ameghiniana, 43, 399–412, 2006.
Borromei, A. M. and Quattrocchio, M.: Holocene sea-level change inferred from palynological data in the Beagle Channel, southern Tierra del Fuego, Argentina, Ameghiniana, 44, 161–171, 2007.
Cancelli, R. R.: Evolução paleoambiental da Planície Costeira sul catarinense (Lagoa do Sombrio) durante o Holoceno, com base em dados palinológicos, Ph.D. thesis, Universidade Federal do Rio Grande do Sul, Brazil, 159 pp., http://hdl.handle.net/10183/70405 (last access: 15 May 2023), 2012.
Cancelli, R. R., Souza, P. A., and Neves, P. C. P.: Grãos de pólen de angiospermas do Holoceno (7908±30 anos AP-atual) da Planície Costeira sul-catarinense, Brasil, Acta Bot. Bras., 26, 866–885, https://doi.org/10.1590/S0102-33062012000400016, 2012.
Candel, M. S. and Borromei, A. M.: Review of the palaeoenvironmental reconstruction of Late Quaternary marine sequences, Tierra del Fuego (Argentina), Publicación Electrónica la Asoc. Paleontológica Argentina, https://doi.org/10.5710/PEAPA.16.08.2016.112, 2016.
Carr, A. S., Boom, A., Chase, B. M., Meadows, M. E., and Grimes, H. L.: Holocene sea level and environmental change on the west coast of South Africa: evidence from plant biomarkers, stable isotopes and pollen, J. Paleolimnol., 53, 415–432, https://doi.org/10.1007/s10933-015-9833-7, 2015.
Caruso Jr., F.: Mapa geológico da Ilha de Santa Catarina: texto explicativo e mapa, Notas Técnicas, 6, 1–28, 1993.
Chemello, R. and Silenzi, S.: Vermetid reefs in the Mediterranean Sea as archives of sea-level and surface temperature changes, Chem. Ecol., 27, 121–127, https://doi.org/10.1080/02757540.2011.554405, 2011.
Cohen, M. C. L., Rodrigues, E., Rocha, D. O. S., Freitas, J., Fontes, N. A., Pessenda, L. C. R., de Souza, A. V., Gomes, V. L. P., França, M. C., Bonotto, D. M., and Bendassolli, J. A.: Southward migration of the austral limit of mangroves in South America, Catena, 195, 104–775, https://doi.org/10.1016/j.catena.2020.104775, 2020.
Colinvaux, P., De Oliveira, P. E., and Moreno, J. E.: Amazon Pollen Manual and Atlas: Manual e Atlas Palinológico da Amazônia, Harwood, Amsterdam, ISBN: 9780429080548, https://doi.org/10.1201/9781482283600, 1999.
Cordeiro, S. H. and Lorscheitter, M. L.: Palynology of Lagoa dos Patos sediments, Rio Grande do Sul, Brazil, J. Paleolimnol., 10, 35–42, 1994.
Dillenburg, S. R., Tomazelli, L. J., Hesp, P. A., Barboza, E. G., Clerot, L. C. P., and Da Silva, D. B.: Stratigraphy and evolution of a prograded transgressive dunefield barrier in southern Brazil, J. Coastal Res., 1, 132–135, 2006.
Diniz, D. and Medeanic, S.: Palynomorph records from the Core of Rio Grande Cone, Brazil: approaches for palaeoenvironmental and palaeoclimatic reconstructions in the Middle Holocene, J. Coastal Res., 29, 1351–1360, https://doi.org/10.2112/JCOASTRES-D-11-00121.1, 2013.
Esri, Maxar, Earthstar Geographics, and the GIS User Community, Imagery [basemap], Scale Not Given, World Imagery, https://www.arcgis.com/home/item.html?id=10df2279f9684e4a9f6a7f08febac2a9, 2020.
Faegri, K., Kaland, P. E., and Krzywinski, K.: Textbook of pollen analysis, fourth. ed., John Wiley & Sons Ltd., ISBN: 978-1-930665-01-09, 1989.
Falkenberg, D. B.: Aspectos da flora e da vegetação secundária da restinga de Santa Catarina, sul do Brasil, Insul. Rev. Botânica, 28, 1–30, 1999.
Félix, C. M. and Souza, P. A.: Acritarcos (Grupo Acritarcha Evitt 1963): conceitos gerais, aplicações e importância na análise estratigráfica do intervalo Pennsilvaniano e Permiano da Bacia do Paraná, Revista do Instituto Geológico, 33, 71–88, https://doi.org/10.5935/0100-929X.20120005, 2012.
Folk, R. L. and Ward, W. C.: Brazos River bar [Texas]; a study in the significance of grain size parameters, J. Sediment. Res., 27, 3–26, https://doi.org/10.1306/74D70646-2B21-11D7-8648000102C1865D, 1957.
França, M. C., Pessenda, L. C. R., Cohen, M. C. L., de Azevedo, A. Q., Fontes, N. A., Silva, F. B., de Melo, J. C. F., Piccolo, M. de C., Bendassolli, J. A., and Macario, K.: Late-Holocene subtropical mangrove dynamics in response to climate change during the last millennium, The Holocene, 29, 445–456, https://doi.org/10.1177/0959683618816438, 2019.
Freitas, A. G. and Carvalho, M. A.: Análise morfológica e inferências ecológicas de grãos de pólen e esporos (últimos ∼ 8.000 anos) da Lagoa da Ferradura, Armação dos Búzios, RJ, Brasil, Rev. Bras. Paleontolog., 15, 300–318, https://doi.org/10.4072/rbp.2012.3.06, 2012.
García-Rodríguez, F., Stutz, S., Inda, H., del Puerto, L., Bracco, R., and Panario, D.: A multiproxy approach to inferring Holocene paleobotanical changes linked to sea-level variation, paleosalinity levels, and shallow lake alternative states in Negra Lagoon, SE Uruguay, Hydrobiologia, 646, 5–20, https://doi.org/10.1007/s10750-010-0184-0, 2010.
Grimm, E. C.: TILIA 1.7.16, Illinois State Museum, Springfield, https://www.neotomadb.org/apps/tilia (last access: 1 April 2023), 2011.
Gu, F., Zonneveld, K. A. F., Chiessi, C. M., Arz, H. W., Pätzold, J., Behling, H.: Long-term vegetation, climate and ocean dynamics inferred from a 73,500 years old marine sediment core (GeoB2107-3) off southern Brazil, Quaternary Sci. Rev., 172C, 55–71, https://doi.org/10.1016/j.quascirev.2017.06.028, 2017.
Gu, F., Chiessi, C. M., Zonneveld, K. A. F., and Behling, H.: Shifts of the Brazil-Falklands/Malvinas Confluence in the western South Atlantic during the latest Pleistocene–Holocene inferred from dinoflagellate cysts, Palynology, 43, 483–493, https://doi.org/10.1080/01916122.2018.1470116, 2019.
Hammer, Ø., Harper, D. A. T., and Ryan, P. D.: PAST: Paleontological Statistics software package for education and data analysis, Palaeontol. Electron., 4, 1–9, https://doi.org/10.1016/j.bcp.2008.05.025, 2001.
Herrera, L. F. and Urrego, L. E.: Atlas de polen de plantas útiles y cultivadas de la Amazonia colombiana, Tropenbos, Bogotá, ISBN: 978-958-9365-03-8, 1996.
Hogg, A. G., Heaton, T. J., Hua, Q., Palmer, J. G., Turney, C. S., Southon, J., Bayliss, A., Blackwell, P. G., Boswijk, G., Ramsey, C. B. Pearson, C., Petchey, F., Reimer, P., Reimer, R., and Wacker, L.: SHCal20 southern hemisphere calibration, 0-55,000 years cal BP, Radiocarbon, 62, 759–778, https://doi.org/10.1017/RDC.2020.59, 2020.
Hooghiemstra. H.: Vegetational and climatic history of the high plain of Bogotá, Colombia, J. Cramer, Vaduz, ISBN: 9783768214049, 1984.
Horn Filho, N. O.: Granulometria das praias arenosas da Ilha de Santa Catarina, SC. Gravel, 4, 1–21, 2006.
IBGE: Manual Técnico da Vegetação Brasileira, 2nd ed., Diretoria de Geociências, Rio de Janeiro, Brazil, 275 pp., ISBN: 978-85-240-4272-0, 2012.
INMET: Normais Climatológicas do Brasil 1991–2020, https://portal.inmet.gov.br/normais, last access: 15 September 2022.
INPE: TOPODATA [shaded relief basemap], https://www.webmapit.com.br/inpe/topodata (last access: 15 May 2023), 2020.
JPL MUR MEaSUREs Project: GHRSST Level 4 MUR Global Foundation Sea Surface Temperature Analysis. Ver. 4.1. PO. DAAC, CA, USA, https://doi.org/10.5067/GHGMR-4FJ04, 2015.
Kuhn, L. A., Souza, P. A., Cancelli, R. R., Silva, W. G., and Macedo, R. B.: Paleoenvironmental evolution of the coastal plain of Southern Brazil: Palynological data from a Holocene core in Santa Catarina State, An. Acad. Bras. Cienc, 89, 2581–2595, https://doi.org/10.1590/0001-3765201720160895, 2017.
Kuhn, L. A.: Neotoma Dataset 55958, Santa Catarina Island [data set], https://data.neotomadb.org/55958 (last access: 10 May 2023), 2023.
Leroy, S. A. G., Kakroodi, A. A., Kroonenberg, S., Lahijani, H. K., Alimohammadian, H., and Nigarov, A.: Holocene vegetation history and sea level changes in the SE corner of the Caspian Sea: relevance to SW Asia climate, Quaternary Sci. Rev., 70, 28–47, https://doi.org/10.1016/j.quascirev.2013.03.004, 2013.
Lorente, F. L., Pessenda, L. C. R., Oboh-Ikuenobe, F., Buso Jr., A. A., Cohen, M. C. L., Meyer, K. E. B., Giannini, P. C. F., de Oliveira, P. E., Rossetti, D. de F., Borotti Filho, M. A., França, M. C., de Castro, D. F., Bendassolli, J. A., and Macario, K.: Palynofacies and stable C and N isotopes of Holocene sediments from Lake Macuco (Linhares, Espírito Santo, southeastern Brazil): Depositional settings and palaeoenvironmental evolution, Palaeogeogr. Palaeoecol. 415, 69–82, https://doi.org/10.1016/j.palaeo.2013.12.004, 2014.
Lorscheitter, M. L.: Contribution to the Holocene history of Atlantic rain forest in the Rio Grande do Sul state, southern Brazil, Revista del Museo Argentino de Ciencias Naturales nueva Ser., 5, 261–271, 2003.
Lorscheitter, M. L. and Dillenburg, S.: Holocene palaeoenvironments of the northern 5 coas tal plain of Rio Grande do Sul, Brazil, reconstructed from palynology of Tramandaí lagoon sediments, Quat. South Am., 11, 73–97, 1998.
Lorscheitter, M. L., Ashraf, A. R., Bueno, R. M., and Mosbrugger, V.: Pteridophyte spores of Rio Grande do Sul flora, Brazil. Part I, Palaeontographica, 246, 1–113, 1998.
Macedo, R. B., Souza, P. A., and Bauermann, S. G.: Catálogo de pólens, esporos e demais palinomorfos em sedimentos holocênicos de Santo Antônio da Patrulha, Rio Grande do Sul, Brasil, Iheringia-Ser. Bot., 64, 43–78, 2009.
Magnago, L. F. S., Martins, S. V., Schaefer, C. E. G. R., and Neri, A. V.: Gradiente fitofisionômico-edáfico em formações florestais de Restinga no sudeste do Brasil, Acta Bot. Bras., 24, 734–746, https://doi.org/10.1590/S0102-33062010000300017, 2010.
Martin, L., Suguío, K., and Flexor, J. M.: Relative sea-level reconstruction during the last 7,000 years along the states of Paraná and Santa Catarina coastal plains: additional information derived from shell middens, Quat. South Am., 4, 219–236, 1986.
Martin, L., Dominguez, J. M., and Bittencourt, A. C.: Fluctuating Holocene sea levels in eastern and southeastern Brazil: evidence from multiple fossil and geometric indicators, J. Coastal Res., 19, 101–124, 2003.
Masetto, E. and Lorscheitter, M. L.: Gymnosperm and angiosperm pollen grains in Holocene sediments from a paleolagoon in the coastal plain of extreme southern Brazil, Braz. J. Bot., 39, 709–720, https://doi.org/10.1007/s40415-015-0242-y, 2016.
Masetto, E. and Lorscheitter, M. L.: Vegetation dynamics during the last 7500 years on the extreme southern Brazilian coastal plain, Quatern. Int., 524, 48–56, https://doi.org/10.1016/j.quaint.2019.07.009, 2019.
Melo Jr., J. C. F. and Boeger, M. R. T.: Riqueza, estrutura e interações edáficas em um gradiente de restinga do Parque Estadual do Acaraí, Estado de Santa Catarina, Brasil, Hoehnea, 42, 207–232, https://doi.org/10.1590/2236-8906-40/2014, 2015.
Meyer, K. E. B., Mendonça Filho, J. G., Ashraf, A. R. Souza, P. A., and Reichhart, K.: Análise de palinofácies em sedimentos holocênicos da lagoa dos Quadros, Rio Grande do Sul, Brasil, Rev. Bras. Paleontolog., 8, 57–72, https://doi.org/10.4072/rbp.2005.1.05, 2005.
Meyers, P. A.: Preservation of elemental and isotopic source identification of sedimentary organic matter, Chem. Geol., 114, 289–302, https://doi.org/10.1016/0009-2541(94)90059-0, 1994.
Meyers, P. A.: Organic geochemical proxies of paleoceanographic, paleolimnologic, and paleoclimatic processes, Org. Geochem., 27, 213–250, https://doi.org/10.1016/S0146-6380(97)00049-1, 1997.
Montenari, M. and Leppig, U.: The Acritarcha: their classification morphology, ultrastructure and palaeoecological/palaeogeographical distribution, Palaeontol Z., 77, 173–194, 2003.
Mourelle, D., Prieto, A. R., Pérez, L., García-Rodríguez, F., Borel, C. M.: Mid and late Holocene multiproxy analysis of environmental changes linked to sea-level fluctuation and climate variability of the Río de la Plata estuary, Palaeogeogr. Palaeocl., 421, 75–88, https://doi.org/10.1016/j.palaeo.2015.01.006, 2015.
Mourelle, D., Macedo, R., and Prieto, A.: Análisis palinológico actual y del Cuaternario tardío en la región de los Campos (Uruguay y sur de Brasil): estado de las investiga- ciones, dificultades y potencialidades, Publicación Electrónica la Asoc, Paleontológica Argentina, 18, 156–170, https://doi.org/10.5710/PEAPA.28.05.2018.258, 2018.
Mudie, P. J., Yanko-Hombach, V. V., and Mudryk, I.: Palynomorphs in surface sediments of the North-Western Black Sea as indicators of environmental conditions, Quaternary Int., 590, 122–145, https://doi.org/10.1016/j.quaint.2020.05.014, 2021.
Neves, P. C. P. and Lorscheitter, M. L.: Palinologia de sedimentos de uma mata tropical paludosa em Terra de Areia, Planície Costeira Norte, Rio Grande do Sul, Brasil. Descrições taxonômicas, parte I: fungos, algas, briófitos, pteridófitos, palinomorfos outros e fragmentos de invertebrados, Acta Geológica Leopoldinense, 15, 83–114, 1992.
Newton, A., Harff, J., You, Z.-J., Zhang, H., and Wolanski, E.: Sustainability of future coasts and estuaries: A synthesis, Estuar. Coast. Shelf S., 183, 271–274, https://doi.org/10.1016/j.ecss.2016.11.017, 2016.
Nimer, E.: Clima, in: Geografia Do Brasil. Região Sul. IBGE, Rio de Janeiro, pp. 151–187, 1990.
Oliveira Filho, A. T. and Fontes, M. A. L.: Patterns of floristic differentiation among Atlantic forests in Southeastern Brazil and the influence of climate, Biotropica, 32, 793–810, https://doi.org/10.1111/j.1744-7429.2000.tb00619.x, 2000.
Peterson, R. G. and Stramma, L.: Upper-level circulation in the South Atlantic Ocean, Prog. Oceanogr., 26, 1–73, https://doi.org/10.1016/0079-6611(91)90006-8, 1991.
Piola, A. R.: The influence of the Plata River discharge on the western South Atlantic shelf, Geophys. Res. Lett., 32, 1–4, https://doi.org/10.1029/2004GL021638, 2005.
Piola, A. R. and Matano, R. P.: Brazil and Falklands (Malvinas) Currents, in: Encyclopedia of Ocean Sciences, Elsevier, 340–349, https://doi.org/10.1006/rwos.2001.0358, 2001.
Piola, A. R. and Matano, R. P.: Ocean currents: Atlantic Western Boundary – Brazil Current/Falkland (Malvinas) Current, in: Cochran, J. K., Bokuniewicz, H. J., and Yager, P. L., Encyclopedia of Ocean Sciences, Academic Press, Amsterdam, 414–420, https://doi.org/10.1016/B978-0-12-409548-9.10541-X, 2019.
Piola, A. R., Campos, E. J. D., Möller, O. O., Charo, M., and Martinez, C.: Subtropical Shelf Front off eastern South America, J. Geophys. Res-Oceans, 105, 6565–6578, https://doi.org/10.1029/1999JC000300, 2000.
Ramesh, R., Chen, Z., Cummins, V., Day, J., D'Elia, C., Dennison, B., Forbes, D. L., Glaeser, B., Glaser, M., Glavovic, B., Kremer, H., Lange, M., Larsen, J. N., Le Tissier, M., Newton, A., Pelling, M., Purvaja, R., and Wolanski, E.: Land-Ocean Interactions in the Coastal Zone: Past, present & future, Anthropocene, 12, 85–98, https://doi.org/10.1016/j.ancene.2016.01.005, 2015.
RStudio Team RStudio: Integrated Development for R. RStudio, Inc., Boston, MA (Computer Software v0.98.1074), http://www.rstudio.com/ (last access: 15 May 2023), 2015.
Scarano, F. R.: Structure, Function and Floristic Relationships of Plant Communities in Stressful Habitats Marginal to the Brazilian Atlantic Rainforest, Ann Bot-London, 90, 517–524, https://doi.org/10.1093/aob/mcf189, 2002.
Scheel-Ybert, R.: Vegetation stability in the Southeastern Brazilian coastal area from 5500 to 1400 14C yr BP deduced from charcoal analysis, Rev. Palaeobot. Palyno., 110, 111–138, https://doi.org/10.1016/S0034-6667(00)00004-X, 2000.
Sebag, D., Copard, Y., Di-Giovanni, C., Durand, A., Laignel, B., Ogier, S., and Lallier-Verges, E.: Palynofacies as useful tool to study origins and transfers of particulate organic matter in recent terrestrial environments: Synopsis and prospects, Earth-Sci. Rev., 79, 241–259, https://doi.org/10.1016/j.earscirev.2006.07.005, 2006.
Sevegnani, L. and Schroeder, E.: Biodiversidade catarinense: características, potencialidades, ameaças, Edifurb, Blumenau, 2013.
Shepard, F. P.: Nomenclature based on sand-silt-clay ratios, J. Sediment. Res., 24, 151–158, 1954.
Silva, W. G., Souza, P. A., Garcia, M. J., Carvalho, M. A., Dillenburg, S. R., Cancelli, R. R., and Kuhn, L. A.: Middle to Late Holocene paleoenvironmental changes in the coastal plain of southern Brazil, J. S. Am. Earth Sci., 111, 103514, https://doi.org/10.1016/j.jsames.2021.103514, 2021.
Silveira, I. C. A. da, Schmidt, A. C. K., Campos, E. J. D., Godoi, S. S., and Ikeda, Y.: A corrente do Brasil ao largo da costa leste brasileira, Revista Brasileira de Oceanografia, 48, 171–183, https://doi.org/10.1590/S1413-77392000000200008, 2000.
Šmilauer, P. and Lepš, J.: Multivariate analysis of ecological data using CANOCO 5, 2nd ed., Cambridge university press, New York, https://doi.org/10.1017/CBO9781139627061, 2014.
Souza, R. B. and Robinson, I. S.: Lagrangian and satellite observations of the Brazilian Coastal Current, Cont. Shelf Res., 24, 241–262, https://doi.org/10.1016/j.csr.2003.10.001, 2004.
Stockmarr, J.: Tablets with spores used in absolute pollen analysis, Pollen Spores, 13, 615–621, 1971.
Suguio, K., Martin, L., Bittencourt, A. C. S. P., Dominguez, J. M. L., Flexor, J. M., and Azevedo, A. E. G.: Flutuações do nível relativo do mar durante o Quaternário superior ao longo do litoral brasileiro e suas implicações na sedimentação costeira, Revista Brasileira de Geociências, 15, 273–286, https://doi.org/10.25249/0375-7536.1985273286, 1985.
Toniolo, T. de F., Giannini, P. C. F., Angulo, R. J., de Souza, M. C., Pessenda, L. C. R., and Spotorno-Oliveira, P.: Sea-level fall and coastal water cooling during the Late Holocene in Southeastern Brazil based on vermetid bioconstructions, Mar. Geol., 428, 106281, https://doi.org/10.1016/j.margeo.2020.106281, 2020.
Tyson, R. V.: Sedimentary organic matter: organic facies and palynofacies, Chapman and Hall, London, ISBN: 978-94-017-0763-3, https://doi.org/10.1007/978-94-017-0763-3_5, 1995.
UNESCO: Rede Mundial de Reservas da Biosfera: lugares de desarrollo sostenible, 2010, Organização das Nações Unidas para a Educação, Ciência e Cultura, 597 pp., Paris, 2011.
Val-Peón, C., Cancelli, R. R., Santos, L., and Soares, A. L. R.: Prehistoric occupation and palaeoenvironmental changes along Santa Catarina's Coastal Plain, Brazil: An integrated approach based on palynological data, J. Archaeol. Sci. (Reports 23), 983–992, https://doi.org/10.1016/j.jasrep.2017.11.017, 2019.
Van Soelen, E. E., Lammertsma, E. I., Cremer, H., Donders, T. H., Sangiorgi, F., Brooks, G. R., Larson, R. A., Sinninghe Damsté, J. S., Wagner-Cremer, F., and Reichart, G. J.: Late Holocene sea-level rise in Tampa Bay: Integrated reconstruction using biomarkers, pollen, organic-walled dinoflagellate cysts, and diatoms, Estuar. Coast. Shelf S., 86, 216–224, https://doi.org/10.1016/j.ecss.2009.11.010, 2010.
Vilanova, I. and Prieto, A. R.: Historia de la Vegetación de Las Llanuras Costeras de la Bahía Samborombón (∼35,5∘S), Argentina, Desde 7800 14C Años, Ameghiniana, 49, 303–318, https://doi.org/10.5710/AMGH.v49i3(499), 2012.
Wilson, G. P., Lamb, A. L., Leng, M. J., Gonzalez, S., and Huddart, D.: Variability of organic δ13C and in the Mersey Estuary, U.K. and its implications for sea-level reconstruction studies, Estuar. Coast. Shelf S., 64, 685–698, https://doi.org/10.1016/j.ecss.2005.04.003, 2005.
Ybert, J. P., Bissa, W. M., Catharino, E. L. M., and Kutner, M.: Environmental and sea-level variations on the southeastern Brazilian coast during the Late Holocene with comments on prehistoric human occupation, Palaeogeogr. Palaeocl., 189, 11–24, https://doi.org/10.1016/S0031-0182(02)00590-4, 2003.
Zazo, C., Goy, J. L., Dabrio, C. J., Lario, J., González-Delgado, J. A., Bardají, T., Hillaire-Marcel, C., Cabero, A., Ghaleb, B., Borja, F., Silva, P. G., Roquero, E., Soler, V.: Retracing the Quaternary history of sea-level changes in the Spanish Mediterranean–Atlantic coasts: Geomorphological and sedimentological approach, Geomorphology, 196, 36–49, https://doi.org/10.1016/j.geomorph.2012.10.020, 2013.
Zonneveld, K. A. F. and Pospelova, V.: A determination key for modern dinoflagellate cysts, Palynology, 39, 387–409, https://doi.org/10.1080/01916122.2014.990115, 2015.
Zonneveld, K. A. F., Marret, F., Versteegh, G. J. M., Bogus, K., Bonnet, S., Bouimetarhan, I., Crouch, E., de Vernal, A., Elshanawany, R., Edwards, L., Esper, O., Forke, S., Grøsfjeld, K., Henry, M., Holzwarth, U., Kielt, J. F., Kim, S. Y., Ladouceur, S., Ledu, D., Chen, L., Limoges, A., Londeix, L., Lu, S. H., Mahmoud, M. S., Marino, G., Matsouka, K., Matthiessen, J., Mildenhal, D. C., Mudie, P., Neil, H. L., Pospelova, V., Qi, Y., Radi, T., Richerol, T., Rochon, A., Sangiorgi, F., Solignac, S., Turon, J. L., Verleye, T., Wang, Y., Wang, Z., and Young, M.: Atlas of modern dinoflagellate cyst distribution based on 2405 data points, Rev. Palaeobot. Palynol., 191, 1–197, https://doi.org/10.1016/j.revpalbo.2012.08.003, 2013.