the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Organic matter contents and degradation in a highly trawled area during fresh particle inputs (Gulf of Castellammare, southwestern Mediterranean)
Antonio Pusceddu
Pere Masqué
Pere Puig
Davide Moccia
Tommaso Russo
Claudio Lo Iacono
Bottom trawling in the deep sea is one of the main drivers of sediment resuspension, eroding the seafloor and altering the content and composition of sedimentary organic matter (OM). The physical and biogeochemical impacts of bottom trawling were studied on the continental slope of the Gulf of Castellammare, Sicily (southwestern Mediterranean), through the analysis of two triplicate sediment cores collected at trawled and untrawled sites (∼550 m water depth) during the summer of 2016. Geochemical and sedimentological parameters (excess 210Pb, excess 234Th, 137Cs, dry bulk density, and grain size), elemental (organic carbon and nitrogen) and biochemical composition of sedimentary OM (proteins, carbohydrates, lipids), as well as its freshness (phytopigments) and degradation rates were determined in both coring locations. The untrawled site had a sedimentation rate of 0.15 cm yr−1 and presented a 6 cm thick surface mixed layer that contained siltier sediment with low excess 210Pb concentrations, possibly resulting from the resuspension, posterior advection, and eventual deposition of coarser and older sediment from adjacent trawling grounds. In contrast, the trawled site was eroded and presented compacted century-old sediment highly depleted in OM components, which were between 20 % and 60 % lower than those in the untrawled site. However, the upper 2 cm of the trawled site consisted of recently accumulated sediments enriched in excess 234Th, excess 210Pb, and phytopigments, while OM contents were similar to those from the untrawled core. This fresh sediment supported protein turnover rates of 0.025 d−1, which doubled those quantified in surface sediments of the untrawled site. The enhancement of remineralization rates in surface sediment of the trawled site was associated with the arrival of fresh particles on a chronically trawled deep-sea region that is generally deprived of OM. We conclude that the detrimental effects of bottom trawling can be temporarily and partially abated by the arrival of fresh and nutritionally rich OM, which stimulate the response of benthic communities. However, these ephemeral deposits are likely to be swiftly eroded due to the high trawling frequency over fishing grounds, highlighting the importance of establishing science-based management strategies to mitigate the impacts of bottom trawling.
- Article
(4419 KB) - Full-text XML
-
Supplement
(450 KB) - BibTeX
- EndNote
Bottom trawling is among the most extensive forms of anthropogenic activities affecting marine ecosystems (Amoroso et al., 2018; Eigaard et al., 2017), and it is one of the most harmful in terms of fish stock overexploitation (Pauly et al., 2002), destruction of habitats (Kaiser et al., 2002; Simpson and Watling, 2006), and the physical impact it exerts on the sediments (Martín et al., 2014a; Oberle et al., 2018; Puig et al., 2012). Since bottom trawling targets benthic and demersal fisheries, its gear is designed to be in continuous contact with the seafloor, scraping the bottom, resuspending large volumes of sediment (O'Neill and Ivanović, 2016; Palanques et al., 2014), and causing significant erosion (Martín et al., 2014b; Oberle et al., 2016). The resuspension of sediment releases nutrients and organic matter (OM) to the overlying water column, and degradation of OM can be accelerated through enhanced microbial activity (Durrieu de Madron et al., 2005; Pusceddu et al., 2005b, 2015). The effects that these perturbations generate on sedimentary OM can vary in cohesive (i.e., muddy sediment with high clay content) and non-cohesive (i.e., sandy seafloor) sediment. For instance, trawling on cohesive sediments can increase superficial concentrations of sedimentary OM (Palanques et al., 2014; Pusceddu et al., 2005a; Sciberras et al., 2016; Polymenakou et al., 2005), whereas trawling on coarse non-cohesive sediments can exert null or minimal effects on OM contents and benthic community metabolism (Hale et al., 2017; Tiano et al., 2019; Trimmer et al., 2005).
Most of these impacts have been documented in shallow environments, where sediment and OM fluxes are generally high and sediment resuspension and OM remineralization induced by bottom trawling can be comparable to those induced by natural high-energy events such as storms (Buscail et al., 1990; Dellapenna et al., 2006; Ferré et al., 2008; Pusceddu et al., 2005b). Since these natural physical disturbances are persistent, shallow benthic communities generally present higher resilience to the impacts of bottom trawling than communities that live in less disturbed areas, such as the deep sea (Kaiser, 1998).
However, bottom trawling has been progressively expanding to deeper environments (>200 m depth) since the 1950s (Morato et al., 2006; Roberts, 2002), driven by technological advances in parallel with an ongoing depletion of shallow-water fisheries (Koslow et al., 2000; Martín et al., 2014a). At such depths, natural sediment fluxes to the seafloor and resuspension processes tend to be low. Hence, bottom trawling has become a major mechanism of sediment resuspension on continental slopes, leading to eroded fishing grounds (Martín et al., 2014b, c; Puig et al., 2012). Resuspended particles can then be exported by ambient currents across- and along-margin as enhanced nepheloid layers (Arjona-Camas et al., 2019; Wilson et al., 2015), or as sediment gravity flows (Martín et al., 2014c; Puig et al., 2012) ultimately generating anthropogenic sedimentary depocenters (Puig et al., 2015; Paradis et al., 2017, 2018).
Contrary to the observed increases in total organic carbon on muddy continental shelf trawling grounds (Palanques et al., 2014; Polymenakou et al., 2005; Pusceddu et al., 2005a), the continuous removal of sediment by trawlers on continental slopes has significantly impoverished bulk organic carbon as well as its labile and fresh pools (Martín et al., 2014b; Pusceddu et al., 2014; Sañé et al., 2013). The loss of organic matter has also reduced organic carbon turnover rates on slope trawling grounds, severely impacting the meiofauna and, at the same time, promoting the abundance of taxa with opportunistic life strategies (Pusceddu et al., 2014). However, the combined effect of bottom trawling erosion along with the alteration of sedimentary organic matter, which usually represents the fundamental energy source for commercial deep-sea benthic species, is not fully understood.
The Gulf of Castellammare holds one of the most important bottom trawling grounds in the northern Sicilian shore (southwestern Mediterranean Sea). Bottom trawlers in this gulf operate using otter trawl gear, a trawling technique which consists of dragging a wide net that is held open and in contact with the seafloor by two otter doors (Martín et al., 2014a). The first data of bottom trawlers in the area go back to the 1960s, but this fishing technique has became more active since the 1980s (European Commission Fisheries & Maritime Affairs, 2014), as a result of the modernization of the Sicilian trawling fleet (L.R. 1/1980, 1980; L.R. 26/1987, 1987). Fishing stocks within this gulf were declining alarmingly until the Sicilian government established a trawling ban area in the inner shelf in 1990 (L.R. 25/1990, 1990), delimited by the junction between Capo Rama and Torre dell'Uzzo (Fig. 1). Since the establishment of this closure, both demersal biomass and catch per unit effort (CPUE) of artisanal fisheries (non-towed bottom gear and pelagic gear) have increased in that area (Pipitone et al., 2000; Whitmarsh et al., 2002). However, bottom trawlers have been concentrating their efforts beyond the restricted area in the mid-continental slope (>500 m depth), leading to a decrease in CPUE since the trawl ban as a result of the continuous overexploitation of fishing stocks (Arculeo et al., 2014; Whitmarsh, 2002). A more recent management strategy aimed to prevent the collapse of Sicilian fisheries established a 30 d bottom trawling closure per year, which can occur from 1 August to 31 October (Decreto 1339/2001, 2001).
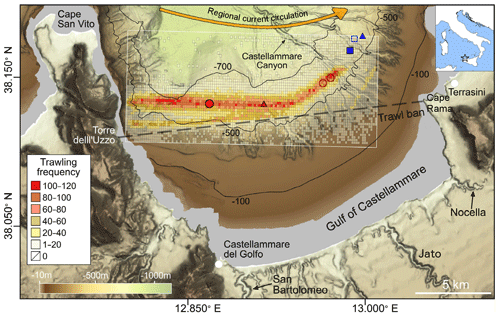
Figure 1Bathymetric map of the Gulf of Castellammare. Location of the sediment cores sampled at the trawled (red circles) and untrawled (blue squares) sites. Unfilled sampling points indicate unsuccessful sediment core deployments. Seafloor images obtained from ROV dives are shown with triangles (see Fig. S1). The distribution of trawling grounds as trawling frequency (number of total hauls per grid area) in 2016 (1 January to 10 August) was calculated for a 200 m × 200 m grid. The limit of the trawl banned area between Torre dell'Uzzo and Cape Rama is indicated by a dashed line. The main trawling harbors (Castellammare del Golfo and Terrasini) and the most relevant ephemeral rivers (San Bartolomeo, Nocella, and Jato) are also annotated. The yellow arrow illustrates the direction of the regional surface current.
Despite the numerous studies that address the effects of the trawl ban in the Gulf of Castellammare (Fanelli et al., 2008; Romano et al., 2016; Pipitone et al., 2000; Whitmarsh et al., 2002), no studies have yet assessed the impacts of bottom trawling on the gulf's sedimentary environment. This study aims to reveal whether erosion prevails in bottom trawling grounds and what the consequent alterations on sedimentary organic matter are, by comparing sediment cores collected at a trawled and untrawled site in the Gulf of Castellammare. The degree of erosion will be estimated based on sedimentological parameters and radioactive tracers with different half-lives (210Pb, years; 234Th, d), whereas the alterations on sedimentary organic matter will be determined based on its quantity, composition, and nutritional quality. The coupled analyses of radioactive tracers and biomarkers will also provide insights into the effects of the arrival of fresh sediment on impacted trawling grounds.
2.1 Study area
The Gulf of Castellammare is one of the widest bays of the northern coast of Sicily, with over 70 km of coastline, enclosed by Cape Rama to the east and Cape San Vito to the west (Fig. 1). A cyclonic along-slope current dominates the gulf's circulation at an average speed of 0.1–0.2 m s−1 on the upper continental shelf, which can sometimes reach maximum speeds of 0.4 m s−1 (Sarà et al., 2006). The seafloor morphology consists of a subhorizontal gently sloping continental shelf that extends approximately 5 km offshore. The continental slope is around 11∘ steep down to 500 m water depth, and it then gradually decreases to around 1.5∘ at 1300 m water depth (Lo Iacono et al., 2014). Several small, narrow submarine canyons cut the slope, breaching the shelf break at 120 to 140 m depth (Lo Iacono et al., 2014). Small seasonal torrents discharge into the gulf, namely the Nocella, Jato, and San Bartolomeo rivers, with annual average discharges between 0.24 and 0.32 m3 s−1 (Regione Siciliana, 2007). Storm-induced flash floods can cause short flushing events of up to 1.2 m3 s−1 that transport significant amounts of nutrients into the sea (Calvo and Genchi, 1989).
2.2 Sediment core sampling
In the framework of the FP7 EU-Eurofleets 2 ISLAND (ExplorIng SiciLian CAnyoN Dynamics) cruise on board the R/V Angeles Alvariño, sediment cores were collected in August 2016 from trawled and untrawled sites in the Gulf of Castellammare. Sampling locations were selected based on the distribution of operating trawlers using data from a vessel monitoring system (VMS) (see Sect. 2.8). Sampling took place prior to the temporal fishing closure in the Gulf of Castellammare, which took place that year between 17 September and 16 October. This ensured that sediment cores reflect the alterations caused by bottom trawling persisting in this deep environment.
A total of five multicore deployments were conducted using a K/C Denmark A/X six-tube multicorer (inner diameter 9.4 cm) at trawled and untrawled sites along the 550 m contour line. However, only one trawled and one untrawled site could be sampled (Fig. 1), possibly due to high sediment compaction at the trawled sites, as experienced by Martín et al. (2014b), and/or due to the swell during the coring operation that could hamper a successful triggering of the multicorer.
Triplicate sediment cores were retrieved at each site from three independent multicore deployments to account for spatial variability of organic matter analyses. The sediment cores were sliced on deck (0–1, 1–3, 3–5, 5–7, and 7–9 cm) and stored in calcinated aluminum foil at −20 ∘C until analysis of organic matter content. At each site, a single sediment core from one of the three deployments was reserved for sedimentary, radiochemical, and elemental analyses. This sediment core was sliced on deck at 1 cm intervals and kept in sealed plastic bags at −20 ∘C until being freeze-dried in the laboratory for analyses.
Prior to sediment recovery, the remotely operated vehicle (ROV) Seaeye Falcon, from the University of Plymouth (UK), collected visual evidence of trawling impact at the trawled sampling site and of no impact at the control site to corroborate the sampling strategy (Fig. S1 in the Supplement).
2.3 Sedimentary characteristics
Dry bulk densities of sediment cores were calculated by dividing the net dry weight corrected for salt content by the sample volume. Grain size fractions of sand (>63 µm), silt (4–63 µm), and clay (<4 µm) were obtained using a Horiba Partica LA-950V2 particle-size analyzer, with an accuracy of 0.6 % and a precision of 0.1 %. Prior to analysis, 1–4 g of sample was oxidized using 20 % H2O2 and sediment particles were disaggregated with .
2.4 Radiochemical analyses
Concentrations of 210Pb were determined through the analysis of its decay product 210Po by alpha spectrometry following the method described by Sanchez-Cabeza et al. (1998), assuming secular equilibrium of both radionuclides at the time of analysis. Between 150 and 300 mg of homogenized ground sample was spiked with 209Po as a chemical yield and microwave-digested using concentrated HNO3, HF, and HBO3. The resulting solutions were evaporated and reconditioned with 1 M HCl. Polonium isotopes were spontaneously deposited onto silver disks while stirring at 70 ∘C for at least 8 h. Alpha emissions of 209Po (4883 keV) and 210Po (5304 keV) were quantified using passivated implanted planar silicon (PIPS) detectors (CANBERRA, model PD-450.18 A.M.) and the Genie™ data acquisition software. Supported concentrations of 210Pb in the sediment cores were obtained by averaging constant concentrations of total 210Pb from the bottom of the core, from 2 to 37 cm for the trawled core and from 27 to 49 cm for the untrawled core, assuming complete decay of excess 210Pb at these depths. Supported 210Pb concentrations were corroborated by measuring 226Ra concentrations through its decay product 214Pb (295 and 352 keV) in several samples along each core using gamma spectroscopy, using calibrated geometries in a well-type high-purity germanium detector (CANBERRA, model GCW3523).
Concentrations of 234Th were also measured with gamma spectroscopy through the 63 keV emission line. Given its short half-life (24.1 d), samples were measured within two half-lives (∼6 weeks) since sampling, which only allowed the measurement of the upper 5 cm of the trawled and untrawled cores. Samples were remeasured at least 6 months later, after excess 234Th had decayed, to obtain supported 234Th concentrations, equivalent to 238U concentrations. Excess 234Th was calculated by subtracting supported 234Th from total 234Th, accounting for 234Th decay and in-growth from 238U since sampling.
Concentrations of 137Cs were also quantified using gamma spectroscopy through the emission line at 662 keV. Gamma measurements of the untrawled sediment core were extended in depth to 20 cm, whereas in the trawled core measurements were limited to the upper 5 cm since no 137Cs was detected in that core.
2.5 Elemental analyses
Analyses of total carbon, organic carbon (OC), and total nitrogen (TN) were carried out with an elemental analyzer (Costech ECS analyzer 4010), according to the procedure described in Nieuwenhuize et al. (1994). Samples for OC analysis were first decarbonated by acid-fuming the samples in the presence of 12 N HCl for 24 h and repeatedly adding 100 µL of 2 N HCl to the sample until effervescence ceased. Inorganic carbon (IC), quantified as the difference between total carbon and organic carbon, was converted to calcium carbonate (CaCO3) concentrations using the molecular mass ratio of CaCO3:IC (100 ∕ 12), assuming all inorganic carbon present is in the form of CaCO3. To account for analytical error, replicate analyses were performed for samples every 5 cm throughout the cores. An average percentage error of 1.2 % was obtained for carbon whereas nitrogen presented a slightly higher average percentage error of 1.9 %.
2.6 Biochemical composition of sedimentary organic matter
Total proteins, carbohydrates, and lipids were quantified spectrophotometrically (Varian Cary® 50 UV-Vis) according to the methods described in Hartree (1972) and modified by Rice (1982), Gerchakov and Hatcher (1972), Bligh and Dryer (1959), and Marsh and Weinstein (1966). The analyses of proteins and lipids were carried out on 0.1–0.6 g of frozen sediment, whereas carbohydrate analyses were performed on previously dried sediment. Protein, carbohydrate and lipid contents were transformed into carbon equivalents using 0.49, 0.4, and 0.75 mgC mg−1 as conversion factors, respectively, and their sum reported as biopolymeric C (Fabiano et al., 1995). Chlorophyll a and phaeopigments, after extraction with 90 % acetone, were quantified fluorometrically (Shimadzu RF-6000) according to Lorenzen and Jeffrey (1980) and modified by Danovaro (2010) for sediments. Total phytopigment concentrations were defined as the sum of chlorophyll a and phaeopigment concentrations and converted into carbon equivalents using a conversion factor of 40 (Pusceddu et al., 2010).
2.7 Sedimentary OM freshness and degradation rates
The contribution of phytopigment to biopolymeric C was used as a proxy to estimate OM freshness: since in the deep sea there is no in situ primary production, higher values of this ratio are associated with recently deposited material of algal origin (Pusceddu et al., 2010).
Since N is the most limiting factor for heterotrophic nutrition and proteins are N-rich products, sedimentary OM degradation was estimated using the degradation rate of proteins, obtained from the analysis of extracellular aminopeptidase activities. Aminopeptidase activity was estimated fluorometrically after incubation of approximately 0.1 g of sediment with 100 µM L-leucine-4-methylcumarinyl-7-amide for 1 h in the dark. This substrate, when exposed to extracellular aminopeptidase, produces fluorescence with an intensity proportional to the enzyme activity. Fluorometric analyses were carried out before and after incubation, and the difference was used to calculate protease activities (Danovaro, 2010). The results were converted to carbon equivalents using the conversion factor of 72 ngC nmol protease−1 (Fabiano and Danovaro, 1998). Turnover rates were then calculated by dividing protein-C degradation rates by protein-C sedimentary contents.
2.8 Trawling effort from VMS data
Fishing intensity of the Italian bottom trawling fleet was obtained using data provided by the vessel monitoring system (VMS), the main tracking device used for monitoring fishing activities. According to the Common Fisheries Policy of the European Union (European Commission, 2003), fishing vessels with overall length equal to or larger than 15 m must be equipped with a VMS transmitter, called “Blue Box”. It estimates the position of the vessel using the Global Positioning System and sends this information, along with the speed and heading of the vessel, to the network of the coastal guard by Inmarsat C to the Fishing Monitoring Centre in less than 10 min at 2 h time intervals. Fishing intensity was calculated using yearly VMS data from 2007 to 2015, whereas for 2016, only VMS data from 1 January to 10 August were taken into account, prior to sampling. Trawling frequency was represented as the number of times trawled per grid cell (200 m × 200 m) during each year. The native VMS data were processed using the R package VMSbase (Russo et al., 2014). The size of the grid was defined considering the error associated with the reconstruction of the trawling hauls as described by Russo et al. (2011).
2.9 Statistical analyses
Statistical analyses were used to test whether OM quantity and biochemical composition (protein, carbohydrate, lipid, and phytopigment concentrations), freshness (the phytopigment-to-biopolymeric C ratio), and degradation rates (sedimentary protein turnover rates) were statistically different between trawled and untrawled sites in the upper 9 cm of sediment cores. The analysis consisted of two orthogonal factors: site (trawled vs. untrawled) and depth in the sediment (five levels: 0–1, 1–3, 3–5, 5–7, 7–9 cm). Permutational analyses of variance (PERMANOVA), in either the univariate (variable by variable) or multivariate contexts, were based on Euclidean distances of previously normalized data using 999 permutations of residuals with unrestricted permutation of raw data (univariate tests) or under a reduced model (multivariate tests) (Anderson, 2001). Since for almost all tests the interaction between factors was significant, we conducted post hoc permutational pairwise comparison tests between trawled and untrawled sites for each sediment layer and among sediment layers for trawled and untrawled sites, separately. Given the restricted number of unique permutations, p values were obtained from Monte Carlo simulations (Anderson and Robinson, 2003). Bi-plots produced after canonical analysis of principal components (CAP) were used to visualize the differences between trawled and untrawled samples in terms of organic matter biochemical composition (Anderson and Willis, 2003). All statistical analyses were performed using the routines included in the PRIMER 6+ software.
Table 1Summary of the main parameters of the trawled and untrawled cores. Data for grain size, CaCO3, OC, TN, and OC∕TN correspond to average values ±1 standard deviation of that layer. MAR: mass accumulation rate; SAR: sediment accumulation rate.

* Section depleted of excess 210Pb, corresponding to sediment deposited before ∼1900.
Bottom trawling in the Gulf of Castellammare is limited to the mid-slope (>500 m), beyond the trawling-ban area. In the main bottom trawling ground, where the trawled core was retrieved, hauls generally follow the contour lines in a W–E direction (Fig. 1). Fishing effort has generally increased since 2007, with a predominating trawling frequency of 1 to 40 hauls per grid cell during that year, which then increased to around 60 to 100 hauls per grid cell since 2013 (Fig. S2). A smaller trawling ground has opened since 2012 towards the eastern side of the gulf, close to Cape Rama (Figs. 1, S2). Although trawling frequency for 2016 was only computed from January to August, this year presented the highest fishing effort in comparison to the previous years.
3.1 Physical characteristics
The untrawled sediment core presented an excess 210Pb concentration profile that extended to 25 cm in depth, with a total inventory of 17 900 ± 900 Bq m−2 (Fig. 2a; Table 1). In the upper 6 cm, excess 210Pb concentrations slightly decreased towards the surface from 372±22 Bq kg−1 to 272±15 Bq kg−1 (Fig. 2a). Below, excess 210Pb concentrations presented a continuous decrease between 6 and 25 cm, from which an average sediment accumulation rate of 0.090±0.003 g cm−2 yr−1, equivalent to 0.151±0.005 cm yr−1 (R2=0.995) (Table 1), was calculated applying the Constant Flux : Constant Sedimentation model (CF : CS; Krishnaswamy et al., 1971). This sedimentation rate was independently validated by 137Cs. Detectable concentrations of 137Cs appeared at 17 cm depth, which was ascribed to the first detonations of thermonuclear weapons in the early 1950s. Above, 137Cs concentrations depicted a broad concentration maximum at 8–13 cm, centered at 10–11 cm. This maximum was attributed to the combined accumulation of the maximum fallout prior to the cessation of nuclear atmospheric testing in 1963 as well as the deposition of 137Cs emitted from the Chernobyl accident in 1986 (Fig. 2a). The deposition of 137Cs from each of these events could not be distinguished due to the low sedimentation rate and the sampling resolution of this core. Concentrations of excess 234Th ranged between 71 and 97 Bq kg−1 between 2 and 5 cm, decreased to 37±7 Bq kg−1 at 1–2 cm and was not detected on surface sediments (0–1 cm) of the core (Fig. 2b). The penetration depth of excess 234Th could be greater than the upper 5 cm analyzed, leading to an inventory of at least 1080 Bq m−2 (Table 1). Dry bulk density of the untrawled site remained constant in the upper 7 cm at ∼0.5 g cm−3, gradually increased to ∼0.7 g cm−3 at 15 cm, and then remained relatively constant with depth (Fig. 2c). The upper 6 cm presented coarser grain size consisting of higher silt (77 %) and lower clay (22 %) fractions, in comparison to the rest of the core, which had lower silt (44 %) and higher clay (54 %) fractions (Fig. 2c, Table 1). CaCO3 concentrations were constant in the upper 10 cm, with an average concentration of 18.4±0.4 %, decreased to ∼15 % at 15–20 cm, and then slightly increased with depth until ∼20 % at 30 cm (Fig. 4d).
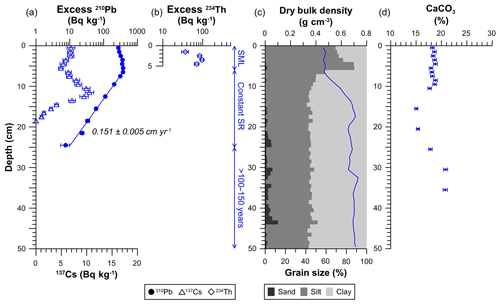
Figure 2Sedimentological and geochemical characteristics of the untrawled sediment core. (a) Concentration profiles of excess 210Pb (circles), indicating the average sedimentation rate below the upper 6 cm, and 137Cs (triangles). (b) Concentration profile of excess 234Th in the upper 5 cm. (c) Dry bulk density and grain size. (d) CaCO3 concentration profile. The analytical error is given in horizontal error bars. Vertical scales indicate the process that has occurred in the depth range given. SML: surface mixed layer; SR: sedimentation rate.
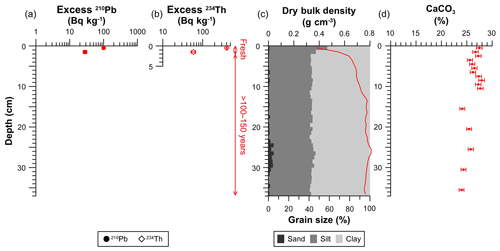
Figure 3Sedimentological and geochemical characteristics of the trawled sediment core. (a) Concentration profiles of excess 210Pb. (b) Concentration profile of excess 234Th in the upper 5 cm. (c) Dry bulk density and grain size profile. (d) CaCO3 concentration profile. The analytical error is given in horizontal error bars. Vertical scales indicate the process that has occurred in the depth range given. SML: Surface Mixed Layer; SR: Sedimentation Rate.
In the sediment core collected from the trawled site, both excess 210Pb and excess 234Th were only present in the upper 2 cm, with inventories of 340±30 and 1000±50 Bq m−2, respectively (Table 1, Fig. 3a, b). 137Cs was not detected in the upper 5 cm analyzed. Dry bulk density rapidly increased from 0.4 to 0.7 g cm−3 in the upper 3 cm and kept increasing to ∼0.8 g cm−3 at 13 cm, from where it remained relatively constant with depth (Fig. 3c). Despite the slight coarsening of grain size in the upper 2 cm, this core presented constant grain size along the upper 37 cm, consisting of higher clay (57 %) and lower silt (41 %) fractions (Fig. 3c, Table 1), similar to grain sizes below 6 cm of the untrawled site. Concentrations of CaCO3 averaged at 26.9±0.7 % in the upper 10 cm, whereas the deeper layers had lower concentrations of ∼24 % (Fig. 3d).
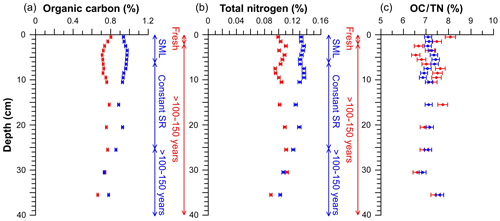
Figure 4Profiles from elemental analyses of untrawled (blue) and trawled (red) cores. Organic carbon (a), total nitrogen (b), and the OC∕TN ratio (c). The analytical error is represented by error bars. Vertical scales indicate the process that has occurred in the depth range given for both untrawled (blue) and trawled (red) sediment cores. SML: surface mixed layer; SR: sedimentation rate.
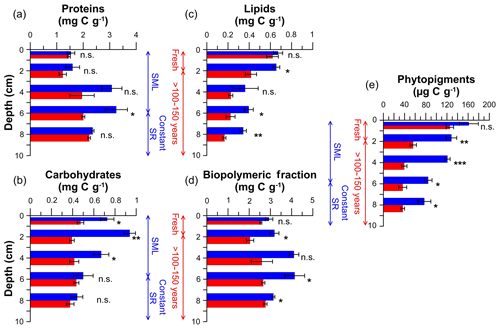
Figure 5Organic matter quantity of untrawled (blue) and trawled (red) sites. Proteins (a), carbohydrates (b), lipids (c), biopolymeric carbon (d), and phytopigments (e). Bar graphs and error bars illustrate the mean values and standard errors at each site and depth of the triplicate sediment cores collected. Asterisks next to bars denote significant difference of post hoc permutational pairwise tests between trawled and untrawled sites: ; ; ; n.s. denotes not significant. Vertical scales indicate the process that has occurred in the depth range given for both untrawled (blue) and trawled (red) sediment cores. SML: surface mixed layer; SR: sedimentation rate.
3.2 Sedimentary organic matter
Trawled and untrawled sediment cores presented different OC and TN concentration profiles (Fig. 4). The untrawled core had OC concentrations of ∼0.9 % in the upper 20 cm, which then decreased to ∼0.8 % at 35 cm. In contrast, the trawled core presented OC concentrations that fluctuated between ∼0.8 % and ∼0.7 % with depth. Similarly, concentrations of TN in the untrawled core were ∼0.13 % in the upper 20 cm, which then decreased to ∼0.10 % at 35 cm, whereas the trawled core presented TN concentrations varying between ∼0.11 % and ∼0.09 % with depth. For both OC and TN, concentrations in the upper 20 cm were ∼20 % lower at the trawled site in comparison to the untrawled site, reaching similar concentrations at 30 cm in depth (Fig. 4). Profiles of the OC∕TN ratio presented similar values at both sites, varying between 6.5 and 7.5 throughout the core (Fig. 4c).
In general, organic matter quantity was higher at the untrawled site than at the trawled site, with the exception of surface layers, which presented similar concentrations at both sites (Fig. 5). Protein concentrations at the untrawled site were 1.5 mgC g−1 in the upper 3 cm, increased to maximum concentrations of ∼3 mgC g−1 at 3–7 cm, and decreased to 2.4 mgC g−1 at 7–9 cm. The trawled site had similar ∼1.5 mgC g−1 protein concentrations in surface sediment that increased to almost 2.5 mgC g−1 in the deepest layer analyzed (Fig. 5a). Carbohydrate concentrations at the untrawled site presented decreasing concentrations from between 0.7 and 0.9 mgC g−1 in the upper 3 cm to 0.44 mgC g−1 in the deepest layer, whereas the trawled site had constant concentrations of 0.42 mgC g−1 in the upper 9 cm of the trawled site (Fig. 5b). Untrawled and trawled sites had similar lipid concentrations of ∼0.6 mgC g−1 in the topmost sediment layer, decreasing to 0.34±0.03 and 0.17±0.01 mgC g−1 at 7–9 cm of the untrawled and trawled cores, respectively (Fig. 5c). Biopolymeric C concentrations were constant (2.5±0.1 mgC g−1) along the upper 9 cm of the trawled core, whereas concentrations increased in the untrawled core from 2.9±0.2 mgC g−1 in the topmost layer to ∼4 mgC g−1 in the 3–7 cm layer, before returning to similar values observed in the surface layer (Fig. 5d). At both sites, phytopigment profiles showed decreasing trends, with a sharper decrease at the trawled site from 126±7 µgC g−1 in the top layer to relatively constant values of 39±5 µgC g−1 below 3 cm, whereas the untrawled site showed a gradual decrease from 161±18 µgC g−1 in the topmost layer to 77±12 µgC g−1 in the deepest one (Fig. 5e).
Statistical analyses of the data indicated that both sampling site and core depth had significant effects on the quantity of sedimentary OM (Table S1). The post hoc comparison tests (Table S2) demonstrated that OM contents between trawled and untrawled sites were generally statistically different only below the topmost sediment layer (0–1 cm), with the exception of carbohydrates.
3.3 Organic matter biochemical composition, freshness, and turnover rates
Variations in the biochemical composition of sedimentary OM in terms of protein, carbohydrate, lipid, and phytopigment contents were assessed through PERMANOVA testing (Table S3). The results revealed that the biochemical composition of sedimentary OM not only differed between trawled and untrawled sites, but that these differences varied depending on the depth in the core. The consequent post hoc pairwise tests between the trawled and untrawled sites at all sediment layers indicated that the biochemical composition of sedimentary OM, similar to the OM content, varied significantly between trawled and untrawled sites at all depths, excluding surface sediments (Table S4). The bi-plot produced after the canonical analysis of principal components (Fig. 6) revealed that the vertical variations in the biochemical composition of sedimentary OM in the untrawled sediment core are greater than those observed at the trawled site, as observed from the greater spatial distribution of these samples. However, the proximity of superficial samples of trawled cores to most of the untrawled samples in the bi-plot illustrates a resemblance in their biochemical composition.
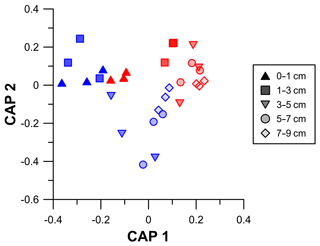
Figure 6Variations in the biochemical composition of the sedimentary organic matter. Bi-plot after canonical analysis of the principal coordinates. Note that symbols represent the same core depth for both trawled (red) and untrawled (blue) sites and that increasing depth is also illustrated by a fading filling.
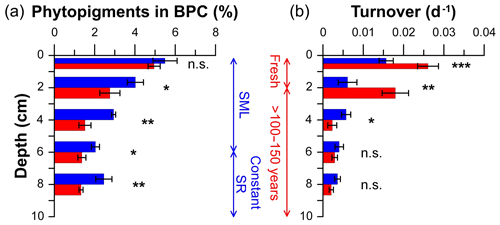
Figure 7Organic matter freshness and degradation rates of trawled (red) and untrawled (blue) cores. Relative contribution of phytopigments to biopolymeric C (a) and protein turnover rates (b). Bar graphs and error bars illustrate the mean values and standard errors at each site and depth of the triplicate sediment cores collected. Asterisks next to bars denote significance of post hoc permutational pairwise tests between trawled and untrawled sites: ; ; ; n.s. denotes not significant. Vertical scales indicate the process that has occurred in the depth range given for both untrawled (blue) and trawled (red) sediment cores. SML: surface mixed layer; SR: sedimentation rate.
The contributions of phytopigment to biopolymeric C and the protein turnover rates at trawled and untrawled sites are given in Table S5 and illustrated in Fig. 7. The relative contribution of phytopigments to biopolymeric C was similar in superficial sediments of trawled and untrawled sites, with relatively high values (∼5 %) that decreased with depth at both sites, although more pronouncedly at the trawled site, reaching ∼1.4 % at 5–7 cm, than at the untrawled site, reaching 2.5 % at 7–9 cm (Fig. 7a). Turnover rates were significantly higher in the upper 3 cm of the trawled site (0.017–0.025 d−1) in comparison to ∼0.015 d−1 of the superficial sediment of the untrawled site (Table S5b, Fig. 7b). Below, turnover rates decreased to ∼0.005 d−1 for both sites.
4.1 Long-term impacts of intense bottom trawling
Evidence of the long-term impacts of intense bottom trawling are clear at a trawled site of the Gulf of Castellammare. ROV images from the trawled area present a barren seafloor, with several deep linear furrows hundreds of meters long and up to 70 cm deep, presumably caused by the trawling gear's heavy otter doors (Fig. S1a). The shallow penetration depth of excess 210Pb in the trawled core suggests that only the upper 2 cm of sediment had been recently deposited on top of highly compacted sediments (0.7–0.8 g cm−3) that had accumulated more than 100 years ago, considering the absence of excess 210Pb (Fig. 3a, c). These sedimentological patterns are characteristic of a seafloor eroded by trawling activities, as observed in other trawled regions with shallow horizons of excess 210Pb and exposed over-consolidated, century-old sediments (Martín et al., 2014b; Oberle et al., 2016; Paradis et al., 2017, 2018). The uprooting of old sediment at this trawled site, with only a thin accumulation of recent sediment in superficial layers, reveals that the rate of sediment erosion induced by the high trawling frequency is greater than sediment accumulation rates in the Gulf of Castellammare. Furthermore, the coincident penetration depth of both excess 210Pb and excess 234Th at the trawled site (Fig. 3a, b) indicates that the accumulation of fresh sediment would have occurred after the passage of the last trawler over the sampling area and thus reveals that trawling frequency controls the residence time of fresh sediment in these trawling grounds.
In contrast, excess 210Pb inventories were 2 orders of magnitude higher in the untrawled core. This core had a sedimentation rate of 0.151±0.005 cm yr−1, comparable to those quantified at similar depths in the Mediterranean Sea (DeGeest et al., 2008; Miralles et al., 2005; Sanchez-Cabeza et al., 1999). The upper 6 cm of this core had excess 210Pb concentrations that slightly decreased towards the surface, a deep penetration depth of excess 234Th, and low dry bulk densities (Fig. 2a, b), altogether signs of biological mixing (Arias-Ortiz, et al., 2018; Pope et al., 1996; van Weering et al., 1998). The influence of bioturbation at this site is corroborated by the presence of several burrows directly observed during ROV dives prior to sampling (Fig. S1b). Sediment mixing caused by bioturbation could explain the broad 137Cs concentration maximum observed at 8–13 cm, attributed to the combined accumulation of 137Cs from the 1986 Chernobyl accident and from the 1963 global fallout, as well as diluting the 1954 signal with depth (Fig. 2a). However, bioturbation alone cannot account for the coarsening of sediment observed in the upper 6 cm (Fig. 2c; Table 1). Provided the high capacity of bottom trawling gear to resuspend sediments (Martin et al., 2014a, c; Oberle et al., 2018; Puig et al., 2012), the siltation of superficial sediments on the untrawled site could be explained by the preferential deposition of siltier particles resuspended from an adjacent trawling ground located ∼1 km up-current from this sampled site (Fig. 1). Finer clay particles resuspended by bottom trawlers can be advected to farther distances along the margin (Linders et al., 2017). This siltier sediment would have been posteriorly redistributed and preserved within the surface mixed layer of the untrawled site (Fig. 1).
Excluding the fresh superficial layers of the trawled site, the chronic erosion induced by bottom trawling resuspension considerably depleted the trawled site of both OC and TN by ∼20 % in comparison to the untrawled site (Fig. 4; Table 1). Concentrations of OC and TN were similar in the deeper layers of both sites, where sediments would have accumulated more than a century ago, revealing that bottom trawling re-exposes old sediment impoverished in OM (Fig. 4; Table 1). Similarly, the trawled site had lower proteins (−5 % to −38 %), carbohydrates (−13 % to −58 %), lipids (−36 % to −52 %), biopolymeric carbon (−12 % to −37 %), and phytopigments (−53 % to −67 %) than the untrawled site, with the exception of superficial layers (Fig. 5). These results are in accordance with previous studies that showed comparable losses of organic matter in slope trawling grounds, reinforcing the concept that chronic and intensive bottom trawling depletes these deep-sea sedimentary habitats of organic matter, promoting their degradation over time (Martín et al., 2014b; Pusceddu et al., 2014; Sañé et al., 2013).
4.2 Effects of the arrival of fresh sediment
During the ISLAND cruise, ROV dives showed high settling fluxes of large particulate matter aggregates to the seafloor (Fig. S1). At both sampled sites, evidence of recent accumulation of surface sediments was provided by the presence of excess 234Th and high concentrations of phytopigments (Fig. 5e), a compound that usually represents the most important food source for deep-sea heterotrophic consumption (Pusceddu et al., 2010; Stephens et al., 1997). This indicates that the Gulf of Castellammare was receiving highly nutritious organic matter inputs during the sampling period. Indeed, both the composition of sedimentary organic matter and the relative contribution of phytopigment to biopolymeric C were similar in the fresh superficial sediments at both trawled and untrawled sites. However, the subsurface, century-old sediments of the trawled site have distinctively different organic matter composition and significantly lower nutritional quality in comparison to its untrawled counterpart (Figs. 6, 7a). This suggests that, aside from the ephemeral deposition of fresh OM that will be swiftly eroded by bottom trawlers' gear, deep-sea trawling grounds are generally characterized by nutritionally poor organic matter contents (Pusceddu et al., 2014; Sañé et al., 2013), which increases the dependence on the supply of fresh OM in order to sustain benthic communities. This hypothesis is corroborated by the higher OM turnover rates in surface sediment of the trawled site in comparison to the untrawled site (Fig. 7b), which reveal a prompt stimulation of microbial activities resulting from the recent accumulation of fresh and nutritionally enriched OM on the trawled site. In fact, benthic communities in areas that have severe nutrient limitations, such as in eroded sediment of this trawled site or in oligotrophic deep-sea regions, react instantaneously to food pulses (Bett et al., 2001; Fabiano et al., 2001; Witte et al., 2003). In contrast, the untrawled site is characterized by relatively higher total organic matter contents as well as fresh and bioavailable compounds throughout the core, possibly due to a deeper penetration of labile material through biological mixing (Fig. 5). However, these sections also presented slowly decreasing concentrations of phytopigment to biopolymeric C with depth as well as lower protein turnover rates, revealing that this unstressed environment has a lower consumption of labile OM and a relatively reduced dependence on the arrival of fresh OM (Fig. 7a).
The higher OM turnover rates observed in the freshly deposited surface layers of the trawled site also indicate a greater efficiency of OM consumption and remineralization in the trawling ground in comparison to the untrawled site. This enhanced mineralization through self-priming can occur when fresh and degradable organic matter, such as fresh phytoplankton, arrives to areas with more refractory compounds (Canfield, 1994; van Nugteren et al., 2009), as was observed in a shallow trawling-disturbed area in the southern North Sea, off the Belgian coast (van de Velde et al., 2018). Similarly, bottom trawling in the shallow Thermaikos Gulf (Aegean Sea) intensified microbial activities, which enhanced nutrient cycling and organic carbon mineralization (Polymenakou et al., 2005; Pusceddu et al., 2005a). This could have been attributed to the combined effect of trawling-induced mixing of superficial labile OM with more degraded subsurface OM, along with the continuous arrival of fresh OM to these shallow continental shelves (Buscail et al., 1990; Tselepides et al., 2000).
In contrast, surface sediments collected in a deeper and intensely trawled flank of La Fonera Canyon, in the NW Mediterranean margin, presented significantly lower turnover rates than the nearby untrawled grounds (Pusceddu et al., 2014). However, those sediment cores did not present signs of recently accumulated sediment as observed at our sampling sites, further proving the dependence on the arrival of fresh and nutritionally rich sediment in intensely trawled grounds to support benthic organisms living in these impacted deep-sea environments.
The high nutritional quality and OM turnover rates in recently accumulated sediments from the trawled site suggest that high OM fluxes could help deep bottom trawling grounds recover the nutritional characteristics of sedimentary OM. These results confirm that actions aimed at mitigating the impacts of bottom trawling include the implementation of temporary fishing closures, allowing for a longer-lived deposition of fresh OM on the seafloor. However, such temporary trawling closures would most probably not allow the full restoration of fresh sediment from trawl-induced erosion, given the low sedimentation rates found in these deep environments. Further management strategies would need to be implemented to mitigate the impacts of bottom trawling erosion (Depestele et al., 2018), which would magnify the effect of temporary closures on the restoration of sedimentary OM in nutrient-deprived trawling grounds.
Chronic and intense deep bottom trawling in the Gulf of Castellammare erodes large volumes of sediment, exposing century-old, compacted sediment that is depleted in OM. This continuous erosion limits the accumulation of fresh sediment since any recently deposited particulate matter is promptly removed due to the high trawling frequency. Nevertheless, we present evidence that the short-lived deposition of recent and nutritionally rich organic matter leads to high turnover rates of labile OM. Our results emphasize that nutrient-deprived and eroded deep bottom trawling grounds are highly dependent on the arrival of fresh and nourishing particulate organic matter to sustain benthic communities, which can temporarily and partially abate the detrimental effects of bottom trawling in superficial sediment.
All data included in this study are available upon request by contact with the corresponding author.
The supplement related to this article is available online at: https://doi.org/10.5194/bg-16-4307-2019-supplement.
SP, AP, PP, and CLI designed the scientific study. SP and CLI retrieved the samples. SP and DM performed the analyses and TR processed the fishing effort data. SP wrote the paper. All authors contributed to the interpretation and discussion of the results, as well as the revision of the paper.
The authors declare that they have no conflict of interest.
The results presented in this study were obtained within the Exploring SiciLian CAnyoN Dynamics (ISLAND) project and the Assessment of Bottom-trawling Impacts in DEep-sea Sediments (ABIDES) Spanish Research Project. We would like to thank the crew of the R/V Ángeles Alvariño and the ISLAND cruise team that helped collect the samples. This work is contributing to the Institut de Ciència i Tencologia Ambientals “Unit of Excellence”. Sarah Paradis is supported by a predoctoral FPU grant from the Spanish government.
This research has been supported by the European Commission, Seventh Framework Programme (EUROFLEETS2 (grant no. 312762)), the Ministerio de Economía y Competitividad (grant no. CTM2015-65142-R), the Generalitat de Catalunya (2017 SGR-863 and 1588), the Australian Research Council (LIEF project (grant no. LE170100219)), the Ministerio de Economía y Competitividad (grant no. MDM2015-0552), and the Ministerio de Educación, Cultura y Deporte (grant no. FPU14/07039).
This paper was edited by Jack Middelburg and reviewed by Sebastiaan van de Velde and Xavier Durrieu de Madron.
Amoroso, R. O., Pitcher, C. R., Rijnsdorp, A. D., McConnaughey, R. A., Parma, A. M., Suuronen, P., Eigaard, O. R., Bastardie, F., Hintzen N. T., Althaus, F., Baird, S. J., Black, J., Buhl-Mortensen, L., Campbell, A. B., Catarino, R., Collie, J. S., Cowan, J. H., Durholtz, D., Engstrom, N., Fairweather, T. P., Fock, H. O., Ford, R., Gálvez, P. A., Gerritsen, H. D., Góngora, M. E., González, J., Hiddink, J. G., Hughes, K. M., Intelmann, S. S., Jenkins, C., Jonsson, P., Kainge, P., Kangas, M., Kathena, J. N., Kavadas, S., Leslie, R. W., Lewis, S. G., Lundy, M., Makin, D., Martin, J., Mazor, T., Mirelis, G. G., Newman, S. J., Papadopoulou, K. N., Posen, P. E., Rochester, W., Russo, T., Sala, A., Semmens, J. M., Silva, C., Tsolos, A., Vanelsander, B., Wakefield, C. B., Wood, B. A., Hilborn, R., Kaiser, M. J., and Jennings, S.: Bottom trawl-fishing footprints on the world's continental shelves, P. Natl. Acad. Sci. USA, 115, E10275–E10282, https://doi.org/10.1073/pnas.1802379115, 2018.
Anderson, M. J.: A new method for non-parametric multivariate analysis of variance, Austral Ecol., 26, 32–46, https://doi.org/10.1111/j.1442-9993.2001.01070.pp.x, 2001.
Anderson, M. J. and Robinson, J.: Generalized discriminant analysis based on distances, Aust. N.Z. J. Stat., 45, 301–318, https://doi.org/10.1111/1467-842X.00285, 2003.
Anderson, M. J. and Willis, T. J.: Canonical analysis of principal coordinates: A useful method of constrained ordination for ecology, Ecology, 84, 511–525, https://doi.org/10.1890/0012-9658(2003)084[0511:CAOPCA]2.0.CO;2, 2003.
Arculeo, M., Lo Brutto, S., Cannizzaro, L., and Vitale, S.: Growth and reproduction of the deep-water rose shrimp, Parapenaeus longirostris (Lucas, 1846) (Decapoda, Penaeidae), in the Southern Tyrrhenian Sea, Crust., 87, 1168–1184, https://doi.org/10.1163/15685403-00003334, 2014.
Arias-Ortiz, A., Masqué, P., Garcia-Orellana, J., Serrano, O., Mazarrasa, I., Marbà, N., Lovelock, C. E., Lavery, P. S., and Duarte, C. M.: Reviews and syntheses: 210Pb-derived sediment and carbon accumulation rates in vegetated coastal ecosystems – setting the record straight, Biogeosciences, 15, 6791–6818, https://doi.org/10.5194/bg-15-6791-2018, 2018.
Arjona-Camas, M., Puig, P., Palanques, A., Emelianov, M., and Durán, R.: Evidence of trawling-induced resuspension events in the generation of nepheloid layers in the Foix submarine canyon (NW Mediterranean), J. Marine Syst., 196, 86–96, https://doi.org/10.1016/j.jmarsys.2019.05.003, 2019.
Bett, B. J., Malzone, M. G., Narayanaswamy, B., and Wigham, B. D.: Temporal variability in phytodetritus and megabenthic activity at the seabed in the deep Northeast Atlantic, Prog. Oceanogr., 50, 349–368, https://doi.org/10.1016/S0079-6611(01)00066-0, 2001.
Bligh, E. G. and Dyer, W. J.: A rapid method of total lipid extraction and purification, Can. J. Biochem Phys., 37, 911–917, https://doi.org/10.1139/o59-099, 1959.
Buscail, R., Pocklington, R., Daumas, R., and Guidi, L.: Fluxes and budget of organic matter in the benthic boundary layer over the northwestern Mediterranean margin, Cont. Shelf Res., 10, 1089–1122, https://doi.org/10.1016/0278-4343(90)90076-X, 1990.
Calvo, S. and Genchi, G.: Carico organico ed effete eutrofici nel Golfo di Castellammare (Sicilia nord-occidentale), OEbalia, 15, 397–408, 1989.
Canfield, D. E.: Factors influencing organic carbon preservation in marine sediments, Chem. Geol., 114, 315–329, https://doi.org/10.1016/0009-2541(94)90061-2, 1994.
Danovaro, R.: Methods for the study of deep-sea sediments, their functioning and biodiversity, edited by: Danovaro, R., CRC Press, Boca Raton, 2010.
Decreto 1339/2001: DECRETO 11 luglio 2001 – Disposizioni relative all'interruzione tecnica delle attività di pesca nei compartimenti marittimi siciliani per il triennio 2001/2003, Gazzetta Ufficiale della Regione Siciliana, Palermo, Italy, 2001.
DeGeest, A. L., Mullenbach, B. L., Puig, P., Nittrouer, C. A., Drexler, T. M., Durrieu de Madron, X., and Orange, D. L.: Sediment accumulation in the western Gulf of Lions, France: the role of Cap de Creus Canyon in linking shelf and slope sediment dispersal systems, Cont. Shelf Res., 28, 2031–2047, https://doi.org/10.1016/j.csr.2008.02.008, 2008.
Dellapenna, T. M., Allison, M. A., Gill, G. A., Lehman, R. D., and Warnken, K. W.: The impact of shrimp trawling and associated sediment resuspension in mud dominated, shallow estuaries, Estuar. Coast. Shelf Sc., 69, 519–530, https://doi.org/10.1016/j.ecss.2006.04.024, 2006.
Depestele, J., Degrendele, K., Esmaeili, M., Ivanović, A., Kröger, S., O'Neill, F. G., Parker, R., Polet, H., Roche, M., Teal, L. R., Vanelslander, B., and Rijnsdorp, A. D.: Comparison of mechanical disturbance in soft sediments due to tickler-chain SumWing trawl vs. electro-fitted PulseWing trawl, ICES J Mar. Sci., 76, 312–329, https://doi.org/10.1093/icesjms/fsy124, 2018.
Durrieu de Madron, X., Ferré, B., Le Corre, G., Grenz, C., Conan, P., Pujo-Pay, M., Buscail, R., and Bodiot, O.: Trawling-induced resuspension and dispersal of muddy sediments and dissolved elements in the Gulf of Lion (NW Mediterranean), Cont. Shelf Res., 25, 2387–2409, https://doi.org/10.1016/j.csr.2005.08.002, 2005.
Eigaard, O. R., Bastardie, F., Niels, T., Hintzen, T., Buhl-Mortensen, L., Buhl-Mortensen, M., Catarino, R., Dinesen, G. E., Egekvist, J., Fock, H., Geitner, K., Gerritsen, H., Marín González, M., Jonsson, P., Kavadas, S., Laffargue, P., Lundy, M., Mirelis, G. G., Rasmus Nielsen, J., Papadopoulou, N., Posen, P. E., Pulcinella, J., Russo, T., Sala, A., Silva, C., Smith, C. J., Vanelslander, B., and Rijnsdorp, A. D.: The footprint of bottom trawling in European waters: distribution, intensity, and seabed integrity, ICES J. Mar. Sci., 74, 847–865, https://doi.org/10.1093/icesjms/fsw194, 2017.
European Commission: Commission Regulation (EC) No. 2244/2003 of 18 December 2003 laying gown detailed provisions regarding satellite-based Vessel Monitoring Systems, Off. J. Eur. Union, L333, 17–27, 2003.
European Comission Fisheries & Maritime Affairs: Fleet Register on the Net, available at: http://ec.europa.eu/fisheries/fleet/index.cfm and https://ec.europa.eu/fisheries/cfp/fishing_rules/fishing_fleet_en (last access: 31 January 2018), 2014.
Fabiano, M. and Danovaro, R.: Enzymatic activity, bacterial distribution, and organic matter composition in sediments of the ross sea (Antarctica), Appl. Environ. Microb., 64, 3838–3845, 1998.
Fabiano, M., Danovaro, R., and Fraschetti, S.: A three-year time series of elemental and biochemical composition of organic matter in subtidal sandy sediments of the Ligurian Sea (north western Mediterranean), Cont. Shelf Res., 15, 1453–1469, 1995.
Fabiano, M., Pusceddu, A., Dell'Anno, A., Armeni, M., Vanucci, S., Lampitt, R. S., Wolff, G. A., and Danovaro, R.: Fluxes of phytopigments and labile organic matter to the deep ocean in the NE Atlantic Ocean, Prog. Oceanogr., 50, 89–104, 2001.
Fanelli, E., Cartes, J. E., Badalamenti, F., Rumolo, P., and Sprovieri, M.: Trophodynamics of suprabenthic fauna on coastal muddy bottoms of the southern Tyrrhenian Sea (western Mediterranean), J. Sea Res., 61, 174–187, https://doi.org/10.1016/j.seares.2008.10.005, 2008.
Ferré, B., Durrieu de Madron, X., Estournel, C., Ulses, C., and Le Corre, G.: Impact of natural and anthropogenic (trawl) resuspension on the export of particulate matter to the open ocean. Application to the Gulf of Lion (NW Mediterranean), Cont. Shelf Res., 28, 2071–2091, https://doi.org/10.1016/j.csr.2008.02.002, 2008.
Gerchakov, S. M. and Hatcher, P. G.: Improved technique for analysis of carbohydrates in sediments, Limnol. Oceanogr., 17, 938–943, https://doi.org/10.4319/lo.1972.17.6.0938, 1972.
Hale, R., Godbold, J. A., Sciberras, M., Dwight, J., Wood, C., Hiddink, J. G., and Solan, M.: Mediation of macronutrients and carbon by post-disturbance shelf sea sediment communities, Biogeochemistry, 135, 121–133, https://doi.org/10.1007/s10533-017-0350-9, 2017.
Hartree, E. F.: Determination of protein: A modification of the lowry method that gives a linear photometric response, Anal. Biochem., 48, 422–427, https://doi.org/10.1016/0003-2697(72)90094-2, 1972.
Kaiser, M. J.: Significance of Bottom-Fishing Disturbance, Conserv. Biol., 12, 1230–1235, https://doi.org/10.1046/j.1523-1739.1998.0120061230.x, 1998.
Kaiser, M. J., Collie, J. S., Hall, S. J., Jennings, S., and Poiner, I. R.: Modification of marine habitats by trawling activities: prognosis and solutions, Fish Fish., 3, 114–136, https://doi.org/10.1046/j.1467-2979.2002.00079.x, 2002.
Koslow, J., Boehlert, G. W., Gordon, J. D. M., Haedrich, R. L., Lorance, P., and Parin, N.: Continental slope and deep-sea fisheries: implications for a fragile ecosystem, ICES J. Mar. Sci., 57, 548–557, https://doi.org/10.1006/jmsc.2000.0722, 2000.
Krishnaswamy, S., Lal, D., Martin, J. M., and Meybeck, M.: Geochronology of lake sediments, Earth Planet. Sc. Lett., 11, 407–414, https://doi.org/10.1016/0012-821X(71)90202-0, 1971.
Linders, T., Nilsson, P., Wikström, A., and Sköld, M.: Distribution and fate of trawling-induced suspension of sediments in a marine protected area, ICES J. Mar. Sci., 75, 785–795, https://doi.org/10.1093/icesjms/fsx196, 2017.
Lo Iacono, C., Sulli, A., and Agate, M.: Submarine canyons of north-western Sicily (Southern Tyrrhenian Sea): Variability in morphology, sedimentary processes and evolution on a tectonically active margin, Deep-Sea Res. Pt. II, 104, 93–105, https://doi.org/10.1016/J.DSR2.2013.06.018, 2014.
Lorenzen, C. and Jeffrey, J.: Determination of chlorophyll in seawater, Technical Paper in Marine Science (UNESCO), Technical Papers in Marine Science, UNESCO, Paris, France, 35, 1–20, 1980.
L. R. 1/1980: Provvedimenti per la razionalizzazione della pesca in Sicilia, Gazzetta Ufficiale della Regione Siciliana, Palermo, Italy, 1980.
L. R. 25/1990: Modificazioni e integrazioni della legislazione regionale in materia di pesca, Gazzetta Ufficiale della Regione Siciliana, Palermo, Italy, 1990.
L. R. 26/1987: Interventi nel settore della pesca, Gazzetta Ufficiale della Regione Siciliana, Palermo, Italy, 1987.
Marsh, J. B. and Weinstein, D. B.: Simple charring method for determination of lipids, J. Lipid Res., 7, 574–576, 1966.
Martín, J., Puig, P., Palanques, A., and Giamportone, A.: Commercial bottom trawling as a driver of sediment dynamics and deep seascape evolution in the Anthropocene, Anthropocene, 7, 1–15, https://doi.org/10.1016/j.ancene.2015.01.002, 2014a.
Martín, J., Puig, P., Masqué, P., Palanques, A., and Sánchez-Gómez, A.: Impact of bottom trawling on deep-sea sediment properties along the flanks of a submarine canyon, PLoS One, 9, e104536, https://doi.org/10.1371/journal.pone.0104536, 2014b.
Martín, J., Puig, P., Palanques, A., and Ribó, M.: Trawling-induced daily sediment resuspension in the flank of a Mediterranean submarine canyon, Deep-Sea Res. Pt. II, 104, 174–183, https://doi.org/10.1016/j.dsr2.2013.05.036, 2014c.
Miralles, J., Radakovitch, O., and Aloisi, J. C.: 210Pb sedimentation rates from the Northwestern Mediterranean margin, Mar. Geol., 216, 155–167, https://doi.org/10.1016/j.margeo.2005.02.020, 2005.
Morato, T., Watson, R., Pitcher, T. J., and Pauly, D.: Fishing down the deep, Fish Fish., 7, 24–34, 2006.
Nieuwenhuize, J., Maas, Y. E. M., and Middelburg, J. J.: Rapid analysis of organic carbon and nitrogen in particulate materials, Mar. Chem., 45, 217–224, 1994.
Oberle, F. K. J., Swarzenski, P. W., Reddy, C. M., Nelson, R. K., Baasch, B., and Hanebuth, T. J. J.: Deciphering the lithological consequences of bottom trawling to sedimentary habitats on the shelf, J. Marine Syst., 159, 120–131, https://doi.org/10.1016/j.jmarsys.2015.12.008, 2016.
Oberle, F. K. J., Puig, P., and Martín, J.: Fishing Activities, 503–534, Springer, Cham., 2018.
O'Neill, F. G. and Ivanović, A.: The physical impact of towed demersal fishing gears on soft sediments, ICES J. Mar. Sci. J. Cons., 73, i5–i14, https://doi.org/10.1093/icesjms/fsv125, 2016.
Palanques, A., Puig, P., Guillén, J., Demestre, M., and Martín, J.: Effects of bottom trawling on the Ebro continental shelf sedimentary system (NW Mediterranean), Cont. Shelf Res., 72, 83–98, https://doi.org/10.1016/j.csr.2013.10.008, 2014.
Paradis, S., Puig, P., Masqué, P., Juan-Díaz, X., Martín, J., and Palanques, A.: Bottom-trawling along submarine canyons impacts deep sedimentary regimes, Sci. Rep.-UK, 7, 43332, https://doi.org/10.1038/srep43332, 2017.
Paradis, S., Sanchez-Vidal, A., Masqué, P., Garcia-Orellana, J., Calafat, A., and Canals, M.: Spatial distribution of sedimentation-rate increases in Blanes Canyon caused by technification of bottom trawling fleet, Prog. Oceanogr., 169, 241–252, https://doi.org/10.1016/J.POCEAN.2018.07.001, 2018.
Pauly, D., Christensen, V., Guénette, S., Pitcher, T. J., Sumaila, U. R., Walters, C. J., Watson, R., and Zeller, D.: Towards sustainability in world fisheries, Nature, 418, 689–695, https://doi.org/10.1038/nature01017, 2002.
Pipitone, C., Badalamenti, F., D'Anna, G., and Patti, B.: Fish biomass increase after a four-year trawl ban in the Gulf of Castellammare (NW Sicily, Mediterranean Sea), Fish. Res., 48, 23–30, https://doi.org/10.1016/S0165-7836(00)00114-4, 2000.
Polymenakou, P. N., Pusceddu, A., Tselepides, A., Polychronaki, T., Giannakourou, A., Fiordelmondo, C., Hatziyanni, E., and Danovaro, R.: Benthic microbial abundance and activities in an intensively trawled ecosystem (Thermaikos Gulf, Aegean Sea), Cont. Shelf Res., 25, 2570–2584, https://doi.org/10.1016/J.CSR.2005.08.018, 2005.
Pope, R. H., Demaster, D. J., Smith, C. R., and Seltmann, H.: Rapid bioturbation in equatorial Pacific sediments: evidence from excess 234Th measurements, Deep-Sea Res. Pt. II, 43, 1339–1364, https://doi.org/10.1016/0967-0645(96)00009-4, 1996.
Puig, P., Canals, M., Company, J. B., Martín, J., Amblas, D., Lastras, G., and Palanques, A.: Ploughing the deep sea floor, Nature, 489, 286–289, https://doi.org/10.1038/nature11410, 2012.
Puig, P., Martín, J., Masqué, P., and Palanques, A.: Increasing sediment accumulation rates in La Fonera (Palamós) submarine canyon axis and their relationship with bottom trawling activities, Geophys. Res. Lett., 42, 8106–8113, https://doi.org/10.1002/2015GL065052, 2015.
Pusceddu, A., Fiordelmondo, C., Polymenakou, P., Polychronaki, T., Tselepides, A., and Danovaro, R.: Effects of bottom trawling on the quantity and biochemical composition of organic matter in coastal marine sediments (Thermaikos Gulf, northwestern Aegean Sea), Cont. Shelf Res., 25, 2491–2505, https://doi.org/10.1016/j.csr.2005.08.013, 2005a.
Pusceddu, A., Grémare, A., Escoubeyrou, K., Amouroux, J. M., Fiordelmondo, C., and Danovaro, R.: Impact of natural (storm) and anthropogenic (trawling) sediment resuspension on particulate organic matter in coastal environments, Cont. Shelf Res., 25, 2506–2520, https://doi.org/10.1016/j.csr.2005.08.012, 2005b.
Pusceddu, A., Bianchelli, S., Canals, M., Sanchez-Vidal, A., Durrieu de Madron, X., Heussner, S., Lykousis, V., deStigter, H., Trincardi, F., and Danovaro, R.: Organic matter in sediments of canyons and open slopes of the Portuguese, Catalan, Southern Adriatic and Cretan Sea margins, Deep-Sea Res. Pt. I, 57, 441–457, https://doi.org/10.1016/j.dsr.2009.11.008, 2010.
Pusceddu, A., Bianchelli, S., Martín, J., Puig, P., Palanques, A., Masqué, P., and Danovaro, R.: Chronic and intensive bottom trawling impairs deep-sea biodiversity and ecosystem functioning, P. Natl. Acad. Sci. USA, 111, 8861–8866, https://doi.org/10.1073/pnas.1405454111, 2014.
Pusceddu, A., Bianchelli, S., and Danovaro, R.: Quantity and biochemical composition of particulate organic matter in a highly trawled area (Thermaikos Gulf, Eastern Mediterranean Sea), Adv. Oceanogr. Limnol., 6, 21–32, https://doi.org/10.4081/aiol.2015.5448, 2015.
Regione Siciliana: Piano di tutela delle acque della Sicilia, SOGESID, Palermo, Sicilia, 243 pp., 2007.
Rice, D. L.: The Detritus Nitrogen Problem: New Observations and Perspectives from Organic Geochemistry, Mar. Ecol.-Prog. Ser., 9, 153–162, 1982.
Roberts, C. M.: Deep impact: The rising toll of fishing in the deep sea, Trends Ecol. Evol., 17, 242–245, 2002.
Romano, C., Fanelli, E., D'Anna, G., Pipitone, C., Vizzini, S., Mazzola, A., and Badalamenti, F.: Spatial variability of soft-bottom macrobenthic communities in northern Sicily (Western Mediterranean): Contrasting trawled vs. untrawled areas, Mar. Environ. Res., 122, 113–125, https://doi.org/10.1016/j.marenvres.2016.10.002, 2016.
Russo, T., Parisi, A., and Cataudella, S.: New insights in interpolating fishing tracks from VMS data for different métiers, Fish. Res., 108, 184–194, https://doi.org/10.1016/j.fishres.2010.12.020, 2011.
Russo, T., Parisi, A., Garofalo, G., Gristina, M., Cataudella, S., and Fiorentino, F.: SMART: A Spatially Explicit Bio-Economic Model for Assessing and Managing Demersal Fisheries, with an Application to Italian Trawlers in the Strait of Sicily, edited by: MacKenzie, B. R., PLoS One, 9, e86222, https://doi.org/10.1371/journal.pone.0086222, 2014.
Sanchez-Cabeza, J. A., Masqué, P., and Ani-Ragolta, I.: Pb-210 and Po-210 analysis in sediments and soils by microwave acid digestion, J. Radioanal. Nucl. Ch., 227, 19–22, https://doi.org/10.1007/BF02386425, 1998.
Sanchez-Cabeza, J. A., Masqué, P., Ani-Ragolta, I., Merino, J., Frignani, M., Alvisi, F., Palanques, A., and Puig, P.: Sediment accumulation rates in the southern Barcelona continental margin (NW Mediterranean Sea) derived from 210Pb and 137Cs chronology, Prog. Oceanogr., 44, 313–332, https://doi.org/10.1016/S0079-6611(99)00031-2, 1999.
Sañé, E., Martín, J., Puig, P., and Palanques, A.: Organic biomarkers in deep-sea regions affected by bottom trawling: pigments, fatty acids, amino acids and carbohydrates in surface sediments from the La Fonera (Palamós) Canyon, NW Mediterranean Sea, Biogeosciences, 10, 8093–8108, https://doi.org/10.5194/bg-10-8093-2013, 2013.
Sarà, G., Scilipoti, D., Milazzo, M., and Modica, A.: Use of stable isotopes to investigate dispersal of waste from fish farms as a function of hydrodynamics, Mar. Ecol.-Prog. Ser., 313, 261–270, https://doi.org/10.3354/meps313261, 2006.
Sciberras, M., Parker, R., Powell, C., Robertson, C., Kröger, S., Bolam, S., and Hiddink, J. G.: Impacts of bottom fishing on the sediment infaunal community and biogeochemistry of cohesive and non-cohesive sediments, Limnol. Oceanogr., 61, 2076–2089, https://doi.org/10.1002/lno.10354, 2016.
Simpson, A. and Watling, A.: An investigation of the cumulative impacts of shrimp trawling on mud-bottom fishing grounds in the Gulf of Maine: effects on habitat and macrofaunal community structure, ICES J. Mar. Sci., 63, 1616–1630, https://doi.org/10.1016/j.icesjms.2006.07.008, 2006.
Stephens, M. P., Kadko, D. C., Smith, C. R., and Latasa, M.: Chlorophyll-a and pheopigments as tracers of labile organic carbon at the central equatorial Pacific seafloor, Geochim. Cosmochim. Ac., 61, 4605–4619, https://doi.org/10.1016/S0016-7037(97)00358-X, 1997
Tiano, J. C., Witbaard, R., Bergman, M. J. N., van Rijswijk, P., Tramper, A., van Oevelen, D., and Soetaert, K.: Acute impacts of bottom trawl gears on benthic metabolism and nutrient cycling, ICES J. Mar. Sci., https://doi.org/10.1093/icesjms/fsz060, online first, 2019.
Trimmer, M., Petersen, J., Sivyer, D. B., Mills, C., Young, E., and Parker, E. R.: Impact of long-term benthic trawl disturbance on sediment sorting and biogeochemistry in the southern North Sea, Mar. Ecol.-Prog. Ser., 298, 79–94, https://doi.org/10.3354/meps298079, 2005.
Tselepides, A., Polychronaki, T., Marrale, D., Akoumianaki, I., Dell'Anno, A., Pusceddu, A., and Danovaro, R.: Organic matter composition of the continental shelf and bathyal sediments of the Cretan Sea (NE Mediterranean), Prog. Oceanogr., 46, 311–344, https://doi.org/10.1016/S0079-6611(00)00024-0, 2000.
van de Velde, S., Van Lancker, V., Hidalgo-Martinez, S., Berelson, W. M., and Meysman, F. J. R.: Anthropogenic disturbance keeps the coastal seafloor biogeochemistry in a transient state, Sci. Rep.-UK, 8, 5582, https://doi.org/10.1038/s41598-018-23925-y, 2018.
van Nugteren, P., Moodley, L., Brummer, G.-J., Heip, C. H. R., Herman, P. M. J., and Middelburg, J. J.: Seafloor ecosystem functioning: the importance of organic matter priming, Mar. Biol., 156, 2277–2287, https://doi.org/10.1007/s00227-009-1255-5, 2009.
van Weering, T. C. E., Hall, I. R., de Stigter, H. C., McCave, I. N., and Thomsen, L.: Recent sediments, sediment accumulation and carbon burial at Goban Spur, N.W. European Continental Margin (47–50∘ N), Prog. Oceanogr., 42, 5–35, https://doi.org/10.1016/S0079-6611(98)00026-3, 1998.
Whitmarsh, D., Carl, J., Pickering, H., Pipitone, C., Badalamenti, F., and D'Anna, G.: Economic Effects of Fisheries Exclusion Zones: A Sicilian Case Study, Mar. Resour. Econ., 17, 239–250, https://doi.org/10.1086/mre.17.3.42629366, 2002.
Wilson, A. M., Kiriakoulakis, K., Raine, R., Gerritsen, H. D., Blackbird, S., Allcocke, A. L., and White, M.: Anthropogenic influence on sediment transport in the Whittard Canyon, NE Atlantic, Mar. Pollut. Bull., 101, 320–329, https://doi.org/10.1016/j.marpolbul.2015.10.067, 2015.
Witte, U., Aberle, N., Sand, M., and Wenzhöfer, F.: Rapid response of a deep-sea benthic community to POM enrichment: an in situ experimental study, Mar. Ecol.-Prog. Ser., 251, 27–36, https://doi.org/10.3354/meps251027, 2003.





