the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Calibration of Mg ∕ Ca and Sr ∕ Ca in coastal marine ostracods as a proxy for temperature
Maximiliano Rodríguez
Christelle Not
The and ratios of marine shells have been widely used in environmental paleoreconstructions to understand past marine conditions. Temperature calibrations to ostracod ratios are known to be species-specific but only available for a few species, despite the large number of known ostracod species. Here, we develop temperature calibrations for two shallow marine ostracods, Sinocytheridea impressa and Neomonoceratina delicata, using modern sediment samples. Our results show that adult specimens of these two species might be useful as a paleothermometer. We observed significant correlations using the ratios of both species to the annual (S. impressa=3.7 ⋅ T−62.7; N. delicata=1.6 ⋅ T−16.8) and April (S. impressa=2.8 ⋅ T−39.2; N. delicata=1.6 ⋅ T−15.7) temperatures. The correlation of temperature to the ratio of S. impressa is more significant and therefore should be preferred for paleoreconstructions. Re-analysis from satellite data allows us to validate our temperature calibration to an extended area around the Pearl River estuary. Our results show that of S. impressa and N. delicata ostracods can be used to reconstruct water temperature at a regional scale, which provides information on the oceanic circulation in coastal areas of the South China Sea. ratios of both species do not correlate with any of the 24 water parameters recorded by the Environmental Protection Department of Hong Kong, including temperature (21.7–24.1 ∘C), salinity (23.8–33.7 PSU), dissolved oxygen (4.3–7.1 mg L−1), suspended solids (1.9–35.4 mg L−1) and pH (7.7–8.2).
- Article
(2238 KB) - Full-text XML
-
Supplement
(2004 KB) - BibTeX
- EndNote
Element calcium ratios () of secreted biogenic calcium carbonate by marine organisms, such as foraminifera and corals, have been used as proxies for past environmental marine parameters (Hendy et al., 2002; Lea, 2003; Lea and Boyle, 1989; Linsley et al., 2000; Martin et al., 2002). in foraminifera and in corals have been frequently used to reconstruct water temperatures (Beck et al., 1992; Cohen et al., 2001, 2002; Lea, 2003; Rosenthal et al., 2006; Sinclair et al., 1998; Yu and Elderfield, 2008). In addition, of ostracod shells has also provided valuable information about water temperatures (Chivas et al., 1983, 1986a; De Deckker and Forester, 1988; Dwyer et al., 2002). The thermodependency of the Mg incorporation into calcite has been observed in natural environments (Cadot and Kaesler, 1977; Corrège and De Deckker, 1997; Cronin et al., 2005a; Dwyer et al., 1995) and culture experiments (Chivas et al., 1986b; Kondo et al., 2005). Several studies have focused their efforts on the development of calibrations for deep water genera, such as Krithe, which are found in the Pacific, Atlantic and Arctic oceans (Corrège and De Deckker, 1997; Cronin et al., 1996; Dwyer et al., 1995, 2002; Elmore et al., 2012; Farmer et al., 2011, 2012; Rodriguez et al., 2020). Other studies have investigated species found in shallow marine environments, such as those from the genus Loxoconcha (Cronin et al., 2003, 2005a) and Cyprideis (Holmes and De Deckker, 2016; Roberts et al., 2020). However, there is uncertainty in temperature calibrations to of marine ostracods due to the low number of calibrations developed in comparison to the number of known species (Holmes and De Deckker, 2012) and the contrasting calibration slopes observed in different species (Yamada et al., 2014). Developing calibrations for new ostracod species can enhance our understanding of the processes controlling ostracod uptake and broaden its use in other areas of the globe (Holmes and De Deckker, 2012; Lea, 2003). The comparison of and with multiple water parameters can also improve our understanding of the variables controlling the incorporation of trace elements into the ostracod carapace. and of ostracod shells have also been used as a proxy for salinity in enclosed water bodies (Chivas et al., 1983) based on two main assumptions: (1) water and water exert a control on shell and shell (Dettman and Dwyer, 2012) and (2) salinity increases simultaneously with the removal of Ca by low-Mg calcite precipitation during evaporation, increasing the water content of Mg relative to Ca (Ito et al., 2003). However, there are several variables that limit their applicability, such as groundwater inputs and the non-equilibrium state of calcite precipitation (Ito et al., 2003). A weak control of water temperature and alkalinity on ratios has also been documented in lacustrine species (De Deckker et al., 1999; Gouramanis and De Deckker, 2010). In spite of this use in lacustrine systems, of ostracod shells does not seem to be related to salinity or temperature in shallow marine environments (Dettman and Dwyer, 2012; Gouramanis and De Deckker, 2010; Ingram, 1998; Roberts et al., 2020). Here, we develop parametric calibrations for and of two geochemically unknown species, Sinocytheridea impressa and Neomonoceratina delicata.
Sinocytheridea impressa and N. delicata are shallow marine ostracods from the superfamily Cytheroidea (Brandão and Karanovic, 2020), which are mainly distributed in Asian waters (Hong et al., 2019; Tanaka et al., 2011). Species of S. impressa and N. delicata have been reported in sediment records from the Miocene and Pleistocene respectively (Irizuki et al., 2005, 2009). The abundance of both species has been used as an indicator of the paleoenvironmental response to sea-level transgressions (Chunlian et al., 2013). A high abundance of both species rarely occurs simultaneously as S. impressa is commonly found in hypoxic environments with low salinity and high turbidity, while N. delicata is common in well-ventilated, polyhaline bays (Hong et al., 2019; Irizuki et al., 2005). Paleoenvironmental investigations have focused on the study of assemblages of these two species but not on the elemental composition of their shells as an indicator of environmental marine variables, such as temperature or salinity. The calibration of ratios of their shells with ocean parameters in an estuary-dominated system may be used to reconstruct shallow marine environments in Asia and complement previous studies on the paleoenvironmental response to sea-level transgressions (Chunlian et al., 2013).
Estuaries are ecosystems with high productivity and biodiversity (Day et al., 2013). Water temperature and salinity are important physical properties of these ecosystems. These parameters are controlled by the combined effects of oceanic currents, upwelling waters, river discharge, and atmospheric forcings such as winds and precipitation. The study of water temperature and salinity can help us understand the evolution of marine currents and global atmospheric patterns, which can improve our understanding of glaciation cycles and sea-level transgressions. For example, of Loxoconcha specimens from Chesapeake Bay has been used as a temperature proxy to evaluate anthropogenic and North Atlantic Oscillation (NAO) impacts on past and present climates (Cronin et al., 2005b). Hong Kong (HK) and the surrounding waters of the Pearl River estuary (PRE) are similar. The local waters are affected by multi-annual oceanic and atmospheric patterns such as El Niño–Southern Oscillation (Niu, 2013; Zhang et al., 2013). The development of a new water temperature and salinity proxy will give us a tool to improve our understanding of past marine conditions as well as better knowledge of oceanic and atmospheric regional patterns. However, the calibration of marine shells with ocean parameters in estuarine systems is challenging due to the highly dynamic chemical and physical variabilities (Snedden et al., 2013) and the combined presence of ostracod populations from different years. Here, we investigate the applicability of ostracod and ratios as proxies for temperature in a freshwater-influenced marine system and several factors that control the robustness of the parametric calibrations including (1) number of shells per site, (2) seasonal variability in ocean parameters, (3) ostracod molting time and (4) the life stage of the ostracods.
2.1 Water parameters
2.1.1 Marine stations
The main dataset consists of measurements of water parameters around the PRE and Hong Kong waters. Water parameters from Hong Kong were obtained from the Environmental Protection Department of Hong Kong (EPD), which records 24 parameters of water quality, including temperature, salinity, pH and dissolved oxygen, around HK (Fig. 1). Monthly single measurements are performed in eight water control zones: Tolo Harbour, Southern Waters, Port Shelter, Junk Bay, Deep Bay, North Western, Mirs Bay, Western Buffer, Eastern Buffer and Victoria Harbour (Fig. S1). The records correspond to one or two daily values per month from 1986 to the present date (EPD, 2018). The sedimentation rate in Hong Kong varies from 0.2 to 5 cm yr−1 (Owen and Lee, 2004; Tanner et al., 2000), which suggests the presence of specimens from different years. For the calibration, we calculated monthly mean values using the last 20 years of data from the collection time of our sediment samples (i.e., 2012) in order to determine a robust monthly mean value with the maximum number of available data. Extreme values with a probability of exceedance higher than 99 % and lower than 1 % were removed from each parameter, in order to consider the most probable values. This was calculated by organizing the historical dataset in descending order and estimating the probability of occurrence of each value regarding the whole dataset. Seawater Mg, Sr and Ca concentrations are available in the environmental impact assessment (EIA) study of the desalination plant in Tseung Kwan O submitted to the EPD in 2013 (Black and Veatch Hong Kong Limited and Water Supplies Department, 2013).
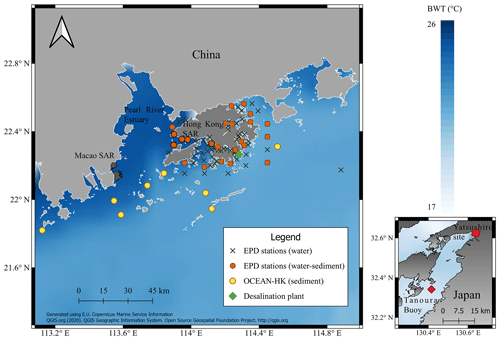
Figure 1Sampling sites around the Pearl River estuary (PRE) and the Yatsushiro bay in Japan. Ostracods were collected from sediment samples obtained by the Environmental Protection Department of Hong Kong (EPD) and OCEAN-HK in Hong Kong. Monthly water quality data were obtained from different sampling locations across Hong Kong (EPD stations). Mean bottom-water temperature (BWT) for the year 2011 was obtained from the satellite product of the Copernicus Marine Environment Monitoring Service (CMEMS).
Additionally, seawater surface temperatures at a 0.5 m depth were retrieved
from Tanoura buoy ![]() to estimate water temperatures at the site of
ostracod collection in the Yatsushiro Sea. These data are available on the
web page of the Kumamoto Prefectural Fisheries Research Center
(2021).
to estimate water temperatures at the site of
ostracod collection in the Yatsushiro Sea. These data are available on the
web page of the Kumamoto Prefectural Fisheries Research Center
(2021).
2.1.2 Copernicus products
Worldwide potential bottom-water temperature (BWT), provided by the Copernicus Marine Environment Monitoring Service (CMEMS) of the European Union, is available from 1993 to 2018 at a spatial resolution of ∘ grid. Potential BWT is a product calculated from re-analysis, which is intended to be as close as possible to real observations (Drévillon et al., 2018). The product is obtained after the assimilation of satellite observations through real-time marine observations and the modeling of atmospheric and oceanic variables, such as tidal and heat fluxes (CMEMS, 2020). The product has low biases at regional and global scales (<0.4 and <0.1 ∘C respectively), but higher errors are present in coastal regions due to land cover and river inputs. We calculated the bias and correction factors between the BWTs from the CMEMS and EPD in areas where the data overlap (Fig. S1). These factors were applied to correct the potential BWT in areas where we do not have direct measurements such as in the sampling locations of OCEAN-HK.
2.2 Ostracod samples
We investigated adult specimens of S. impressa (Brady, 1869; Whatley and Zhao, 1988) and N. delicata (Ishizaki and Kato, 1976) from the uppermost 1 cm sediment layer collected in HK by the EPD in January and July 2012 (Hong et al., 2019; Rodriguez, 2021). The temperature calibration was developed using only samples from HK waters. We also used samples collected around the PRE by OCEAN-HK in July 2017 to validate the calibrations. Ostracods were collected from sediment samples sieved in a 150 µm mesh. Most of the specimens collected from HK and PRE consist of single valves without animal appendages (Fig. 1). For S. impressa, we distinguished adult ostracods as those larger than 600 µm (Irizuki et al., 2005) and with a well-developed inner lamella (Fig. S2). For N. delicata, we distinguished adult specimens by size (>450 µm) based on Wang et al. (2018) and Fig. S2. We additionally included S. impressa specimens from the Yatsushiro Sea collected on 7 June and 7 November 2020 from the intertidal zone (32.624∘ N, 130.640∘ E; 0.5 m depth; Fig. 1) in order to test the calibration developed. These specimens present the animal appendages and intact right and left valves. The size of these ostracods ranged between 550 and 650 µm, and they each had a well-developed inner lamella.
2.3 Trace-element analyses
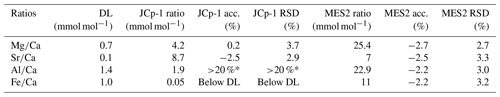
Ostracod shells were sonicated in a methanol bath, rinsed twice with Milli-Q water, bleached with 5 % sodium hypochlorite for 12–24 h, and rinsed twice again with Milli-Q water to limit potential contamination affecting the carbonate and (Rodriguez, 2021). Then, elemental concentrations on individual shells were measured by inductively coupled plasma mass spectrometry (ICP-MS) Agilent 7900 in the School of Biological Sciences at the University of Hong Kong. We measured 48Ca, 43Ca, 24Mg, 25Mg, 86Sr and 88Sr. In addition, we measured 27Al and 56Fe to control for potential contamination in our samples. The data were corrected by a blank (2 % HNO3) measured every third sample and a multi-element standard measured every sixth sample, which was prepared from individual pure elemental solutions (MES1; Mg = 28.1 ppb; Sr = 29.6 ppb; Al = 28.1 ppb; Fe = 29.4 ppb; Ca = 1830 ppb). A multi-element standard prepared from a pure multi-element solution (MES2; Mg = 28.6 ppb; Sr = 28.6 ppb; Al = 28.6 ppb; Fe = 28.6 ppb; Ca = 1861 ppb) and a carbonate standard JCp-1 (Hathorne et al., 2013; Inoue et al., 2004; Okai et al., 2002) were used to check the quality of the analysis. Our precision and accuracy improved using 48Ca and 24Mg. The accuracy and precision (RSD) of the analysis for JCp-1 and MES2 standards are shown in Table 1 (n = 90). The detection limit of the concentrations was initially estimated using the blank as 3σ, resulting in a value lower than 0.3 ppb for Mg, Sr, Al and Fe. Then, the detection limit of the ratios was calculated as 3σ of a solution with a concentration of 0.1 ppb for Mg, Sr, Al and Fe and 990 ppb of Ca (Yu et al., 2005). The precision and accuracy for Al and Fe could not be determined for JCp-1 because of the low ratios within the standard and contamination associated with our analytical procedure.
3.1 Marine waters
Considering Hong Kong sampling sites, annual mean BWT ranges between 21.8 and 23.9 ∘C (Fig. 1). The maximum monthly temperature has been recorded close to the PRE (28.6 ∘C) in August, while the minimum monthly temperature has been recorded eastward of Hong Kong Island (16.2 ∘C) in February. Mean annual salinity ranges from 24.7 to 33.7 PSU. The maximum monthly salinity was measured eastward of Hong Kong Island in August (34.3 PSU), while the minimum salinity was recorded close to the Pearl River in July (15.6 PSU). A negative linear correlation is observed between the annual temperature and salinity considering all EPD stations in Hong Kong waters (, Fig. S4). BWT and salinities from the EPD grouped by water control zones are shown in Table 2. A summary of annual mean values of other parameters such as dissolved oxygen and suspended solids can be found in Table S1.
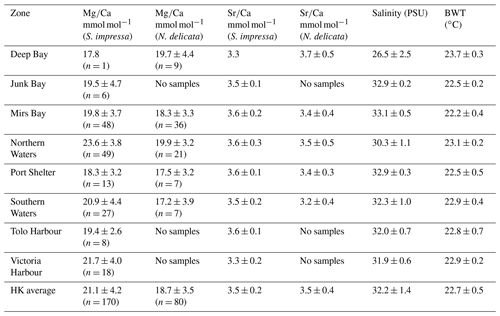
Table 2Mean and standard deviation (1 SD) of and ratios of adult S. impressa and N. delicata. The number of shells for and are the same. Last two columns show mean and standard deviation (1 SD) of annual bottom-water salinity and temperature by water control zone of HK.
Average surface water temperatures at the Tanoura buoy recorded in the months of ostracod collection were 23 and 20.7 ∘C for June and November respectively. Satellite images reveal a temperature difference lower than 1 ∘C between the surface temperature in the Tanoura buoy and the sampling site in the Yatsushiro Sea. Therefore, we considered surface water temperatures from Tanoura buoy (0.5 m depth) as representative of the water temperature collected in the sampling site in the Yatsushiro Sea.
3.2 Ostracod ratios
mean values of adult S. impressa and N. delicata ostracods from EPD samples are 21.1 ± 4.2 mmol mol−1 (n = 170; min = 13.6 mmol mol−1; max = 34.8 mmol mol−1) and 18.7 ± 3.5 mmol mol−1 (n = 80; min = 10.3 mmol mol−1; max = 28.1 mmol mol−1) respectively. mean values of adult S. impressa and N. delicata ostracods from OCEAN-HK are 20.1 ± 3.7 mmol mol−1 (n = 51; min = 14.4 mmol mol−1; max = 35.9 mmol mol−1) and 19 mmol mol−1 (n = 30, 14.1 to 24.7 ± 2.8 mmol mol−1) respectively. mean values in each EPD water control zone are in Table 2. mean values of S. impressa specimens from the Yatsushiro Sea were 21.6 ± 3.8 mmol mol−1 (n = 14; min = 15.3 mmol mol−1; max = 28 mmol mol−1) and 18.3 ± 5 mmol mol−1 (n = 9; min = 12.1 mmol mol−1; max = 26.1 mmol mol−1) for June and November respectively.
mean values of adult S. impressa and N. delicata ostracods from EPD samples are 3.5 ± 0.2 mmol mol−1 (n = 170; min = 2.9 mmol mol−1; max = 4.3 mmol mol−1) and 3.5 ± 0.4 mmol mol−1 (n = 80; min = 2.3 mmol mol−1; max = 4.7 mmol mol−1) respectively, while for OCEAN-HK samples are 3.6 ± 0.2 mmol mol−1 (n = 51; min = 2.9 mmol mol−1; max = 4 mmol mol−1) and 3.5 ± 0.4 mmol mol−1 (n = 30; min = 2.6 mmol mol−1; max = 4.4 mmol mol−1) respectively. mean values in each EPD water control area are in Table 2. mean values of S. impressa specimens from the Yatsushiro Sea were 3.9 ± 0.6 mmol mol−1 (n = 14; min = 2.8 mmol mol−1; max = 4.5 mmol mol−1) and 3.3 ± 0.5 mmol mol−1 (n = 9; min = 2.7 mmol mol−1; max = 4.4 mmol mol−1) for June and November respectively.
mean values of adult S. impressa and N. delicata from EPD samples are 1 ± 1.3 mmol mol−1 (n = 160; min = 0 mmol mol−1; max = 11.7 mmol mol−1) and 1.8 ± 1.2 mmol mol−1 (n = 76; min = 0 mmol mol−1; max = 5.5 mmol mol−1) respectively. mean values of adult S. impressa and N. delicata from OCEAN-HK samples are 1.2 ± 0.7 mmol mol−1 (n = 51; min = 0 mmol mol−1; max = 3.4 mmol mol−1) and 1.5 ± 1.7 (n = 28; min = 0 mmol mol−1; max = 7 mmol mol−1) respectively. and ratios of S. impressa and N. delicata specimens do not correlate (Fig. S3). The low ratios indicate the absence of clays in the ostracods. mean values of S. impressa specimens from the Yatsushiro Sea were 2.3 ± 0.8 mmol mol−1 (n = 14; min = 1 mmol mol−1; max = 3.6 mmol mol−1) and 3.3 ± 1.4 mmol mol−1 (n = 9; min = 1.5 mmol mol−1; max = 6 mmol mol−1) for June and November respectively.
mean values of adult S. impressa and N. delicata from EPD samples are 1.1 ± 1.8 mmol mol−1 (n = 160; min = 0 mmol mol−1; max = 11.9 mmol mol−1) and 1.5 ± 1.4 mmol mol−1 (n = 67; min = 0 mmol mol−1; max = 6.1 mmol mol−1) respectively. mean values of adult S. impressa and N. delicata from OCEAN-HK samples are 0.6 ± 0.4 mmol mol−1 (n = 47; min = 0 mmol mol−1; max = 2.1 mmol mol−1) and 1.3 ± 1.4 mmol mol−1 (n = 26; min = 0 mmol mol−1; max = 6.5 mmol mol−1) respectively. The low values indicate the absence of Mn–Fe oxides in the ostracods. mean values of S. impressa specimens from the Yatsushiro Sea were 0.2 ± 0.2 mmol mol−1 (n = 14; min = 0 mmol mol−1; max = 0.8 mmol mol−1) and 1 ± 0.6 mmol mol−1 (n = 9; min = 0.4 mmol mol−1; max = 2.1 mmol mol−1) for June and November respectively.
3.3 calibrations to water parameters
ratios of S. impressa significantly correlate to annual water temperatures in HK waters (; pS. impressa=0.007), but the ratios of N. delicata ostracods do not correlate if the full dataset is used (Table 3). However, the removal of sampling sites with only one shell allows us to produce significant temperature calibrations for both species (Table 3). The highest R2 at 95 % significance for the temperature calibration to ratios for S. impressa and N. delicata was obtained considering a minimum number (η) of 11 and 3 shells per sampling site respectively (Table 3). The highest R2 at 99.9 % significance for S. impressa was obtained considering a minimum number of 7 shells per sampling site, while temperature calibrations for N. delicata did not reach this level of significance. Juvenile ostracods from both species do not correlate with temperature. Juvenile ostracods of S. impressa obtained from samples from the eastern side of HK have similar ratios to adults, but juveniles close to the PRE have lower ratios than adults (Fig. 2).
Table 3Correlation fit and significance of temperature calibrations using different number of shells per sampling site to calculate the mean value of ostracod ratios for S. impressa and N. delicata.
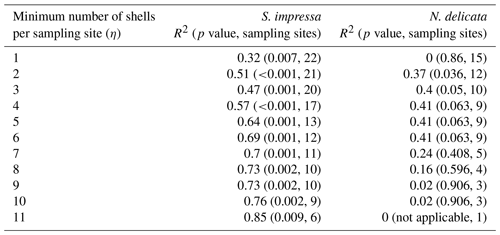
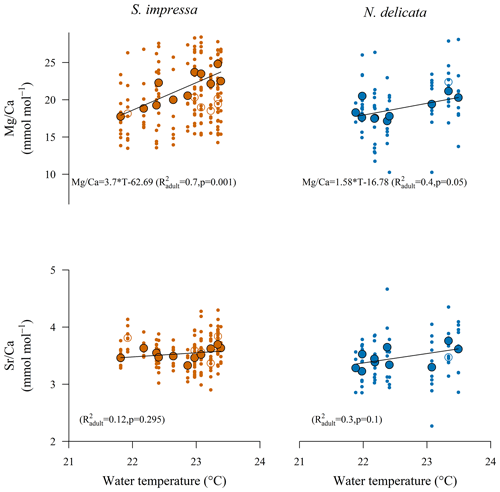
Figure 2Relationships of and with annual temperature for adult (filled circles) and A-1 juvenile (open circles) ostracod samples of S. impressa (η = 7) and N. delicata (η = 3). Single-shell samples are shown as small dots. Solid lines show the linear regressions for adult specimens.
The ratio of S. impressa and N. delicata also correlates with other water parameters (from Figs. S5–S12) such as volatile suspended solids (; pS. impressa<0.001; ; pN. delicata=0.037), turbidity (; pS. impressa<0.001; ; pN. delicata=0.033), suspended solids (; pS. impressa=0.001; ; pN. delicata=0.034), silica (; pS. impressa=0.002; ; pN. delicata=0.041), salinity (; pS. impressa<0.001; ; pN. delicata=0.037), nitrite (; pS. impressa<0.001; ; pN. delicata=0.031) and nitrate (; pS. impressa<0.001; ; pN. delicata=0.02).
The temperature calibration to is more significant in spring–summer months for both species (Fig. 3) when the ocean temperature increases. The highest correlation occurred in April for S. impressa and N. delicata (ηS. impressa=7; ; pS. impressa<0.001; ηN. delicata=3; ; pN. delicata=0.015).
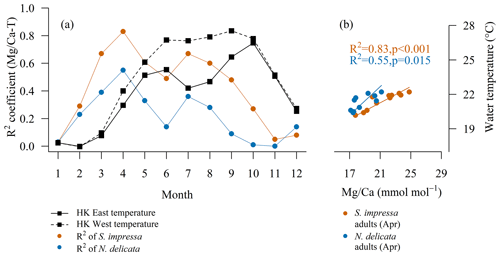
Figure 3(a) Monthly temperature calibrations to of S. impressa (η = 7) and N. delicata (η = 3) for adult ostracod. (b) The calibrations for S. impressa and N. delicata for April are S. impressa=2.8 ⋅ T−39.2 and N. delicata=1.6 ⋅ T−15.7.
A one-way ANOVA determined that ratios do not show significant differences between sampling sites for N. delicata (p = 0.16, Type II). ratios were significantly different for S. impressa (p = 0.02). However, this was only the result of samples collected from Victoria Harbour (n = 18). After removal of these samples, ratios of S. impressa were no longer significantly different between sampling sites (p = 0.96). ratios of both species do not correlate with any of the 24 water parameters measured by the EPD.
4.1 Control on and ratios of S. impressa and N. delicata
The strongest linear correlation between the 24 parameters (Table S1) measured by the EPD and ratio was for the annual mean temperature, which suggests that water temperature is the main control of uptake in adult ostracods of S. impressa and N. delicata (Fig. 2). Correlations with other parameters are also significant but are likely caused by multicollinearity of temperature with other water parameters, such as turbidity, suspended solids, salinity, pH and nitrite (Fig. S4). The temperature control on the ratio of biogenic material is well-known and has been usually described linearly for ostracods, even though inorganic calcite follows an exponential relationship (Lea, 2003). We observed a linear correlation between water temperature and ratios of S. impressa and N. delicata ostracods (Fig. 2), but the significance of the linear calibration is higher for the former species, making this species more suitable for temperature reconstructions.
We evaluated the sensitivity of the calibration using the natural variability in ostracod ratios and seawater temperatures in Hong Kong sampling sites. The error (range) of mean values in each station used for the calibration can be established at a certain confidence level (Holmes, 2008). For S. impressa, the error in mean values in each station ranged from 1.5 to 4.1 mmol mol−1 at a 95 % confidence level. For annual and spring calibrations, the temperature mean errors are 0.7 ∘C (0.4 to 1 ∘C) and 0.9 ∘C (0.5 to 1.5 ∘C) respectively. These values are lower than the temperature difference between the stations (1.6 ∘C), which indicates that differences higher than 1 ∘C can be estimated using S. impressa calibration curves. For N. delicata, the error in mean values ranged from 1.1 to 5.7 mmol mol−1. Annual and spring calibrations have the same slope, which produce temperature mean errors of 1.9 ∘C (0.7 to 3.6 ∘C). This error is similar to the difference in temperatures between the stations. Therefore, more shells of ostracods living at different temperatures would be needed to estimate differences at 1 ∘C for this species. We also investigated the potential impact of daily temperature fluctuations on the calibrations. We estimated daily BWT variability by using the Copernicus satellite products. Using the daily data from 1993 to 2018, we calculated the standard deviation for each month. We then determined the average variation for each month across all the years. We performed this calculation on three Hong Kong sampling stations located (a) at the lower section of the PRE, (b) outside the PRE and south of Hong Kong Island, and (c) on the eastern side of Hong Kong Island. We found the variations were 1 ± 0.4, 0.8 ± 0.5 and 0.7 ± 0.5 ∘C respectively. Therefore, daily bottom-water fluctuations in Hong Kong waters are unlikely impacting the calibrations obtained.
The of A-1 juvenile ostracods of S. impressa does not correlate to April temperature (Fig. 2). A significant weak correlation is only observed using June temperature (; pS. impressa=0.031; η = 2). Juvenile ostracods usually show higher ratios than adults (Chivas et al., 1986b; Dwyer et al., 2002). However, juvenile specimens of S. impressa from sampling sites with salinity lower than 32 PSU have lower ratios than adults (Fig. 4). We hypothesize that environmental factors may increase the bias between ostracod life stages. For example, strong currents can produce preferential post-mortem transport of lighter shells (Boomer et al., 2003), which may foster the mixing of ostracod shells calcified at different temperatures. Our results suggest that juvenile and adult ostracods of S. impressa cannot be used indistinctly to reconstruct April temperature, supporting previous findings about the incompatible use of ratios of different ostracod stages (Dwyer et al., 2002).
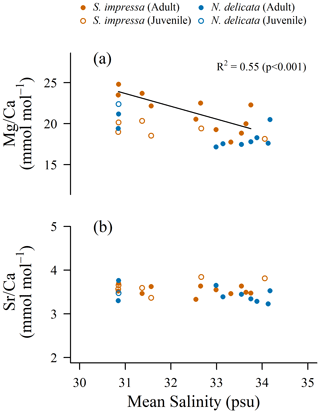
Figure 4 and correlation to salinity for S. impressa (η = 7) and N. delicata (η = 3) for salinity recorded in April considering mean values.
None of the 24 parameters measured by the EPD, including temperature and salinity, exert control on the ratios in adult specimens of either species. The low variabilities in S. impressa and N. delicata suggest that the potential control variables of this ratio, such as water or vital effects, do not change considerably across different locations within the PRE. A positive correlation in with chlorophyll a and dissolved oxygen is observed for S. impressa specimens, but this relationship is mainly produced by specimens with low from one sampling site in Victoria Harbour. Dissolved oxygen in this sampling site was particularly low, probably as a result of discharge from the nearby Stonecutters Island sewage treatment plant.
ratios of both species are negatively correlated to salinity (Fig. 4). A potential control of seawater on ostracod would be possible if seawater decreases with salinity. Marine waters have higher Mg and Ca concentrations in comparison to freshwater (Open University, 1992; Bruland and Lohan, 2006). Previous studies have shown mostly conservative behavior of Mg and Ca in estuaries and surrounding areas (Millero, 2006; Patra et al., 2012), where these concentrations increase linearly with salinity. Therefore, a higher Ca concentration over Mg concentration or a lower Mg concentration over Ca concentration toward more saline waters is unlikely in Hong Kong waters. Moreover, ostracod is similar at different salinities (Fig. 4), supporting the idea that changes in seawater Ca concentrations are not the main control on and ostracod ratios. Measurements during 2013 and 2017 at the desalination plant in Tseung Kwan O (Figs. 1 and S13) show water and ranging between 4 and 6 mol mol−1, and between 8 and 9 mol mol−1 respectively during the year. These values are mostly stable even when monthly salinity decreases to 25 PSU in some Hong Kong stations during summer, suggesting that the seawater chemistry may not be a primary control on ostracod and ratios of Hong Kong waters. Our dataset does not allow us to explore in more detail the potential relationship between these two variables as more data of the seawater chemical composition are needed.
4.2 Factors controlling the robustness of the temperature calibration to
4.2.1 Number of shells per sampling site
The robustness of the annual temperature calibration to with respect to R2 and p values depends on the number of individual shells available to calculate the mean value of each sampling site (Table 3). The removal of sampling sites with a low number of shells increases the R2 of temperature calibrations in both species, suggesting that a low number of samples does not allow us to capture all the natural variability exerted by the temperature on ostracod ratios. The most significant calibrations were obtained using at least two to four shells for S. impressa and at least two to three shells per sampling site for N. delicata respectively (Table 3). The margin of error (z ⋅ SE, where z is critical value and SE is standard error) of confident intervals can be used to provide an estimation of the number of shells needed to obtain a desired level of error. Using temperature calibrations for Bythocypris and Krithe specimens, Corrège and De Deckker (1997) showed that the use of four shells provides an error in the temperature prediction lower than 1.4 ∘C at a 95 % confidence level. In stratigraphic studies the use of three to five shells is a common practice (Holmes and De Deckker, 2012). Holmes (2008) studied the critical sample size of ostracod specimens to keep a desired error at a 0.99 significance level in and from a stratigraphic sequence. The author concluded that in modern specimens of Cyprideis torosa from shallow brackish environments the optimal sample size may vary from 3 to 16 shells. We can use this approach to estimate the number of shells needed per sampling site to keep the error at an acceptable level to produce a significant correlation. The use of one, two, three, four, five, six and seven shells produces margin of errors of 3.4, 2.4, 2.0, 1.7, 1.5, 1.4 and 1.3 ∘C at 95 % significance respectively, considering the highest standard deviation observed in our sampling sites (σ=4.7 mmol mol−1; n = 15) and normally distributed samples (Rodriguez, 2021). Our results suggest that in shallow marine waters the calculation of mean ratios with at least four individual shells in more than 10 sampling sites would likely produce a significant temperature calibration at a 99 % significance level (p<0.01), accounting for short-term temperature variations. In the following analyses, we restricted the minimum number of shells per sampling site to η = 7 for S. impressa and η = 3 for N. delicata as we obtained the highest R2 at a significance of 99.9 % and 95 % respectively.
4.2.2 Temporal variability
Most temperature calibrations to are produced with the annual mean temperature as they are intended to reflect long-term changes in paleoceanographic studies. However, ostracods molt their shells in a short period of time (usually days or weeks). Thus, it may be possible to record ocean parameters at a shorter timescale if the correct molting time is known. Our monthly analysis of linear regressions shows that the best temperature calibrations to in terms of high R2 and significant p values were found with the water temperature measured in April for S. impressa and N. delicata (Fig. 3). This suggests that adult ostracods of both species may inhabit HK waters mainly during spring or early summer, as has been documented for other shallow marine species (Cronin et al., 2005a; Kamiya, 1988). July temperature also correlates with ratios in both species, showing a second peak (Fig. 3). Two periods of calcification have recently been shown for C. torosa (Roberts et al., 2020), which may also occur in S. impressa and N. delicata. The high correlation between April water temperature and ratios indicates the possibility of reconstructing water temperature at a finer temporal scale, which can help to unravel coastal ocean circulation patterns. For example, the interaction of the Pearl River and Hainan, Taiwan and Kuroshio currents (Morton and Wu, 1975) determines the temperature and salinity of Hong Kong waters. The Hainan current is dominant during summer, while the Taiwan and Kuroshio currents affect Hong Kong waters during winter. Sinocytheridea impressa and N. delicata may become important tools to determine the currents' interaction by providing information on water temperature during the transition between these currents and freshwater from the Pearl River.
4.3 Validation of temperature calibration
April BWTs obtained from the Copernicus product are on average −1.5 ± 1.8 ∘C (2σ) below the measurements from the EPD (Fig. S1). BWT differences between EPD stations and the Copernicus product range from 0 to 3 ∘C, being the largest in stations close to the Pearl River (Fig. S1). We corrected the BWTs obtained from the Copernicus product in all OCEAN-HK sampling locations by −1.5 ∘C.
The BWTs estimated from ostracod ratios in OCEAN-HK sampling locations are in good agreement with BWTs obtained from the Copernicus product for April (Fig. 5 and Table S2). The difference between the estimated temperature by the linear regressions and the potential BWT from the Copernicus product is lower than 0.7 ∘C in seven out of the eight sites for S. impressa. Only one sampling site shows an error of 1.8 ∘C (Table S2). For N. delicata, the error was lower than 2.3 ∘C in the three stations considered. This suggests that S. impressa and N. delicata specimens around the PRE follow the regression line developed with ostracods collected from HK waters. Our findings also suggest that calibrations may be done without direct measurements of BWT but using the potential BWT from satellite products. We highlight that the improvement in the quality of BWT products derived from satellite images may facilitate the calibration of multiple species across the world as scientists have global coverage of BWT with a high resolution ( km).
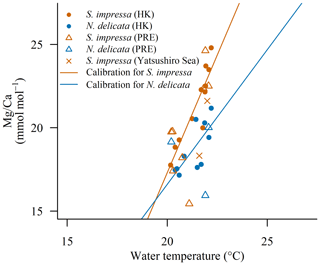
Figure 5 ratios of ostracods from the PRE (OCEAN-HK) and Yatsushiro bay over the calibration for S. impressa (S. impressa=2.8 ⋅ T−39.2) and N. delicata (N. delicata=1.6 ⋅ T−15.7) performed with samples from Hong Kong (EPD). The BWT from the Copernicus product was used for OCEAN-HK samples. Surface water temperature from the Tanoura buoy in the last 15 d in June (21.8 ∘C) and November (21.9 ∘C) were considered for the Yatsushiro Sea samples.
The ratios of S. impressa ostracods from the Yatsushiro Sea are within the range of ratios found in Hong Kong ostracods (June 21.6 ± 3.8 mmol mol−1 and November 18.3 ± 5 mmol mol−1, Fig. 5). Using the Hong Kong calibration developed for April, temperatures in the Yatsushiro Sea were estimated to be 21.5 ∘C in June and 20.4 ∘C in November. We compared these temperatures with seawater temperatures recorded (a) during the day of sampling (June 21.6 ∘C and November 15 ∘C, in situ) and as the mean of (b) 10 d (June 22.0 ∘C and November 21.6 ∘C, buoy), (c) 20 d (June 21.5 ∘C and November 22.1 ∘C, buoy), (d) 30 d (June 20.7 ∘C and November 22.6 ∘C, buoy) and (e) 60 d (June 18.8 ∘C and November 23.9 ∘C, buoy) before the specimen collection. The estimation of water temperatures using ostracod ratios in November is not similar to the temperatures recorded on the same day as sampling. This is likely due to the exposure of the site to freshwater inputs, which may have affected local water temperatures. In addition, the ostracod calcification may have occurred several days before its collection. The consideration of the mean temperature across the 10 d before the ostracod collection produced the greatest agreement with -estimated temperatures. Thus, our results suggest that ostracods from the Yatsushiro Sea may have calcified the shells during the last few days before their collection.
4.4 Calibrations at a superfamily level
Sinocytheridea impressa and N. delicata are related at a superfamily level (Cytheroidea). A few studies have developed calibrations for ostracod species from the same superfamily. A unique calibration for ostracods from the same superfamily cannot be performed according to our results. Temperature calibrations to ratios have been developed for specimens of the same superfamily, including the genus Krithe (Cadot and Kaesler, 1977; Corrège and De Deckker, 1997; Cronin et al., 1996; Dwyer et al., 1995, 2002; Elmore et al., 2012; Farmer et al., 2012), Loxoconcha (Cronin et al., 2003), Cytheropteron (Yamada et al., 2014) and Xestoleberis (Kondo et al., 2005). Sinocytheridea impressa and N. delicata show lower ratios at the same temperature in comparison to other species (Fig. 6). Similar ratios to those of S. impressa and N. delicata have been observed in Krithe specimens at seawater temperatures ranging between 10 and 20 ∘C. The residual standard errors (RSEs) for the calibration of S. impressa and N. delicata are 1.1 mmol mol−1 for both species, showing one of the best regression fits of all calibrations for the superfamily Cytheroidea (Fig. 6). The calibration of N. delicata has a similar slope in comparison to Xestoleberis specimens (Fig. 6), but S. impressa has the highest slope of all species from the superfamily, which shows that different species from the same superfamily do not share the same calibration slope and intercept. In addition, ostracods from the same superfamily under similar conditions of temperature and salinity may show significant differences in their calibration parameters, such as S. impressa and N. delicata specimens in HK waters or Xestoleberis specimens (Kondo et al., 2005). Factors such as the development of temperature calibrations from multiple species, the number of shells considered per site and variability induced after the burial of ostracods might impoverish the calibration fit. Our study suggests that when species-specific calibrations are performed, the residual standard error in the temperature calibration to may be around 1 mmol mol−1. This happens in shells which have not been buried, are oxide-free, and were treated with non-corrosive cleaning reagents such as 5 % sodium hypochlorite and methanol (Rodriguez, 2021). The of ostracods is temperature-sensitive at a very small temperature range and may be useful to understand variation in a localized region. Sinocytheridea impressa and N. delicata ostracods from other locations can improve the calibration shown in this study by expanding the temperature range.
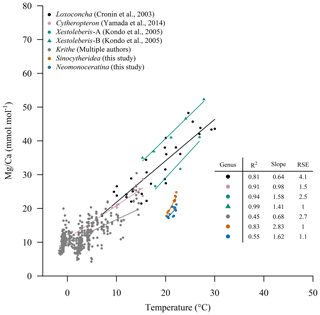
Figure 6Temperature versus ratios for specimens of the superfamily Cytheroidea. Krithe samples were retrieved from Farmer et al. (2012) and the study of Elmore et al. (2012) which contains the collection of and BWT from different studies including Cadot and Kaesler (1977), Corrège and De Deckker (1997), Dwyer et al. (1995), Cronin et al. (1996), and Dwyer et al. (2002).
The lower in S. impressa and N. delicata ostracods in comparison to other species from the same superfamily such as Krithe or Loxoconcha may be the result of other variables such as pH, dissolved inorganic carbon (DIC), CO3 concentration or the calcite saturation index (Ω). These variables have been suggested to partially control the incorporation of ostracod ratios (Elmore et al., 2012; Farmer et al., 2012; Holmes and De Deckker, 2012). Loxoconcha from Chesapeake Bay (Cronin et al., 2003, 2005a) most likely dwells in the environment most similar to that of Sinocytheridea and Neomonoceratina due to its presence in a large estuary in a polyhaline system at similar depths. Measurements of pH and DIC in HK and Chesapeake Bay waters allow us to compare the saturation index in both basins. A bottom-water pH ranging from 7.8 to 8.2 (EPD, 2018) and DIC over 1850 µM (Guo et al., 2008; Yuan et al., 2011) indicate that the seawater is oversaturated with calcite (Ω∼3) in our sampling sites for most of the year. On the other hand, pH and DIC in the bottom waters of the Chesapeake lower bay in 2006 ranged from 7.92 to 7.96 and 1717.4 to 1865.4 µM respectively, with a calcite saturation index over 2.8 (Brodeur et al., 2019), suggesting that the lower ostracod ratios in S. impressa and N. delicata ostracods are not a direct result of different conditions of pH, DIC or the calcite saturation index. Species-specific biomineralization types (i.e., ostracod calcification) may also play an important role in the temperature response to ostracod ratios of different species, but our measurements do not allow us to explore this factor.
The ratios of S. impressa and N. delicata ostracods can be used as proxies for water temperature in shallow marine environments. The temperature dependence of S. impressa is higher, and therefore it is more suitable for temperature reconstructions. This study shows that (1) the number of shells per sampling site has an important impact on the calibration robustness due to the strong seasonal variability in temperature in estuaries and coastal areas, and therefore we recommend the use of four or more shells per sampling site; (2) ostracods can give information on monthly water temperatures; (3) the temperature reconstruction based on ratios of S. impressa and N. delicata specimens has the potential to give insight into past ocean circulation in coastal areas of the South China Sea; and (4) ostracods from the same superfamily show different calibration curves, which do not seem to be controlled by the ratios of marine waters, temperature or the carbonate system. Better understanding of ostracod molting time will likely improve calibrations and the identification of the calcification temperature for S. impressa and N. delicata ostracods.
Data used for this study are available from EarthChem (https://doi.org/10.26022/IEDA/111891; Rodriguez and Not, 2021).
The supplement related to this article is available online at: https://doi.org/10.5194/bg-18-1987-2021-supplement.
CN and MR designed the project and wrote the manuscript. MR carried out the experiments, calculations and interpretation of the results under the supervision of CN.
The authors declare that they have no conflict of interest.
We are thankful to the Theme-based Research Scheme project of OCEAN-HK (T21-602/16R) for providing access to sediment samples and CTD data. This study has been conducted using EU Copernicus Marine Service information. We wish to thank Moriaki Yasuhara and Yuanyuan Hong for their contribution in the provision of sediment samples and the identification of ostracod species and stages. We greatly thank Gengo Tanaka for providing us with ostracod specimens from the Yatsushiro Sea in Japan as well. Finally, we would like to thank Kayi Chan for proofreading a version of this paper.
This research has been supported by the seed funding program for basic research of the University of Hong Kong (project code no. 104003882) and the start-up fund for new staff from the Science Faculty of the University of Hong Kong, awarded to Christelle Not. Maximiliano Rodriguez was supported by the HKU SPACE Research Fund (no. 200004912) granted to Christelle Not. The sample collection in Hong Kong waters was also partially supported by the Environment and Conservation Fund of Hong Kong (project code 19/2012), the General Research Fund of the Research Grants Council of Hong Kong (project codes HKU 17302518 and HKU 17303115), and the seed funding program for Basic Research of the University of Hong Kong (project codes 201111159140 and 201611159053) awarded to Moriaki Yasuhara.
This paper was edited by Hiroshi Kitazato and reviewed by Thomas M. Cronin and one anonymous referee.
Beck, J. W., Edwards, R. L., Ito, E., Taylor, F. W., Rougerie, F., Joannot, P., and Henin, C.: Sea-Surface Temperature from Coral Skeletal Strontium/Calcium Ratios, Science, 257, 644–647, 1992.
Black and Veatch Hong Kong Limited and Water Supplies Department: Desalination Plant at Tseung Kwan O – Feasibility Study, EIA Project Profile, Hong Kong SAR, available at: https://www.epd.gov.hk/eia/register/report/eiareport/eia_2292015/Table of Content_PDF.html (last access: 21 May 2020), 2013.
Boomer, I., Horne, D. J., and Slipper, I. J.: The use of ostracods in palaeoenvironmental studies, or what can you do with an Ostracod shell?, Paleontol. Soc. Pap., 9, 153–180, https://doi.org/10.1017/S1089332600002199, 2003.
Brady, G. S.: Les entomostraces de Hong Kong, in: Les fonds de la mer, edited by: de. Folin, L. and Périer, L., pp. 155–159, 1869.
Brandão, S. N. and Karanovic, I.: World Ostracoda Database, World Register of Marine Species, https://doi.org/10.14284/364, 2020.
Brodeur, J. R., Chen, B., Su, J., Xu, Y. Y., Hussain, N., Scaboo, M. M., Zhang, Y., Testa, J. M., and Cai, W. J.: Chesapeake bay Inorganic Carbon: Spatial distribution and Seasonal Variability, Front. Mar. Sci., 6, 1–17, https://doi.org/10.3389/fmars.2019.00099, 2019.
Open University: Seawater: Its Composition, Properties, and Behaviour, Oxford: Pergamon, Exeter, UK, in Association with the Open U, 1992.
Bruland, L. W. and Lohan, M. C.: Controls of Trace Metals in Seawater, in: The oceans and marine geochemistry, edited by: Elderfield, H., Elsevier Pergamon, Amsterdam, The Netherlands, 23–45, 2006.
Cadot, H. M. and Kaesler, R. L.: Magnesium content of calcite in carapaces of benthic marine ostracoda, Paleontol. Contrib., 87, 1–23, 1977.
Chivas, A. R., De Deckker, P., and Shelley, J. M. G.: Magnesium, strontium, and barium partitioning in nonmarine ostracode shells and their use in paleoenvironmental reconstructions – a preliminary study, Proc. Eighth Int. Symp. Ostracoda. Dep. Geosci., 10, 238–249, 1983.
Chivas, A. R., De Deckker, P., and Shelley, J. M. G.: Magnesium and strontium in non-marine ostracod shells as indicators of palaeosalinity and palaeotemperature, Hydrobiologia, 143, 135–142, https://doi.org/10.1007/BF00026656, 1986a.
Chivas, A. R., De Deckker, P., and Shelley, J. M. G.: Magnesium content of non-marine ostracod shells: A new palaeosalinometer and palaeothermometer, Palaeogeogr. Palaeoclimatol. Palaeoecol., 54, 43–61, https://doi.org/10.1016/0031-0182(86)90117-3, 1986b.
Chunlian, L., Fürsich, F. T., Jie, W., Yixin, D., Tingting, Y., and Jian, Y.: Late Quaternary palaeoenvironmental changes documented by microfaunas and shell stable isotopes in the southern Pearl River Delta plain, J. Palaeogreography, 2, 344–361, https://doi.org/10.3724/SP.J.1261.2013.00035, 2013.
CMEMS: Global ocean physics reanalysis, available at: https://marine.copernicus.eu (last access: 1 January 2020), 2020.
Cohen, A. L., Layne, G. D., Hart, S. R., and Lobel, P. S.: Kinetic control of skeletal in a symbiotic coral: Implications for the paleotemperature proxy, Paleoceanography, 16, 20–26, 2001.
Cohen, A. L., Owens, K. E., Layne, G. D., and Shimizu, N.: The effect of algal symbionts on the accuracy of paleotemperatures from coral, Science, 296, 331–333, https://doi.org/10.1126/science.1069330, 2002.
Corrège, T. and De Deckker, P.: Faunal and geochemical evidence for changes in intermediate water temperature and salinity in the western Coral Sea (northeast Australia) during the Late Quaternary, Palaeogeogr. Palaeoclimatol. Palaeoecol., 131, 183–205, https://doi.org/10.1016/S0031-0182(97)00003-5, 1997.
Cronin, T. M., Dwyer, G. S., Baker, P. A., Rodriguez-Lazaro, J., and Briggs, W. M.: Deep-sea ostracode shell chemistry (Mg:Ca ratios) and late Quaternary Arctic Ocean history, in: Late Quaternary Palaeoceanography of North Atlantic Margins, edited by: Andrews, J. T., Geological Society Special Publications, London, UK, 117–134, 1996.
Cronin, T. M., Dwyer, G. S., Kamiya, T., Schwede, S., and Willard, D. A.: Medieval Warm Period, Little Ice Age and 20th century temperature variability from Chesapeake Bay, Glob. Planet. Change, 36, 17–29, https://doi.org/10.1016/S0921-8181(02)00161-3, 2003.
Cronin, T. M., Kamiya, T., Dwyer, G. S., Belkin, H., Vann, C. D., Schwede, S., and Wagner, R.: Ecology and shell chemistry of Loxoconcha matagordensis, Palaeogeogr. Palaeoclimatol. Palaeoecol., 225, 14–67, https://doi.org/10.1016/j.palaeo.2005.05.022, 2005a.
Cronin, T. M., Thunell, R., Dwyer, G. S., Saenger, C., Mann, M. E., Vann, C., and Seal, I. R.: Multiproxy evidence of Holocene climate variability from estuarine sediments, eastern North America, Paleoceanography, 20, PA4006, https://doi.org/10.1029/2005PA001145, 2005b.
Day, J., Yanez-Arancibia, A., Kemp, W. M., and Crump, B.: Introduction to Estuarine ecology, in: Estuarine ecology, edited by: Day, J., Crump, B., Kemp, W. M., and Yanez-Arancibia, A., John Wiley and Sons, Hoboken, New Jersey, USA, 1–18, 2013.
De Deckker, P. and Forester, R. M.: The use of ostracods to reconstruct continental palaeoenvironmental records, in: Ostracoda in the Earth Sciences, edited by: De Deckker, P., Colin, J. P., and Peypouquet, J. P., Elsevier, Amsterdam, The Netherlands, 175–199, 1988.
De Deckker, P., Chivas, A. R., and Shelley, J. M. G.: Uptake of Mg and Sr in the euryhaline ostracod Cyprideis determined from in vitro experiments, Palaeogeogr. Palaeoclimatol. Palaeoecol., 148, 105–116, 1999.
Dettman, D. L. and Dwyer, G. S.: The Calibration of Environmental Controls on Elemental Ratios in Ostracod Shell Calcite: A Critical Assessment, Dev. Quatern. Sci., 17, 145–163, 2012.
Drévillon, M., Régnier, C., Lellouche, J.-M., Garric, G., Bricaud, C., and Hernandez, O.: Quality Information Document for Global Ocean Reanalysis Products, GLOBAL-REANALYSIS-PHY-001-030, available at: http://marine.copernicus.eu/services-portfolio/access-to-products/?option=com_csw&view=details&product_id=GLOBAL_REANALYSIS_PHY_001_030 (last access: 4 March 2020), 2018.
Dwyer, G. S., Cronin, T. M., Baker, P. A., and Raymo, E.: North Atlantic deepwater temperature change during late Pliocene and late Quaternary climatic cycles, Science, 270, 1347–1351, 1995.
Dwyer, G. S., Cronin, T. M., and Baker, P. A.: Trace elements in marine ostracodes, Appl. Quatern. Res., 131, 205–225, 2002.
Elmore, A. C., Sosdian, S., Rosenthal, Y., and Wright, J. D.: A global evaluation of temperature and carbonate ion control on ratios of ostracoda genus Krithe, Geochem. Geophy. Geosy., 13, 1–20, https://doi.org/10.1029/2012GC004073, 2012.
EPD: Marine water quality in Hong Kong in 2018, Hong Kong SAR, available at: http://wqrc.epd.gov.hk/pdf/water-quality/annual-report/MarineReport2015eng.pdf (last access: 7 January 2020), 2018.
Farmer, J. R., Cronin, T. M., De Vernal, A., Dwyer, G. S., Keigwin, L. D., and Thunell, R. C.: Western Arctic Ocean temperature variability during the last 8000 years, Geophys. Res. Lett., 38, 4–9, https://doi.org/10.1029/2011GL049714, 2011.
Farmer, J. R., Cronin, T. M., and Dwyer, G. S.: Ostracode paleothermometry in the North Atlantic and Arctic oceans: Evaluation of a carbonate ion ef, Paleoceanography, 27, PA2212, https://doi.org/10.1029/2012PA002305, 2012.
Gouramanis, C. and De Deckker, P.: Alkalinity control on the partition coefficients in lacustrine ostracodes from Australia, Geology, 38, 359–362, https://doi.org/10.1130/G30235.1, 2010.
Guo, X., Cai, W. J., Zhai, W., Dai, M., Wang, Y., and Chen, B.: Seasonal variations in the inorganic carbon system in the Pearl River (Zhujiang) estuary, Cont. Shelf Res., 28, 1424–1434, https://doi.org/10.1016/j.csr.2007.07.011, 2008.
Hathorne, E. C., Gagnon, A., Felis, T., Adkins, J., Asami, R., Boer, W., Caillon, N., Case, D., Cobb, K. M., Douville, E., Demenocal, P., Eisenhauer, A., Garbe-Schönberg, D., Geibert, W., Goldstein, S., Hughen, K., Inoue, M., Kawahata, H., Kölling, M., Cornec, F. L., Linsley, B. K., McGregor, H. V., Montagna, P., Nurhati, I. S., Quinn, T. M., Raddatz, J., Rebaubier, H., Robinson, L., Sadekov, A., Sherrell, R., Sinclair, D., Tudhope, A. W., Wei, G., Wong, H., Wu, H. C., and You, C. F.: Interlaboratory study for coral and other element/Ca ratio measurements, Geochem. Geophy. Geosy., 14, 3730–3750, https://doi.org/10.1002/ggge.20230, 2013.
Hendy, E. J., Gagan, M. K., Alibert, C. A., Mcculloch, M. T., Lough, J. M., and Isdale, P. J.: Abrupt Decrease in Tropical Pacific Sea Surface Salinity at End of Little Ice Age, Science, 295, 1511–1514, 2002.
Holmes, J. A.: Sample-size implications of the trace-element variability of ostracod shells, Geochim. Cosmochim. Ac., 72, 2934–2945, https://doi.org/10.1016/j.gca.2008.03.020, 2008.
Holmes, J. A. and De Deckker, P.: The Chemical Composition of Ostracod Shells: Applications in Quaternary Palaeoclimatology, Dev. Quatern. Sci., 17, 131–143, 2012.
Holmes, J. A. and De Deckker, P.: Trace-element and stable-isotope composition of the Cyprideis torosa (Crustacea , Ostracoda) shell, J. Micropalaeontology, 36, 38–49, https://doi.org/10.1144/jmpaleo2015-024, 2016.
Hong, Y., Yasuhara, M., Iwatani, H., and Mamo, B.: Baseline for ostracod-based northwestern Pacific and Indo-Pacific shallow-marine paleoenvironmental reconstructions: ecological modeling of species distributions, Biogeosciences, 16, 585–604, https://doi.org/10.5194/bg-16-585-2019, 2019.
Ingram, C.: Palaeoecology and geochemistry of Shallow Marine ostracoda from the Sand Hole Formation, inner silver pit, southern North Sea, Quat. Sci. Rev., 17, 913–929, https://doi.org/10.1016/S0277-3791(98)00025-0, 1998.
Inoue, M., Nohara, M., Okai, T., Suzuki, A., and Kawahata, H.: Concentrations of Trace Elements in Carbonate Reference Materials Coral JCp-1 and Giant Clam JCt-1 by Inductively Coupled Plasma-Mass Spectrometry, Geostand. Geoanal. Res., 28, 411–416, https://doi.org/10.1111/j.1751-908X.2004.tb00759.x, 2004.
Irizuki, T., Matsubara, T., and Matsumoto, H.: Middle Pleistocene Ostracoda from the Takatsukayama member of the Meimi Formation, Hyogo prefecture, western Japan: significance of the occurrence of Sinocytheridea impressa, Paleontol. Res., 9, 37–54, https://doi.org/10.2517/prpsj.9.37, 2005.
Irizuki, T., Taru, H., Taguchi, K., and Matsushima, Y.: Paleobiogeographical implications of inner bay Ostracoda during the Late Pleistocene Shimosueyoshi transgression, central Japan, with significance of its migration and disappearance in eastern Asia, Palaeogeogr. Palaeoclimatol. Palaeoecol., 271, 316–328, https://doi.org/10.1016/j.palaeo.2008.11.002, 2009.
Ishizaki, K. and Kato, M.: The basin development of the Diluvium Furuya mud basin, Shizuoka Prefecture, Japan, based on faunal analysis of fossil ostracodes, 118–143, Progress in Micropaleontology, Micropaleontology Press, Amer. Mus. Nat. Hist., New York, 422 pp., 1976.
Ito, E., De Deckker, P., and Eggins, S. M.: Ostracodes and their shell chemistry: Implications for paleohydrologic and paleoclimatologic applications, Paleontol. Soc. Pap., 9, 119–152, https://doi.org/10.1017/s1089332600002187, 2003.
Kamiya, T.: Contrasting population ecology of two species of Loxoconcha (Ostracoda, Crustacea) in recent Zostera (Eelgrass) beds: Adaptive Differences between Phytal and Bottom-Dwelling species, Micropaleontology, 34, 316–331, https://doi.org/10.2307/1485600, 1988.
Kondo, H., Toyofuku, T., and Ikeya, N.: ratios in the shells of cultured specimens and natural populations of the marine ostracode Xestoleberis hanaii (Crustacea), Palaeogeogr. Palaeoclimatol. Palaeoecol., 225, 3–13, https://doi.org/10.1016/j.palaeo.2004.05.026, 2005.
Kumamoto Prefectural Fisheries Research Center: Automatic sea state observation, available at: http://www.nanotech.co.jp/kumamoto/Main2020.asp (last access: 5 February 2021), 2021.
Lea, D. W.: Elemental and isotopic proxies of marine temperatures, Ocean. Mar., 6, 365–390, https://doi.org/10.1016/B0-08-043751-6/06114-4, 2003.
Lea, D. W. and Boyle, E.: Barium content of benthic foraminifera controlled by bottom-water composition, Nature, 338, 751–753, https://doi.org/10.1038/338751a0, 1989.
Linsley, B. K., Wellington, G. M., Schrag, D. P., Matthews, H. E., Owen, T., Meier, R., and Nifio-southem, E.: Decadal Sea Surface Temperature Variability in the Subtropical South Pacific from 1726 to 1997 A.D., Science, 290, 1145–1148, 2000.
Martin, P. A., Lea, D. W., Rosenthal, Y., Shackleton, N. J., Sarnthein, M., and Papenfuss, T.: Quaternary deep sea temperature histories derived from benthic foraminiferal , Earth Planet. Sci. Lett., 198, 193–209, https://doi.org/10.1016/S0012-821X(02)00472-7, 2002.
Millero, F. J.: Physicochemical controls on seawater, in: The oceans and marine geochemistry, edited by: Elderfield, H., Elsevier, Pergamon, Amsterdam, The Netherlands, 2006.
Morton, B. and Wu, S. S.: The hydrology of the coastal waters of Hong Kong, Environ. Res., 10, 319–347, https://doi.org/10.1016/0013-9351(75)90029-8, 1975.
Niu, J.: Precipitation in the Pearl River basin, South China: Scaling, regional patterns, and influence of large-scale climate anomalies, Stoch. Environ. Res. Risk Assess., 27, 1253–1268, https://doi.org/10.1007/s00477-012-0661-2, 2013.
Okai, T., Suzuki, A., Kawahata, H., Terashima, S., and Imai, N.: Preparation of a new Geological Survey of Japan geochemical reference material: Coral JCp-1, Geostand. Newsl., 26, 95–99, https://doi.org/10.1111/j.1751-908X.2002.tb00627.x, 2002.
Owen, R. B. and Lee, R.: Human impacts on organic matter sedimentation in a proximal shelf setting, Hong Kong, Cont. Shelf Res., 24, 583–602, https://doi.org/10.1016/j.csr.2003.11.004, 2004.
Patra, S., Liu, C. Q., Wang, F. S., Li, S. L., and Wang, B. L.: Behavior of major and minor elements in a temperate river estuary to the coastal sea, Int. J. Environ. Sci. Technol., 9, 647–654, https://doi.org/10.1007/s13762-012-0097-8, 2012.
Roberts, L. R., Holmes, J. A., and Horne, D. J.: Tracking the seasonal calcification of Cyprideis torosa (Crustacea, Ostracoda) using -inferred temperatures, and its implications for palaeotemperature reconstruction, Mar. Micropaleontol., 156, 101838, https://doi.org/10.1016/j.marmicro.2020.101838, 2020.
Rodriguez, M.: Trace-elements in ostracods as proxies of environmental parameters in shallow marine waters, PhD thesis, The University of Hong Kong, Hong Kong SAR, China, 123 pp., 2021.
Rodriguez, M. and Not, C.: Ostracod Mg/Ca, Sr/Ca, Al/Ca and Fe/Ca ratios of Sinocytheridea impressa and Neomonoceratina delicata, Version 1.0, Interdisciplinary Earth Data Alliance (IEDA), https://doi.org/10.26022/IEDA/111891, 2021.
Rodriguez, M., Doherty, J. M., Hilary Man, H. L., Wang, R., Xiao, W., Zhou, B., Thibodeau, B., and Not, C.: Intra-valve elemental distributions in Arctic marine ostracodes: Implications for and paleothermometry, Geochem. Geophy. Geosy., 22, e2020GC009379, https://doi.org/10.1029/2020GC009379, 2020.
Rosenthal, Y., Lear, C. H., Oppo, D. W., and Linsley, B. K.: Temperature and carbonate ion effects on and ratios in benthic foraminifera: Aragonitic species Hoeglundina elegans, Paleoceanography, 21, PA1007, https://doi.org/10.1029/2005PA001158, 2006.
Sinclair, D. J., Kinsley, L. P. J., and Mcculloch, M. T.: High resolution analysis of trace elements in corals by laser ablation ICP-MS, Geochim. Cosmochim. Ac., 62, 1889–1901, https://doi.org/10.1016/S0016-7037(98)00112-4, 1998.
Snedden, G., Cable, J., and Kjerfve, B.: Estuarine Geomorphology and coastal hydrology, in: Estuarine ecology, edited by: Day, J., Crump, B., Kemp, M., and Yanez-Arancibia, A., John Wiley and Sons, Hoboken, New Jersey, USA, 19–38, 2013.
Tanaka, G., Komatsu, T., Saito, Y., Phong, D., Lan, Q., Nguyen, D. P., and Vu, Q. L.: Temporal changes in ostracod assemblages during the past 10,000 years associated with the evolution of the Red River delta system, northeastern Vietnam, Mar. Micropaleontol., 81, 77–87, https://doi.org/10.1016/j.marmicro.2011.08.001, 2011.
Tanner, P. A., Leong, L. S., and Pan, S. M.: Contamination of heavy metals in marine sediment cores from Victoria harbour, Hong Kong, Mar. Pollut. Bull., 40, 769–779, https://doi.org/10.1016/S0025-326X(00)00025-4, 2000.
Wang, H. E., Zhang, H., Cao, M., and Horne, D. J.: Holocene Ostracods from the Hang Hau Formation in Lei Yue Mun, Hong Kong, and their palaeoenvironmental implications, Alcheringa An Australas, J. Palaeontol., 43, 320–333, https://doi.org/10.1080/03115518.2018.1511830, 2018.
Whatley, R. and Zhao, Q.: A revision of Brady's 1869 study of the Ostracoda of Hong Kong, J. Micropalaeontology, 7, 21–29, https://doi.org/10.1144/jm.7.1.21, 1988.
Yamada, K., Irizuki, T., Ikehara, K., and Okamura, K.: Calibration of past water temperature in the Sea of Japan based on of ostracode shells of two shallow marine species in the genus Cytheropteron, Palaeogeogr. Palaeoclimatol. Palaeoecol., 410, 244–254, https://doi.org/10.1016/j.palaeo.2014.05.042, 2014.
Yu, J. and Elderfield, H.: in the benthic foraminifera Cibicidoides wuellerstorfi and Cibicidoides mundulus: Temperature versus carbonate ion saturation, Earth Planet. Sci. Lett., 276, 129–139, https://doi.org/10.1016/j.epsl.2008.09.015, 2008.
Yu, J., Day, J., Greaves, M., and Elderfield, H.: Determination of multiple element/calcium ratios in foraminiferal calcite by quadrupole ICP-MS, Geochem. Geophy. Geosy., 6, 1–9, https://doi.org/10.1029/2005GC000964, 2005.
Yuan, X.-C., Yin, K., Cai, W.-J., Ho, A. Y., Xu, J., and Harrison, P. J.: Influence of seasonal monsoons on net community production and CO2 in subtropical Hong Kong coastal waters, Biogeosciences, 8, 289–300, https://doi.org/10.5194/bg-8-289-2011, 2011.
Zhang, Q., Li, J., Singh, V. P., Xu, C. Y., and Deng, J.: Influence of ENSO on precipitation in the East River basin, south China, J. Geophys. Res.-Atmos., 118, 2207–2219, https://doi.org/10.1002/jgrd.50279, 2013.