the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Microbial biobanking – cyanobacteria-rich topsoil facilitates mine rehabilitation
Wendy Williams
Angela Chilton
Mel Schneemilch
Stephen Williams
Brett Neilan
Colin Driscoll
Restoration of soils post-mining requires key solutions to complex issues through which the disturbance of topsoil incorporating soil microbial communities can result in a modification to ecosystem function. This research was in collaboration with Iluka Resources at the Jacinth–Ambrosia (J–A) mineral sand mine located in a semi-arid chenopod shrubland in southern Australia. At J–A, assemblages of microorganisms and microflora inhabit at least half of the soil surfaces and are collectively known as biocrusts. This research encompassed a polyphasic approach to soil microbial community profiling focused on “biobanking” viable cyanobacteria in topsoil stockpiles to facilitate rehabilitation. We found that cyanobacterial communities were compositionally diverse topsoil microbiomes. There was no significant difference in cyanobacterial community structure across soil types. As hypothesised, cyanobacteria were central to soil microprocesses, strongly supported by species richness and diversity. Cyanobacteria were a significant component of all three successional stages with 21 species identified from 10 sites. Known nitrogen-fixing cyanobacteria Symploca, Scytonema, Porphyrosiphon, Brasilonema, Nostoc, and Gloeocapsa comprised more than 50 % of the species richness at each site and 61 % of the total community richness. In the first study of its kind, we have described the response of cyanobacteria to topsoil stockpiling at various depths and ages. Cyanobacteria are moderately resilient to stockpiling at depth and over time, with average species richness greatest in the top 10 cm of the stockpiles of all ages and more viable within the first 6 weeks, indicating potential for biocrust re-establishment. In general, the resilience of cyanobacteria to burial in topsoil stockpiles in both the short and long term was significant; however, in an arid environment recolonisation and community diversity could be impeded by drought. Biocrust re-establishment during mine rehabilitation relies on the role of cyanobacteria as a means of early soil stabilisation. At J–A mine operations do not threaten the survival of any of the organisms we studied. Increased cyanobacterial biomass is likely to be a good indicator and reliable metric for the re-establishment of soil microprocesses.
- Article
(2959 KB) - Full-text XML
-
Supplement
(1330 KB) - BibTeX
- EndNote
Following the destruction of the soil profile in a post-mining landscape there is a critical need to restore ecological integrity to the system. Mine rehabilitation is a complex process that involves many levels of understanding of difficult issues relating to ecosystem function. Harsh disturbance disrupts the spatial structure of soil microbial communities. Not least is the removal or burial of bioactive soils that will have knock-on effects for rehabilitation efforts such as nutrient cycling and plant re-establishment (Jasper, 2007; Tongway and Ludwig, 1996). Microorganisms are critical components of soils that drive assorted microprocesses and impact soil ecosystem function on several levels. The successful ecological restoration of arid mining sites relies on a holistic approach in which microbial recolonisation can serve as an indicator of the integrity of the wider ecosystem (Tongway, 1990). In this sense, recent restoration approaches in arid landscapes include the re-establishment of surficial crusts that develop as a protective skin across the bare soil or stony interspaces between plants. These encrusted surfaces can be physical, chemical, or biological (microbial) in nature. Well-established biological crusts (biocrusts) are intricately interwoven and structured high-functioning communities that are variable in composition (incorporating cyanobacteria, algae, lichens, mosses, liverworts, microfauna, and bacteria). Biocrusts are a significant asset to arid soil ecosystems providing a protective nutrient-rich layer closely integrated into the soil surface (Delgado-Baquerizo et al., 2013; Maestre et al., 2012).
However, biocrusts are very sensitive to disturbance, and mining severely disrupts and compromises their structure and function. The recognition of the absolute importance of microbial communities in the topsoil has led to the accepted practice of stripping and storing in stockpiles as a form of “biobanking” for redistribution at the cessation of mining. During this process the crushing and burial of biocrusts results in the inability of crust organisms to photosynthesise due to the lack of light. Massive soil disturbance results in a loss of structure and resources and often has long-lasting effects on energy transfers, soil stability, nutrient cycling, and surface hydrology (Bowker, 2007; Tongway and Hindley, 2004). Physical disturbance can profoundly disrupt biocrust integrity, composition, and physiological function whereby the impact is governed by site characteristics, severity, frequency, and timing (Belnap and Eldridge, 2001). Biocrust burial as a result of environmental stress and disturbance can result in serious impacts on biocrust microorganisms' viability and function. For example, grazing and drought caused significant declines in cyanobacterial richness and abundance, which resulted in a reduction in soil nutrient concentrations (Rao et al., 2012; Williams and Eldridge, 2011).
Following disturbance, restoration and regrowth of biocrusts can take place unassisted and seasonally driven generally over many years (Belnap and Eldridge, 2001; Belnap and Gillette, 1998). Should biocrust organisms remain inactive while they are wet, cell death and decomposition commonly occur (Kidron et al., 2012; Rao et al., 2012). Nevertheless, in dry conditions, cyanobacteria and algae are known to remain desiccated and viable for millions of years (Vishnivetskaya et al., 2003). Alternatively, assisted biocrust restoration places emphasis on the recovery of ecosystem function and necessarily addresses environmental constraints. This incorporates knowledge of the “potential condition” based on experience with sites of ecological similarity that have undergone disturbance and recovery (Bowker, 2007). Biocrust recovery can be altered by dust deposition, fire, and climatic conditions (Weber et al., 2016). When biocrusts recover naturally soil properties change. For example, in southern African and the Spanish rangelands an incremental accumulation of soil nutrients and organic matter and a build-up of silt and clay lead to the development of a resilient and multifunctional biocrust (Büdel et al., 2009; Maestre et al., 2012; Weber et al., 2016).
Restoration of ecosystem function post-disturbance requires an appreciation of the dynamic functional status of the landscape prior to disturbance (Tongway and Ludwig, 1996), as well as an understanding of the net accumulative effects of disturbance on the components of the system. On the micro-scale, cyanobacterial species richness contributes to soil ecosystem function through microprocesses including carbon fixation through photosynthesis, atmospheric nitrogen fixation in a biological-available form, micronutrient breakdown and release, soil particle cohesion, regulation of moisture, and soil surface structure (Delgado-Baquerizo et al., 2013; Elbert et al., 2012; Hu et al., 2002; Maestre et al., 2012 and others). Consequently, it is necessary to appreciate the microprocesses that will assist in the restoration of soil function and to monitor recovery along the way.
Cyanobacterial re-establishment is a key indicator of early soil surface re-stabilisation, regulation of soil moisture, and the balancing of soil carbon and nitrogen (Chamizo et al., 2012; Mager and Thomas, 2011). Cyanobacteria in arid landscapes are exceptionally well-adapted to desiccation. Their polysaccharide sheaths and EPS production perform a vital role in maintaining cyanobacterial cell integrity, exchange of information, and absorption of water during rehydration (Rossi et al., 2017). EPS has adhesive properties that bind non-aggregated soil particles into a protective encrusted surface that reduces the destructive impacts of wind and water (Eldridge and Leys, 2003; Rossi et al., 2017). Cyanobacterial biofilms provide stabilisation of initially disturbed surfaces that pave the way for diverse microbial communities and form bioactive crust-like layers assimilated into the soil (e.g. Büdel et al., 2009; Rossi et al., 2017; Bowker et al., 2014). As biocrusts develop in structural complexity, the diversity of organisms is regulated by water infiltration, temperature, light, and additional disturbance (Belnap and Eldridge, 2001; Büdel et al., 2009; Elbert et al., 2012).
Research into biocrust disturbance with a focus on recovery post-mining is rare. In the Namaqualand arid lands (Namibia, South Africa) low rainfall and high winds impact the rehabilitation of degraded lands following diamond mining and grazing (Carrick and Krüger, 2007). Researchers have found that cyanobacteria and non-vascular plants that form a living and protective surface crust were crucial to surface stabilisation. Jasper (2007) also recognised the importance of soil microbial communities, including cyanobacteria, in post-mine rehabilitation in the Jarrah forests of south-western Australia. In the Czech Republic and Germany chrono-sequential studies of old brown coal mine sites found that green algal biofilms and a diverse range of cyanobacteria initiated the rehabilitation of the soils (Lukešová, 2001). In serpentinite mine tailings (New South Wales, Australia), McCutcheon et al. (2016) showed that filamentous cyanobacteria accelerated carbonate mineral precipitation and stabilised the tailings. They demonstrated that cyanobacteria had the capacity to adsorb magnesium while acting as a nucleation site and sequestering carbon. In our current study, preliminary research found that in undisturbed chenopod shrublands at the edge of the Nullarbor Plain (South Australia) biocrusts cover more than 45 % of the soil surface between the grass plants, and post-mining rehabilitation needs to take their role into account (Doudle et al., 2011). It follows that there is a real need to focus on practical approaches that contribute to the restoration of soil function and measure relevant aspects of success through soil microbial communities and biocrust re-establishment, especially cyanobacteria (for example: Setyawan et al., 2016; Mazor et al., 1996; Fischer et al., 2014; Chiquoine et al., 2016; Doherty et al., 2015; Harris, 2003; Tongway and Hindley, 2004; Zhao et al., 2014).
To our knowledge, the effects of topsoil stockpiling on biocrust organisms, such as cyanobacteria, and their recovery time following topsoil spreading has not been previously investigated. In this research we focused on the cyanobacterial component of the biocrusts. This was in keeping with the mining framework in the ongoing development of informed rehabilitation plans that focus on improved long-term outcomes. Overall, we sought to determine whether shallow biobanks of cyanobacterial-enriched topsoil would facilitate the recovery of essential soil microprocesses when re-spread following mine disturbance. The specific aims of the microbial biobanking research programme were to (a) define the cyanobacterial community structure applying a polyphasic approach with a special focus on species that drive early colonisation, nutrient cycling, and soil stabilisation and (b) to examine the effects of stockpiling topsoil on cyanobacterial resilience to crushing and burial and their recovery following spreading of topsoil back across mined land. We hypothesised that cyanobacterial survival within a topsoil stockpile would be reduced with both depth within the stockpile and elapsed time before topsoil re-spreading.
2.1 Background and site description
The Jacinth–Ambrosia (J–A) heavy mineral sand mine is located on the eastern edge of the Nullarbor Plain, South Australia, across the boundary of two regional reserves within the Eucla Basin region. The climate is semi-arid with a mean rainfall of 185 mm, mean maximum temperature of 27.6 ∘C, and minimum temperature of 12.1 ∘C (further detail provided in Fig. S1 in the Supplement; http://www.bom.gov.au, last access: 14 June 2018). Tertiary sediments deposited in marine and terrestrial settings and the soil distribution of the area reflect the geological history, with at least five marine transgression and regression events depositing 40–50 m of sediments (Hou and Warland, 2005). The landscape is broadly undulating with low open woodlands that have a shrub understorey with chenopod shrublands as well as dune fields that consist of parallel dunes and inter-dune swales (Doudle et al., 2011; Gillieson et al., 1996). Prior to mining disturbance, the landscape is superficially homogenous chenopod-dominated vegetation but functionally patchy on the fine scale at which the soil surfaces are extensively colonised by biocrusts. At J–A, biocrusts cover around 45 % of the landscape surfaces, equating to 2000 hectares of the mining lease, and had been previously classified into three successional stages representative of the five biocrust types found growing across the landscape (Tables S1, S2 in the Supplement) (Doudle et al., 2011). Types 1–2 are light-coloured, patchy, thin, and fragile cyanobacterial crusts corresponding to early stages of development; type 3 includes well-established cyanobacterial crusts with the establishment of some mosses and lichens corresponding to intermediate stages of development; type 4–5 biocrusts are well-established with cyanolichens and/or green algal lichens and mosses corresponding to late stages of development (additional descriptions available in Table S1). In this study the term biocrust covers whole crust samples that incorporated lichens, cyanobacteria, and mosses in varying proportions; however, the cyanobacterial component of this crust was the focus in terms of our polyphasic approach to community structure, succession, and its biophysiochemical properties.
At J–A the landscape has been characterised into three distinct soil types that were associated with vegetation communities identified as soil management units (SMUs) (Doudle et al., 2011; Hou and Warland, 2005). SMU 1 is characterised by deep calcareous yellow sands associated with dune ridges and dominated by red mallee: Eucalyptus oleosa ssp. oleosa, with open mallee–myall woodland. SMU 2 is made up of shallow calcareous sandy loams dominated by chenopod shrubland: Maireana sedifolia and Atriplex vesicaria. SMU 3 is comprised of western myall: deep calcareous sandy loam dominated by Acacia papyrocarpa Maireana sedifolia, with open myall woodland. Site 1 occurs in a transition between SMU 2 and SMU 3 but was treated as most like SMU 2 (see also Table S1). Sample site locations (Fig. 1) were selected based on these SMUs and a 2-year-old stockpile (Table S1). Secondarily, sites within these SMUs were selected for subsequent detailed studies of cyanobacterial succession and its resilience to longer-term stockpiling (Fig. 2 and Sect. 2.4). On two occasions (July 2011 and March 2012) we sampled the biocrusts across the three main SMU sites (10 sites; see Fig. 1).
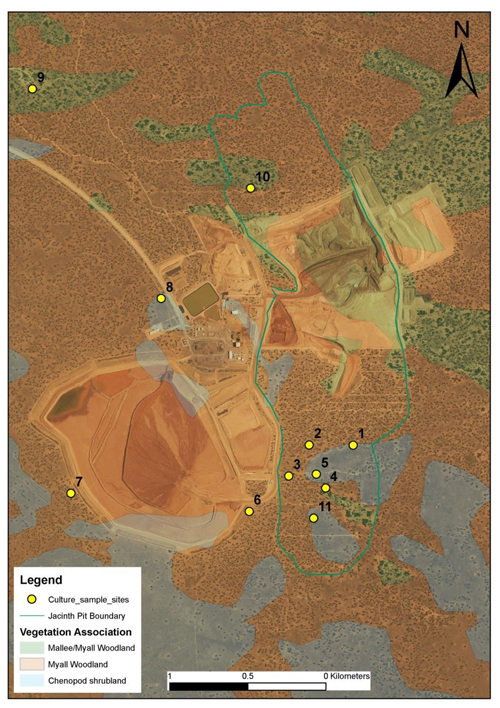
Figure 1Image of biocrust sample sites located within the vegetation associations described in Table S1 (supplied by Samantha Doudle). Site 11 was initially investigated but later discarded as it was a fourth replicate of the chenopod shrubland.

Figure 2(a) SMU 1 (Sites 4, 9, and 10): type 1–3 biocrusts on deep calcareous yellow sands (dunes). (b) SMU 2 (Sites 1, 5, and 8): primarily type 4 and 5 biocrusts on shallow calcareous sandy loam. (c) SMU 3 (Sites 2, 3, and 7): type 1–5 biocrusts on deep calcareous sandy loam (photographs by Samantha Doudle, 2011). (d) Different biocrust stages (top) north of stockpile 19, adjacent to Site 9; (e) bare, (f) early, and (g) late stages, also showing biocrust sample already removed from the late stage (photographs by Angela Chilton).
In the process of mining and preparation of the J–A site for future rehabilitation, topsoil (0–100 mm) is stored in low stockpiles, generally less than 2 m in depth, and stockpiles are comprised of topsoil sourced from only one vegetation type. Later stockpiles will be returned to the surface of mined areas in the rehabilitation process, and the cyanobacterial activity therein must be quantified to enable educated planning and decision-making regarding biocrust re-establishment. If low levels of biocrust organisms are detected below the top few centimetres, the addition of propagated biocrust organisms (e.g. cyanobacteria) to returned topsoil may be warranted. Activity and species richness within the stockpiled topsoils may also vary with age, and this may also influence the establishment of vegetation in the rehabilitation process.
2.2 Biophysical characteristics of biocrusts and cyanobacteria
2.2.1 Field sampling
The preliminary identification of biocrust types had been determined by Doudle et al. (2011) and provided the baseline data for biocrust sampling from Sites 1–10 within J–A (Fig. 1). The sites selected encompassed the three SMUs and five crust types (Table S1). All sites are of naturally occurring biocrusts except for Site 6, which is from a 2-year-old topsoil stockpile regarded as an early (type 1) biocrust representing recovery 2 years post-disturbance (Fig. 2a–c). Within each site, eight 10 cm diameter samples were selected at random and removed to a depth of 1 cm using a metal scraper (n=80), air-dried (>40 ∘C), and stored in Petri dishes. Each Petri dish contained approximately 80 g of crust. The samples were packed to avoid crust disruption and transported to the University of Queensland's Central Analytical Laboratory at Gatton.
2.2.2 Biocrust biophysiochemical properties
For each site, about half the sample was removed from each Petri dish and fine-sieved (1.70 mm). Duplicate subsamples (∼2 g) were analysed for total C and N and the C:N ratio using a high-temperature digestion in a vario MACRO elemental analyser (Elementar) (n=6 per SMU). Duplicate samples (10 g) for the purpose of analysing soil pH and electrical conductivity (EC) were prepared using a 1:5 (soil to water) ratio and shaken for 1 h. Following shaking, samples were left to stand for 30 min and EC was measured using a Crison conductivity meter 525. The sample was mixed again, and pH was measured with a TPS pH meter MC-80 using an ionode IJ44C electrode. The remaining half of the Petri dish was used to determine the chlorophyll a concentration of the biocrusts. Following resurrection (by moistening) the crust was lightly homogenised and a 5 g sample used at a 1:5 ratio of (dry weight) biocrust to dimethyl sulfoxide (DMSO) (Barnes et al., 1992) with samples placed in a warm bath (65 ∘C) for a 2 h of dark extraction, followed by centrifuging for 5 min (5000 g RCF). Chlorophyll a concentration was determined using Wellburn's (1994) equations.
Prior to the use of the samples for analysis a pocket penetrometer (8 mm foot) was used to determine the compressive strength (kg cm2) of the dry intact biocrust samples. Overall, the crust thickness was <0.5 cm. Each sample (10 cm diameter × 2 cm of depth) was placed on a solid surface and a total of 12 measurements (three readings for each of four reps) were taken for each site. The measurement was taken at the point when the crust was broken and the foot penetrated the crust surface.
At J–A we measured the photosynthetic performance (recorded as yield, YII) of the biocrusts using a pulse-amplitude modulated (PAM) fluorometer (Pocket PAM; Gademann Instruments, Germany). The goal was to demonstrate photosynthetic yield (YII) indicative of active growth of the biocrusts using the detection of chlorophyll fluorescence from photosystem II (PSII). The sensor was placed onto the biocrust and, once started, a series of short pulses of excitation light at high intensity was amplified, resulting in a brief closure of PSII and the measurement of fluorescence yield based on the Genty parameter, which is the quantum yield (YII) of the charge separation of PSII (Genty et al., 1989) recorded on a scale of 0–1 for all photosynthesis. In the field this process was completed at least six times adjacent to each sampled location (6×4 reps per site).
Significant differences in C, N, and C:N between SMUs as well as differences in chlorophyll a and YII between SMUs and the 2-year-old stockpile were tested by one-way ANOVA and Tukey's post hoc tests (Minitab 18).
2.3 Cyanobacterial community structure
2.3.1 Microscopy of biocrust cyanobacteria
Prior to the destruction of the samples for analysis, a portion of the field sample (Sect. 2.2) subsections were removed from six of the eight Petri dishes, placed into six-cell plates, used to enumerate cyanobacterial richness and diversity, and used to classify colonies. These dried crust samples were resurrected in the glasshouse for 3 to 5 days. For each of the 10 sites, 12 replicates (subsampled from the eight samples) were analysed via light microscopy. For each replicate, a minimum of two wet-mount slides incorporating six representative portions of the cyanobacterial colonies were examined (n=144 colonies per site). For the dominant land type, chenopod shrubland (Site 8), there were an additional 10 × 6-cell multi-well plates. These were treated similarly whereby two slides were examined from each of the 60 multi-wells (n=120). In total >2184 cyanobacterial colonies were examined. Initial inspection of the biocrust and the separation of individual species were made using an Olympus SZH10 microscope at 70 × magnification. Cyanobacterial filaments or colonies were carefully extracted with forceps to recover sufficient material that included important morphological features such as their colour, encasing sheaths, and cellular structure. Live material was examined by Nomarski differential interference contrast (DIC) microscopy with a Jenaval (Jena Zeiss) and an Olympus BX51 compound microscope (magnifications 400–1000 ×). Photomicrographs were taken using an Olympus SC100 digital microscope camera, and morphological measurements of vegetative cells were made from digital images of live material taken at 400 × magnification using Olympus cellSens® digital imaging software.
Identification was performed to a species level (wherever possible) in the laboratory using the following taxonomic references: Anagnostidis and Komarek (2005), Komárek and Anagnostidis (2005), Sant'Anna et al. (2011), and Skinner and Entwisle (2002). It was often necessary to record the closest named species as attributes varied somewhat-to temperate climate and aquatic specimens described in the literature. Nitrogen fixing cyanobacteria were identified based on the three recognised types: (1) heterocyte-forming species (those with specialised N-fixing cells); (2) non-heterocyte-forming species that fix N aerobically; and (3) non-heterocyte-forming species that fix N anaerobically (Bergman et al., 1997; Stal, 1995). Using a graticule, abundance was ranked on a scale of 1–8 and the main taxa are ranked in decreasing order of the relative percentage area occupied in a single view (Biggs and Kilroy, 2000). More than one species could be dominant, and all other taxa were ranked in relation to the dominant taxa as abundant, common, occasional, and rare. To determine similarities between cyanobacterial communities, cluster analysis, SIMPROF, and non-metric multidimensional scaling (nMDS) were conducted using Primer v6 (Clarke and Gorley, 2001).
2.3.2 16S rDNA profiling of native undisturbed biocrust microbiomes
For genomic profiling of naturally occurring successional biocrust communities, a location adjacent to Site 9 was visually determined to contain bare, early (crust types 1–2, SMU 1), or late (crust types 4–5, SMU 2, SMU 3) stages of development (Table S2). Biocrust successional features were determined by morphological attributes of pigmentation, thickness, and surface roughness as well as the presence–absence of lichens and mosses (Fig. 2d) (Chilton et al., 2017). The bare stage was characterised by loose soil particles with no visible biocrust structure. Samples were collected in July 2014. For each successional stage (representative of SMUs 1–3) , three replicates were collected, and a 10 cm2 plot with 95 % coverage of the desired biocrust stage was excised to the depth of the crust and non-aggregated soil was discarded (Fig. 2e–g). Samples were processed at UNSW, Sydney.
Each biocrust replicate for the bare, early, and late stages of development was homogenised and genomic DNA extraction performed using the FASTDNA Spin Kit for Soil (MP Bio Laboratories, USA) according to the manufacturer's instructions. Molecular libraries of the 16S rDNA V123 hypervariable region were generated via PCR as per Chilton et al. (2017) and submitted to the Ramaciotti Centre for Genomics (UNSW, Australia) for a 2×300 bp sequencing run on an Illumina MiSeq instrument. Sequencing data were processed using Mothur version 1.34.0 (Schloss et al., 2009) and are described in detail in Chilton et al. (2017). Singleton and doubleton OTUs were removed and samples rarefied to 8598 sequences each across 3785 OTUS. The curated Greengenes database (McDonald et al., 2012) was used to assign taxonomy to OTUs. Diversity values were derived using the DIVERSE function within the Primer package (Anderson et al., 2008) based upon standardised OTU values. ANOVA with post hoc Tukey's tests was used to test for significant differences between stages. Multivariate analyses were performed in Primer upon a Bray–Curtis dissimilarity matrix generated from square-root-transformed abundance data. Samples were represented in two- and three-dimensional space within an nMDS plot. Pair-wise, a posteriori comparisons of factor stage were performed using the PERMANOVA function with 9999 Monte Carlo permutations. The homogeneity of dispersion for each stage was tested using PERMDISP.
Table 1Intact biocrust soil physicochemical descriptions for all sites. EC: electrical conductivity in µS cm−1; total percentage of nitrogen present (N%), total percentage of carbon present (C%), and carbon-to-nitrogen ratios (C:N) for all sites. Different letters indicate significant differences in columns.
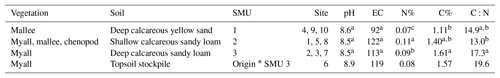
* SMU 3 is the origin of the topsoil stockpile aged 2 years, which was not included in the data analysis with SMUs 1–3 as there were only two samples.
2.4 Cyanobacterial tolerance to stockpiling
Stockpile sampling was carried out in March 2012 with samples sourced from SMU 3 areas with Acacia papyrocarpa (western myall) overstorey (Table S3). Three different age stockpiles (9, 20, and 29 months) were sampled in triplicate at six depths (0–2, 2–4, 4–6, 10, 25, and 50 cm). This resulted in 18 replicates for each soil depth for each stockpile age. Holes were dug to >50 cm of depth with a shovel and the exposed profile was removed with an ethanol-wiped spatula before taking a soil sample. A second sample at 50 cm was autoclaved to serve as a culturing control. Samples were stored in paper bags and processed at the University of Queensland. For each sample, 20 g of soil was set up in Petri dishes with 9 mL of water and sealed with Parafilm®. Petri dishes were incubated for 6 weeks at 26 ∘C under a 12 h photoperiod regime and rotated weekly to prevent site-specific effects. Sample moisture was maintained by the addition of sterile water within a laminar flow cabinet. To determine cyanobacterial growth and species richness, wet mounts for each sample were examined under 16 × magnification. Identification was carried out as per the methods detailed in Sect. 2.2.1. Cyanobacterial thalli and colony sizes were estimated using the area of coverage of the field of view.
A two-way ANOVA was initially run with stockpile age and depth as independent variables and species richness as the effect. If there was no interaction between age and depth a one-way ANOVA with Tukey's post hoc test was run between the significant independent variable and species richness. Similarity analysis (hierarchical agglomerative clustering with the SIMPROF test and non-metric multidimensional scaling; Primer 7) was used to determine whether there was a significant difference in relative species abundance at each depth.
3.1 Biophysical characteristics of biocrusts and cyanobacteria
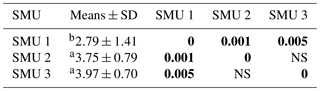
Across the three SMUs mean soil pH ranged from 8.5–8.6, while the 2-year-old topsoil stockpile was higher at pH 8.9 (Table 1). Electrical conductivity ranged from 92–140 µS cm−1. Total nitrogen was typically <0.1 % across all sites and total carbon ranged between 1 % and 2 % with higher percentages generally found across SMU 2 and 3 (Table 1). The ratio between carbon and nitrogen was the greatest across SMU 3 and the 2-year-old topsoil stockpile, also originating from SMU 3, whereas the lowest values were found on SMU 2. There were no significant differences between SMU 1, SMU 2, and SMU 3. Compressive strengths across Sites 4, 9, and 10 (SMU 1) were significantly lower than SMU 2, SMU 3, and the 2-year-old topsoil stockpile (Table 2).
Table 2Biocrust compressive strengths measured with a penetrometer; means and standard deviations (SDs) for SMUs 1–3 (kg cm−2). Tests with p values (in bold) that are different from 0 with a significance level of alpha = 0.05; letters indicate significant differences.
Mean chlorophyll a concentration ranged between 10.34 and 13.64 µg g−1 of soil for the intact undisturbed biocrusts. From the three different SMUs the lowest values were found at SMU 1 (10.34±2.13, p=0.000), whereas SMU 2 and SMU 3 showed no significant differences between them (Fig. 3). The mean chlorophyll concentrations of biocrusts sourced from the 2-year-old topsoil stockpile (7.49±1.01 µg g−1 soil) were almost half those of SMU 3 (13.53±1.74 µg g−1 soil), which was the origin of the topsoil stockpile. The photosynthetic yields recorded ranged between 0.073 (stockpile) and 0.147 (SMU 2). SMU 3 and the 2-year-old stockpile showed lower values than SMU 1 and SMU 2 (p=0.017), whereas SMU 1 and SMU 2 were not significantly different (Fig. 3).
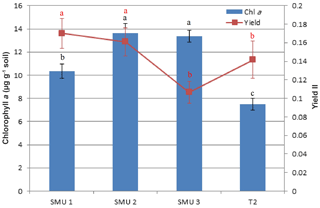
Figure 3Chlorophyll a concentration (µg g−1 soil) following resurrection (2 weeks) after a natural desiccation (dry) period of 6 months. Soil management units: SMUs 1–3 and T2 (2-year-old topsoil stockpile); photosynthetic yield (YII) of photosystem II (PSII), with mean values and standard error of the mean (SEM). Different letters indicate significant differences between SMUs.
3.2 Cyanobacterial community structure
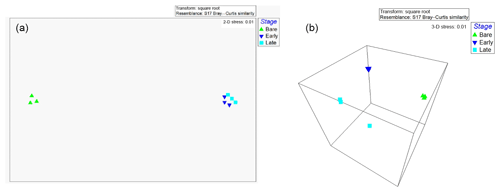
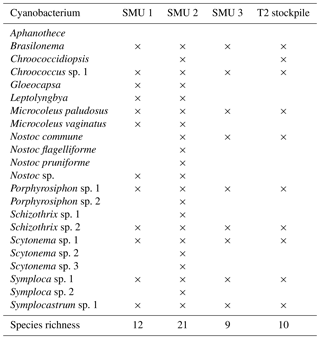
A total of 21 cyanobacterial species were identified across the 10 sites using microscopy (Table 5; Figs. S2–S9). The majority of species richness and abundance was comprised of four filamentous genera: Symploca (18 %), Schizothrix (16 %), Porphyrosiphon (16 %), and Scytonema (16 %). The secondary cyanobacteria present variously occupied <1 % to 10 % and overall made up 34 % of the community. Symploca occurred more frequently and was more abundant in the majority of the samples examined; therefore, it was the most significant contributor to the community (data not shown). The known nitrogen-fixing cyanobacteria Symploca, Scytonema, Porphyrosiphon, Brasilonema, Nostoc, and Gloeocapsa comprised more than 50 % of the species richness at each site and formed 61 % of the total community richness (Fig. 4). When the cyanobacterial community structure of different soil types was compared we found no significant difference in cyanobacterial community structure across soil types; however, the results do suggest that some spatial structuring exists across the three SMUs (Fig. 5a). Moreover, the structural relationship between all samples shows greater similarities rather than dissimilarities, and there are only a small number of samples that are significantly different (Fig. 5b). Soil type did not explain these differences. Individual sites generally displayed similar trends although there was some variability occurring between sites.
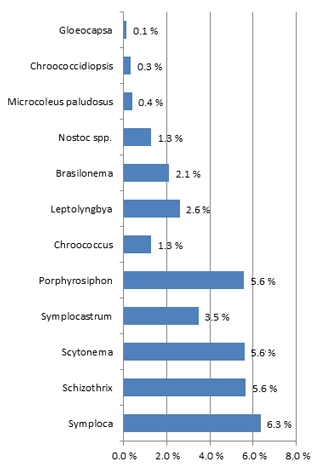
Figure 4Cyanobacterial community structure across all sites expressed as a percentage of the total community based on mean richness and abundance scores. N-fixing cyanobacteria contributed to 61 % of the community structure.
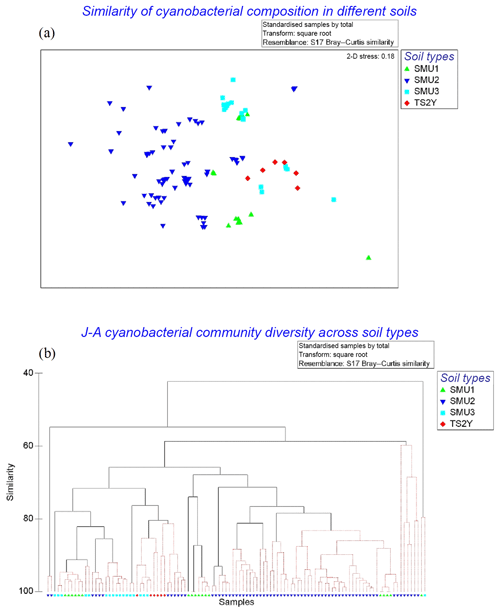
Figure 5(a) Cyanobacterial community structure based on indexed abundance and diversity across all sites displayed in an nMDS plot (Bray–Curtis similarity). Soil management units represented by SMUs 1–3 and 2-year-old topsoil stockpile (TS2Y). (b) Similarities between samples within their SMU are displayed in a Bray–Curtis dendrogram. Black continuous lines show significant differences between samples (p=0.05), and lighter lines indicate that most samples were not significantly different to each other.
Of the 21 cyanobacteria species found at J–A, more than half (12 species) were identified in SMU 1 where four primary genera made up 75 % of the community: Symploca, Schizothrix, Scytonema, and Symplocastrum (for more detail see Fig. S10). Cyanobacterial crusts from the dune regions on SMU 1 (deep calcareous yellow sands) were representative of crust types 1–3: patchy, brittle (when dry), early successional crusts as well as formed dark crusts that were middle to late successional and included cyanolichens (see also Doudle et al., 2011).
Cyanobacterial crusts from the chenopod shrublands and open woodlands in SMU 2 (shallow calcareous sandy loam) represented a broad range of crust types (2–5) but overall could be described as late successional. Lichens and mosses were highly visible (see also Doudle et al., 2011). There were 21 cyanobacteria recorded: four were primary genera that made up 63 % of the community, including Schizothrix, Porphyrosiphon, Scytonema, and Symploca (for more detail see Fig. S11). Cyanobacterial crusts from the open woodlands in SMU 3 (deep calcareous sandy loam, Fig. 2c) represented a broad range of crust types (2–5), but like SMU 2 could also be described as late successional. Lichens and mosses were also highly visible (see Doudle et al., 2011). There were nine cyanobacteria recorded, four of which were primary genera that made up 85 % of the community: Symploca, Porphyrosiphon, Scytonema, and Schizothrix (for more detail see Fig. S12). Cyanobacteria with the capacity to fix nitrogen contributed to 77 % of the community structure.
Cyanobacterial crusts from the 2-year-old topsoil stockpile that had originated from SMU 3 (deep calcareous sandy loam) would be described as early successional crusts with some seasonal mosses. There were eight cyanobacteria recorded, four of which were primary genera that made up 84 % of the community: Symploca, Symplocastrum, Porphyrosiphon, and Scytonema (see also Fig. S13). It was interesting to note that Symplocastrum was co-dominant with Symploca, whereas in the other communities it ranged between 8 % and 13 %. The subsurface species Schizothrix (found in top 5 mm) only contributed to 4 % of the richness compared to 10 %–20 % elsewhere. Cyanobacteria with the capacity to fix nitrogen (Symploca, Porphyrosiphon, Scytonema, and Brasilonema) contributed to 61 % of the community (Fig. 4).
3.3 16S rDNA profiling of native undisturbed biocrust microbiomes
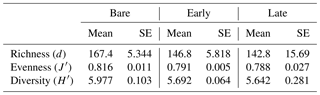
Microbial community profiling using high-throughput sequencing revealed that cyanobacteria comprised a significant component of all three stages, forming the majority of sequences in the early and late stages (Fig. 6a). There was a diversity of morphotypes observed including simple filamentous, heterocyte-forming, and unicellular types (Fig. 6b). The most abundant genera identified were Leptolyngbya, Phormidium, Tolypothrix, Nostoc, Brasilonema, Chroococcidiopsis, and Acaryochloris. Unclassified Nostocaceae were dominant within bare soils, while the early stages observed a relative even increase in Phormidium, Brasilonema, and the unicellular genera (e.g. Chroococcidiopsis, Acaryochloris, Xenococcaceae). Late-stage biocrusts showed a slight resurgence of Nostocaceae. There was no significant difference in the richness, evenness, or diversity among the three stages (Table 3). However, there were significant differences in the composition and structure of the communities of each stage. PERMANOVA analysis showed that there were significant differences in the composition and structure of the communities of each stage (pseudo-F = 4.9544, P(perm) = 0.004, unique perms: 273). Post hoc pair-wise comparisons showed that the bare stage was significantly different to the crusted stages (Fig. 7, Table 4), while the resolution between the early and late stages was less clear. The three-dimensional ordination of the samples showed a separation and grouping between the early and late stages (Fig. 7).
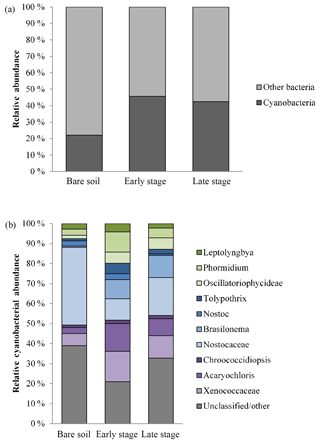
Figure 6(a) Relative abundance of cyanobacteria to other bacteria within bare soil and early- and late-stage biocrusts. (b) Abundance of cyanobacterial genera and groups. Green: simple filamentous types, blue: heterocystic types, purple: unicellular. grey: unclassified or other (includes chloroplasts).
3.4 Cyanobacterial tolerance to stockpiling
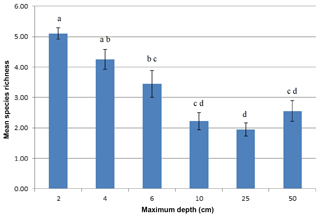
Cyanobacteria species richness in the stockpiles was affected by sampling depth (p=0.000), whereas stockpile age and its interaction with sampling depth did not exert any significant effect (p=0.378). As shown in Fig. 8, species richness drastically decreased from 2 cm (5.11±0.79) to 10 cm (2.22±1.18). Then it fluctuated around 1.94–2.56 with no significant change between 10 and 50 cm (Fig. 8).
Five identifiable cyanobacterial genera were found in the stockpile soil samples: Nostoc, Scytonema, Microcoleus, Porphyrosiphon, and Leptolyngbya. Nostoc cf. commune, Porphyrosiphon, Microcoleus and Scytonema were the first genera to develop to an identifiable stage. Filaments of the Stigonema genus were found in low numbers and appeared to be recently formed. It was only present in samples examined in the latter stages of the identification process. Nostoc sp. (yellow) exhibited a much slower rate of development than Nostoc cf. commune and could only be definitively determined as a form of Nostoc when examined after 13 weeks of incubation.
Table 4Permutational analysis of variance (PERMANOVA) of pair-wise comparisons of Bray–Curtis dissimilarity between biocrust stages and bare soil. P(MC): probability values obtained using 9999 Monte Carlo permutations. A test for homogeneity of multivariate dispersions (PERMDISP) showed no significant differences in variation for the spread of samples (pseudo-F=3.8068, P(perm) = 0.068). Significant pair-wise differences are in bold.
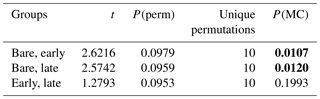
Table 5Diversity across sites on a presence–absence basis for all seasons and the 2-year-old stockpile (T2). Different species attributed to a genus (i.e. sp. 1, 2, 3) have all been separated based on their morphological features and size but could not be positively identified.
This research has demonstrated cyanobacteria to be a key component of soil microbial communities at J–A. These were compositionally diverse topsoil microbiomes that substantially contributed to the myall–chenopod landscape. In this study cyanobacterial community richness and abundance were not related to soil and landform type, and this was further supported through the sequenced samples for which no significant differences were observed. At J–A any of the cyanobacteria could conceivably occur anywhere across the landscape. Their relative abundance was most probably determined by microenvironments and microhabitats such as light (sun and shade) and chemical gradients (Stal, 2003), as well as moisture availability (Büdel et al., 2018) and soil particle size (Büdel et al., 2009). We had hypothesised that cyanobacteria would be central to soil microprocesses, and this was strongly supported by extensive species richness and diversity values. The results have demonstrated how these microprocesses provide a strong foundation for the restoration of soil function. At J–A cyanobacteria contributed to soil structure and function during the early developmental stages of the biocrust. Photosynthesis drove the productivity and growth of the biocrust that initiated carbon and nitrogen cycling and resulted in increases in soil nutrient concentrations right where vascular plants might use them.
Yet we found that various cyanobacteria responded differently to high-level disturbance. In the first study of its kind we have shown that there were no time–depth interactions over the different ages of the stockpiles. The greatest determination of species richness occurred at quite shallow depths with highly significant decreases in richness beyond 6 cm of depth, even in the 9-month-old stockpile. Nonetheless, the resilience of the individual cyanobacterial species to burial in topsoil stockpiles appeared good; however, in an arid environment recolonisation and community diversity could be impeded by drought (Williams and Büdel, 2012).
4.1 Cyanobacterial community structure
Overall, at J–A there was a rich cyanobacterial community comprised of 21 species recorded from 13 genera. Four species were unicellular and the remaining 17 were filamentous. Some cyanobacteria found at J–A (Microcoleus paludosus, Nostoc sp., Gloeocapsa) had also been recorded at Lake Gilles (SA) about 400 km south-east of J–A (Ullmann and Büdel, 2001). Surprisingly though, Microcoleus species that were recorded at J–A did not dominate the biocrust compared with many reports from the United States, Asia, and elsewhere (see e.g. Belnap and Eldridge, 2001). This implies that the early colonisers such as Microcoleus would not necessarily play a dominant role in early stabilisation and colonisation of the soil. At J–A, Symploca and Scytonema appeared to be important as early colonising cyanobacteria in the biocrusts. These species have also been recorded as playing a key role in carbon sequestration in northern Australian cyanobacterial crusts (Büdel et al., 2018).
Elsewhere, multiple studies have demonstrated the high value of biocrust attributes as drivers of soil microprocesses that restore soil function (e.g. Barger et al., 2016; Belnap and Eldridge, 2001; Bowker et al., 2014; Büdel et al., 2009; Chilton et al., 2017; Chiquoine et al., 2016; Weber et al., 2016). Similarly, in south-western Queensland and northern Australia cyanobacterial species richness was strongly linked to increased bioavailable nitrogen and carbon uptake (Büdel et al., 2018; Williams et al., 2018; Williams and Eldridge, 2011). Environmentally induced strategies, which have developed over a long evolutionary history, reflect their habitat. Cyanobacterial richness at J–A was determined according to their morphological features (e.g. outer protective sheaths, UV protection, EPS production), which in many cases provided the basis for attributes that pertained to fundamental survival strategies.
Filamentous cyanobacteria formed the major part of the J–A crust structure with tufts, webs, or creeping masses closely intertwined (e.g. Porphyrosiphon, Symploca, Scytonema, Schizothrix, Microcoleus). These are often assimilated with unicellular forms (e.g. Gloeocapsa, Chroococcus, Chroococcidiopsis) or gelatinous colonies of Nostoc (see Fig. S16 for images of growth habits). Simple filamentous types are often attributed with the primary crust-building role, able to span inter-particle gaps within the soil via supra-cellular structures (e.g. Microcoleus; see Garcia-Pichel and Wojciechowski, 2009). Important crust-building cyanobacteria in this study also appeared to be Symploca that was associated with EPS production, a principal feature of early colonising crust formation (Hu et al., 2002). Sequencing data showed that Phormidium was the dominant cyanobacterium for this role and it is likely that Symploca, identified though microscopy, was the principal Phormidium present. Microcoleus sp. and Porphyrosiphon were also identified as early colonisers; however, these genera are currently poorly resolved phylogenetically (Garcia-Pichel et al., 2013) but share critical morphological features enabling biocrust formation and maintenance. Other key cyanobacteria indicative of biocrust formation and development were Leptolyngbya, Phormidium, Tolypothrix, Nostoc, Brasilonema, Chroococcidiopsis, and Acaryochloris. Notably, the identification of Brasilonema, not been previously recorded in Australian soils, was supported with sequencing data. The taxonomic status of Brasilonema remained uncertain and may be a variety of Scytonema; however, genomic data supported morphological identification and the type has also been recorded in other terrestrial habitats globally. Due to its similar morphological attributes and genomic data, in this study we called this cyanobacterium Brasilonema (Fiore et al., 2007; Vaccarino and Johansen, 2012). Many primary (common to abundant) and secondary (uncommon) cyanobacteria recorded at J–A exhibited thick gelatinous sheaths (Porphyrosiphon, Schizothrix, Microcoleus, Nostoc) or were associated with the production of EPS (Symploca, Nostoc, Schizothrix, Leptolyngbya).
Nostoc commune var. flagelliforme had been recorded at J–A along with Nostoc commune across the shallow and deep sandy loams. Although N. flagelliforme appeared rarely, it had been previously documented from sites in south-western South Australia, Western Australia, Northern Territory (Skinner and Entwisle, 2002), and Victoria (Wendy Williams, unpublished data). Nevertheless, it has now been documented that both Nostoc commune and N. flagelliforme are not separate species; rather, the spaghetti-like tubes are unique to the ecotype and likely associated with aridity (Aboal et al., 2016). This is supported by the semi-arid environment at J–A, and it may be more widespread in Australia than previously recorded as it is often only clearly visible following rain.
4.2 Cyanobacterial tolerance to stockpiling
Physical disturbance of biocrusts occurs on a large scale at the J–A mine site with the removal and temporary stockpiling of topsoil. This type of mechanical disturbance results in burial and translocation of the biocrust. The survival of cyanobacteria following burial has rarely been studied and has never been analysed in topsoil stockpiles. In China, artificial sand burial at shallow depths showed significant reductions in chlorophyll concentration, UV synthesis, total carbohydrates (EPS), and damage to photosynthetic activity (Rao et al., 2012). In a semi-arid grassland in Australia, wind-borne sand burial of cyanobacterial crusts during a severe drought resulted in a significant reduction in surface-dwelling cyanobacteria and significant reductions in biological-available nitrogen (Williams and Eldridge, 2011). In this study we have demonstrated that without further disturbance a range of cyanobacterial species survived stockpiling for over 2 years, an important feature underpinning the concept of biobanking soil microbial communities. Yet we also discovered that species richness was significantly greater only in the shallow surfaces of the stockpiles (i.e. <6 cm) and that there were significant losses between 0 and 10 cm. The depletion appeared to be rapid in that it had already taken effect in the youngest stockpile (9 months) with no further age effects.
In previous studies, cyanobacteria had been cultivated from samples sourced at 18 cm depths in Japanese rice paddy soils (Fujita and Nakahara, 2006), 50 cm in the UK (Esmarch, 1914), and 70 cm depths in the USA (Moore and Karrer, 1919). Microcoleus and Leptolyngbya have survived and remained viable after up to 3 million years of being frozen in lake sediments in permafrost (Vishnivetskaya et al., 2003). Vegetative Nostoc commune material retains viability following several decades of storage in desiccated form (Bristol, 1919; Lipman, 1941). Reactivation of vegetative material after decades of storage was successful but several months (Lipman, 1941) to a year (Bristol, 1919) of incubation can be necessary for growth to take place. These results were reflected in the current study in which growth was not observed for a prolonged period of time although conditions were optimal. It is likely that the longer the period of inactivity, the longer the time taken for reactivation to occur (Billi and Potts, 2002; Williams et al., 2014) or less material is viable; therefore, it takes longer to rebuild colonies (Agrawal and Singh, 2002).
Taking into account the length of time it took for buried cyanobacteria to resurrect suggested they had regrown from vegetative material that had been inactive since the stockpiling process, which in turn resulted in long lag times for growth (Bristol, 1919; Lipman, 1941; Shaw et al., 2003). In addition, there may be potential for photo-damage to occur as many subsurface cyanobacteria would now be exposed in the topsoil removal and stockpiling process. This could also disrupt and slow down the recovery process once re-spread; this was observed in the laboratory following an out-of-season heat wave during which many subsurface species were trapped on the surface and died (Wendy Williams, unpublished data). In the context of rehabilitation, it is not practical to store stockpiles at very shallow depths due to the land area they would occupy. We have shown that under optimum laboratory conditions a diverse range of cyanobacteria recovered from a range of stockpile depths. It would be important to monitor the recovery of cyanobacteria from stockpiled soil to ascertain whether there is adequate inoculum to support the regrowth of biocrusts.
4.3 Limitations
As this was the first study of its kind, further investigation would be needed to determine what factors are influential in the decline of species richness following burial. We do not yet understand whether there may be a critical time frame for holding the topsoil or whether rainfall penetration into the stockpile results in the demise of cyanobacteria (possibly consumed by bacteria). On the other hand, in the longer term, provided conditions are favourable, there may be adequate cyanobacterial survival, at least down to 50 cm, to recolonise the soil surfaces. Additionally, dust containing cyanobacteria from nearby locations might also be blown across the rehabilitated site, thereby facilitating recovery (e.g. Burrows et al., 2009; McKenna Neuman et al., 1996). In this study the sampled buried cyanobacteria in the topsoil were provided optimum conditions of moisture, light, and warmth in the laboratory. Under field conditions, rainfall deficiencies, especially drought, could be a limiting factor following the spreading of the topsoil during rehabilitation. This leads to the need for additional studies on increased depth, younger stockpiles, and areas of regeneration under natural weather conditions to determine in what time frame the biocrust recolonises. The role of bacteria in biocrust re-establishment should also be investigated further as they clearly exist as a poorly understood component of these microbial communities (Chilton et al., 2017; Zhang et al., 2016).
The Eucla Basin, in which J–A is located, is situated on the eastern edge of the Nullarbor Plain. Naturally occurring biocrusts occupy at least 45 % of the landscape with a diverse community structure that contributes to landscape resilience and function. Cyanobacteria naturally occur within the first few millimetres of the surface of the biocrust where access to resources such as light and moisture are essential. Biocrusts contain cyanobacterial species such as Phormidium and Leptolyngbya that are regarded as early colonisers and other species such as Scytonema and Nostoc that contribute bioavailable nitrogen to the nutrient cycle.
In these studies, we found that at J–A cyanobacteria were a diverse community that had a proven capacity as ecosystem engineers. Many of these cyanobacteria were early colonisers and were represented by a high proportion of filamentous and N-fixing species. Biocrust functionality is aided by a diverse cyanobacterial population and its connective properties via EPS whereby nutrients and resources can be exchanged. The destruction of structural integrity and fragmentation presents hurdles; however, biocrust resilience, especially cyanobacteria, can consolidate over time and recolonise.
At J–A, rare earth mining processes require the removal of the topsoil; this soil is mechanically scraped off and relocated to topsoil stockpiles. These stockpiles act as a biobank in that they contain biocrust microorganisms and are valuable eco-resources. A major component of these biocrusts is cyanobacteria known for their capacity to survive extreme environments with the ability to remain in a desiccated state for long periods of time. Nevertheless, the stockpiling process destroys the macrostructure of the soil profile, thus fragmenting and translocating the biocrusts throughout the stockpiles. In the process the burial of cyanobacteria at depth in soil stockpiles showed that species richness diminished substantially below the top few centimetres across all ages. Limited access to light for photosynthesis while buried can cause cyanobacterial death. Here, our results support the early return of topsoil stockpiles to facilitate the re-establishment of biocrusts and soil microbial community function. A direct-return process planned at J–A would improve outcomes by reducing the associated impacts of topsoil storage on key microbial communities such as cyanobacteria. Further research should focus on the (1) establishment of an optimum time for topsoil storage and (2) the relationship of N-fixing cyanobacteria to seedling recruitment and plant establishment.
Currently, data can only be accessed in the form of Excel spreadsheets via the corresponding author.
The supplement related to this article is available online at: https://doi.org/10.5194/bg-16-2189-2019-supplement.
WW, SW, and CD contributed to microbial biobanking and background, biophysiochemical properties, cyanobacterial community structure, and paper compilation. AC and BN performed 16S rDNA profiling, and MS assessed cyanobacterial tolerance to stockpiling.
The authors declare that they have no conflict of interest.
This article is part of the special issue “Biological soil crusts and their role in biogeochemical processes and cycling”. It is a result of the BIOCRUST3 conference, Moab, USA, 26 to 30 September 2016.
This project was funded and supported by Iluka Resources Ltd. and the
Jacinth–Ambrosia rehabilitation team. Thanks to Sam Doudle,
Iluka Rehabilitation Specialist, for her recognition of the importance of
biocrusts at Jacinth–Ambrosia and for initiating this research programme.
This was a UniQuest project that was carried out at the University
of Queensland's School of Agriculture and Food Sciences, Gatton Campus, and
the University of New South Wales. Especially appreciated was the strong
support, project advice, and laboratory resources provided by Vic Galea and
Katherine Raymont. Special thanks to Glenn McGregor for technical advice,
assistance with cyanobacterial identification, and the use of specialist
microscopes and facilities at the Ecosciences Precinct (Qld), as well as
report editing. Special thanks to David Tongway, who assisted in landscape
function analysis fieldwork, and two anonymous reviewers who provided
valuable insights and editing to greatly improve this paper.
Edited by: Emilio Rodriguez-Caballero
Reviewed
by: Eli Zaady and one anonymous referee
Aboal, M., Werner, O., García-Fernández, M. E., Palazón, J. A., Cristóbal, J. C., and Williams, W.: Should ecomorphs be conserved? The case of Nostoc flagelliforme, an endangered extremophile cyanobacteria, J. Nat. Conserv., 30, 52–64, https://doi.org/10.1016/j.jnc.2016.01.001, 2016.
Agrawal, S. C. and Singh, V.: Viability of dried filaments, survivability and reproduction under water stress, and survivability following heat and UV exposure in Lyngbya martensiana, Oscillatoria agardhii, Nostoc calcicola, Hormidium fluitans, Spirogyra sp. and Vaucheria geminata, Folia Microbiol., 47, 61–67, https://doi.org/10.1007/BF02818567, 2002.
Anagnostidis, K. and Komarek, J.: Cyanoprokariota. Teil 2: Oscillatoriales, Gustav Fischer Verlag, Berlin, 2005.
Anderson, M., Gorley, R. N., and Clarke, R. K.: Permanova+ for primer: Guide to software and statisticl methods, Primer-E Limited, Plymouth, UK, 2008.
Barger, N. N., Weber, B., Garcia-Pichel, F., Zaady, E., and Belnap, J.: Patterns and Controls on Nitrogen Cycling of Biological Soil Crusts, in Biological Soil Crusts: An Organizing Principle in Drylands, 257–285, Springer, Cham, 2016.
Barnes, J. D., Balaguer, L., Manrique, E., Elvira, S., and Davison, A. W.: A reappraisal of the use of DMSO for the extraction and determination of chlorophylls a and b in lichens and higher plants, Environ. Exp. Bot., 32, 85–100, https://doi.org/10.1016/0098-8472(92)90034-Y, 1992.
Belnap, J. and Eldridge, D.: Disturbance and Recovery of Biological Soil Crusts, in Biological Soil Crusts: Structure, Function, and Management, 363–383, Springer, Berlin, Heidelberg, 2001.
Belnap, J. and Gillette, D. A.: Vulnerability of desert biological soil crusts to wind erosion: the influences of crust development, soil texture, and disturbance, J. Arid Environ., 39, 133–142, https://doi.org/10.1006/jare.1998.0388, 1998.
Bergman, B., Gallon, J. R., Rai, A. N., and Stal, L. J.: N2 Fixation by non-heterocystous cyanobacteria, FEMS Microbiol. Rev., 19, 139–185, https://doi.org/10.1111/j.1574-6976.1997.tb00296.x, 1997.
Biggs, B. J. F. and Kilroy, C.: Stream Periphyton Monitoring Manual, National Institute for Water and Atmospheric Research, Wellington, NZ, 2000.
Billi, D. and Potts, M.: Life and death of dried prokaryotes, Res. Microbiol., 153, 7–12, https://doi.org/10.1016/S0923-2508(01)01279-7, 2002.
Bowker, M. A.: Biological Soil Crust Rehabilitation in Theory and Practice: An Underexploited Opportunity, Restor. Ecol., 15, 13–23, https://doi.org/10.1111/j.1526-100X.2006.00185.x, 2007.
Bowker, M. A., Maestre, F. T., Eldridge, D., Belnap, J., Castillo-Monroy, A., Escolar, C., and Soliveres, S.: Biological soil crusts (biocrusts) as a model system in community, landscape and ecosystem ecology, Biodivers. Conserv., 23, 1619–1637, https://doi.org/10.1007/s10531-014-0658-x, 2014.
Bristol, B. M.: On the retention of vitality by algae from old stored soils, New Phytol., 18, 92–107, 1919.
Büdel, B., Darienko, T., Deutschewitz, K., Dojani, S., Friedl, T., Mohr, K. I., Salisch, M., Reisser, W., and Weber, B.: Southern African biological soil crusts are ubiquitous and highly diverse in drylands, being restricted by rainfall frequency, Microb. Ecol., 57, 229–247, https://doi.org/10.1007/s00248-008-9449-9, 2009.
Büdel, B., Williams, W. J., and Reichenberger, H.: Annual net primary productivity of a cyanobacteria-dominated biological soil crust in the Gulf Savannah, Queensland, Australia, Biogeosciences, 15, 491–505, https://doi.org/10.5194/bg-15-491-2018, 2018.
Burrows, S. M., Butler, T., Jöckel, P., Tost, H., Kerkweg, A., Pöschl, U., and Lawrence, M. G.: Bacteria in the global atmosphere – Part 2: Modeling of emissions and transport between different ecosystems, Atmos. Chem. Phys., 9, 9281–9297, https://doi.org/10.5194/acp-9-9281-2009, 2009.
Carrick, P. J. and Krüger, R.: Restoring degraded landscapes in lowland Namaqualand: Lessons from the mining experience and from regional ecological dynamics, J. Arid Environ., 70, 767–781, https://doi.org/10.1016/j.jaridenv.2006.08.006, 2007.
Chamizo, S., Cantón, Y., Miralles, I., and Domingo, F.: Biological soil crust development affects physicochemical characteristics of soil surface in semiarid ecosystems, Soil Biol. Biochem., 49, 96–105, https://doi.org/10.1016/j.soilbio.2012.02.017, 2012.
Chilton, A. M., Neilan, B. A., and Eldridge, D. J.: Biocrust morphology is linked to marked differences in microbial community composition, Plant Soil, 429, 65–75, https://doi.org/10.1007/s11104-017-3442-3, 2017.
Chiquoine, L. P., Abella, S. R., and Bowker, M. A.: Rapidly restoring biological soil crusts and ecosystem functions in a severely disturbed desert ecosystem, Ecol. Appl., 26, 1260–1272, https://doi.org/10.1002/15-0973, 2016.
Clarke, K. R. and Gorley, R. N.: PRIMER v6 PRIMER-E Ltd, Plymouth, UK, 2001.
Delgado-Baquerizo, M., Morillas, L., Maestre, F. T., and Gallardo, A.: Biocrusts control the nitrogen dynamics and microbial functional diversity of semi-arid soils in response to nutrient additions, Plant Soil, 372, 643–654, https://doi.org/10.1007/s11104-013-1779-9, 2013.
Doherty, K. D., Antoninka, A. J., Bowker, M. A., Ayuso, S. V., and Johnson, N. C.: A Novel Approach to Cultivate Biocrusts for Restoration and Experimentation, Ecol. Restor., 33, 13–16, 2015.
Doudle, S., Williams, W., and Galea, V.: Improving rehabilitation outcomes using biocrusts, 8th Int. Heavy Miner. Conf. 2011, 85–97, 2011.
Elbert, W., Weber, B., Burrows, S., Steinkamp, J., Büdel, B., Andreae, M. O., and Pöschl, U.: Contribution of cryptogamic covers to the global cycles of carbon and nitrogen, Nat. Geosci., 5, 459–462, https://doi.org/10.1038/ngeo1486, 2012.
Eldridge, D. J. and Leys, J. F.: Exploring some relationships between biological soil crusts, soil aggregation and wind erosion, J. Arid Environ., 53, 457–466, https://doi.org/10.1006/jare.2002.1068, 2003.
Esmarch, F.: Untersuchungen über die Verbreitung der Cyanophyceen auf und in Verschiedenen Böden, Hedwigia. B., 55, 224–273, 1914.
Fiore, M. F., Sant'Anna, C. L., Azevedo, M. T. de P., Komárek, J., Kaštovský, J., Sulek, J., and Lorenzi, A. A. S.: The Cyanobacterial Genus Brasilonema, Gen. Nov., a Molecular and Phenotypic Evaluation1, J. Phycol., 43, 789–798, https://doi.org/10.1111/j.1529-8817.2007.00376.x, 2007.
Fischer, T., Gypser, S., Subbotina, M., and Veste, M.: Synergic hydraulic and nutritional feedback mechanisms control surface patchiness of biological soil crusts on tertiary sands at a post-mining site, J. Hydrol. Hydromech., 62, 293–302, https://doi.org/10.2478/johh-2014-0038, 2014.
Fujita, Y. and Nakahara, H.: Variations in the microalgal structure in paddy soil in Osaka, Japan: comparison between surface and subsurface soils, Limnology, 7, 83–91, https://doi.org/10.1007/s10201-006-0167-z, 2006.
Garcia-Pichel, F. and Wojciechowski, M. F.: The evolution of a capacity to build supra-cellular ropes enabled filamentous cyanobacteria to colonize highly erodible substrates, PLoS One, 4, e7801, https://doi.org/10.1371/journal.pone.0007801, 2009.
Garcia-Pichel, F., Loza, V., Marusenko, Y., Mateo, P., and Potrafka, R. M.: Temperature drives the continental-scale distribution of key microbes in topsoil communities, Science, 340, 1574–1577, 2013.
Genty, B., Briantais, J.-M., and Baker, N. R.: The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence, Biochim. Biophys. Acta, 990, 87–92, https://doi.org/10.1016/S0304-4165(89)80016-9, 1989.
Gillieson, D., Wallbrink, P., and Cochrane, A.: Vegetation change, erosion risk and land management on the Nullarbor Plain, Australia, Environ. Geol., 28, 145–153, https://doi.org/10.1007/s002540050087, 1996.
Harris, J. A.: Measurements of the soil microbial community for estimating the success of restoration, Eur. J. Soil Sci., 54, 801–808, https://doi.org/10.1046/j.1351-0754.2003.0559.x, 2003.
Hou, B. and Warland, I.: Heavy mineral sands potential of the Eucla Basin in South Australia-a world-class palaeobeach placer province, Mesa J., 37, 4–12, 2005.
Hu, C., Liu, Y., Zhang, D., Huang, Z., and Paulsen, B. S.: Cementing mechanism of algal crusts from desert area, Chinese Sci. Bull., 47, 1361–1368, https://doi.org/10.1360/02tb9301, 2002.
Jasper, D. A.: Beneficial Soil Microorganisms of the Jarrah Forest and Their Recovery in Bauxite Mine Restoration in Southwestern Australia, Restor. Ecol., 15, S74–S84, https://doi.org/10.1111/j.1526-100X.2007.00295.x, 2007.
Kidron, G. J., Barinova, S., and Vonshak, A.: The effects of heavy winter rains and rare summer rains on biological soil crusts in the Negev Desert, Catena, 95, 6–11, https://doi.org/10.1016/j.catena.2012.02.021, 2012.
Komárek, J. and Anagnostidis, K.: Cyanoprokaryota, 2. Teil Oscillatoriales, in: Süßwasserflora von Mitteleuropa 19/2, edited by: Büdel, B., Krientz, L., Gärtner, G., and Schagerl, M., Gustav Fischer Verlag, Jena, Germany, 2005.
Lipman, C. B.: The successful revival of Nostoc commune from a herbarium specimen eighty-seven years old, B. Torrey Bot. Club, 68, 664–666, 1941.
Lukešová, A.: Soil Algae in Brown Coal and Lignite Post-Mining Areas in Central Europe (Czech Republic and Germany), Restor. Ecol., 9, 341–350, https://doi.org/10.1046/j.1526-100X.2001.94002.x, 2001.
Maestre, F. T., Castillo-Monroy, A. P., Bowker, M. A., and Ochoa-Hueso, R.: Species richness effects on ecosystem multifunctionality depend on evenness, composition and spatial pattern, J. Ecol., 100, 317–330, https://doi.org/10.1111/j.1365-2745.2011.01918.x, 2012.
Mager, D. M. and Thomas, A. D.: Extracellular polysaccharides from cyanobacterial soil crusts: A review of their role in dryland soil processes, J. Arid Environ., 75, 91–97, https://doi.org/10.1016/j.jaridenv.2010.10.001, 2011.
Mazor, G., Kidron, G. J., Vonshak, A., and Abeliovich, A.: The role of cyanobacterial exopolysaccharides in structuring desert microbial crusts, FEMS Microbiol. Ecol., 21, 121–130, https://doi.org/10.1016/0168-6496(96)00050-5, 1996.
McCutcheon, J., Wilson, S. A., and Southam, G.: Microbially accelerated carbonate mineral precipitation as a strategy for in situ carbon sequestration and rehabilitation of asbestos mine sites, Environ. Sci. Technol., 50, 1419–1427, 2016.
McDonald, D., Price, M. N., Goodrich, J., Nawrocki, E. P., DeSantis, T. Z., Probst, A., Andersen, G. L., Knight, R., and Hugenholtz, P.: An improved Greengenes taxonomy with explicit ranks for ecological and evolutionary analyses of bacteria and archaea, ISME J., 6, 610–618, https://doi.org/10.1038/ismej.2011.139, 2012.
McKenna Neuman, C., Maxwell, C. D., and Boulton, J. W.: Wind transport of sand surfaces crusted with photoautotrophic microorganisms, CATENA, 27, 229–247, https://doi.org/10.1016/0341-8162(96)00023-9, 1996.
Moore, G. T. and Karrer, J. L.: A subterranean algal flora, Ann. Mo. Bot. Gard., 6, 281–307, 1919.
Rao, B., Liu, Y., Lan, S., Wu, P., Wang, W., and Li, D.: Effects of sand burial stress on the early developments of cyanobacterial crusts in the field, Eur. J. Soil Biol., 48, 48–55, https://doi.org/10.1016/j.ejsobi.2011.07.009, 2012.
Rossi, F., Mugnai, G., and Philippis, R. D.: Complex role of the polymeric matrix in biological soil crusts, Plant Soil, 429, 19–34, https://doi.org/10.1007/s11104-017-3441-4, 2017.
Sant'Anna, C. L., Azevedo, M. T. P., Fiore, M. F., Lorenzi, A. S., Kaštovský, J., and Komárek, J.: Subgeneric diversity of Brasilonema (Cyanobacteria, Scytonemataceae), Braz. J. Bot., 34, 51–62, 2011.
Schloss, P. D., Westcott, S. L., Ryabin, T., Hall, J. R., Hartmann, M., Hollister, E. B., Lesniewski, R. A., Oakley, B. B., Parks, D. H., and Robinson, C. J.: Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities, Appl. Environ. Microbiol., 75, 7537–7541, 2009.
Setyawan, D., Napoleon, A., and Hanum, H.: Measuring Soil Recovery after Coal Minesite Rehabilitation in South Sumatra, SICEST Proc., 293–295, ISSN 979-587-621-1, 2016.
Shaw, S. L., Chisholm, S. W., and Prinn, R. G.: Isoprene production by Prochlorococcus, a marine cyanobacterium, and other phytoplankton, Mar. Chem., 80, 227–245, 2003.
Skinner, S. and Entwisle, T.: Non-marine algae of Australia: 1. Survey of colonial gelatinous blue-green macroalgae (Cyanobacteria), Telopea, 9, 573–599, https://doi.org/10.7751/telopea20024003, 2002.
Stal, L. J.: Physiological ecology of cyanobacteria in microbial mats and other communities, New Phytol., 131, 1–32, https://doi.org/10.1111/j.1469-8137.1995.tb03051.x, 1995.
Stal, L. J.: Microphytobenthos, their Extracellular Polymeric Substances, and the Morphogenesis of Intertidal Sediments, Geomicrobiol. J., 20, 463–478, https://doi.org/10.1080/713851126, 2003.
Tongway, D. and Hindley, N.: Landscape function analysis: a system for monitoring rangeland function, Afr. J. Range For, Sci., 21, 109–113, https://doi.org/10.2989/10220110409485841, 2004.
Tongway, D. J.: Soil and landscape processes in the restoration of rangelands, Rangel. J., 12, 54–57, https://doi.org/10.1071/rj9900054, 1990.
Tongway, D. J. and Ludwig, J. A.: Rehabilitation of Semiarid Landscapes in Australia. I. Restoring Productive Soil Patches, Restor. Ecol., 4, 388–397, https://doi.org/10.1111/j.1526-100X.1996.tb00191.x, 1996.
Ullmann, I. and Büdel, B.: Ecological determinants of species composition of biological soil crusts on a landscape scale, Biol. Soil Crusts Struct. Funct. Manag., Springer, Berlin, Heidelberg, 203–213, 2001.
Vaccarino, M. A. and Johansen, J. R.: Brasilonema Angustatum Sp. Nov. (nostocales), a New Filamentous Cyanobacterial Species from the Hawaiian Islands1, J. Phycol., 48, 1178–1186, https://doi.org/10.1111/j.1529-8817.2012.01203.x, 2012.
Vishnivetskaya, T. A., Spirina, E. V., Shatilovich, A. V., Erokhina, L. G., Vorobyova, E. A., and Gilichinsky, D. A.: The resistance of viable permafrost algae to simulated environmental stresses: implications for astrobiology, Int. J. Astrobiol., 2, 171–177, https://doi.org/10.1017/S1473550403001575, 2003.
Weber, B., Bowker, M., Zhang, Y., and Belnap, J.: Natural Recovery of Biological Soil Crusts After Disturbance, in Biological Soil Crusts: An Organizing Principle in Drylands, 479–498, Springer, Cham, 2016.
Wellburn, A. R.: The Spectral Determination of Chlorophylls a and b, as well as Total Carotenoids, Using Various Solvents with Spectrophotometers of Different Resolution, J. Plant Physiol., 144, 307–313, https://doi.org/10.1016/S0176-1617(11)81192-2, 1994.
Williams, W., Büdel, B., and Williams, S.: Wet season cyanobacterial N enrichment highly correlated with species richness and Nostoc in the northern Australian savannah, Biogeosciences, 15, 2149–2159, https://doi.org/10.5194/bg-15-2149-2018, 2018.
Williams, W. J. and Büdel, B.: Species diversity, biomass and long-term patterns of biological soil crusts with special focus on Cyanobacteria of the Acacia aneura Mulga Lands of Queensland, Australia, Algol. Stud., 140, 23–50, 2012.
Williams, W. J. and Eldridge, D. J.: Deposition of sand over a cyanobacterial soil crust increases nitrogen bioavailability in a semi-arid woodland, Appl. Soil Ecol., 49, 26–31, https://doi.org/10.1016/j.apsoil.2011.07.005, 2011.
Williams, W. J., Büdel, B., Reichenberger, H., and Rose, N.: Cyanobacteria in the Australian northern savannah detect the difference between intermittent dry season and wet season rain, Biodivers. Conserv., 23, 1827–1844, https://doi.org/10.1007/s10531-014-0713-7, 2014.
Zhang, B., Kong, W., Wu, N., and Zhang, Y.: Bacterial diversity and community along the succession of biological soil crusts in the Gurbantunggut Desert, Northern China, J. Basic Microbiol., 56, 670–679, 2016.
Zhao, Y., Zhu, Q., Li, P., Zhao, L., Wang, L., Zheng, X., and Ma, H.: Effects of artificially cultivated biological soil crusts on soil nutrients and biological activities in the Loess Plateau, J. Arid Land, 6, 742–752, https://doi.org/10.1007/s40333-014-0032-6, 2014.