the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Role of phosphorus concentration and the nitrogen to phosphate ratio in the synergistic stimulation of alkaline phosphatase activity in Laizhou Bay, China, coastal waters
Yanqun Yang
Xiaomeng Duan
Shengkang Liang
Mingzheng Zhang
Shanshan Li
Hongguan Li
Guoling Zhang
Haoyang Ma
Xiurong Han
Xiulin Wang
In coastal ecosystems, microbial alkaline phosphatase (AP) production is primarily induced by low phosphate (PO4-P) availability but is additionally regulated by the dissolved inorganic nitrogen to phosphate (DIN : PO4-P) ratio and seasonal temperature variation. However, the dominant driver of alkaline phosphatase activity (APA) surges and potential synergistic effects among these factors remain unclear. Through integrated seasonal field surveys and enclosure experiments in Laizhou Bay, China, we demonstrate that PO4-P seawater concentration serves as the primary control for APA induction, with a consistent threshold of 0.05 µmol L−1. Significant positive correlation was observed between APA and the DIN : PO4-P ratio below this threshold (0.05 µmol L−1), as analyzed in the combined dataset of field and enclosure experiments (p<0.01, n=36). Notably, phytoplankton-dominated APA was evidenced in autumn. Genetic analysis confirms that AP-related gene expression increases only when PO4-P falls below the identified threshold. These findings refine the conceptual framework for AP regulation in coastal ecosystems, highlighting the hierarchical control of phosphorus limitation over stoichiometric effects.
- Article
(10906 KB) - Full-text XML
-
Supplement
(2515 KB) - BibTeX
- EndNote
With the intensification of nitrogen (N) inputs and the control of phosphorus (P), P limitation has become widespread in global coastal waters (Zhang et al., 2024; Maavara et al., 2020; Liang et al., 2023) and has triggered adverse ecological consequences, including exacerbated eutrophication and shifts in phytoplankton community structure (Xin et al., 2019; Peñuelas and Sardans, 2022). To cope with P limitation, microorganisms have evolved a range of strategies, including an increase in inorganic phosphate transporters, induction of hydrolases for scavenging organophosphates (OP, organic phosphorus compounds), and a reduction in P demand by replacing phospholipids with sulfur- or nitrogen-containing lipids (Van et al., 2006; Karl, 2014; Lin et al., 2016). Among these strategies, the extracellular enzymatic hydrolysis of dissolved organic phosphorus (DOP) to bioavailable inorganic P represents a critical pathway for microbial communities to overcome P limitation in coastal waters (Zhang et al., 2017; Jin et al., 2024). Understanding OP utilization mechanisms is essential to allow prediction and management ecological responses to increasing P stress in coastal ecosystems.
Alkaline phosphatase (AP) is regarded as the most common extracellular enzyme for the utilization of dissolved organophosphates (DOP) (Yamaguchi et al., 2016; Zhang et al., 2018; Jin et al., 2024). In general, alkaline phosphatase activity (APA) is induced under P-restricted conditions and inhibited at high phosphate (PO4-P) concentrations (Dyhrman and Ruttenberg, 2006; Zhang et al., 2018). Nausch (1998) found that APA increased significantly when PO4-P concentrations were <0.2 µmol L−1. Since then, the PO4-P threshold for the APA surge in coastal waters has been reported to range from 0.01 to 0.5 µmol L−1 in various coastal systems (Jin et al., 2024). Temperature, microbial species dominance, trace metal concentrations, macronutrient levels and their ratio may affect APA levels and the PO4-P threshold (Dyhrman and Ruttenberg, 2006; Mahaffey et al., 2014; Browning et al., 2017; Zhang et al., 2018; Ivancic et al., 2021; Jin et al., 2024). Temperature has also been shown to greatly influence APA via both direct effects on enzyme kinetics and indirect effects on the composition of the microbial community (Toseland et al., 2013; Thomas et al., 2017). A high DIN : PO4-P ratio (indicating N excess and P limitation) has been confirmed to be positively correlated with APA (Bogé et al., 2017; Jin et al., 2024). Freshwater inputs, which often carry nutrients with intrinsically high N:P ratios, can thereby enhance APA (Kang et al., 2019; Ivancic et al., 2021). Trace metals (Zn, Ca, etc.) affect APA because they are core enzymatic structural components, but trace metals are unlikely to limit APA in offshore waters (Jin et al., 2024). The complexity of the variables influencing APA likely contributes to the lack of consensus on the conditions for its induction in coastal waters, and the interactive effects of these factors are poorly understood. Moreover, there is currently controversial research evidence as to whether there is a PO4-P threshold concentration for APA surges to occur. Qin et al. (2021) found that APA values were very high in PO4-rich environments during the pre-algal bloom of the dinoflagellate Prorocentrum obtusidens Schiller, and APA showed no detectable correlation with PO4-P or DOP concentrations. These conclusions indicated that the stimulatory effect of P-stress on APA in coastal waters can be variable.
At the molecular level, microbial adaptation to P stress involves comprehensive physiological restructuring, including regulation of PO4-P acquisition systems, extracellular enzyme synthesis, protein turnover, lipid metabolism, and adjustments to photosynthetic and respiratory pathways (Feng et al., 2015). The genetic basis of the diversity in AP responses is particularly relevant, as there are three distinct AP types (PhoAEC, PhoX, and PhoD) showing limited sequence homology yet performing similar ecological functions (Luo et al., 2009; Lin et al., 2015). Molecular adaptations provide critical insight into microbial strategies to cope with P limitation.
This study aimed to elucidate the drivers of APA surges in Laizhou Bay by identifying the phosphate (PO4-P) concentration threshold for APA induction, evaluating the synergistic role of the DIN : PO4-P ratio under P-limited conditions, assessing seasonal and community-level APA partitioning, and validating these dynamics with genetic evidence. The combined field and experimental approach seeks to refine the conceptual framework for AP regulation in coastal ecosystems affected by anthropogenic nutrient imbalances.
2.1 Study area
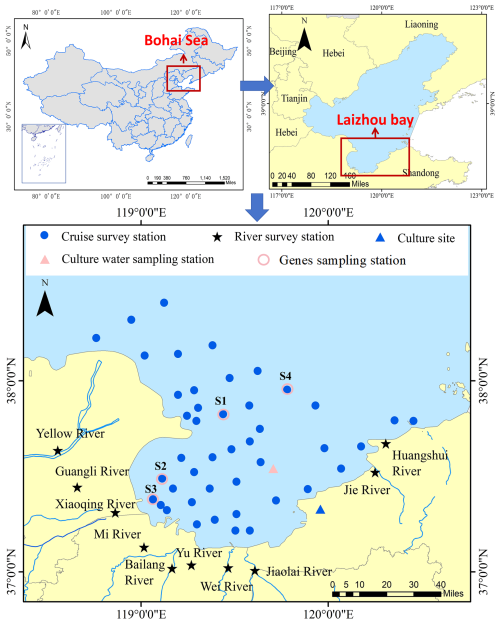
Laizhou Bay (LZB), a typical semi-enclosed bay, is situated south of the Bohai Sea and north of the Shandong Peninsula (Fig. 1). It covers an area of approximately 7000 km2 with a coastline of 320 km, a mean depth of less than 10 m, and a water half-exchange time (i.e., the time for 50 % water renewal) of 55 d (Wu et al., 2023). Water quality in LZB has deteriorated due to high land-based riverine inputs, including those from the Yellow River (YR) and Xiaoqing River (XQR), as well as discharges from mariculture. Since 1980, the dissolved inorganic nitrogen (DIN) concentration has increased from 3 to 25 µmol L−1, and the ratio of DIN : PO4-P ratio increased from 5:1 to 110:1 (Xin et al., 2019). The resulting increase in N availability, coupled with intensifying P limitation, has triggered significant ecological shifts in the bay, including more frequent harmful algal blooms and changes in dominant phytoplankton species from diatoms to dinoflagellates (Song et al., 2017; Xin et al., 2019).
2.2 Land and sea synchronous surveys
To investigate the distribution of APA in LZB, four land-sea synchronous surveys were conducted on 10–12 May 2019 (spring), 18–21 August 2019 (summer), 2–14 November 2021 (autumn), and 22–27 March 2021 (winter). The land-sea synchronous survey stations covered the entire LZB and the adjacent waters of the Yellow River Estuary (YRE) (Fig. 1). Additional riverine monitoring stations were located at the mouths of ten rivers, including YR and XQR (Fig. 1). The monitored pollutant load accounted for >95 % of the total discharge load in the LZB's basin (Li et al., 2022). Surface seawater samples were collected using a 5 L Niskin bottle. For subsequent analysis of nutrients, chlorophyll a (Chl a), bacterial abundance, phytoplankton, and alkaline phosphatase activity, water was processed immediately upon collection: aliquots for nutrient analysis were filtered through acetate membranes (0.45 µmol L−1); aliquots for enzymatic activity assays were processed as either unfiltered or size-fractionated as described in Sect. 2.4.3; and samples for molecular analysis were filtered onto 0.22 µmol L−1 polycarbonate membranes. For riverine water samples collected at the mouths of ten major rivers, sampling was conducted following standard protocols for surface water monitoring in China (specifically, HJ 494-2009). An organic glass water sampler or a pre-cleaned polyethylene container was used to collect subsurface water at a depth of approximately 0.5 m. All samples were preserved according to the specific protocols detailed in Sect. 2.4.1–2.4.4 and transported on ice to the laboratory within 12 h. The August 2019 survey occurred after the landfall of the strong typhoon “Lekima” in Shandong. From late August to October 2021, the middle and lower reaches of the YR experienced a historically rare autumn flood, and the November 2021 survey took place after this flood. Samples were collected in the river and sea areas in near-synchrony.
2.3 Field enclosure experiments
An enclosure experiment was conducted from 23 July to 21 August 2021 to explore the effects of phosphate concentration and DIN : PO4-P ratio on APA secretion. Two enclosed ecosystems were designed. Water samples were collected at a representative station (119.71° E, 37.54° N) at the mouth of LZB on 23 July 2021 (Fig. 1). Seawater samples were siphoned into containment bags on the shore. Incubation experiments were carried out in the nearshore sea area (120.00° E, 37.36° N) for 29 d (Fig. 1). The initial seawater nutrient environment with a DIN : PO4-P ratio of 55 (Table S1 in the Supplement) was maintained in the control group (CG). In the treatment group (TG) the DIN : PO4-P seawater ratio was adjusted to 28 by adding NaNO3 and KH2PO4 (Tables S1 and S2). This choice (TG, DIN : PO4-P = 28) was guided by two key considerations: (1) to establish a significant contrast (approximately halved) to the ambient ratio of 55, and (2) to approach the classical Redfield ratio of 16:1, thereby testing microbial responses under a scenario of mitigated stoichiometric imbalance. Furthermore, the higher absolute nutrient concentrations in the TG (DIN : 50.01 µmol L−1, PO4-P: 1.78 µmol L−1) compared to the control (CG) (DIN: 1.90 µmol L−1, PO4-P: 0.04 µmol L−1) were necessary to ensure that nutrients remained non-limiting throughout the incubation, allowing the target N:P ratio to be the primary variable influencing the system over time. In parallel, culture water samples were collected 17 times at fixed intervals. Samples were collected 17 times over the 29 d incubation to cover the full phytoplankton growth and senescence cycle. During the exponential growth phase, sampling was conducted daily. After nutrient depletion and population decline, the interval was extended to monitor the stabilized system. The detailed sampling days and weather conditions are presented in Table S2.
2.4 Sample analysis
2.4.1 Nutrients
Nutrient samples were filtered through acetate membranes (0.45 µm pore size) and stored at −20 °C until analysis. After thawing, nutrients were measured using an automatic nutrient analyzer (SEAL-AA3, Germany). The analyzed parameters included: DIN (sum of ammonium, NH4-N [detection limit 0.02 µM, relative standard deviation (RSD) <2 %], nitrate, NO3-N [0.02 µM, RSD <2 %], and nitrite, NO2-N [0.01 µM, RSD <3 %]), PO4-P [0.01 µM, RSD <2 %], dissolved silica, DSi [0.05 µM, RSD <3 %]), total dissolved nitrogen, TDN [0.05 µM, RSD <3 %]), total dissolved phosphorus (TDP [0.02 µM, RSD <3 %]), and particulate nutrients (particulate nitrogen, PN [0.1 µM, RSD <5 %]; particulate phosphorus, PP [0.05 µM, RSD <5 %]) (Murphy and Riley, 1962). TDN, TDP, and particulate nutrient concentrations were measured after the filtrates and filter membranes were oxidized by persulfate at 121 °C for 30 min (Armstrong et al., 1966). Dissolved organic N and P (DON [≈0.1 µM, RSD 5 %–10 %]; DOP [≈0.05 µM, RSD 5 %–10 %]) were calculated as the difference between TDN/TDP and . respectively.
2.4.2 Chlorophyll a, bacterial abundance, and phytoplankton community structure
Chl a was extracted using 90 % acetone for 24 h in the dark at 4 °C and centrifuged for 10 min at 4000 rpm; Chl a concentration was measured using a fluorometer (Turner Designs-Trilogy, USA) with a detection limit of 0.025 µg L−1, following methods of Parsons et al. (1984) within one month after sampling, with filters stored at −20 °C prior to analysis. Samples for determination of bacterial abundance were stained with SYBR Green I fluorescent dye for 20 min and measured using a BD FACSCalibur flow cytometer (BD, USA) (Marie et al., 1997). Phytoplankton community structure was determined using an inverted microscope at 400× magnification.
2.4.3 Alkaline phosphatase activity and kinetics
The fluorometric method using 4-methylumbelliferyl phosphate (MUF-P) as the substrate was employed on unfiltered (APAtotal) and two pre-filtered (through 3 or 0.22 µm filters, Millipore) seawater samples. This fractionation approach allows distinguishing phosphatase activity from different microbial compartments: APAphy was represented by the difference between unfiltered water samples and 3 µm-filtered water samples (mainly microalgal-associated), APAbac by the difference between 3- and 0.22 µm-filtered water samples (bacterial-associated), and APAfree by the activity in the 0.22 µm filtrate (extracellular enzymes), thus providing complementary information on P acquisition strategies across microbial size classes (Hoppe, 1983; Labry et al., 2005). Samples were then processed and cryopreserved at −20 °C until analysis. The fluorescence intensity of the samples was measured using a fluorescence photometer (F4700, Japan; excitation wavelength, Ex = 365 nm, emission wavelength, Em = 445 nm) immediately after thawing. Enzyme kinetic parameters Vmax and Km were calculated using non-linear least squares regression fitted to the Michaelis–Menten equation (Brooks, 1992). The relationship between APA and substrate concentration followed the Michaelis–Menten equation:
where V represents the rate of enzymatic substrate hydrolysis (in nmol L−1 h−1), and S the substrate concentration (in nmol L−1). Vmax represents the maximum hydrolysis rate and Km represents the half- saturation constant, i.e., the substrate concentration at , (in nmol L−1).
2.4.4 Functional genomics
DNA was extracted from the samples to investigate the microbial P metabolism pathway. The E.Z.N.A.® Soil DNA Kit (Omega Bio-tek, U.S.) was used for DNA extraction. The concentration, purity, and integrity of DNA were assessed using TBS-380, Nanodrop2000, and 1 % agarose gel electrophoresis, respectively (Mäki et al., 2017). DNA fragments were segmented using a Covaris M220 sonicator (Gene Company, China); fragments of approximately 400 bp were screened and paired-end libraries were constructed using the NEXTFLEX Rapid DNASeq (Bioo Scientific, U.S.) library preparation kit. Metagenomic sequencing was performed on the Illumina NovaSeq (Illumina, U.S.) sequencing platform following bridge PCR amplification. Amino acid sequences were compared with the Kyoto Encyclopedia of Genes and Genomes (KEGG) gene database (http://www.genome.jp/kegg/, last access: 26 February 2026) using BLASTP to obtain corresponding KEGG function information for the genes. The sum of gene abundances corresponding to Kegg Orthology (KO), Pathway, Enzyme Commission (EC) number, and Module, was used to calculate the abundances of corresponding functional categories.
2.5 Statistical analysis
Using Origin software (version 2024), the non-parametric Spearman correlation test (two-sided) was conducted to create a heat map of the correlation between APA and environmental parameters. The PO4-P threshold for APA induction was subsequently determined using a segmented regression approach on the relevant dataset. Differences between experimental groups (e.g., CG vs. TG) were assessed for significance using t test and Mann-Whitney U test. Linear regression analysis was applied to determine whether the data were linearly correlated. The spatial distribution maps of temperature, salinity, nutrients, APA, and Chl a were generated using ordinary kriging interpolation in Surfer (version 16, Golden Software LLC), which accounts for spatial autocorrelation among sampling points to produce unbiased continuous surfaces.
3.1 LZB terrigenous nutrient inputs
Seasonal investigation of rivers emptying into LZB revealed that the average annual DIN : PO4-P ratio of riverine loads into LZB was 39:1, with a maximum of 125:1 (summer in the Jiaolai River) and a minimum of 2:1 (spring in the Bailang River) (Fig. 2). The average annual total nitrogen to total phosphorus ratio (TN : TP) was 90:1, with a maximum of 263:1 (autumn in the Bailang River) and a minimum of 16:1 (spring in the Guangli River) (Fig. 2). The DIN : PO4-P ratio of riverine loads was maximal (70:1) in winter (Fig. 2). For the P components of riverine loads, PP accounted for the highest percent contribution, with an average of 53±7 % (mean ± SD, standard deviation), while DOP and PO4-P accounted for 27±17 % and 21±16 %, respectively (Fig. 2). Regarding nitrogen components, DON accounted for the highest percent contribution, with an average of 52±17 %, and DIN and PN accounted for 39±19 % and 9±9 %, respectively (Fig. 2).
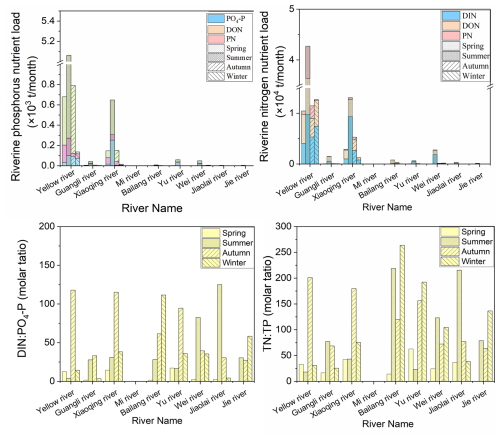
Figure 2Seasonal phosphorus (P) and nitrogen (N) load and nutrient ratios of various rivers discharging into Laizhou Bay (LZB). The spring, summer, autumn, and winter surveys were conducted in May 2019, August 2019, November 2021, and March 2021, respectively. DIN and DON: dissolved inorganic and organic nitrogen, respectively; PN and TN: particulate and total nitrogen, respectively.
3.2 Bulk parameters in LZB
3.2.1 Seawater temperature and salinity
The seawater surface layer temperature of LZB exhibited significant seasonal variation (P<0.05), with mean values of 26.2±1.2 °C in summer, 16.8±2.3 °C in spring, 12.9±3.6 °C in autumn, and 6.7±1.8 °C in winter (Fig. S1 in the Supplement). The salinity of LZB showed limited seasonal variation, although it was higher in spring (27.4±4.9) and winter (27.6±4.5) than in summer (26.1±7.1) and autumn (22.7±3.6) (Fig. S1).
3.2.2 Nutrients and chlorophyll a
During our seasonal field investigation, the average PO4-P concentration in LZB was 0.13±0.01, 0.32±0.03, 0.06±0.14, 0.08±0.1 µmol L−1 in spring, summer, autumn and winter, respectively (Fig. 1). PO4-P in LZB gradually decreased with increasing distance from the shore (Fig. 1). Average TP concentrations were 1.35±0.26, 0.97±1.42, 0.64±0.42, and 0.49±0.34 µmol L−1 in spring, summer, autumn, and winter, respectively. The percentage contributions of PO4-P, DOP, and PP in TP were 7 %, 70 %, and 24 % in spring; 33 %, 16 %, and 51 % in summer; 11 %, 38 %, and 51 % in autumn; and 24 %, 14 %, and 62 % in winter, respectively (Fig. S2). The average DIN : PO4-P ratios of the seawater surface layer across the four seasons were 711.5±423.1, 452.9±500.2, 1083.6±1043.7, and 363.7±308.5, respectively, all far exceeding the Redfield ratio of 16:1 (Fig. 3).
The average Chl a concentration in spring, summer, autumn, and winter was 1.20±0.98, 6.82±6.14, 5.56±6.28, and 6.30±5.99 µg L−1, respectively. The ranges were 0.4 to 4.3, 0.95 to 22.10, 0.18 to 27.49, and 0.37 to 24.94 µg L−1, respectively. Areas with relatively high Chl a values was mainly distributed in the adjacent sea area of the Xiaoqing River and Yellow River estuaries (Fig. 3).
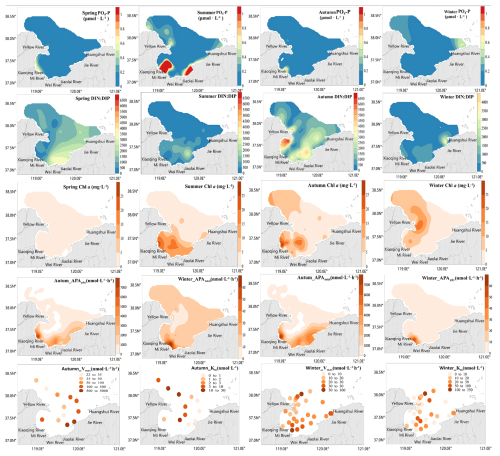
Figure 3Planar distribution of dissolved inorganic phosphorus (PO4-P) (µmol L−1) and chlorophyll a (Chl a) concentrations (in µg L−1), total and phytoplankton-derived alkaline phosphatase activity (APAtotal and APAphy, respectively), (nmol L−1 h−1), and AP enzyme kinetic parameters(Vmax, Km) in Laizhou Bay in May (spring) and August (Summer) 2019, and November (autumn) and March (winter) 2021.
3.3 Alkaline phosphatase activity and kinetics in the LZB
The distribution of APA in LZB showed a gradual decrease from the coastline to offshore waters, with the highest values observed in nearshore coastal waters (Fig. 3). The mean value of APAtotal reached 795.8±1749.8 in autumn and was thus 50 times higher than in winter (Fig. 3). Mean values of APAphy, APAbac, and APAfree in autumn were 715.1±1592.6, 55.1±286.2, and 25.7±61.1 , respectively, and thus 78, 12, and 17 times higher, respectively, than in winter (Figs. 3, S3). Accordingly, in autumn APAphy contributed most to APAtotal while APAfree contributed the least (Fig. S4), whereas in winter APAfree and APAbac made the highest and lowest contribution, respectively (Fig. S4). The spatial distributions of high Chl a concentrations and high APAtotal values showed consistency, especially in the adjacent areas of the XQR and YR estuaries (Fig. 3).
The mean value of the AP kinetic parameter Km was nmol L−1 in autumn and was thus 3.6 times lower than in winter ( nmol L−1). In contrast, the AP kinetic parameter Vmax averaged 754±1474.7 in autumn, approximately an order of magnitude higher than in winter (26.1±17.1 ) (Fig. 4).
In autumn, correlation analysis of field data revealed that APAphy exhibited a significant positive correlation with the ratio (p<0.05) (Fig. 4). However, there was no detectable correlation between APA and PO4-P (Fig. 4). In winter, APAtotal, APAbac, and APAfree were negatively correlated with PO4-P and Chl a, but not with the ratio (Fig. 4). And APAtotal was positively correlated with APAphy, APAbac, and APAfree. Additionally, APAfree was positively correlated with APAphy (p<0.05) and APAbac (p<0.05). APAtotal, APAbac, and APAfree were also positively correlated with the DON TDN ratio (p<0.05) (Fig. 4).

Figure 4Heatmap of Spearman correlations between measured variables in autumn (left) and winter (right) in Laizhou Bay. Abbreviations denote: T (temperature), S (salinity), Chl a (chlorophyll a), PO4-P (phosphate), DIN (dissolved inorganic nitrogen), DON (dissolved organic nitrogen), DOP (dissolved organic phosphorus), TDN (total dissolved nitrogen), PP (particulate phosphorus), and PN (particulate nitrogen). Vmax and Km represent the kinetic parameters of alkaline phosphatase. APAtotal, APAphy, APAbac, and APAfree denote total, phytoplankton-derived, bacteria-derived, and free alkaline phosphatase activity, respectively.
3.4 Variation in APA, nutrients, and microorganisms during enclosure experiments
3.4.1 Nutrients
At the beginning of the field enclosure experiment (days 0–3, no rainfall), the DIN and PO4-P in the TG (treatment group) were absorbed rapidly and in large quantities, and the DIN : PO4-P ratio increased from the initial adjusted value (28:1) (Fig. 5a–c). In contrast, the CG (control group; initial DIN : PO4-P = 55) showed a decrease in the DIN : PO4-P ratio from 0 to day 3 (Fig. 5c). Meanwhile, DON and DOP concentrations in the TG increased rapidly from 0 to day 10 (Fig. 5d, e). From day 10 to 29, DON concentrations remained high while those of DOP slowly declined. However, in the CG, DON accumulated gradually throughout the experiment starting from day 0, but DOP concentrations remained relatively constant (Fig. 5d, e). Initial DON and DOP concentrations in both the CG and the TG were roughly comparable, but average DON and DOP concentrations over the experiment were 1.6 and 3.6 times higher in the TG than in the CG, respectively (Fig. 5d, e).
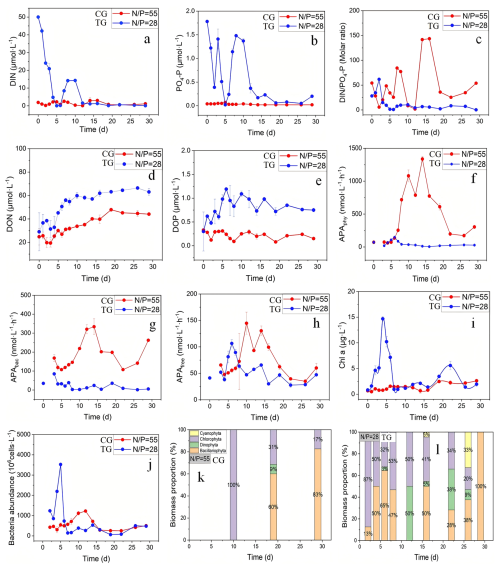
Figure 5Dissolved nutrient concentrations, alkaline phosphatase activity (APA), chlorophyll a (Chl a) concentration, bacterial numerical abundance, and changes in percent biomass of major phytoplankton taxonomic groups over selected intervals during the 29 d enclosure experiments. indicates the time-averaged nutrient ratio in the control group (CG, red line) and the treatment group (TG, blue line).
3.4.2 Variation in phytoplankton and bacterioplankton
Chl a concentrations and bacterial abundance in the TG were significantly higher than those in the CG throughout most of the experiment. Thus, the average Chl a and bacterial abundance in the TG were 2.9 and 1.4 times greater than those in the CG, respectively (Fig. 5i). In the TG, Chl a peaked during the early culture stage (0–6 d), reaching levels indicative of a red tide outbreak (Chl a ∼15 µg L−1) (Fig. 5i). Based on the analysis of phytoplankton species composition, the Chl a peak in the TG mainly comprised diatoms (Bacillariophyta) and chlorophytes, and the percentage of the former also peaked during the 0–6 d-period (Fig. 5l). Coincidentally, bacterial abundance also attained maximum levels during this period (Fig. 5j). In the CG, Chl a only increased slightly (by 25 %) during mid- and late culture stages, when the phytoplankton was mainly composed of diatoms and chlorophytes (Fig. 5k). Bacterial biomass increased gradually during the 5 to 13 d period to attain a maximum that occurred later than that in the TG (Fig. 5j).
3.4.3 Alkaline phosphatase activity
Average values of APAphy, APAbac, APAfree, and APAtotal in the CG were 47.5, 22.6, 54.8, and 687.6 , respectively. Average values of APAphy, APAbac, APAfree, and APAtotal in the TG were 5.1, 2.9, 42.2, and 122.8 , respectively (Figs. 5f–h, S5). In the CG, peak values of APAphy, APAbac, APAfree, and APAtotal were 1335.8, 333.9, 144.6, and 1800.0 , respectively, and the peaks appeared between days 10 and 16. In the TG, the respective peak values of APAphy, APAbac, APAfree, and APAtotal were 147.38, 85.00, 106.50, and 274.75 , occurring between days 3 and 6 (Figs. 5f–h, S5). The APA maximum in the CG was mainly contributed by APAphy (77 %), while that in the TG was mainly contributed by APAphy (50 %) and APAfree (37 %).
3.5 Regulation of APA by both PO4-P concentration and the DIN : PO4-P ratio
Both field and experimental data in LZB clearly showed that APAphy, APAbac, and APAfree were synthesized and secreted when PO4-P <0.05 µmol L−1, and their concentrations and activity increased rapidly as PO4-P decreased (Fig. 6). The PO4-P threshold was consistent between the field study and enclosure experiment. Moreover, when PO4-P was lower than its concentration threshold (0.05 µmol L−1), the enzyme kinetic parameters Km and Vmax also increased sharply (Fig. S6). There was a significant linear positive correlation (P<0.01) between APAtotal and the DIN : PO4-P ratio during both the autumn survey and the experiment (Fig. 6). During the autumn survey, this linear relationship could be further divided into two groups by K-means cluster analysis based on APA, such that the slope of the fitted straight line was significantly higher in the high APA group (P<0.01; red line in Fig. 6) relative to that of low APA group (P<0.01; black line in Fig. 6). However, no obvious linear relationship was found between APA and the DIN : PO4-P ratio in winter (Fig. S7).
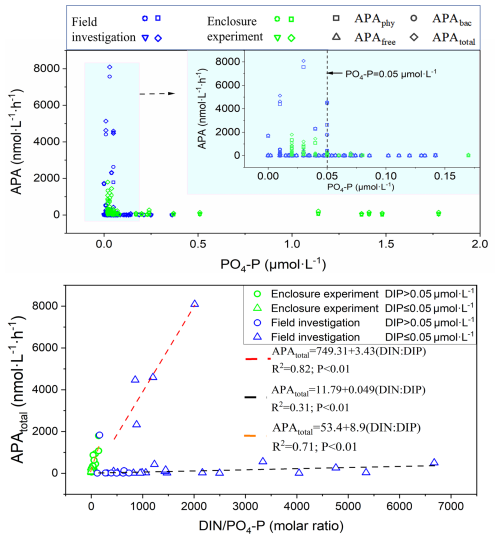
Figure 6Phosphate (PO4-P) concentration threshold for alkaline phosphatase activity (APA) (upper graph), and fitted linear regression lines between APAtotal and the ratio of dissolved inorganic nitrogen-to-phosphate () (lower graph) based on data from the field study and the enclosure experiment. Fitted linear equations and the coefficient of determination (R2) are shown; APA components as in Fig. 4.
3.6 Functional genes for phosphorus nutrient utilization
During the autumn field study, the AP regulatory gene phoD had higher read counts at stations with lower PO4-P concentrations (S2_11 vs. S1_11). Station S2_11 also showed a significant increase in APA compared to S1_11. During the winter survey, station S4_3 had relatively low PO4-P concentrations and high APA, along with high read counts for AP regulatory genes including phoD.
In the CG, the PO4-P concentration gradually decreased during the 29 d experiment (Fig. 5), while the phoD and phoABAP (AP synthesis genes) and phoP (regulatory gene) increased, accompanied by an increase in APA (Fig. 7). Comparing the two groups groups with different DIN : PO4-P ratios, the PO4-P concentration in the CG over the 29 d was lower than that in the TG (Fig. 5). Additionally, the read counts of phoD, phoAB, and phoP were higher in the CG by the end of the experiment (day 29), and APA was significantly higher than in the TG over the same period (P<0.01; Fig. 7).
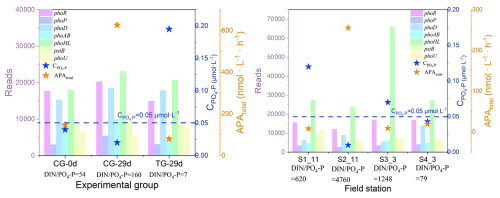
Figure 7Changes in phosphorus (P) nutrient utilization genes and active phosphate concentration () and total alkaline phosphatase activity (APAtotal) in Laizhou Bay and the experimental culture group. S1_11 and S2_11 represent data from two monitoring stations in November 2021, and S3_3 and S4_3 represent data from two monitoring stations in March 2021. CG-0d is the initial day for the control group, while CG-29d and TG-29d represent the 29th day for the control and treatment groups, respectively (see Table S1). Genes in figure are phoR (phosphate regulon sensor histidine kinase), phoP (alkaline phosphatase synthesis response regulator), phoD (alkaline phosphatase D), phoAB (alkaline phosphatase), phoHL (phosphate starvation-inducible protein and related proteins), phoB (phosphate transport system ATP-binding protein), and phoU (phosphate transport system protein). Dashed horizontal lines indicate the threshold PO4-P concentration.
4.1 Phosphorus limitation driven by the anthropogenic nitrogen pump in Laizhou Bay
While the Yellow River and Xiaoqing River are indeed the dominant sources of nutrients to Laizhou Bay (Fig. 2), our comprehensive analysis of ten major rivers provides a complete picture of the terrigenous nutrient load spectrum. Although these smaller tributaries contribute less to the total nutrient load, their impact on local nutrient concentrations and ratios, particularly in adjacent estuarine and coastal waters, remains significant. This basin-wide analysis reveals that highly elevated N:P ratios are a regional scale of anthropogenic nutrient imbalance (Figs. 2–3). The nutrient regime of LZB has changed markedly due to anthropogenic perturbations over the past six decades, shifting from a N-limited oligotrophic state before the 1990s to a potentially P-limited eutrophic state (Xin et al., 2019). Phosphorus limitation in LZB may be influenced by both anthropogenic inputs and shifts in biogeochemical processes. From the 1980s to 2020s, the ratio increased sharply from 200:1 to ca. 1600:1 (Xin et al., 2019), mainly due to the effects of the YR which contributes the largest river discharge in the LZB. Our study also showed that the average N and P flux ratio of the main rivers discharging into LZB is ca. 5.6× higher than the Redfield ratio (Fig. 2). The high values of the DIN : PO4-P ratio in LZB were distributed in the nearshore area and gradually decreased moving offshore, suggesting that the terrigenous nutrient input may significantly influence the spatial distribution of the DIN : PO4-P ratio in LZB (Fig. 3). The pronounced increase in the N:P ratio of riverine nutrient loads is driven by anthropogenic activities, especially the heavy application of N fertilizer, and industrial and domestic P restrictions (Wang et al., 2019; Liang et al., 2023). Atmospheric deposition contributes ca. 30 % to 50 % of the total DIN of LZB (Shou et al., 2018), and only about 10 % of total phosphate (Zhang et al., 2024), leading to an N:P ratio far exceeding the 16:1 Redfield ratio (Chen et al., 2021; Zhang et al., 2024). Thus, the imbalanced input of N and P from both terrigenous and atmospheric deposition is the critical factor aggravating the ratio of LZB.
The increasing DIN concentration in coastal waters, driven by anthropogenic N inputs, promotes phytoplankton growth, leading to elevated Chl a levels (Fig. 3). Prior studies demonstrate that phytoplankton nutrient uptake under such conditions does not adhere to the classical 16:1 Redfield ratio but rather exhibits higher DIN : PO4-P uptake ratios (Fransner et al., 2018; Macias et al., 2019). This pattern is also observed in our study where phytoplankton culture groups, with high DIN : PO4-P ratios showed correspondingly high nutrient absorption ratios (Fig. 5). Under high DIN : PO4-P ratio conditions, phytoplankton upregulate high-affinity phosphate transport systems (e.g., pstSCAB), whose Km value (Michaelis-Menten constant, reflecting the affinity of the transport system for phosphate, with lower Km indicating higher affinity) can be as low as 0.1 µmol L−1 (Dyhrman and Haley, 2006). This enables efficient dissolved phosphate uptake even under extremely low P concentrations (<1 µmol L−1). When P is sufficient, a delayed negative feedback mechanism in the PhoB PhoR two-component system allows continuous P uptake, with rapid conversion to storage forms such as polyphosphate (polyP) (Martin et al., 2011). Concurrently, phytoplankton secrete AP to mineralize DOP, further enhancing P acquisition (Lin et al., 2016). This highly efficient P competition strategy not only exacerbates P limitation for non-storing species but also widens the range of the N:P ratio of residual dissolved inorganic nutrients due to their high N:P uptake demands (Fransner et al., 2018), creating a positive feedback loop that ultimately favors the dominance of P-storing species (Klausmeier et al., 2004).
Therefore, rather than improving the DIN : PO4-P imbalance, biological adaptations to anthropogenic high DIN : PO4-P inputs appear to amplify and perpetuate elevated DIN : PO4-P conditions in coastal ecosystems. This creates an ecological “trap” where the ecosystem becomes increasingly dominated by high adapted species that reinforce the imbalance through their nutrient acquisition and storage strategies. Management strategies must prioritize the reduction of nitrogen inputs at their source to mitigate the N:P imbalance and restore ecological balance, rather than relying on intrinsic biological compensation within the coastal zone.
4.2 AP regulation by PO4-P concentration and the DIN : PO4-P ratio
APA is regulated by multiple environmental factors, such as PO4-P concentration, the DIN : PO4-P ratio and temperature. The PO4-P threshold (0.05 µmol L−1) identified in this study aligns with reported values from both coastal and open-ocean waters, including the semi-enclosed Bohai and Yellow Seas (Jin et al., 2024), the Sargasso Sea (Lomas et al., 2010), the eastern subtropical Atlantic (Mahaffey et al., 2014), and the subtropical Pacific (Suzumura et al., 2012). In the present study, both data from field surveys and enclosure experiment data confirmed this concentration threshold, with the field data demonstrating a significant increase in APA and substrate affinity below this concentration. Moreover, enzyme kinetics data from field investigations in LZB further validated the phosphate concentration threshold, showing that AP activity and substrate affinity increased significantly when phosphate concentration dropped below the threshold (Fig. S6). The Km in autumn was lower than in winter, while Vmax was much higher in autumn than in winter. The lower Km (indicating higher substrate affinity) and much higher Vmax (indicating higher maximum hydrolysis capacity) in autumn compared to winter collectively resulted in higher APA (Fig. 3).
When PO4-P concentrations fell below the threshold in autumn, a strong positive APA-DIN : PO4-P ratio correlation was found (P<0.01), consistent with observations in the Bohai Sea (Jin et al., 2024) and NW Mediterranean (Bogé et al., 2017). In winter, however, this relationship was undetectable in LZB. The absence of a significant APA response to low PO4-P or high DIN : PO4-P ratios in winter may be attributed to several factors. Low temperatures likely directly suppress microbial metabolic rates and enzyme production, including APA. Additionally, seasonal shifts in phytoplankton community composition – such as a predominance of diatoms in winter, which may exhibit an inherently lower responsiveness to phosphorus stress compared to the autumn assemblages – could also contribute to the observed seasonal discrepancy in APA regulation (Ivancic et al., 2016; Ou et al., 2024). Furthermore, genetic analysis showed no significant AP gene upregulation at high DIN : PO4-P ratios when PO4-P remained above the identified threshold (station S3_3, Fig. 7), confirming that sub-threshold PO4-P concentrations are essential for APA induction. Below this threshold, APA increases proportionately with increasing DIN : PO4-P ratios.
Studies indicate that interspecific differences in APA expression capacity, induction thresholds, and regulatory patterns may influence phytoplankton adaptation to phosphorus limitation and community succession. Laboratory cultures show that APA induction thresholds vary widely (10.1–16.4 µmol L−1), with the dinoflagellate P. minimum requiring significantly higher phosphate concentrations than diatoms (Meseck et al., 2009). In natural systems, diatom taxa that dominate under phosphorus limitation commonly express APA, whereas few dinoflagellates do so, and their APA expression is not linked to dominance (Ivancic et al., 2016). Furthermore, diatoms exhibit phosphate-regulated APA, while dinoflagellate APA appears less responsive to phosphate levels, possibly regulated by other factors (Dyhrman and Ruttenberg, 2006). Such heterogeneity in APA strategies suggests that species with lower induction thresholds or stronger expression may gain a competitive advantage under fluctuating phosphorus supply, potentially influencing succession (Fig. 5). However, APA represents only one of multiple phosphorus-acquisition strategies, and its role is likely a contributing factor rather than the sole driver of community shifts.
The phenomenon of APA surge in coastal phytoplankton reveals complex regulatory mechanisms and ecological adaptation strategies. When ambient PO4-P concentrations fall below the 0.05–0.2 µmol L−1 threshold (Dyhrman and Ruttenberg, 2007), phytoplankton activate the pho regulon (a gene suite responsible for phosphorus scavenging) operon (a gene cluster containing the pstSCAB phosphate transport system and phoA/phoX alkaline phosphatase genes) through the PhoB/PhoR two-component system (a P-sensing system consisting of histidine kinase PhoR and the response regulator PhoB) (Lin et al., 2016). Notably, our findings demonstrate a significant positive correlation between APA and the DIN : PO4-P ratio when PO4-P concentrations fall below the threshold. This result may stem from the following fundamental mechanisms: (1) High DIN : PO4-P conditions induce an enhanced capacity for P acquisition to maintain elemental stoichiometric balance, manifested as increased APA with rising DIN : PO4-P ratios; (2) Elevated DIN : PO4-P ratios may indirectly modulate APA through N-sensitive regulators (e.g., PII protein) in the pho signaling pathway (Ustick et al., 2021).
It is noteworthy that prior studies have found that under the same N:P ratio and active phosphate concentration, phytoplankton APA in DON-enriched cultures was significantly higher than that in cultures without DON (Fitzsimons et al., 2020). Our winter survey data revealed a significant positive correlation between DON and APAbac (Fig. 4), similar to findings of a correlation between APAphy and DON concentration (Ou et al., 2024), indicating that not only PO4-P but also nutrient composition and ratios, can affect APA. Regulation of APA by DON may potentially operate through specific DON components or their degradation products influencing P metabolic pathways, or through DON serving as an alternative N source that stimulates phytoplankton growth and P demand (Ma et al., 2018; Forchhammer et al., 2022). Meanwhile, DON serving as alternative nitrogen source may enhance phytoplankton growth and phosphorus demand.
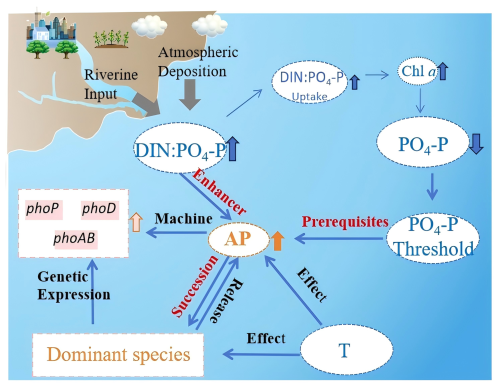
4.3 Conceptual framework of anthropogenic N pump-driven APA regulatory cascades
Human activities have significantly altered coastal nutrient dynamics via multi-level ecological feedback mechanisms that regulate APA activity patterns (Fig. 8). The “anthropogenic N pump” driven by riverine discharge and atmospheric deposition (Jin et al., 2024) has induced a disproportionate increase in DIN inputs, leading to relative PO4-P depletion and elevated DIN : PO4-P ratios (Figs. 2 and 3). This nutrient imbalance triggers phytoplankton adaptive responses, i.e., when PO4-P concentrations fall below the identified threshold, the pho regulon and other APA-related gene clusters are activated (Fig. 7), while the combined effects of high DIN : PO4-P ratios and P limitation significantly enhance APA expression and DOP mineralization capacity (Fig. 6). These biochemical responses initiate ecological cascades that favor phytoplankton with high APA activity and superior DOP utilization capabilities (Fig. 3; Hackett et al., 2005; Nicholson et al., 2006; Ivancic et al., 2016; Ou et al., 2024). Variation in seasonal temperature regulates this process both through direct suppression of APA activity (via reduced enzymatic kinetics and microbial metabolism) and indirect modulation of community composition (reflecting thermal niche differentiation among phytoplankton groups) (Toseland et al., 2013; Thomas et al., 2017).
The above system ultimately establishes a positive feedback loop: APA-mediated DOP utilization increases phytoplankton biomass (as reflected in elevated Chl a), which accelerates PO4-P depletion and exacerbates the DIN : PO4-P imbalance, thereby continuously selecting for APA-enhanced genotypes. This conceptual framework demonstrates how anthropogenic alteration of DIN : PO4-P stoichiometry triggers cross-scale responses from molecular to ecosystem levels, creating self-reinforcing ecological restructuring.
This study confirmed the existence of a PO4-P threshold for APA activation in both phytoplankton and bacteria in LZB, China, using a combination of field studies and enclosure experimentation. The thresholds for APA surges in phytoplankton and bacteria were comparable, with no significant differences observed between them. Results of the relationship between enzyme kinetic parameters and environmental parameters further validate the existence of a PO4-P threshold. When PO4-P levels fell below this threshold, AP-related gene expression increased at survey stations and in culture experiments resulting in high APA. We conclude that in LZB, APA was primarily regulated by PO4-P concentration, the DIN : PO4-P ratio, and phytoplankton biomass. Typically, a linear positive correlation was found between APA and the DIN : PO4-P ratio under low P stress. Most importantly, this study refines the conceptual theory of AP's role in the P cycle under the influence of anthropogenic activities: PO4-P concentrations falling below the threshold value are a prerequisite for observed APA surges, and the DIN : PO4-P ratio is the primary environmental factor driving the increase in APA when PO4-P concentration is below the identified threshold.
The data are available upon reasonable request.
The supplement related to this article is available online at https://doi.org/10.5194/bg-23-1609-2026-supplement.
YY: Writing-original draft, Methodology, Data analysis and curation. XD: Methodology, Data analysis and curation; SL: Methodology, Resources, Writing-review & editing, Supervision; MZ, SL, HL, GZ, HM, and XH: Data curation; XW: Supervision.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
This research was funded by the National Natural Science Foundation of China (grant no. 42576040), the National Key Research and Development Project of China (grant no. 2018YFC1407602), and the Fundamental Research Funds for the Central Universities (grant nos. 202464007 and 202472007).
This research was funded by the National Natural Science Foundation of China (grant no. 42576040), the National Key Research and Development Project of China (grant no. 2018YFC1407602), and the Fundamental Research Funds for the Central Universities (grant nos. 202464007 and 202472007).
This paper was edited by Wei Wen Wong and reviewed by Dongliang Lu and one anonymous referee.
Armstrong, F. A. J., Williams, P. M., and Strickland, J. D. H.: Photo-oxidation of organic matter in sea water by ultra-violet radiation, analytical and other applications, Nature, 211, 481–483, https://doi.org/10.1038/211481a0, 1966.
Bogé, G., Lespillette, M., Jamet, D., and Jamet, J. L.: Role of DOP on the alkaline phosphatase activity of size fractionated plankton in coastal waters in the NW Mediterranean Sea (Toulon Bay, France), Mar. Pollut. Bull., 117, 264–273, https://doi.org/10.1016/j.marpolbul.2016.11.037, 2017.
Brooks, S. P. J.: A simple computer program with statistical tests for the analysis of enzyme kinetics, Biotechnology, 13, 906–911, 1992.
Browning, T. J., Achterberg, E. P., Yong, J. C., Rapp, I., Utermann, C., Engel, A., and Moore, C. M.: Iron limitation of microbial phosphorus acquisition in the tropical North Atlantic, Nat. Commun., 8, 15465, https://doi.org/10.1038/ncomms15465, 2017.
Chen, H. Y., Huang, L. M., Ho, T. Y., Chiang, K. P., and Chou, W. C.: A study of the nitrogen and phosphorus imbalance in East Asia based on the distribution patterns of and stoichiometric variation in global atmospheric nitrogen and phosphorus, Atmos. Environ., 266, 118691, https://doi.org/10.1016/j.atmosenv.2021.118691, 2021.
Dyhrman, S. T. and Haley, S. T.: Phosphorus scavenging in the unicellular marine diazotroph Crocosphaera watsonii, Appl. Environ. Microbiol., 72, 1452–1458, https://doi.org/10.1128/AEM.72.2.1452-1458.2006, 2006.
Dyhrman, S. T. and Ruttenberg, K. C.: Presence and regulation of alkaline phosphatase activity in eukaryotic phytoplankton from the coastal ocean: Implications for dissolved organic phosphorus remineralization, Limnol. Oceanogr., 51, 1381–1390, https://doi.org/10.4319/lo.2006.51.3.1381, 2006.
Dyhrman, S. T. and Ruttenberg, K. C.: Presence and regulation of alkaline phosphatase activity in eukaryotic phytoplankton from the coastal ocean: Implications for dissolved organic phosphorus remineralization, Limnol. Oceanogr., 51, 1381–1390, https://doi.org/10.4319/lo.2006.51.3.1381, 2007.
Feng, T. Y., Yang, Z. K., Zheng, J. W., Xie, Y., Li, D. W., Murugan, S. B., Yang, W. D., Liu, J. S., and Li, H. Y.: Examination of metabolic responses to phosphorus limitation via proteomic analyses in the marine diatom Phaeodactylum tricornutum, Sci. Rep., 5, 10373, https://doi.org/10.1038/srep10373, 2015.
Fitzsimons, M. F., Probert, I., Gaillard, F., and Rees, A. P.: Dissolved organic phosphorus uptake by marine phytoplankton is enhanced by the presence of dissolved organic nitrogen, J. Exp. Mar. Biol. Ecol., 530, 151434, https://doi.org/10.1016/j.jembe.2020.151434, 2020.
Forchhammer, K., Selim, K. A., and Huergo, L. F.: New views on PII signaling: from nitrogen sensing to global metabolic control, Trends Microbiol., 30, 722–735, https://doi.org/10.1016/j.tim.2021.12.014, 2022.
Fransner, F., Gustafsson, E., Tedesco, L., Vichi, M., Hordoir, R., Roquet, F., Spilling, K., Kuznetsov, I., Eilola, K., Mörth, C. M., Humborg, C., and Nycander, J.: Non-Redfieldian Dynamics Explain Seasonal pCO2 Drawdown in the Gulf of Bothnia, J. Geophys. Res.-Oceans, 123, 166–188, https://doi.org/10.1002/2017JC013019, 2018.
Hackett, J. D., Scheetz, T. E., Yoon, H. S., Soares, M. B., Bonaldo, M. F., Casavant, T. L., and Bhattacharya, D.: Insights into a dinoflagellate genome through expressed sequence tag analysis, BMC Genomics, 6, 80, https://doi.org/10.1186/1471-2164-6-80, 2005.
Hoppe, H.-G.: Significance of exoenzymatic activities in the ecology of brackish water: measurements by means of methylumbelliferyl-substrates, Mar. Ecol. Prog. Ser., 11, 299–308, https://doi.org/10.3354/meps011299, 1983.
Ivancic, I., Pfannkuchen, M., Godrijan, J., Djakovac, T., Pfannkuchen, D. M., Korlevic, M., Gasparovic, B., and Najdek, M.: Alkaline phosphatase activity related to phosphorus stress of microphytoplankton in different trophic conditions, Prog. Oceanogr., 146, 175–186, https://doi.org/10.1016/j.pocean.2016.07.003, 2016.
Ivancic, I., Kraus, R., Najdek, M., and Cozzi, S.: Ecological Importance of Alkaline Phosphatase Activity in Changing Marine Environmental Conditions, Water, 13, 2750, https://doi.org/10.3390/w13192750, 2021.
Jin, H., Zhang, C., Meng, S., Wang, Q., Ding, X., Meng, L., Zhuang, Y., Yao, X., Gao, Y., Shi, F., Mock, T., and Gao, H.: Atmospheric deposition and river runoff stimulate the utilization of dissolved organic phosphorus in coastal seas, Nat. Commun., 15, 658, https://doi.org/10.1038/s41467-024-44838-7, 2024.
Kang, W., Wang, Z. H., Liu, L., and Guo, X.: Alkaline phosphatase activity in the phosphorus-limited southern Chinese coastal waters, J. Environ. Sci., 86, 38–49, https://doi.org/10.1016/j.jes.2019.04.026, 2019.
Karl, D. M.: Microbially Mediated Transformations of Phosphorus in the Sea: New Views of an Old Cycle, in: Annual Review of Marine Science, vol. 6, edited by: Carlson, C. A. and Giovannoni, S. J., Annual Reviews, Palo Alto, 279–337, https://doi.org/10.1146/annurev-marine-010213-135046, 2014.
Klausmeier, C. A., Litchman, E., Daufresne, T., and Levin, S. A.: Optimal nitrogen-to-phosphorus stoichiometry of phytoplankton, Nature, 429, 171–174, https://doi.org/10.1038/nature02454, 2004.
Labry, C., Delmas, D., and Herbland, A.: Phytoplankton and bacterial alkaline phosphatase activities in relation to phosphate and DOP availability within the Gironde plume waters (Bay of Biscay), J. Exp. Mar. Biol. Ecol., 318, 213–225, https://doi.org/10.1016/j.jembe.2004.12.017, 2005.
Li, H. G., Li, X. R., Xu, Z. H., Liang, S. K., Ding, Y., Song, D. H., and Guo, H.: Nutrient budgets for the Bohai Sea: Implication for ratio imbalance of nitrogen to phosphorus input under intense human activities, Mar. Pollut. Bull., 179, 113665, https://doi.org/10.1016/j.marpolbul.2022.113665, 2022.
Liang, W., Wang, Y., Mu, J. L., Wu, N., Wang, J. Y., and Liu, S. M.: Nutrient changes in the Bohai Sea over the past two decades, Sci. Total Environ., 903, 166696, https://doi.org/10.1016/j.scitotenv.2023.166696, 2023.
Lin, S. J., Litaker, R. W., and Sunda, W. G.: Phosphorus physiological ecology and molecular mechanisms in marine phytoplankton, J. Phycol., 52, 10–36, https://doi.org/10.1111/jpy.12365, 2016.
Lin, X., Wang, L., Shi, X. G., and Lin, S. J.: Rapidly diverging evolution of an atypical alkaline phosphatase (PhoAaty) in marine phytoplankton: insights from dinoflagellate alkaline phosphatases, Front. Microbiol., 6, 868, https://doi.org/10.3389/fmicb.2015.00868, 2015.
Lomas, M. W., Burke, A. L., Lomas, D. A., Bell, D. W., Shen, C., Dyhrman, S. T., and Ammerman, J. W.: Sargasso Sea phosphorus biogeochemistry: an important role for dissolved organic phosphorus (DOP), Biogeosciences, 7, 695–710, https://doi.org/10.5194/bg-7-695-2010, 2010.
Luo, H. W., Benner, R., Long, R. A., and Hu, J. J.: Subcellular localization of marine bacterial alkaline phosphatases, P. Natl. Acad. Sci. USA, 106, 21219–21223, https://doi.org/10.1073/pnas.0907586106, 2009.
Ma, S. N., Wang, H. J., Wang, H. Z., Li, Y., Liu, M., Liang, X. M., Yu, Q., Jeppesen, E., and Sondergaard, M.: High ammonium loading can increase alkaline phosphatase activity and promote sediment phosphorus release: A two-month mesocosm experiment, Water Res., 145, 388–397, https://doi.org/10.1016/j.watres.2018.08.043, 2018.
Maavara, T., Akbarzadeh, Z., and Van Cappellen, P.: Global Dam-Driven Changes to Riverine Ratios Delivered to the Coastal Ocean, Geophys. Res. Lett., 47, e2020GL088288, https://doi.org/10.1029/2020GL088288, 2020.
Macias, D., Huertas, I. E., Garcia-Gorriz, E., and Stips, A.: Non-Redfieldian dynamics driven by phytoplankton phosphate frugality explain nutrient and chlorophyll patterns in model simulations for the Mediterranean Sea, Prog. Oceanogr., 173, 37–50, https://doi.org/10.1016/j.pocean.2019.02.005, 2019.
Mahaffey, C., Reynolds, S., Davis, C. E., and Lohan, M. C.: Alkaline phosphatase activity in the subtropical ocean: insights from nutrient, dust and trace metal addition experiments, Front. Mar. Sci., 1, 13, https://doi.org/10.3389/fmars.2014.00073, 2014.
Mäki, A., Salmi, P., Mikkonen, A., Kremp, A., and Tiirola, M.: Sample Preservation, DNA or RNA Extraction and Data Analysis for High-Throughput Phytoplankton Community Sequencing, Front. Microbiol., 8, 1848, https://doi.org/10.3389/fmicb.2017.01848, 2017.
Marie, D., Partensky, F., Jacquet, S., and Vaulot, D.: Enumeration and cell cycle analysis of natural populations of marine picoplankton by flow cytometry using the nucleic acid stain SYBR Green I, Appl. Environ. Microbiol., 63, 186–193, https://doi.org/10.1128/AEM.63.1.186-193.1997, 1997.
Martin, P., Van Mooy, B. A. S., Heithoff, A., and Dyhrman, S. T.: Phosphorus supply drives rapid turnover of membrane phospholipids in the diatom Thalassiosira pseudonana, ISME J., 5, 1057–1060, https://doi.org/10.1038/ismej.2010.192, 2011.
Meseck, S. L., Alix, J. H., Wikfors, G. H., and Ward, J. E.: Differences in the soluble, residual phosphate concentrations at which coastal phytoplankton species up-regulate alkaline-phosphatase expression, as measured by flow-cytometric detection of ELF-97® fluorescence, Estuar. Coasts, 33, 137–151, https://doi.org/10.1007/s12237-009-9211-7, 2009.
Murphy, J. and Riley, J. P.: A modified single solution method for the determination of phosphate in natural waters, Anal. Chim. Acta, 27, 31–36, https://doi.org/10.1016/S0003-2670(00)88444-5, 1962.
Nausch, M.: Alkaline phosphatase activities and the relationship to inorganic phosphate in the Pomeranian bight (southern Baltic Sea), Aquat. Microb. Ecol., 16, 87–94, https://doi.org/10.3354/ame016087, 1998.
Nicholson, D., Dyhrman, S., Chavez, F., and Paytan, A.: Alkaline phosphate activity in the phytoplankton communities of Monterey Bay and San Francisco Bay, Limnol. Oceanogr., 51, 874–883, https://doi.org/10.4319/lo.2006.51.2.0874, 2006.
Ou, L. J., Wang, Z., Ding, G. M., Han, F. X., Cen, J. Y., Dai, X. F., Li, K. Q., and Lu, S. H.: Organic nutrient availability and extracellular enzyme activities influence harmful algal bloom proliferation in a coastal aquaculture area, Aquaculture, 582, 740530, https://doi.org/10.1016/j.aquaculture.2023.740530, 2024.
Parsons, T. R., Maita, Y., and Lalli, C. M.: A Manual of Chemical and Biological Methods for Seawater Analysis, Pergamon Press, Oxford, 173 pp., ISBN 0080302874, 1984.
Peñuelas, J. and Sardans, J.: The global nitrogen-phosphorus imbalance, Science, 375, 266–267, https://doi.org/10.1126/science.abl4827, 2022.
Qin, X., Shi, X., Gao, Y., Dai, X., Ou, L., Guan, W., Lu, S., Cen, J., and Qi, Y.: Alkaline phosphatase activity during a phosphate replete dinoflagellate bloom caused by Prorocentrum obtusidens, Harmful Algae, 103, 101979, https://doi.org/10.1016/j.hal.2021.101979, 2021.
Shou, W. W., Zong, H. B., Ding, P. X., and Hou, L. J.: A modelling approach to assess the effects of atmospheric nitrogen deposition on the marine ecosystem in the Bohai Sea, China, Estuar. Coast. Shelf Sci., 208, 36–48, https://doi.org/10.1016/j.ecss.2018.04.025, 2018.
Song, D. B., Gao, Z. Q., Zhang, H., Xu, F. X., Zheng, X. Y., Ai, J. Q., Hu, X. K., Huang, G. P., and Zhang, H. B.: GIS-based health assessment of the marine ecosystem in Laizhou Bay, China, Mar. Pollut. Bull., 125, 242–249, https://doi.org/10.1016/j.marpolbul.2017.08.027, 2017.
Suzumura, M., Hashihama, F., Yamada, N., and Kinouchi, S.: Dissolved phosphorus pools and alkaline phosphatase activity in the euphotic zone of the western North Pacific Ocean, Front. Microbiol., 3, 99, https://doi.org/10.3389/fmicb.2012.00099, 2012.
Thomas, M. K., Aranguren-Gassis, M., Kremer, C. T., Gould, M. R., Anderson, K., Klausmeier, C. A., and Litchman, E.: Temperature-nutrient interactions exacerbate sensitivity to warming in phytoplankton, Glob. Change Biol., 23, 3269–3280, https://doi.org/10.1111/gcb.13641, 2017.
Toseland, A., Daines, S. J., Clark, J. R., Kirkham, A., Strauss, J., Uhlig, C., Lenton, T. M., Valentin, K., Pearson, G. A., Moulton, V., and Mock, T.: The impact of temperature on marine phytoplankton resource allocation and metabolism, Nat. Clim. Change, 3, 979–984, https://doi.org/10.1038/nclimate1989, 2013.
Ustick, L. J., Larkin, A. A., Garcia, C. A., Garcia, N. S., Brock, M. L., Lee, J. A., Wiseman, N. A., Moore, J. K., and Martiny, A. C.: Metagenomic analysis reveals global-scale patterns of ocean nutrient limitation, Science, 372, 287–291, https://doi.org/10.1126/science.abe6301, 2021.
Wang, J. J., Yu, Z. G., Wei, Q. S., and Yao, Q. Z.: Long-Term Nutrient Variations in the Bohai Sea Over the Past 40 Years, J. Geophys. Res.-Oceans, 124, 703–722, https://doi.org/10.1029/2018JC014765, 2019.
Wu, Z. C., Zhou, C. Y., Wang, P., and Fei, Z. H.: Responses of tidal dynamic and water exchange capacity to coastline change in the Bohai Sea, China, Front. Mar. Sci., 10, 1118795, https://doi.org/10.3389/fmars.2023.1118795, 2023.
Xin, M., Wang, B. D., Xie, L. P., Sun, X., Wei, Q. S., Mang, S. K., and Chen, K.: Long-term changes in nutrient regimes and their ecological effects in the Bohai Sea, China, Mar. Pollut. Bull., 146, 562–573, https://doi.org/10.1016/j.marpolbul.2019.07.011, 2019.
Yamaguchi, T., Furuya, K., Sato, M., and Takahashi, K.: Phosphate release due to excess alkaline phosphatase activity in Trichodesmium erythraeum, Plankton Benthos Res., 11, 29–36, https://doi.org/10.3800/pbr.11.29, 2016.
Zhang, C., Luo, H., Huang, L. M., and Lin, S. J.: Molecular mechanism of glucose-6-phosphate utilization in the dinoflagellate Karenia mikimotoi, Harmful Algae, 67, 74–84, https://doi.org/10.1016/j.hal.2017.06.006, 2017.
Zhang, X., Zhang, J. P., Shen, Y., Zhou, C. H., and Huang, X. P.: Dynamics of alkaline phosphatase activity in relation to phytoplankton and bacteria in a coastal embayment Daya Bay, South China, Mar. Pollut. Bull., 131, 736–744, https://doi.org/10.1016/j.marpolbul.2018.05.008, 2018.
Zhang, X. J., Tian, C. G., Sun, Z. Y., Yin, X. H., Sun, R., and Wang, J. Y.: Temporal and Spatial Distribution of DIN and PO4-P Concentrations and Source Apportionment Along the Bohai Sea of China During 2015-2022, Chinese Geogr. Sci., 34, 1004–1016, https://doi.org/10.1007/s11769-024-1464-3, 2024.