the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
The inverted microbial loop stimulates mineralisation of sedimentary organic detritus
Dick van Oevelen
Silvia Hidalgo-Martinez
Filip J. R. Meysman
Respiration is a key process in the organic carbon cycle of marine sediments, the understanding of which is key to future management decisions which aim to maximise sediment carbon storage. The microbial community is typically considered the dominant actor in overall sedimentary respiration, but knowledge is lacking about interactions with other components, particularly the macrofauna. The “inverted microbial loop” hypothesis suggests that macrofaunal activity stimulates the microbial respiration of organic carbon through the mixing of fresh organic carbon to depth, and subsequent priming (i.e. activation of refractory detritus by co-respiration with fresh detritus).
We conducted experimental incubations to partition respiration amongst the microbial and macrofaunal components of the community and investigate interactions between them. We prepared sediment cores with native benthic communities, macrofauna only and microbial communities only. We added 13C labelled fresh organic matter to these cores and measured respiration over 7 d, quantifying both O2 consumption (reflecting remineralisation of all sedimentary organic C) and production of 13C dissolved inorganic C (DIC, reflecting remineralisation of labile organic C).
Consumption of O2, which reflected remineralisation of ambient as well as added fresh organic C, showed greater rates when macrofaunal and microbial communities were present together than the sum of their separate rates. This provides direct experimental evidence that the inverted microbial loop mechanism stimulates mineralisation of less reactive, ambient organic C. Macrofaunal and microbial communities showed approximately equal contributions to the total community respiration, suggesting that faunal respiration should be more routinely included in carbon degradation modelling. The fate of the added fresh organic C in different treatments suggested competition for this resource between macrofauna and microbes, and some functional redundancy amongst different components of the benthic community. The enhanced understanding of sediment respiration generated by this study has implications for management of shelf seafloors to balance carbon storage with other human uses.
- Article
(2411 KB) - Full-text XML
- BibTeX
- EndNote
Marine sediments play a key role in the global carbon cycle, as they serve as the location for long-term burial of organic carbon (C), with shelf sea and deltaic settings being disproportionately important (Berner, 1982). Continental shelf sediments cover only ∼ 7 %–10 % of the ocean's area, but account for up to 80 % of total marine organic C burial, because shelves are locations of high marine primary production (receiving a high nutrient input) and act as critical interfaces between terrestrial and marine ecosystems (processing terrestrial organic material delivered by rivers). After being deposited at the sediment surface, organic C can follow two principal pathways: it may either be mineralised by the respiratory metabolism of resident organisms (macrofauna, meiofauna or microorganisms) and so be converted back to CO2, or it may escape mineralization through burial into deeper sediment horizons and thus be removed from the short-term carbon cycle. Changes in the relative balance of respiration over burial – whether natural or human-induced – may have significant consequences for the longer-term carbon cycle, ocean chemistry, and climate. Shelf sediments comprise an efficient “recycling reactor” for the organic matter that is deposited from the water column onto the continental shelf seafloor (Middelburg and Meysman, 2007). Isotope labelling experiments have shown that ∼ 90 % of the incoming organic carbon is subject to respiration , and is released back to the water column as dissolved inorganic carbon (CO2, ∼ 80 %) or dissolved organic carbon (∼ 10 %), while the remaining part is eventually buried and preserved (Middelburg and Meysman, 2007; Burdige, 2007). This efficient decomposition process is due to the overall metabolic activity of benthic organisms, i.e. sedimentary microbes as well as sediment-dwelling fauna. While we understand some environmental factors that govern the overall rate of benthic respiration, we lack knowledge about how respiration is partitioned between groups of organisms, or how interactions between groups affects the overall respiration rate. Improving this understanding is important to further our knowledge of marine carbon cycling.
Total community respiration is the process by which all heterotrophic organisms living in the sediment consume organic matter and metabolise it to gain energy, converting it ultimately to CO2. It is often measured as the flux of dissolved inorganic carbon (DIC) or oxygen (O2), that crosses the sediment-water interface and is influenced by several external factors. Total community respiration increases with both temperature and organic matter deposition, and consequently it tends to vary with season (Kristensen, 2000) and shows a strong negative relationship with water depth (Middelburg and Duarte, 2005; Stratmann et al., 2019). Strong current and wave activity can also induce higher respiration rates in sandy sediments, as advective porewater exchange supplies both fresh organic matter and oxygen, thus stimulating mineralisation activity (Huettel et al., 2003; Ehrenhauss and Huettel, 2004; Alongi et al., 2011). Light availability has also been suggested to control respiration in shallow environments, as photosynthesis in biofilms at the sediment surface can increase the supply of organic C or oxygen to the sediment community (Kristensen, 2000; Middelburg and Duarte, 2005; Hubas et al., 2007).
While we have reasonable understanding (outlined above) of the external factors that govern total community respiration, we lack an understanding of the internal mechanisms that determine how respiration is partitioned amongst the different groups of organisms that make up the benthic community, and especially, how interactions between those groups can influence the total community respiration. The microbial component of the community is often assumed to be of paramount importance in driving total community respiration, and evidence for this comes from both observational (e.g., Schwinghamer et al., 1986; Hubas et al., 2006) and modelling (van Oevelen et al., 2006a) studies. Other studies have emphasized that macrofaunal activity may also play a major role (e.g. Herman et al., 1999; Heip et al., 2001), either through their direct contribution to respiration, or through indirect interactions (e.g. increased oxygen supply via pore water irrigation) that stimulate the respiration of the microbial community.
For this reason, it is important to consider how interactions between macrofaunal and microbial activity may influence sediment respiration. In the water column, macrofauna and microbes are linked through the `microbial loop', in which organic C that is lost to the dissolved organic carbon pool during macrofaunal metabolism is subsequently assimilated and transformed into new biomass by the microbial community, and becomes available once again to macrofauna as a food source (Kemp, 1988, 1990; Vasquez-Cardenas et al., 2020). This looping stimulates C cycling and increases remineralization efficiency. However, there is little evidence that macrofaunal grazing on microbes plays an important role in the carbon cycle of marine sediments, as studies have shown that bacterial biomass is a rather minor food source for benthic faunal communities, which typically rely on an input of fresh algal detritus from the water column (e.g. Kemp, 1990; van Oevelen et al., 2006b; Guilini et al., 2010). To better capture the effective carbon cycling that occurs in shelf sediments, Middelburg (2018) proposed the `inverted microbial loop' concept, which states that macrofaunal activity can stimulate sedimentary microbial activity and respiration, rather than depressing it by grazing. In this view, the impact of microbes and macrofauna on the carbon cycling is not sequential but occurs in parallel. Macrofauna transport freshly deposited organic matter to depth, thus making it available to the sediment dwelling microbes for respiration. This then prompts an enhancement of the total community respiration via a priming effect, i.e., an increase in the decomposition rate of the resident “old” sedimentary organic carbon at depth through an input of fresh organic matter.
From a conceptual point of view the “inverted microbial loop” makes sense: it is well known that macrofauna can stimulate both the supply of the electron donor (fresh organic C) as well as the electron accepter (O2) used in respiration to the resident microbial community in marine sediments. Bioirrigation refers to the process by which fauna pump fresh seawater through their burrows, which can increase the oxygenated volume of sediment several-fold and supplies respiratory electron acceptors, thus stimulating microbial degradation (Aller and Aller, 1998; Herman et al., 1999; Kristensen, 2000; Glud et al., 2003; Middelburg and Duarte, 2005), and enhancing total respiration by 25 %–271 % (see Kristensen, 2000, and references therein). Likewise, solid particle mixing by macrofauna during burrowing activity transports freshly deposited organic material to depth in the sediment, which brings together labile (fresh, and readily metabolised) and refractory (slow to decompose) types of organic carbon. This enhanced supply of O2 and/or fresh organic C could lead to priming, whereby refractory organic carbon is now decomposed that would otherwise not have been respired . The occurrence of priming however seems very much dependent on the compounds and environment in question (Bengtsson et al., 2018), but it has been previously observed in marine sediments (van Nugteren et al., 2009b; Gontikaki et al., 2015). Priming mechanisms require further investigation but are likely to involve changes to microbial population composition and activity, and associated enzyme production, mutalism and/or co-metabolism (Bianchi, 2011).
Stimulation of microbial processes by macrofaunal activity is also thought to play a role in Earth evolution. It has been proposed that the rise of animals around 540 Myr ago, and the concomitant evolution of burrowing and bioturbation, may have instigated a more efficient mineralisation of organic matter in the seafloor with potential Earth system impacts (Meysman et al., 2006). Recent studies have quantitatively explored this idea using Earth System Models, and propose that this effect may have been large enough to increase atmospheric CO2 levels, inducing global warming and ocean anoxia (van de Velde et al., 2018).
Here we take an experimental approach to investigate the inverted microbial loop effect. Our principal goal was to resolve the total respiration in marine sediment into contributions of microbial respiration and faunal respiration, and to elucidate any microbial-faunal interaction term. Furthermore, we aimed to assess the role of these three components in the mineralisation of both reactive and refractory organic C.
Few studies have experimentally assessed the contribution of the microbial, faunal and microbial-faunal interaction components to total sediment respiration. Previous works have taken a theoretical approach to partitioning respiration (e.g. Schwinghamer et al., 1986; Franco et al., 2010), but these approaches do not account for positive interactions between components of the benthic community, such as the inverted microbial loop. Furthermore, van Nugteren et al. (2009a) found that the resource partitioning of fresh organic matter between macrofauna and microbes depends on the spatial distribution of the organic matter, with only microbes being able to efficiently utilise resources that are mixed throughout (diffusely distributed) the sediment. This leads us to hypothesize that the inverted microbial loop effect may apply predominantly to the ambient, more refractory and “diffusively” distributed sedimentary organic C, and less to the fresh organic C that is concentrated on the sediment surface.
2.1 Experimental approach and rationale
Marine sediment cores were constructed and then incubated for 7 d. The total oxygen uptake (TOU) was measured as the indicator of total community respiration, which primarily represents mineralisation of refractory organic C. In parallel, we quantified fresh organic matter respiration (FOMR) in the same cores by addition of 13C labelled substrates and determining the subsequent release of 13C labelled dissolved inorganic C (DIC).
To obtain insights into the TOU and FOMR of different components of the benthic community, as well as to assess the interaction between microorganisms and macrofauna, we applied the following four treatments when constructing experimental cores: (1) Control: natural, intact sediment cores. Respiration is due to prokaryotes and macrofauna, and their interaction. (2) Defaunated: sediment cores that were defaunated by inducing anoxia, and exposed again to overlying oxygenated water. Respiration is dominated by prokaryotes (with some meiofauna present), but macrofauna are excluded. (3) Restocked: sediment cores were first de-faunated (by inducing anoxia), and then exposed again to overlying oxygenated water and re-stocked with a controlled macrofaunal community. Respiration is due to prokaryotes and a controlled biomass of macrofauna, and their interaction. (4) Fauna: sediment cores were constructed that contain only clean construction sand, to which macrofauna were introduced. Respiration is due to macrofauna. A control with only clean construction sand was run, but TOU data was not acquired due to instrument problems. However, we expect microbial respiration to be small in these construction sand cores compared to that of the macrofauna added.
The experiment with the four treatments was conducted twice using different 13C labelled substrates. In a first experiment, 13C labelled algal detritus from an axenic culture (13C-AA) was added, which allowed tracing of C into the microbial biomass. In the second experiment, we added natural microphytobenthos cultured in the presence of 13C labelled bicarbonate(13C-MPB), thus providing a fully natural fresh C source.
If there were no interactions between components of the benthic community, respiration rates measured in the “fauna” treatment can simply be added to those from the “defaunated” treatment and would equal the rates measured in the re-stocked treatment (macrofauna and microbes + meiofauna together). Deviations from this expectation are indicative of positive (i.e. inverted microbial loop) or negative (i.e. competitive) interactions between components of the benthic community.
2.2 Sediment collection and experimental conditions
Experiments were conducted in June 2010 and June 2011 at the Netherlands Institute for Sea Research (Yerseke, the Netherlands). Sediment cores and filtered seawater were collected from nearby intertidal sites in the Oosterschelde estuary. Key experimental details are listed in Table 1. Surface sediment cores (19.4 and 14.3 cm inner diameter for 13C-AA and 13C-MPB respectively) were collected in acrylic tubes, and after a short transit to the laboratory (< 2 h), they were kept in darkness in a climate-controlled room at ambient temperature with overlying filtered seawater (0.2 µm pore size) at in-situ salinity (Table 1). Overlying water was oxygenated using air stones, except for when periods oxygen consumption rates were determined (see description below).
2.3 Experimental treatments
Three replicate cores were subjected to each of the 4 treatments in the two experiments. De-faunation of sediment cores for the defaunated and restocked treatments was conducted by asphyxiation as in described Rao et al. (2014), which leaves the sediment stratification intact (as opposed to defaunation by sieving). To this end, anoxic conditions were induced by purging the overlying seawater in the core with N2 gas for several hours and then sealing the cores with gas-tight lids for 4–6 d. After this anoxic period, the cores were opened and the overlying water was exchanged and re-aerated with air stones. Dead organisms that had migrated to the sediment surface were first removed with tweezers. The cores were subsequently left undisturbed for one day to allow the re-oxidation of reduced compounds that had accumulated in the surface layer of sediment. After one day of reaeration, a mix of fauna (Table 2 and further below) was added at the surface of restocked treatment cores, and were allowed migrate into the sediment. Cores were then acclimated again for 1–2 d before being amended with 13C labelled organic detritus. After that the cores were incubated for 7 d.
Table 2Biomass of macrofaunal taxa added (g wet weight m−2) in the “restocked” and “fauna only” treatments. Note that the same biomass values was aimed for in the two treatments, so means and standard deviations are reported across both treatments.

De-faunation by inducing anoxia was selected in preference to de-faunation by sieving. It was felt that sieving would cause extensive changes to sediment structure and composition which would have more potential to introduce artefacts than the possibility of live fauna remaining in the sediment following induction of anoxia. Presence of live fauna in the de-faunated treatment was minimal, and is reported below.
Based on background knowledge about the sampling site (Daggers et al., 2020), we knew a priori that the fauna at the sampling location predominantly consists of the polychaetes Hediste diversicolor, Arenicola marina and Heteromastus filiformis, the gastropod Hydrobia ulvae and the bivalve Cerastoderma edule. These species were therefore selected for the re-stocked and fauna treatment and introduced into cores at densities that simulated the natural faunal community (Table 2 and results).
We acknowledge that greater replication will always strengthen an experiment, but benefits have to be balanced against practical constraints (space, volume of sediment, operator time). Although some experiments use 4 or 5 replicates per treatment, the triplicate replication used here is in line with similar experiments in the literature (e.g. Moodley et al., 2000; Sweetman and Witte, 2008; van Nugteren et al., 2009a; Rossi et al., 2009), and is sufficient for showing difference between treatments despite natural variability (see results).
2.4 Experimental procedures
For the “axenic algae” (13C-AA) experiment, the marine diatom Skeletonema costatum was axenically cultivated in 13C-labelled medium. The resulting algal cells were 28.25 atom % and 14.49 atom % 13C for two separate batches. A slurry of freeze-dried, 13C-labelled biomass (395 ± 11 mg C m−2) was carefully mixed into to the water column and allowed to settle onto the sediment-water interface (so that the whole surface area was more or less homogeneously covered with labelled substrate).
For the “microphytobenthos” (13C-MPB) experiment, microphytobenthos was collected at the study site at the same time as the sediment cores. The top millimetres of sediment were scraped off at locations where distinctly brown patches (indicative of high MPB biomass) were present. This sediment/microphytobenthos mixture was enriched with 13C through incubation in a white plastic culture box (0.6 m × 0.4 m) that was placed outside (ambient temperature) and covered with a transparent lid (natural light). The thin layer of sediment in the culture box was topped with a thin layer of ambient seawater (∼ 5 mm) to prevent dehydration. The next day, 0.136 g of 13C-labelled sodium bicarbonate (NaH13CO3, 99 %; Cambridge Isotope Laboratories) was dissolved in 50 mL of filtered seawater and introduced into the culture. This label addition was repeated daily for 7 d, after which the labelled microphytobenthos was harvested by scraping off the top several millimetres. This mixture was homogenised, frozen in liquid nitrogen (to kill the MPB cells and prevent respiration activity by MPB during the sediment core incubations) and stored until further usage at −18 °C. The chlorophyll-a concentration of this slurry was determined on 3 subsamples using standard fluorometry methods (Aminot and Rey, 2001). The resulting concentration (37 ± 5 µg g−1) was converted to C using a conversion factor of 40 (Stephens et al., 1997) resulting in an estimated 1.5 ± 0.2 mg C g−1. Cores were amended with 12.5 cm3 of slurry (density 2.0 g cm−3), which was added using a pipette, and allowed to settle onto the sediment surface over several hours. Each core hence received 3.08 mmol of C from MPB (corresponding to 2.30 g C m−2). The 13C labelling level of the MPB was unknown, but this does not prevent calculation of respiration rates from the measured 13C-DIC production.
In both experiments, cores were incubated for 7 d after addition of labelled algae, with repeated measurements of O2 consumption and 13C-DIC release during this period (see below). At the termination of the experiment, sediment cores were sub-sampled using plastic syringes and samples were frozen at −18 °C. The remaining sediment in each core was sieved through a 1 mm mesh. Fauna retained on the mesh were picked, and their wet biomass was recorded, after which specimens were frozen for further analysis.
2.5 Respiration measurements
Benthic respiration was measured in all cores through total O2 uptake (TOU, i.e. proxy for total community respiration, primarily of refractory organic C) and release of 13C-DIC (i.e. proxy for respiration of fresh, labile algae) at several time points: before and straight after addition of isotopically labelled algae, and every 1.5 d for 7 d thereafter.
At the beginning of each respiration measurement, the overlying water of each core was sampled for dissolved oxygen (DO), dissolved inorganic carbon (DIC), and 13C of DIC. Cores were then sealed with custom-built gas-tight lids, excluding all air bubbles, and incubated for 2–5 h until O2 saturation in the overlying water had fallen to ∼ 70 %. During the closed incubation, core top water was stirred continuously. At the end of each respiration measurement, core top water was again sampled for the parameters listed above. After respiration measurements, the overlying water in each core was exchanged to avoid build-up of (toxic) metabolic products, and kept aerated by gentle bubbling with air.
2.6 Analytical
Samples for O2 analysis were collected in glass Winkler bottles with ground glass stoppers and known volumes. Bottles were allowed to overflow copiously before MnSO4 and KI in KOH solutions were added and stoppers inserted. Samples were shaken for 30 s, and stored in at 4 °C before analysis within 2 d. Samples were titrated against standardised thiosulphate solution using a micro-titration set-up.
Dissolved inorganic carbon (DIC) samples (20 mL) were stored in crimp-cap vials, and preserved with HgCl2 (20 µL of saturated solution). Vials were stored at 4 °C, inverted with the caps standing in water to prevent the exchange of CO2 with the atmosphere. Samples were analysed for DIC concentration and δ13C as detailed in Moodley et al. (2000), using a Carlo Erba MEGA 540 gas chromatograph coupled to a Finnigan Delta S isotope ratio mass spectrometer, following creation of a He headspace in each sample vial. Standards used were acetanilide, and the IAEA standard CH-6. Repeat analysis of standard materials yielded precision of ± 4.4 % for DIC concentrations, and ± 0.09 ‰ for δ13C.
Sediment samples from the axenic algae experiment were analysed for 13C incorporation into bacterial phospholipid fatty acids (PLFAs) using a modified Bligh-Dyer extraction after Middelburg et al. (2000). Lipids were extracted at room temperature in a mixture of chloroform, methanol and water, before being loaded onto silicic acid columns. Phospholipid fatty acids were eluted in methanol and derivatised to fatty acid methyl esters (FAMEs) using methanolic NaOH. The C12:0 and C19:0 FAMEs were used as internal standards. Samples were separated by gas chromatography using a BPX70 column, combusted in a Thermo GC combustion II interface, and isotopic ratios were measured using a Thermo Delta + isotope ratio mass spectrometer.
2.7 Data analysis
The Total Oxygen Uptake (TOU) of the sediment was calculated from the difference in the total amount of dissolved O2 present (i.e. O2 concentration × chamber volume) between the start and end of each closed incubation, divided by the time elapsed in each measurement (Δt), and normalised to the surface area of the cores (SA), i.e., TOU = (O2 end − O2 start) SA.
Release of 13C-DIC was determined from the difference in total amount of 13C in each chamber (i.e. DIC concentration × chamber volume × At % 13C DIC) between the start and end of the incubation, divided by the duration of the incubation (Δt), and normalised to the surface area (SA) of the cores, i.e. 13C-DIC Release = (13C end − 13C start) SA.
Cumulative TOU and 13C-DIC release were calculated by multiplying each of the measured rates described above by the time periods between closed TOU 13C incubations. These were then summed to produce estimates of cumulative TOU and 13C-DIC release over the whole experiment for each treatment.
Uptake of 13C into bacterial biomass in the 13C-AA experiment was calculated by first subtracting naturally present 13C based on analysis of unlabelled sediment. Presence of 13C in the bacterial indicators i-C14:0, i-C15:0, ai-C15:0 and i-C16:0 was then summed, and scaled up based on these compounds representing 14 % of total bacterial PLFAs, and PLFAs representing 5.6 % of total bacterial biomass (Boschker and Middelburg, 2002).
Statistical analysis of data was performed using Minitab 18. Differences between treatments were investigated using either one-way ANOVA or KruskalWallis, depending on whether data were normally distributed, determined using the Anderson-Darling normality test. For tests of respiration rate data values of n ranged from 10 to 24, and for faunal 13C labelling from 5 to 17. In some cases tests for difference were conducted between treatments for data from each day separately, in which case n=3. We recognise that checking the distribution of such a small group of data is not necessarily possible, and also that use of non-parametric tests in this situation does carry a risk of not identifying patterns which are in fact present. We note that the questions of which statistical tests are most appropriate, and whether statistical testing should be included at all, are ones on which different statisticians and readers are unlikely to agree. We feel that the approach we have taken is justifiable, but acknowledge that any approach to statistical testing which we could take would be open to differences of opinion.
3.1 Biomass of fauna
The living macrofaunal biomass recovered from the control treatment at the end of the 13C-AA experiment (4.8 ± 3.2 g wet weight per core) was far greater than that recovered from the defaunated treatment (0.9 ± 1.0 g wet weight per core). This illustrates that asphyxiation removed > 80 % of the fauna, but still a restricted anoxia-tolerant community (Hediste, Arenicola) survived. The natural biomass present (in the control treatment) was lower than anticipated, and so biomass added to the restocked and fauna treatments (18–21 g wet weight per core, Table 3) was four times higher than the control treatment. Very few dead organisms were seen in treatments where fauna were added, with the majority recovered alive at the end of the experiment. Note that macrofauna biomass data were not recorded for the 13C-MPB experiment.
3.2 Total oxygen uptake
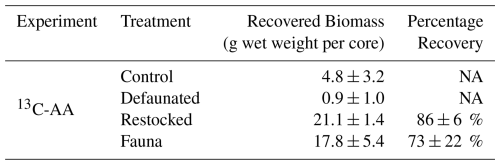
Total Oxygen Uptake rates showed substantial variation and ranged from 9–91 mmol O2 m−2 d−1 in the 13C-AA experiment, and 7–241 mmol O2 m−2 d−1 in the 13C-MPB experiment (Fig. 1). TOU values were generally higher in the 13C-MPB experiment compared to the 13C-AA experiment. Due to problems with the oxygen measurement technique, data is lacking for the control and fauna treatments in the 13C-AA experiment during the first 2 d after feeding.
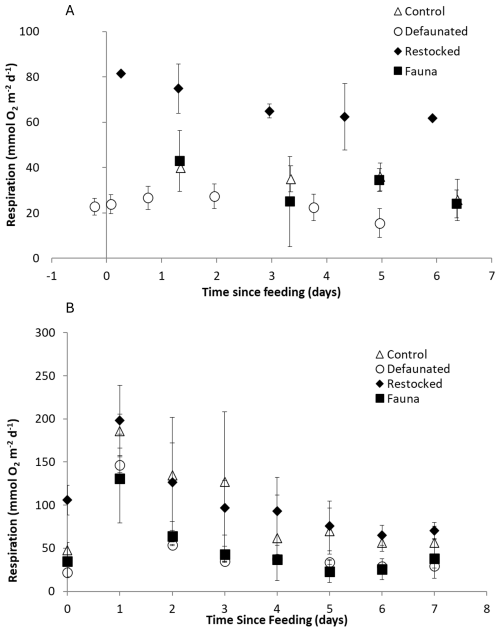
Figure 1Respiration rates, determined as the Total Oxygen Uptake (TOU) of the sediment, in (a) the axenic algae (13C-AA) experiment and (b) the microphytobenthos (13C-MPB) experiment.
In the 13C-AA experiment, TOU showed a slight decrease over time in the restocked treatment, but no clear temporal pattern in the other treatments (Fig. 1a). In the 13C-MPB experiment all treatments displayed a similar temporal pattern, with maximal TOU values immediately after algal addition, and TOU values returning to pre-feeding levels after ∼ 6 d (Fig. 1b). Differences in TOU between treatments were apparent in both experiments (Kruskal-Wallis p < 0.001 for both experiments). In the 13C-AA experiment, TOU values were always higher in the re-stocked cores compared to other treatments (Mann-Whitney pairwise comparisons p < 0.001). There was also a significant difference between the control and defaunated treatments, while other pairs of treatments were not significantly different (Mann-Whitney pairwise comparisons p = 0.004, 0.62, and 0.012 for control vs. defaunated, control vs. fauna, and defaunated vs. fauna, respectively). In the 13C-MPB experiment rates were higher in the control and re-stocked treatments than in the defaunated and fauna only treatments (Kruskal-Wallis, p≤0.001). TOU values in the control and restocked treatments (Mann-Whitney, p=0.130) and in the defaunated and fauna only treatments (Mann-Whitney, p=0.516) were not significantly different from each other.
The cumulative TOU (i.e. the total O2 consumed during each 7 d experiment) was higher in the 13C-MPB experiment compared to the 13C-AA experiment. Cumulative TOU showed a similar pattern between treatments in both experiments (Fig. 2a) and was maximal in the restocked treatment, then followed by the control, and finally the defaunated and fauna only treatments (Fig. 2a). Due to the high variability, significant differences between treatments could be identified for the 13C-AA experiment (ANOVA, p < 0.001, groupings shown in Fig. 2a), but not for the 13C-MPB experiment (ANOVA, p=0.052).
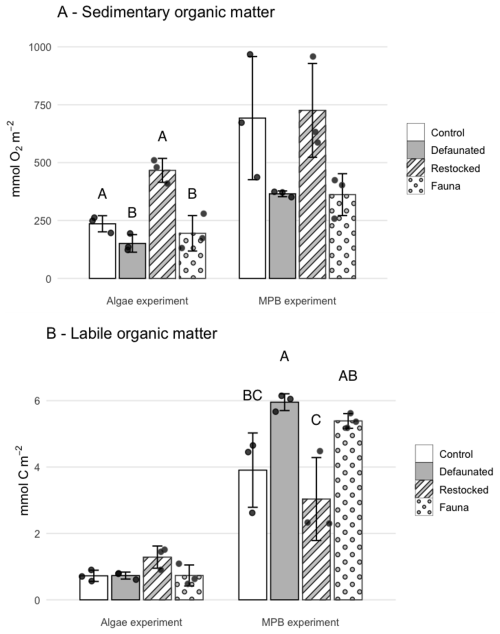
Figure 2Cumulative respiration over the whole of each experiment of (a) total sedimentary organic matter, measured as Total Oxygen Uptake, and (b) of added, fresh organic matter, measured as 13C-DIC release. Error bars are ± 1 standard deviation. Letters indicate significantly different treatments as shown by ANOVA.
3.3 13C-DIC release
Fresh organic matter respiration (FOMR) rates were measured as the release of 13C -DIC and ranged between 0.04–1.85 mmol C m−2 d−1 in the 13C-AA experiment, and 0.01–4.38 mmol C m−2 d−1 in the 13C-MPB experiment (Fig. 3). Fresh organic matter respiration rates were substantially higher in the 13C-MPB experiment, but generally showed a similar time evolution in both experiments. Rates were always highest immediately after feeding, and declined rapidly thereafter, reaching constant levels after ∼ 5 d (Fig. 3). Differences between treatments were most apparent during the first 2 d after feeding. For the 13C-AA experiment, the re-stocked and fauna treatments showed slightly higher initial FOMR rates (Fig. 3a). For the 13C-MPB experiment, the defaunated treatment showed higher initial rates (Fig. 3b). Due to the marked change in rates over time, significant differences in rates between treatments were only apparent on individual days. Significant differences between treatments were present 5 d after feeding in the 13C-AA experiment (Kruskal-Wallis p=0.04), and 1, 2 and 7 d after feeding in the 13C-MPB experiment (Kruskall-Wallis, p=0.029, 0.038 and 0.034, respectively). However, pairwise Mann-Whitney U tests were not sufficiently powerful to show which pairs of treatments were significantly different on those days.
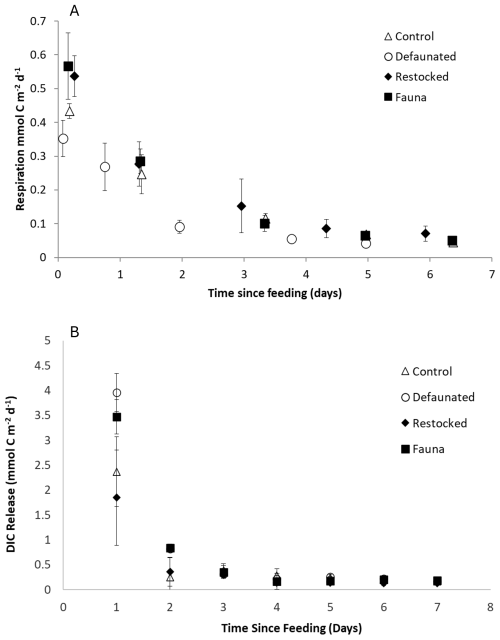
Figure 3Fresh Organic Matter Respiration calculated from 13C-DIC release for (a) the 13C-AA experiment, and (b) the 13C-MPB experiment.
The cumulative FOMR was higher in the 13C-MPB experiment by a factor ∼ 2–8 compared to the 13C-AA experiment for different treatments, and showed different patterns in the two experiments (Fig. 2b). In the 13C-MPB experiment cumulative FOMR was maximal in the defaunated and fauna only treatments (ANOVA, p=0.011, groupings shown in Fig. 2b). In the 13C-AA experiment there was no significant difference in cumulative FOMR between treatments (ANOVA, p=0.061).
3.4 Ratio of oxygen consumption versus 13C-DIC production
For each time point the TOU/FOMR ratio was calculated. The ratio ranged from ∼ 50–1100 for the 13C-AA experiment and from ∼ 19–958 for the 13C-MPB experiment (Fig. 4). There was a significant difference in TOU : FOMR ratios between the treatments in the two experiments (13C-AA experiment ANOVA, p=0.014; 13C-MPB experiment Kruskall-Wallis, p < 0.001). Post-hoc testing showed that for the 13C-AA experiment the restocked treatment had significantly higher ratios than the defaunated treatment, and that the other two treatments were not significantly different from any other. Further, the fauna treatment, although not being statistically significantly different, appeared most similar to the defaunated treatment (Fig. 4). Similarly, in the 13C-MPB experiment, all treatments were significantly different from each other (Mann-Whitney, p < 0.001–0.002), except for the defaunated and fauna treatments, which were not significantly different (Mann-Whitney, p=0.948).
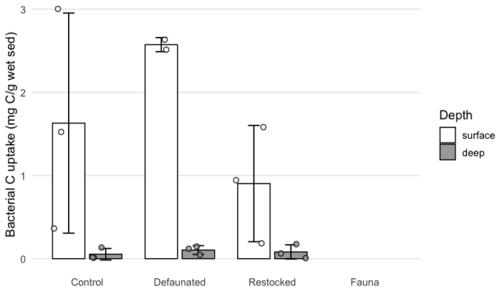
3.5 Bacterial carbon uptake
Uptake of 13C into bacterial biomass was quantified by PLFA analysis in the 13C-AA experiment (Fig. 5) and predominantly occurred in the surface sediment (0–1 cm), with uptake values 10-fold higher than the subsurface sediment (9–10 cm). Differences were notable between treatments: 13C uptake into bacterial biomass was not detectable in the fauna-only treatment, and ranged up to a maximum of 0.052 mg C g−1 of wet sediment in the defaunated treatment (Fig. 5). Bacterial 13C uptake appeared to be maximal in the defaunated treatment (Fig. 5), but due to high variability in the control treatment, the observed differences between the control, defaunated and restocked treatments were not statistically significant.
3.6 Faunal carbon uptake
Uptake of 13C into macrofaunal biomass was quantified in both the 13C-AA and 13C-MPB experiments and varied between the two experiments (Fig. 6). All taxa showed uptake of labelled fresh organic matter in both experiments, providing δ13C values up to 786 ‰ in the 13C-AA experiment and up to 286 ‰ in the 13C-MPB experiment (Fig. 6).
It should be noted that the C dose used in the two experiments varied (395 ± 11 and 2300 mg C m−2 for the 13C-AA and 13C-MPB experiments, respectively), and therefore direct comparison of faunal C uptake or labelling intensity between experiments is not possible. However, comparisons can be made regarding relative labelling levels of different taxa within each experiment, and these showed significant differences in labelling between taxa (Kruskal-Wallis, p< 0.001 for the 13C-AA experiment and ANOVA, p< 0.001 for the 13C-MPB experiment). Hydrobia ulvae and Hediste diversicolor showed the highest labelling in both experiments, consistent with their high motility and surface deposit feeding habits. In contrast, the sessile and deep-living taxa Arenicola marina, Cerastoderma edule and Macoma balthica showed a lower labelling intensity (Fig. 6). Data for Heteromastus filiformis illustrated how variable the feeding can be within a single macrofaunal taxon, with low labelling in the 13C-AA experiment, and high labelling in the 13C-MPB experiment. This may be due to a feeding preference by Heteromastus filiformis, or could be a result of differences between the experiments in terms of C dose or other site-specific factors.
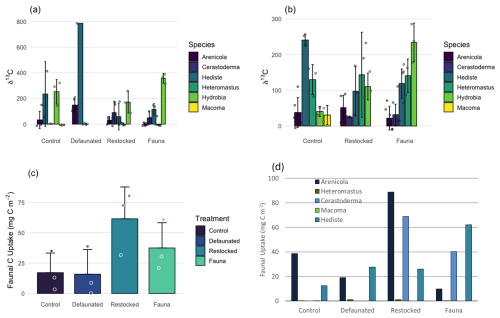
Figure 6Mean 13C isotopic signatures of macrofaunal taxa recovered from each treatment in (a) the 13C-AA experiment, and (b) the 13C-MPB experiment. The magnitude of faunal 13C uptake in the 13C-AA experiment as (c) total faunal uptake, and (d) by taxon. Data for Hydrobia were excluded from panels (c) and (d) due to uncertainties regarding biomass. Bars represent mean ± 1 standard deviation.
For the 13C-AA experiment, the wet weight of the macrofauna were measured, allowing quantification of total added C uptake by the macrofauna. Hydrobia ulvae was excluded from this calculation due to uncertainties in wet weight data. Macrofaunal C uptake ranged from 15.9 mg C m−2 in the defaunated treatment up to 61.5 mg C m−2 in the restocked treatment (Fig. 6c). Macrofaunal uptake was generally higher in the restocked treatment than in other treatments, however variability in faunal biomass meant the differences were not statistically significant (Kruskal-Wallis, p = 0.192). Further, when total C uptake data from the 13C-AA experiment were pooled by taxon, there was a significant difference in uptake accounted for by different taxa (Kruskal-Wallis, p=0.044), with Arenicola marina and Hediste diversicolor each showing significantly more C uptake than Heteromastus filiformis, and Macoma balthica (Mann-Whitney, p = 0.027–0.030, Fig. 6d).
3.7 The fate of added 13C
In the 13C-AA experiment sufficient 13C pools were quantified to allow a carbon budget to be calculated (with Hydrobia excluded from macrofaual uptake, as mentioned above). In the control and defaunated treatments a mean of 17.5 ± 5.5 % of the added 13C was recovered from biologically processed pools (fauna, bacterial biomass and respiration). The restocked treatment showed the highest percentage of biologically processed 13C (24.2 %), with particularly high uptake into macrofauna (Fig. 8). We presume that the 13C that could not be accounted for remained predominantly in the sediment, although a portion will have been converted to DOC. Data are not available to confirm this.
In this section we first discuss findings related to respiration and remineralisation of organic matter and the inverted microbial loop, before taking a broader look at the fate of added organic carbon in our experiments and conclusions that can be drawn about resource partitioning and functional redundancy. Finally we consider wider implications of the work.
4.1 Respiration
4.1.1 Total oxygen uptake and the inverted microbial loop
The “inverted microbial loop” hypothesis, originally proposed by Middelburg (2018), suggests that macrofaunal activity stimulates the microbial community by mixing freshly deposited, bioavailable organic carbon in deeper sediment horizons, thus increasing its availability to microbes for their respiratory metabolism. Therefore, the master response variable in the inverted microbial loop concept is the respiration of organic matter, measured in our experiments as TOU. Total oxygen uptake reflects the respiration of the total sedimentary organic carbon pool, which in our experiments included both the slow-decaying ambient organic matter, as well as the fast-decaying fresh organic detritus that was added and carried the 13C label.
Interactions between components of the benthic community are indicated by differences between the TOU rates measured in the restocked treatment (macrofauna plus microbes), and the sum of those in the defaunated (microbes only) and the fauna (macrofauna only) treatments. Our results indicate a positive interaction, as the sum of TOU in the defaunated and fauna only treatments tended to be less than the TOU of the restocked treatment. This was the case for all days except day 1 in the 13C-MPB experiment, and for the cumulative TOU in the 13C-AA experiment (Fig. 7). In summary, at the majority of timepoints, the co-presence of macrofauna alongside the microbial community enhanced the TOU, supporting the occurrence of the inverted microbial loop in respiration of total sedimentary organic matter.
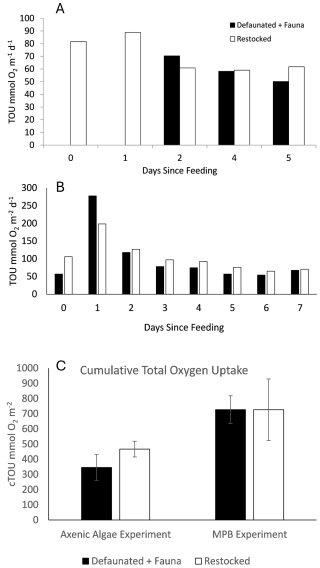
Figure 7Comparison between the sum of de-faunated and fauna rates and re-stocked TOU rates for the (a) 13C-AA and (b) 13C-MPB experiments, and (c) for cumulative TOU (cTOU).
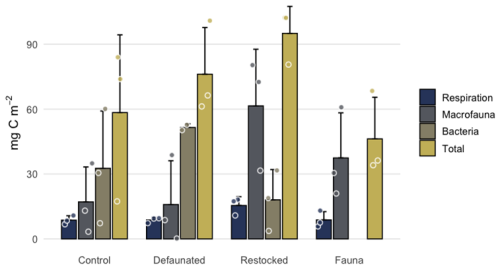
Figure 8The distribution of added 13C labelled OC between different biologically processed pools in the axenic algae experiment. Note that Hydrobia are not included in macrofaunal uptake due to biomass uncertainties.
Furthermore, the cumulative TOU (cTOU) during each experiment was maximal in the control and restocked treatments, where macrofaunal and microbial communities were present together (Fig. 2). In the 13C-AA experiment the cTOU in the restocked treatment was approximately 2-fold higher than that in the fauna and defaunated treatments, and for the 13C-MPB experiment both the restocked and control treatments were ∼ 2-fold higher than the fauna or defaunated treatments (Fig. 2). Thus the cTOU results also supported the occurrence of the inverted microbial loop stimulating total respiration.
The mechanisms behind the inverted microbial loop are relatively well documented. Macrofauna stimulate microbial activity by enhancing the supply of O2 via bioirrigation (Aller and Aller, 1998), as well as through the niche structuring and resource partitioning that result from redistribution of organic matter to deeper sediment layers resulting from particle biomixing, thus increasing the availability of organic matter to microbes (Schwinghamer et al., 1986; van Nugteren et al., 2009a). There is also likely to be a role for priming, whereby the microbial community is activated by addition of a small amount of relatively bioavailable organic C, allowing remineralisation of more of the ambient, less bioavailable organic C than would otherwise have occurred (Bianchi, 2011;van Nugteren et al., 2009b; Hannides and Aller, 2016). Further experiments designed to distinguish between these mechanisms would be informative.
4.1.2 Contrasting ambient and fresh organic C mineralisation
In our experiments TOU reflects remineralisation of all organic C present, including ambient sedimentary organic C, while FOMR reflects remineralisation of only the added, fresh organic C. Thus a comparison of TOU and FOMR rates can inform on the factors controlling remineralisation of different pools of organic C. Ratios of TOU/FOMR (19–1100, Fig. 4) were very high compared to the value of ∼ 1.3 for mineralisation of Redfield Ratio organic matter, and compared to the values of 0.8–2.0 reported by Alongi et al. (2011) in core incubation experiments. This indicates that the majority of the O2 consumption we observed was associated with remineralisation of pre-existing, ambient sedimentary organic C, rather than the 13C which was added as fresh algal detritus or MPB.
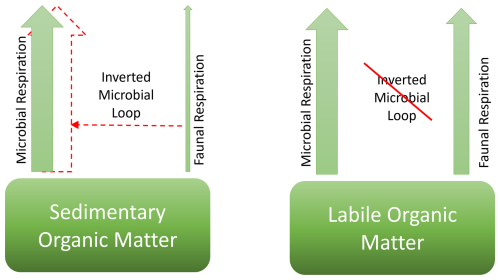
The TOU:FOMR ratios were higher in the restocked treatment than in the other treatments for both experiments (Fig. 4). This suggests that stimulation of ambient C remineralisation occurred by the inverted microbial loop when macrofauna and microbes are both present, but stimulation of mineralisation of fresh organic C did not occur to the same extent. As summarised in a conceptual model in Fig. 9, we suggest that there is a marked difference in operation of the inverted microbial loop (Middelburg, 2018) between remineralisation of different fractions of organic matter. We suggest that the inverted microbial loop works to stimulate the degradation of total sediment organic matter, but does not operate on the degradation of newly deposited, fresh organic matter. This is consistent with the concept of and mechanisms proposed for priming (which is closely related to the inverted microbial loop), whereby introduction of fresh organic matter stimulates microbial activity such that microbial remineralisation is enabled for more refractory ambient organic matter, such as through increased concentrations of extracellular enzymes (Bianchi, 2011). It is notable that the conceptual understanding that sedimentary organic matter consists of different “fractions” with different reactivities is well established, and is incorporated into organic carbon degradation models (Arndt, 2013). However, because those fractions are difficult or impossible to identify and separate analytically or physically it is relatively unusual to find direct experimental evidence that distinguishes how different fractions behave.
4.1.3 Partitioning respiration
Our experiments provide a rare empirical quantified partitioning of sediment respiration between different components of the sediment biological community. Fauna-only respiration (measured as TOU and FOMR) was similar to the respiration measured in the defaunated treatment, which represented only the microbial and meiofaunal communities (Figs. 1 and 3). This implies that, independently, these two compartments of the benthic community make approximately equal contributions to total sediment respiration. This contrasts with some previous studies which found that bacteria dominate sediment respiration (Hubas et al., 2006), production (Schwinghamer et al., 1986), and organic matter degradation (Lillebo et al., 1999). Herman et al. (1999) estimated that macrofauna contributed 15 %–20 % of SCOC. In contrast, on the macrofauna rich Goban Spur, Heip et al. (2001) calculated that macrofauna accounted for a greater proportion of community respiration than bacteria.
Thus our finding that microbial and faunal respiration are of comparable magnitude is relatively unusual but not unprecedented. In combination with the literature cited it suggests that the relative importance of faunal and bacterial respiration varies spatially. In some settings, such as shallower (i.e. coastal, shelf, and some continental margin) sediments where macrofaunal biomass tends to be high (Wei et al., 2010; Stratmann et al., 2019), faunal respiration can be quantitatively important. On the basis of previous isotope tracing experiment results Middelburg (2018) pointed out that fauna cannot be assumed to merely move organic particles around without contributing to carbon processing, and argued that direct C respiration by fauna should now be included in diagenetic models. Our finding on the partitioning of sediment respiration supports that suggestion.
4.2 Biological processing of fresh organic C
4.2.1 Competition and functional redundancy
In both the 13C-AA and 13C-MPB experiments the fauna and defaunated treatments showed similar FOMR rates, measured as production of 13C-DIC (Figs. 2b, 3). Rates in the restocked treatment were never as high as the sum of the rates when either only fauna or only microbes were present, despite the majority of added C remaining in the sediment.
This suggests that access to the fresh organic matter may have been the limiting factor on FOMR rates, with the macrofaunal and microbial plus meiofaunal components of the benthic community competing for the resource that they could reach (notably, the same amount of fresh 13C was added in each treatment, irrespective of the community biomass present). The fact that a considerable amount of the added C remained in the sediment also indicates that it consisted of different fractions with different bioavailability. In line with competition for the fresh organic C, the uptake of added 13C into bacterial biomass (Fig. 5) was greatest in the defaunated treatment, while in the control and re-stocked treatments bacterial uptake was supressed by competition with macrofauna. The suggestion of competition for fresh organic matter is consistent with previous studies which have found that the availability of organic matter exerts a control on benthic respiration rates (Provoost et al., 2013), and that, more generally, the functioning of intertidal ecosystems tends to be food limited (Edgar, 1993). Other studies have also suggested that in marine benthic communities, the macrofaunal and bacterial components may compete for detrital organic matter. In two deep sea settings, reduced bacterial production in the presence of macrofauna has been attributed to competition for organic matter and resource partitioning (Hunter et al., 2012, 2013). Macrofauna are more able than microbes to locate and exploit concentrated food deposits on the sediment surface (van Nugteren et al., 2009a). Macrofauna are also thought to interact with meiofauna regarding organic matter availability, although it is not clear whether this includes competition (Schwinghamer et al., 1983), or enhances its availability to meiofauna through redistribution (Braeckman et al., 2011). Overall, the competition for resources between organisms of different kingdoms is poorly studied in marine sediments, despite the suggestion that microbes versus eukaryotes may represent the most prevalent form of competition on Earth (Hochberg and Lawton, 1990).
The differences in FOMR rates between treatments may also be discussed in terms of functional redundancy within the benthic community, such that fresh organic matter is mineralised at approximately the same rate, irrespective of the identity and (to some extent) biomass of the organisms present. The “redundancy” hypothesis for ecosystem functioning (Walker, 1992) states that an ecosystem function will be delivered by the pool of species in an ecosystem, such that if one species is removed, the function will be taken over by other species. In the case of our experiments this redundancy could be related to a release from competition when some organisms are not present. Functional redundancy stands in contrast to the “rivet” hypothesis (Ehrlich and Ehrlich, 1981), in which every species in an ecosystem supplies a unique function, such that the removal of any one species leads to a loss of function. Evidence for redundancy within marine benthic communities has been found previously. For example, in a cockle removal study (Cesar and Frid, 2009), ecosystem function as measured by sediment surface chlorophyll a and organic matter concentrations remained unchanged, despite a shift in the biological traits of the macrofaunal community. Also, following defaunation of an intertidal site, the carbon flows from microphytobenthos and bacteria into macrofauna recovered months before the full macrofaunal diversity had re-established (Rossi et al., 2009). On an intertidal mudflat, manipulations of species richness were found not to impact any ecosystem functions apart from sediment oxygen consumption (Bolam et al., 2002). This latter effect was thought to be because one species, when present, appeared to have a disproportionately large role in sediment oxygen consumption, and so could be termed a keystone species. Clarke and Warwick (1998) analysed macrofaunal communities from two coastal sites and determined that they contained up to 4 sub-sets of species, each of which alone could deliver the same function as the whole community.
The studies detailed above consider functional redundancy only within macrofaunal communities, and functional redundancy has also been observed within microbial communities (Franklin and Mills, 2006). However, the redundancy suggested by our experiments is between macrofauna and microbes for fresh organic matter remineralisation. As with competition, redundancy between kingdoms is rarely considered. One macrofauna removal study found that defaunated patches showed reduced ammonium flux and reduced gross primary production (Lohrer et al., 2010), indicating lack of functional redundancy between kingdoms. Other studies which consider the recovery of whole benthic community function after disturbance have found that microbial communities recover very rapidly (over 1–2 d, reliant only on redox conditions being re-established), limiting the time available to study their role in functional redundancy during ecosystem recovery (Rossi et al., 2009; Larson and Sundback, 2012). It seems likely that redundancy between microbial and macrofaunal communities, as observed here for fresh organic matter respiration, will operate for some functions to a greater extent than for others. This warrants further study, as it will support predictions of how overall ecosystem functions could change in the future under various anthropogenic pressures.
4.2.2 Utilisation of fresh and ambient organic matter
Comparison of 13C respiration and uptake into biomass in the 13C-AA experiment suggests different biological use of and pathways followed by fresh and total sedimentary organic matter. It is notable that the uptake of 13C into both macrofaunal and bacterial biomass were always higher than 13C respiration (Fig. 8). This observation has not been made in previous isotope tracing experiments (Woulds et al., 2009, 2016), which have usually shown respiration to be the dominant fate of biologically processed 13C, even in relatively shallow, near shore and high faunal biomass settings. The observation of assimilation of 13C exceeding respiration of 13C suggests that carbon from fresh detritus may be more likely to be incorporated into biomass, while older ambient organic C tends to be routed to respiration.
This study provides experimental evidence for the importance of interactions between different fractions of benthic communities and the importance of macrofaunal activities in sedimentary carbon cycling. Our specific findings are that:
-
The inverted microbial loop, in which macrofaunal processes stimulate microbial activity including through priming, was demonstrated to influence the remineralisation of total sediment organic matter (including less reactive organic matter), as revealed by O2 consumption rates.
-
Macrofauna and the microbial community appeared to compete for the added, fresh organic matter, and this was a limiting resource when both communities were present together.
Partitioning of total respiration between fractions of the benthic community showed that the direct contribution by macrofauna can be of a similar magnitude than that of the microbial community
The role of macrofauna elucidated here will need to be considered in decision making about use of shelf seafloors, which are disturbed by activities including cable trenching, wind farm installation and trawling (Sala et al., 2021; Heinatz and Scheffold, 2023), and which could increasingly be managed through marine protected areas. Decision making and management of all such activities will need to consider likely changes in benthic faunal communities, and knock on effects on the fate of organic carbon in the sediment. More explicit consideration of impacts on and changes in faunal processes would be facilitated by their inclusion in Earth System models to a greater extent than is currently the case.
The results presented here also enhance our understanding of the processes involved in respiration and release of organic carbon from the seafloor. This is timely, given the growing awareness amongst policy makers that so called “blue carbon” stores in sub-tidal marine sediments are substantial (Kröger et al., 2018). Potential trade offs must be considered in management decision making in order for blue carbon stores to be maintained and potentially enhanced, to maximise their contribution to climate change mitigation and moves towards net zero.
Finally, while this study focuses on estuarine sediment, and so is most applicable to shallow sub-tidal and shelf settings, it shows that faunal-microbial interactions require further investigation in a wide range of benthic environments from the intertidal to the deep sea.
Data available through University of Leeds Data Repository (https://doi.org/10.5518/1729, Woulds et al., 2026).
All authors contributed to the experimental design. The experiments were performed by CW and SHM. The manuscript was written by CW and DvO, with contributions from other co-authors.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
The authors would like to thank Pieter Van Rijswijk and Bert Sinke for their assistance in the field and laboratory, and two reviewers for comments which helped to improve the manuscript. The experiments were conducted at the Royal Netherlands Institute for Sea Research (NIOZ) in Yerseke.
This research has been supported by the Royal Society and the Nederlandse Organisatie voor Wetenschappelijk Onderzoek (grant no. 864.08.004).
This paper was edited by Mark Lever and reviewed by William Ross Hunter and one anonymous referee.
Aller, R. C. and Aller, J. Y.: The effect of biogenic irrigation intensity and solute exchange on diagenetic reaction rates in marine sediments, Journal of Marine Research, 56, 905–936, 1998.
Alongi, D. M., Trott, L. A., and Mohl, M.: Strong tidal currents and labile organic matter stimulate benthic decomposition and carbonate fluxes on the southern Great Barrier Reef shelf, Continental Shelf Research, 31, 1384–1395, https://doi.org/10.1016/j.csr.2011.05.018, 2011.
Aminot, A. and Rey, F.: Chlorophyll a: Determination by spectroscopic methods, ICES Techniques in Marine Environmental Sciences, 30, 17 pp., https://doi.org/10.25607/OBP-278, 2001.
Arndt, S., Jorgensen, B., LaRowe, D., Middelburg, J., Pancost, R., and Regnier, P.: Quantifying the degradation of organic matter in marine sediments: A review and synthesis, Earth-Sci. Rev., 123, 53–86, https://doi.org/10.1016/j.earscirev.2013.02.008, 2013.
Bengtsson, M. M., Attermeyer, K., and Catalan, N.: Interactive effects on organic matter processing from soils to the ocean: are priming effects relevant in aquatic ecosystems?, Hydrobiologia, 822, 1–17, https://doi.org/10.1007/s10750-018-3672-2, 2018.
Berner, R.: Burial of organic-carbon and pyrite sulfur in the modern ocean - its geochemical and environmental significance, American Journal of Science, 282, 451–473, https://doi.org/10.2475/ajs.282.4.451, 1982.
Bianchi, T. S.: The role of terrestrially derived organic carbon in the coastal ocean: A changing paradigm and the priming effect, Proceedings of the National Academy of Sciences of the United States of America, 108, 19473–19481, https://doi.org/10.1073/pnas.1017982108, 2011.
Bolam, S. G., Fernandes, T. F., and Huxham, M.: Diversity, biomass, and ecosystem processes in the marine benthos, Ecological Monographs, 72, 599–615, https://doi.org/10.2307/3100059, 2002.
Boschker, H. T. S. and Middelburg, J. J.: Stable isotopes and biomarkers in microbial ecology, FEMS Microbiology Ecology, 40, 85–95, 2002.
Braeckman, U., Provoost, P., Moens, T., Soetaert, K., Middelburg, J. J., Vincx, M., and Vanaverbeke, J.: Biological vs. Physical Mixing Effects on Benthic Food Web Dynamics, Plos One, 6, https://doi.org/10.1371/journal.pone.0018078, 2011.
Cesar, C. P. and Frid, C. L. J.: Effects of experimental small-scale cockle (Cerastoderma edule L.) fishing on ecosystem function, Marine Ecology-an Evolutionary Perspective, 30, 123–137, https://doi.org/10.1111/j.1439-0485.2009.00305.x, 2009.
Clarke, K. R. and Warwick, R. M.: Quantifying structural redundancy in ecological communities, Oecologia, 113, 278–289, https://doi.org/10.1007/s004420050379, 1998.
Daggers, T. D., van Oevelen, D., Herman, P. M. J., Boschker, H. T. S., van der Wal, D.: Spatial variability in macrofaunal diet composition and grazing pressure on microphytobenthos in intertidal areas, Limnology and Oceanography, 65, 2819–2834, https://doi.org/10.1002/lno.11554, 2020.
Edgar, G. J.: Measurement of the carrying-capacity of benthic habitats using a metabolic-rate based index, Oecologia, 95, 115–121, https://doi.org/10.1007/bf00649514, 1993.
Ehrenhauss, S. and Huettel, M.: Advective transport and decomposition of chain-forming planktonic diatoms in permeable sediments, Journal of Sea Research, 52, 179–197, https://doi.org/10.1016/j.seares.2004.01.004, 2004.
Ehrlich, P. R. and Ehrlich, A. H.: Extinction: the causes and consequenses of the disappearance of species, Random House, New York, ISBN 978-0394513126, 1981.
Franco, M. d. A., Vanaverbeke, J., Van Oevelen, D., Soetaert, K., Costa, M. J., Vincx, M., and Moens, T.: Respiration partitioning in contrasting subtidal sediments: seasonality and response to a spring phytoplankton deposition, Marine Ecology-an Evolutionary Perspective, 31, 276–290, https://doi.org/10.1111/j.1439-0485.2009.00319.x, 2010.
Franklin, R. B. and Mills, A. L.: Structural and functional responses of a sewage microbial community to dilution-induced reductions in diversity, Microbial Ecology, 52, 280–288, https://doi.org/10.1007/s00248-006-9033-0, 2006.
Glud, R., Gundersen, J., Roy, H., and Jorgensen, B.: Seasonal dynamics of benthic O2 uptake in a semienclosed bay:: Importance of diffusion and faunal activity, Limnology and Oceanography, 48, 1265–1276, https://doi.org/10.4319/lo.2003.48.3.1265, 2003.
Gontikaki, E., Thornton, B., Cornulier, T., and Witte, U.: Occurrence of Priming in the Degradation of Lignocellulose in Marine Sediments, Plos One, 10, https://doi.org/10.1371/journal.pone.0143917, 2015.
Guilini, K., Van Oevelen, D., Soetaert, K., Middelburg, J. J., and Vanreusel, A.: Nutritional importance of benthic bacteria for deep-sea nematodes from the Arctic ice margin: Results of an isotope tracer experiment, Limnology and Oceanography, 55, 1977–1989, https://doi.org/10.4319/lo.2010.55.5.1977, 2010.
Hannides, A. K. and Aller, R. C.: Priming effect of benthic gastropod mucus on sedimentary organic matter remineralization, Limnology and Oceanography, 61, 1640–1650, https://doi.org/10.1002/lno.10325, 2016.
Heinatz, K. and Scheffold, M.: A first estimate of the effect of offshore wind farms on sedimentary organic carbon stocks in the Southern North Sea, Front. Mar. Sci., 9, https://doi.org/10.3389/fmars.2022.1068967, 2023.
Heip, C. H. R., Duineveld, G., Flach, E., Graf, G., Helder, W., Herman, P. M. J., Lavaleye, M., Middelburg, J. J., Pfannkuche, O., Soetaert, K., Soltwedel, T., de Stigter, H., Thomsen, L., Vanaverbeke, J., and de Wilde, P.: The role of the benthic biota in sedimentary metabolism and sediment-water exchange processes in the Goban Spur area (NE Atlantic), Deep-Sea Res. Pt. II, 48, 3223–3243, 2001.
Herman, P. M. J., Middelburg, J. J., Van de Koppel, J., and Heip, C. H. R.: Ecology of estuarine macrobenthos, in: Advances in Ecological Research, Vol 29, Estuaries, edited by: Nedwell, D. B. and Raffaelli, D. G., Advances in Ecological Research, 195–240, https://doi.org/10.1016/S0065-2504(08)60194-4, 1999.
Hochberg, M. E. and Lawton, J. H.: Competition between kingdoms, Trend. Ecol. Evol., 5, 367–371, https://doi.org/10.1016/0169-5347(90)90097-w, 1990.
Hubas, C., Artigas, L. F., and Davoult, D.: Role of the bacterial community in the annual benthic metabolism of two contrasted temperate intertidal sites (Roscoff Aber Bay, France), Marine Ecology Progress Series, 344, 39–48, https://doi.org/10.3354/meps06947, 2007.
Hubas, C., Davoult, D., Cariou, T., and Artigas, L. F.: Factors controlling benthic metabolism during low tide along a granulometric gradient in an intertidal bay (Roscoff Aber Bay, France), Marine Ecology Progress Series, 316, 53–68, https://doi.org/10.3354/meps316053, 2006.
Huettel, M., Roy, H., Precht, E., and Ehrenhauss, S.: Hydrodynamical impact on biogeochemical processes in aquatic sediments, Hydrobiologia, 494, 231–236, https://doi.org/10.1023/a:1025426601773, 2003.
Hunter, W. R., Jamieson, A., Huvenne, V. A. I., and Witte, U.: Sediment community responses to marine vs. terrigenous organic matter in a submarine canyon, Biogeosciences, 10, 67–80, https://doi.org/10.5194/bg-10-67-2013, 2013.
Hunter, W. R., Veuger, B., and Witte, U.: Macrofauna regulate heterotrophic bacterial carbon and nitrogen incorporation in low-oxygen sediments, Isme Journal, 6, 2140–2151, https://doi.org/10.1038/ismej.2012.44, 2012.
Kemp, P. F.: Bacterivory by benthic ciliates – significance as a carbon source and impact on sediment bacteria, Marine Ecology Progress Series, 49, 163–169, https://doi.org/10.3354/meps049163, 1988.
Kemp, P. F.: The fate of benthic bacterial production, Reviews in Aquatic Sciences, 2, 109–124, 1990.
Kristensen, E.: organic matter diagenesis at the oxic/anoxic interface in coastal marine sediments with emphasis on the role of burrowing animals, Hydrobiologia, 426, 1–24, 2000.
Kröger, S., Parker, R., Cripps, G., and Williamson, P. (Eds.): Shelf Seas: The Engine of Productivity, Policy Report on NERC-Defra Shelf Sea Biogeochemistry programme, Cefas, Lowestoft, 2018.
Larson, F. and Sundback, K.: Recovery of microphytobenthos and benthic functions after sediment deposition, Marine Ecology Progress Series, 446, 31–44, https://doi.org/10.3354/meps09488, 2012.
Lillebo, A. I., Flindt, M. R., Pardal, M. A., and Marques, J. C.: The effect of macrofauna, meiofauna add microfauna on the degradation of Spartina maritima detritus from a salt marsh area, Acta Oecologica-International Journal of Ecology, 20, 249–258, https://doi.org/10.1016/s1146-609x(99)00141-1, 1999.
Lohrer, A. M., Halliday, N. J., Thrush, S. F., Hewitt, J. E., and Rodil, I. F.: Ecosystem functioning in a disturbance-recovery context: Contribution of macrofauna to primary production and nutrient release on intertidal sandflats, Journal of Experimental Marine Biology and Ecology, 390, 6–13, https://doi.org/10.1016/j.jembe.2010.04.035, 2010.
Meysman, F., Middelburg, J., and Heip, C.: Bioturbation: a fresh look at Darwin's last idea, Trends In Ecology & Evolution, 21, 688–695, https://doi.org/10.1016/j.tree.2006.08.002, 2006.
Middelburg, J. J.: Reviews and syntheses: to the bottom of carbon processing at the seafloor, Biogeosciences, 15, 413–427, https://doi.org/10.5194/bg-15-413-2018, 2018.
Middelburg, J. and Meysman, F.: Ocean science – Burial at sea, Science, 316, 1294–1295, https://doi.org/10.1126/science.1144001, 2007.
Middelburg, J. and Duarte, C., Respiration in coastal benthic communities. 10.1093/acprof:oso/9780198527084.003.0011, 2005.
Middelburg, J. J., Barranguet, C., Boschker, H. T. S., Herman, P. M. J., Moens, T., and Heip, C. H. R.: The fate of intertidal microphytobenthos carbon: An in situ C-13-labeling study, Limnology and Oceanography, 45, 1224–1234, https://doi.org/10.4319/lo.2000.45.6.1224, 2000.
Moodley, L., Boschker, H. T. S., Middelburg, J. J., Pel, R., Herman, P. M. J., de Deckere, E., and Heip, C. H. R.: Ecological significance of benthic foraminifera: 13C labelling experiments, Marine Ecology Progress Series, 202, 289–295, 2000.
Provoost, P., Braeckman, U., Van Gansbeke, D., Moodley, L., Soetaert, K., Middelburg, J. J., and Vanaverbeke, J.: Modelling benthic oxygen consumption and benthic-pelagic coupling at a shallow station in the southern North Sea, Estuarine Coastal and Shelf Science, 120, 1–11, https://doi.org/10.1016/j.ecss.2013.01.008, 2013.
Rao, A. M. F, Malkin, S. Y., Montserrat F., and Meysman, F. J. R.: Alkalinity production in intertidal sands intensified by lugworm bioirrigation, Estuarine Coastal and Shelf Science, 148, 36–47, 2014.
Rossi, F., Vos, M., and Middelburg, J. J.: Species identity, diversity and microbial carbon flow in reassembling macrobenthic communities, Oikos, 118, 503–512, https://doi.org/10.1111/j.1600-0706.2009.17112.x, 2009.
Sala, E., Mayorga, J., Bradley, D., Cabral, R., Atwood, T., Auber, A., Cheung, W., Costello, C., Ferretti, F., Friedlander, A., Gaines, S., Garilao, C., Goodell, W., Halpern, B., Hinson, A., Kaschner, K., Kesner-Reyes, K., Leprieur, F., McGowan, J., Morgan, L., Mouillot, D., Palacios-Abrantes, J., Possingham, H., Rechberger, K., Worm, B., and Lubchenco, J.: Protecting the global ocean for biodiversity, food and climate, Nature, 592, https://doi.org/10.1038/s41586-021-03371-z, 2021.
Schwinghamer, P., Hargrave, B., Peer, D., and Hawkins, C. M.: Partitioning of production and respiration among size groups of organisms in an intertidal benthic community, Marine Ecology Progress Series, 31, 131–142, https://doi.org/10.3354/meps031131, 1986.
Stephens, M. P., Kadko, D. C., Smith, C. R., and Latasa, M.: Chlorophyll a and pheopigments as tracers of labile organic carbon at the central equatorial Pacific seafloor, Geochimica et Cosmochimica Acta, 61, 4605–4619, https://doi.org/10.1016/S0016-7037(97)00358-X, 1997.
Stratmann, T., Soetaert, K., Wei, C. L., Lin, Y. S., and van Oevelen, D.: The SCOC database, a large, open, and global database with sediment community oxygen consumption rates, Scientific Data, 6, https://doi.org/10.1038/s41597-019-0259-3, 2019.
Sweetman, A. and Witte, U.: Macrofaunal response to phytodetritus in a bathyal Norwegian fjord, Deep-Sea Res. Pt. I, 55, 1503–1514, https://doi.org/10.1016/j.dsr.2008.06.004, 2008.
van de Velde, S., Mills, B. J. W., Meysman, F. J. R., Lenton, T. M., and Poulton, S. W.: Early Palaeozoic ocean anoxia and global warming driven by the evolution of shallow burrowing, Nature Communications, 9, https://doi.org/10.1038/s41467-018-04973-4, 2018.
van Nugteren, P.,Herman, P. M. J., Moodley, L., Middelburg, J. J., Vos, M., and Heip, C. H. R.: Spatial distribution of detrital resources determines the outcome of competition between bacteria and a facultative detritivorous worm, Limnology and Oceanography, 54, 1413–1419, 2009a.
van Nugteren, P., Moodley, L., Brummer, G. J., Heip, C. H. R., Herman, P. M. J., and Middelburg, J. J.: Seafloor ecosystem functioning: the importance of organic matter priming, Marine Biology, 156, 2277–2287, https://doi.org/10.1007/s00227-009-1255-5, 2009b.
van Oevelen, D., Soetaert, K., Middelburg, J. J., Herman, P. M. J., Moodley, L., Hamels, I., Moens, T., and Heip, C. H. R.: Carbon flows through a benthic food web: Integrating biomass, isotope and tracer data, Journal of Marine Research, 64, 453-482, 10.1357/002224006778189581, 2006a.
van Oevelen, D., Moodley, L., Soetaert, K., and Middelburg, J. J.: The trophic significance of bacterial carbon in a marine intertidal sediment: Results of an in situ stable isotope labeling study, Limnology and Oceanography, 51, 2349–2359, 2006b.
Vasquez-Cardenas, D., Meysman, F. J. R., and Boschker, H. T. S.: A Cross – System Comparison of Dark Carbon Fixation in Coastal Sediments, Global Biogeochemical Cycles, 34, https://doi.org/10.1029/2019gb006298, 2020.
Walker, B. H.: Biodiversity and ecological redundancy, Conservation Biology, 6, 18–23, https://doi.org/10.1046/j.1523-1739.1992.610018.x, 1992.
Wei, C. L., Rowe, G. T., Escobar-Briones, E., Boetius, A., Soltwedel, T., Caley, M. J., Soliman, Y., Huettmann, F., Qu, F. Y., Yu, Z. S., Pitcher, C. R., Haedrich, R. L., Wicksten, M. K., Rex, M. A., Baguley, J. G., Sharma, J., Danovaro, R., MacDonald, I. R., Nunnally, C. C., Deming, J. W., Montagna, P., Levesque, M., Weslawski, J. M., Wlodarska-Kowalczuk, M., Ingole, B. S., Bett, B. J., Billett, D. S. M., Yool, A., Bluhm, B. A., Iken, K., and Narayanaswamy, B. E.: Global Patterns and Predictions of Seafloor Biomass Using Random Forests, Plos One, 5, https://doi.org/10.1371/journal.pone.0015323, 2010.
Woulds, C., Andersson, J. H., Cowie, G. L., Middelburg, J. J., and Levin, L. A.: The short-term fate of organic carbon in marine sediments: Comparing the Pakistan margin to other regions, Deep-Sea Res. Pt. II, 56, 393–402, 2009.
Woulds, C., Bouillon, S., Cowie, G., Drake, E., Middelburg, J. J., and Witte, U.: Patterns of carbon processing at the seafloor: the role of faunal and microbial communities in moderating carbon flows, Biogeosciences, 13, 1–15, https://doi.org/10.5194/bg-13-1-2016, 2016.
Woulds, C., van Oevelen, D., Hidalgo-Martinez, S., and Meysman, F.: The Inverted Microbial Loop Stimulates Mineralisation of Sedimentary Organic Detritus, University of Leeds [data set], https://doi.org/10.5518/1729, 2026.