the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Nitrous oxide emissions from pigeon pea–maize rotation in response to conservation agriculture and biochar amendments in a Ferralsol, northern Uganda
Talent Namatsheve
Vegard Martinsen
Jan Mulder
Alfred Obia
Peter Dörsch
Smallholder agriculture in sub-Saharan Africa (SSA) commonly involves limited use of mineral or organic fertilizer, often resulting in severe nutrient limitation. Conservation Agriculture (CA), including crop rotation with legumes and biochar amendments, has been advocated to enhance soil fertility and plant available nitrogen (N). However, CA may affect nitrous oxide (N2O) emissions even in unfertilized agroecosystems. N2O is an important greenhouse (GHG) gas, and understanding the trade-offs between N2O emissions and crop yields in N-poor agroecosystems in SSA is essential. Here we studied crop yield, soil N dynamics and N2O emissions in a double cropping system (pigeon pea–maize rotation) throughout two consecutive cropping seasons (April–October 2023 and October 2023–January 2024) in a Ferralsol in Northern Uganda. The study, conducted at a site which had been left fallow for 3 years, involved pairwise comparison of conventionally tilled systems under crop rotation (Conventional) and continuous maize monocropping (ConventMM). In addition, the effect of tillage systems (Conventional, CA and CA + biochar) under pigeon pea–maize rotation was investigated. We defined CA as reduced tillage with planting basins, crop rotation and residue retention, whereas conventional tillage involved overall ploughing. N2O fluxes were small, ranging from 1.02–51.19 µg N m2 h−1 over the entire period. Short-lived emission peaks were observed following pigeon pea harvest in the crop rotation, which were absent in maize monocropping. Across two growing seasons, area-weighted cumulative N2O emissions for 279 d ranged from 0.46 kg kg N ha−1 in CA + BC treatment to 0.88 kg N ha−1 in the Conventional treatment, respectively. CA + BC reduced area-weighted N2O emissions by 33 % and 66 % compared to Conventional treatment in the first and second season, respectively. In addition, biochar amendments in CA systems also reduced yield-scaled N2O emissions by 48 % across two seasons. In the first season, yield-scaled N2O emissions and N yield scaled N2O emissions were significantly smaller in CA systems with biochar compared to conventional tillage, suggesting that CA and biochar was effective in minimising emissions without reducing pigeon pea yield, in the first year after field clearing.
- Article
(914 KB) - Full-text XML
-
Supplement
(806 KB) - BibTeX
- EndNote
Nitrous oxide (N2O) is a long-lived greenhouse gas (GHG) with a lifetime of 116 years, and a global warming potential approximately 300 times greater than that of carbon dioxide (CO2) (Tian et al., 2020). It is the largest driver of stratospheric ozone depletion (Portmann et al., 2012). Globally, agriculture is a major source of atmospheric N2O, contributing approximately 60 % to global anthropogenic N2O emissions (Adegbeye et al., 2020; Kim et al., 2016). In sub-Saharan Africa (SSA), N2O emissions are mainly associated with forest clearing, livestock manure, and crop production (Boateng et al., 2019; Hickman et al., 2011). Although acidic soils tend to have high N2O emissions (Wang et al., 2018), the limited N inputs in smallholder farming systems in SSA reduce soil N availability, leading to low N2O emissions. Only ∼3 % of globally applied inorganic fertilizer, a key driver for soil N2O emissions, is used in Africa (Hickman et al., 2011). The recent 2024 Nairobi Declaration, targeting increased fertilizer use in Africa (Africa Union, 2024), might change future trajectories of fertilizer consumption in SSA, potentially increasing soil N availability and N2O emissions over time. Plant available N can also be derived from introducing legumes in crop rotations (Jensen et al., 2012), or applying organic fertilizers such as farmyard manure. These strategies are central to conservation agriculture (CA) in subsistence farming systems, but little is known about how they affect baseline N2O emissions. Also, climate smart practices such as biochar amendments, have been proposed to enhance crop yield and soil fertility (Namatsheve et al., 2024; Schmidt et al., 2021) and to reduce N2O emissions (Zhang et al., 2021).
Nitrous oxide is an intermediate or by-product in soil N transformations, that include nitrification, denitrification and nitrifier denitrification (Meier et al., 2020). The biogeochemistry of N2O in soil is to a large extent regulated by complex interactions between environmental and biological factors such as temperature, water, carbon and nitrogen availability, oxygen levels and acidity (Case et al., 2015; Tian et al., 2020). Nitrification occurs under predominately aerobic soil conditions, whereby autotrophic bacteria or archaea oxidize to NO, which is further oxidized to nitrite () by nitrate-oxidizing bacteria (Dick et al., 2008; Fungo et al., 2019). In upland soils denitrification occurs during anaerobic spells, or in anaerobic soil aggregates, and is an anoxic respiratory process mediated by bacteria and some fungi, reducing stepwise to N2 via the intermediates NO, NO and N2O (Saggar et al., 2013; Scheer et al., 2020). Nitrifier denitrification occurs when nitrifying bacteria reduce NO under hypoxic conditions, analogously to the denitrification pathway (Wrage-Mönnig et al., 2018).
In a quest to improve crop production and soil fertility, sustainable agricultural practices such as conservation agriculture (CA) and biochar amendment have been promoted in SSA (Namatsheve et al., 2024). CA is based on three core principles which are reduced tillage, crop diversification and maintenance of a soil cover (Giller et al., 2015; Hobbs et al., 2008). Minimum soil disturbance enhances water retention and soil organic matter (Pittelkow et al., 2015; Powlson et al., 2011). Crop residue retention and incorporating legumes into cereal dominated farming systems can improve soil N availability (Fang et al., 2007; Turmel et al., 2015; Namatsheve et al., 2021). Although CA may stimulate N2O emissions (Abalos et al., 2022; Guenet et al., 2021; Shakoor et al., 2021), their mitigation in low-input crop production systems ultimately depends on synchronizing the release of mineral N from legumes and crop residues with the N uptake by crops.
Biochar, a carbon rich material produced by pyrolysis of organic waste has been claimed to enhance yields in impoverished tropical soils (Cornelissen et al., 2016; Lehmann, 2007). Interest in biochar emerges from pioneering research on Brazil's Terra Preta soils, which were enriched over time by repeated application of charcoal and organic matter, resulting in soils with a stable organic matter pool (Glaser et al., 2001). Biochar tends to increase soil pH which favours N2 over N2O production in denitrification (Wang et al., 2018). Although biochar contributes to the retention of exchangeable plant-available , it may also immobilize soil N (Nguyen et al., 2017), thereby reducing N availability and N2O emission (Jeffery et al., 2015). However, Weldon et al. (2022) reported that the sorption capacity of biochar for is low and variable, rendering N2O mitigation by biochar inherently uncertain. Effect of biochar on N cycling and N2O emissions in unfertilised tropical soils remains unclear (Namatsheve, 2025).
Earlier studies reported increased N2O emissions in SSA under CA (Baggs et al., 2006; Raji and Dörsch, 2020), while biochar amendments have been shown to reduce N2O emissions (Fungo et al., 2017, 2019; Namoi et al., 2019). However, these studies were carried out in systems that received inorganic N fertilizers, which do not represent the realities of unfertilized smallholder tropical agroecosystems typical of Uganda and other countries in SSA. In our recent meta-analysis of CA and biochar effects on N cycling, we found that residue retention increased soil , leading to higher N2O emissions; where at least 23 kg N ha−1 was applied (Namatsheve et al., 2024). We also found that integrating biochar into CA systems enhances biological N2-fixation of pigeon pea in unfertilized systems in Uganda (Namatsheve et al., 2025). This raises the question how the additional N from biological N2-fixation affects soil and N2O emissions in systems with tight N cycling. As far as we know, there are no published studies that examine the synergy of CA and biochar on N2O emissions in unfertilized tropical agroecosystems.
In this study we investigated the effect of CA on grain yield, N2O emissions, mineral N dynamics, and yield-scaled N2O emissions in an unfertilized Ferralsol in northern Uganda over two consecutive cropping seasons. Specifically, we compared crop rotation (pigeon pea–maize) with maize monocropping under conventional tillage (ConventMM). We also compared pigeon pea–maize rotation under three practices: conventional tillage, CA (reduced tillage), and CA in combination with biochar (CA + BC). We hypothesised that rotation with pigeon pea increases N2O emission compared to maize monocropping, while CA + BC reduces N2O emissions, both compared to CA and to conventionally tilled soil.
2.1 Site description
A field experiment was carried out in Gulu, Northern Uganda (2°47′46′′ N, 32°20′45′′ E). Uganda has a bimodal rainfall pattern and eight distinct agro-ecological regions, where Gulu lies in the Northern savannah grasslands (Mubiru et al., 2012). Soils in Gulu are acric Ferralsols, and the texture is a loamy sand (Wortmann and Eledu, 1999). Average soil organic carbon (SOC) and total N are 1.52 % and 0.11 % , respectively, while average soil pH is 6.7 (Namatsheve et al., 2025). The research site has a double cropping system i.e., one during the first season from April to August (long rain season) and a second rain season from August to December (short rain season), and a dry period is from December to February. In this experiment, first season stretched to October because pigeon pea was harvested 6 months after planting, and second season began in October, just after harvesting pigeon pea. Mean annual rainfall in the period 1981–2010 in Gulu was 1460 mm yr−1 (Oriangi et al., 2024). The annual rainfall in 2023 was 1238 mm, of which 818 mm precipitated in the first and 419 mm in the second season, respectively. Average temperature for 2023 was 24 °C. The weather data were obtained from the Gulu weather station which is about 6 km from the experimental site.
2.2 Experimental design, crop establishment and management
The experiment was established on a field that was fallowed for three years; before that it was used for maize and cassava production without fertilization, for at least five years. Prior to establishing the experiment, on 15 March 2023, a dense, naturally grown vegetation of grasses and shrubs was removed by slashing and chemical weeding, using glyphosate [N-(phosphonomethyl) glycine]. On 27 March 2023, plots under conventional management were prepared by overall digging using hand hoeing (100 % tillage) and plots of the same size under CA by manually digging 10 L planting basins (35 cm long × 15 cm wide × 20 cm deep) spaced 70 cm × 35 cm (interrow × within row spacing). The plot size was 6×5 m, and there were 63 basins in each plot, resulting in 21 000 planting basins ha−1. Given the basin dimensions about 10 %–12 % of the land under CA was tilled. The experiment had four treatments, replicated four times, and randomised in a complete block design (RCBD). The treatments were ConventMM, Conventional, CA and CA + BC (Table 1). Biochar was homogeneously mixed into the basins of the CA + BC plots when preparing the planting basins before sowing. In the treatments with crop rotation, pigeon pea was sown in the 1st season, and maize in the 2nd season. Maize monocropping had maize in both seasons. Dates of sowing and harvesting are indicated in Fig. 1a.
A pigeon pea variety SEPI 1 (bred at ICRISAT, Malawi and released by the National Agricultural Research Organisation, Uganda) was sown uniformly in Conventional, CA and CA + BC treatments. SEPI 1 is a medium maturity variety, with 77–87 d to flowering and 105–139 d to 75 % maturity. It is an indeterminate variety with semi-branching growth, the main stem continuing to elongate indefinitely; potential grain yields range from 1.8–3.4 Mg ha−1. The maize variety, Longe 10H, which is a hybrid with 100–120 d to maturity and a yield potential of 7–9 Mg ha−1 was sown in the ConventMM treatment (1st season) and in all treatments in the 2nd season. During sowing, three seeds were planted for both maize and pigeon pea at each planting station spaced 10 cm from each other giving a total planting population of 63 000 plants ha−1 in all treatments. To mimic subsistence farming systems in Uganda, no inorganic fertilizer was applied. For CA, weeds were controlled by spraying glyphosate at a rate of 1.03 L ha−1, immediately after sowing and hand pulling throughout the season. Weed control in the conventional treatment was done by hand hoeing at planting and throughout the season.
2.3 Biochar production and application
Biochar was prepared from pigeon pea stems and twigs using the flame curtain “Kon Tiki” kiln (Cornelissen et al., 2016; Munera-Echeverri et al., 2020). The kiln consists of a conically shaped pit with a depth of 1 m and a diameter of 3 m. The pyrolysis temperature was 600 °C. After weighing, the pigeon pea feedstock was pyrolyzed, quenched with water, covered with banana leaves and soil, and recovered after 3 d. The biochar was weighed (dry matter), ground and packed. The feedstock to biochar conversion ratio was 4:1, and the biochar had a pH of 9.74, carbon (C) concentration of 51 %, nitrogen (N) concentration of 0.76 %, cation exchange capacity (CEC) of 80.94 cmolc kg−1 and plant available P of 703 mg kg−1 (Table S1 in the Supplement). During biochar application, manually dug 10-L planting basins (35 cm long × 15 cm wide × 20 cm deep) spaced 70 cm × 35 cm (interrow × within row spacing) were opened, and 240 g of dry biochar (equivalent to 1 L by volume) was thoroughly mixed with the soil in each basin for the CA + BC treatment. The planting basins were then covered with a thin layer of soil.
2.4 Soil sampling and analysis for chemical characterization
Soils were sampled before establishing the trials in March 2023 (background sampling) for general characterisation of the research site. Using an auger, 3 samples from 0–20 cm depth were randomly taken from the experimental site and mixed into a single composite sample (Table S1). At the onset (April 2023) and end (October 2023) of the first growing season, soils were sampled plot wise (0–20 cm) from planting basins in CA and CA + BC treatments and in the planting rows in conventional treatments to assess the effect of different treatments on soil properties. Prior to analysis, samples from the same treatments in each of the four blocks were bulked (viz., n=4 for the onset and n=4 for the end for each treatment), air dried and passed through a 2 mm sieve. Soil pH was determined in water (2.5:1) (Gee and Bauder, 1986). SOC and N were analysed by a Thermo Finnigan EA attached to Isotope Ratio Mass Spectrometer (EA-IRMS).
2.5 Nitrous oxide flux sampling and analysis
The static chamber method was used to estimate N2O emissions. We used cylindrical 20 cm high, custom-made PVC chambers manufactured from 16 cm diameter, grey opaque sewage pipes with a self-sealing rubber septum on the top for gas sampling. Permanent gas sampling plots were established by inserting 17 cm diameter PVC rings (the base) to a depth of 7 cm into the soil on 19 April 2023, 3 weeks before the first flux sampling on 10 May 2023. We installed two chamber bases in each plot. One was placed in the interrow, between two rows and another in-row between two plants within a row or in the planting basin (CA) (Figs. S1, S2 in the Supplement).
The chambers were deployed by carefully inserting them 3 cm into the pre-installed collars to obtain an airtight fit. To facilitate chamber deployment, the contact area between the collar and chamber was sealed with a thin layer of petroleum jelly. Each chamber covered an area of 0.020 m2 and had a total headspace volume of 0.004 m3. For each flux measurement, four gas samples were drawn from the chamber headspace 1, 15, 30 and 60 min after deployment using a 20 mL polypropylene syringe equipped with a three-way valve. Collected gas samples were transferred to pre-evacuated 12 mL glass vials with crimp-sealed butyl septa. When sampling the chamber headspace, the plunger of the syringe was moved slowly in and out for three times to mix the gas and obtain a representative sample. Air and chamber temperatures were recorded before removing the chambers using a handhold thermometer which was placed inside a chamber, before and after sampling.
Gas sampling was done approximately biweekly, resulting in 17 sampling campaigns between May 2023 and January 2024. The vials were shipped to the Norwegian University of Life Sciences for CO2 and N2O analysis by gas chromatography. He-filled vials were included as blanks to check for contamination during storage and shipment of the vials. Detected concentrations of CO2 and N2O were <5 % of ambient. We used slightly over-pressured glass vials, crimp sealed with thick butyl septa, which were shown previously to maintain pressure and mixing ratios during air transport and storage (Raji and Dörsch, 2020). Therefore, storage and shipment were assumed to result in negligible changes in gas concentration of the samples. The vials were analysed on a gas chromatograph (GC; model 7890A, Agilent, Santa Clara, CA, USA) connected to an auto-sampler (Gilson). N2O was quantified by an electron capture detector and CO2 by a thermal conductivity detector as described by Žurovec et al. (2017).
Plotting measured CO2 and N2O concentrations over time, revealed linear increase in most cases with little saturation observed. In some cases, the N2O concentration in the sample taken right after chamber deployment was substantially higher than 0.336 ppm (ambient N2O concentration), pointing at residual N2O in the chamber. The exalted N2O concentration after chamber deployment usually decreased until the second measurements (15 min) and to avoid fitting negative fluxes, the first sampling point was discarded. Flux rates were estimated by fitting a linear or second order (polynomial) function to the concentration change over time. A quadratic fit was only used in few cases in which N2O accumulation in the chamber showed a convex downwards trend, i.e., decreasing emissions. Changes in gas concentrations were converted to area flux as follows:
where F is the N2O flux (µg N2O-N m−2 h−1), the rate of change in concentration over time in the chamber headspace (ppm min−1), Vc the volume of the chamber, 0.004 m3, A the area covered by the chamber, 0.020 m2, Mn the molar mass of N in N2O (g mol−1) and Vn the molecular volume of N2O or CO2 at chamber temperature (m3 mol−1). Fluxes were cumulated plot-wise by linear interpolation as kg N2O-N ha−1 and per period) as follows:
where f represents the N2O flux (µg N2O-N m−2 h−1), i the ith measurement, the number of days between two subsequent measurements, and was used for unit conversion. We estimated a representative flux for each plot (area-weighted cumulative N2O emission) by calculating area-weighted mean of fluxes from the basin and interrow positions. Weighing factors of 0.12 and 0.88 were used for basin and interrow areas, respectively, in CA treatments (CA and CA + BC), while a factor of 0.50 was applied to both inrow and interrow areas in conventional treatments (Conventional and ConventMM).
2.6 Soil moisture and mineral N content
Directly after each flux sampling, soils were sampled from both planting stations (Conventional and ConventMM) and basins (CA and CA + BC) and from interrow positions (all treatments), for analysing mineral N ( and ) and soil moisture. Soils were sampled from 0–20 cm depth, using a 10 mm diameter corer with a height of 20 cm. Only one core was taken to prevent excessive perturbation, particularly in the planting basins. The cores were stored in a cooling box on ice and shipped to Gulu University which is located 5 km from the experimental site. The soil samples were extracted the same day, within 5 h after sampling. Mineral N was extracted from 11 g of field moist soil in 40 mL of 1 M potassium chloride (KCl), after 1 h of horizontal shaking at 200 strokes per minute using an automatic shaker (SHKE4450CC-1CE, USA) and passing the supernatant through Whatman filters grade 589/3. The supernatants were frozen for subsequent analysis of and at the Norwegian University of Life Sciences by flow injection analysis (FIA star 5020, Tecator, Sweden).
The remaining soil was dried at 105 °C for 72 h to determine gravimetric moisture content and bulk density (BD). BD was calculated by dividing weight of oven dried soil with the volume of the soil core (15.714 cm3) and its gravimetric soil moisture content calculated by dividing weight of water (difference between fresh soil weight and oven dried soil) by the weight of oven dried soil.
The bulk density (BD) was then used to calculate water filled pore space (WFPS) as follows:
where θg is the gravimetric water content, BD the soil bulk density (1.29±0.01 g cm−3) and PD the soil particle density (2.65 g cm−3). Daily rainfall and temperature data were obtained from the Gulu meteorological station which is located 6 km from the experimental site.
2.7 Yield and yield-scaled emissions
Crops were harvested at physiological maturity, 6 months after sowing for pigeon pea and 4 months for maize. To compare crop yields under conventional and CA management and in CA + BC treatments, all values for dry biomass and grain (moisture content of 12.5 % for maize and 15 % for pigeon pea) were extrapolated from the plot to the hectare. Yield-scaled N2O emissions (kg N2O-N kg−1 grain yield) and N-yield scaled emissions (kg N2O-N kg−1 grain N) were estimated for each season by dividing the area-weighted cumulative N2O emissions with grain yield or N content of the grain (N concentration × grain yield).
2.8 Data analysis
All data were analysed using R software, version 4.3.2. The full dataset is available at NMBU dataverse repository (Namatsheve et al., 2026). A random intercept, fixed slope linear mixed-effect model using the lmer function from lme4 packages (Bates, 2010) with treatment, chamber position (interrow and inrow) and season as fixed factors was used to evaluate treatment effects on N2O emissions and soil mineral N. On soil parameters, yield and yield-scaled emissions, fixed factors were treatments and seasons while block was a random factor (Tables S2–S13). Variation associated with chamber ID was modelled by introducing random effects to account for repeated measurement on the same chamber, on N2O fluxes and soil mineral N. The most parsimonious model was selected after model comparisons based on goodness of-fit assessed by the Akaike Information Criterion (AIC) and the Bayesian Information Criterion (BIC), and stepwise model reduction (Aho et al., 2014). We validated model assumptions by checking quantile plots of residuals against fitted values. Visual inspection of QQ plots showed that residuals were approximately normally distributed for cumulative N2O data, but not for daily N2O fluxes. However, N2O flux data were transformed and did not substantially improve model fit, so we retained the original scale of interpretability. Mixed-effects models are robust to mild non-normality (Zuur et al., 2009). Model parameters (estimated marginal means) were extracted using the “emmeans” package (Lenth, 2016), and multiple comparisons were performed using multcomp (Hothorn et al., 2008) with adjusted pvalues (Tukey post-hoc test at 0.05 probability level) (Lenth, 2016). The 95 % confidence intervals (CI) were retrieved using lsmeans function. Linear regression analyses were performed to analyse the relationship between N2O fluxes with WFPS and mineral N. Visualization of the fitted models was achieved using the package ggplot2 (Wickham, 2016).
3.1 Soil parameters
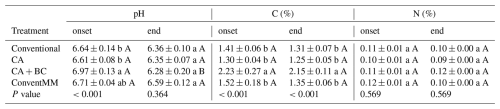
Soils were near neutral with a background pH of 6.71 (Table S1). During the onset of the first season, soil pH ranged from 6.71–6.97. CA + BC significantly (p<0.001) increased pH compared to CA systems at the onset of the first season (Table 2). Generally, pH decreased from the beginning to the end of the first season, after which no significant (p=0.364) pH differences among treatments were found. SOC ranged from 1.25 %–2.23 % and biochar significantly (p<0.001) increased SOC, from 1.30 % in CA to 2.23 % in the CA + BC treatment (Table 2). At the end of the first season, CA + BC had significantly (p<0.001) more SOC than other non-BC treatments. Different treatments did not affect (p=0.57) soil N at the beginning and end of the first season (Table 2).
Table 2Treatment effects on in-row (conventional treatments) and within planting basin (CA treatments) soil properties (pH, C, N, and BD). Soils were sampled at sowing (onset) and at the end of the first season, in Gulu, Uganda. Shown are arithmetic means with standard errors (N=4). Lowercase letters compare treatments at onset or end of sampling, while uppercase letters compare change between samples taken at onset and end of the season. Different letters represent significant differences (p<0.05), determined at 5 % level using Tukey test.
3.2 N2O emission dynamics
Interaction of treatment and chamber position significantly (p=0.03) affected N2O emission rates (Tables S2, S3). N2O emission rates were small in all treatments throughout the entire observation period (Fig. 1a) with treatment averages ranging from 1.02–51.19 . The fluxes peaked in mid-October following pigeon pea harvest and sowing of maize. At this point in time, conventionally tilled soil with pigeon pea–maize rotation (Conventional) had the largest emissions, while conventional tillage with maize monocropping (ConventMM) showed a far less pronounced N2O emission peak. N2O emissions levelled off towards the end of the second season, in January, coinciding with a longer dry spell (Fig. 1a).
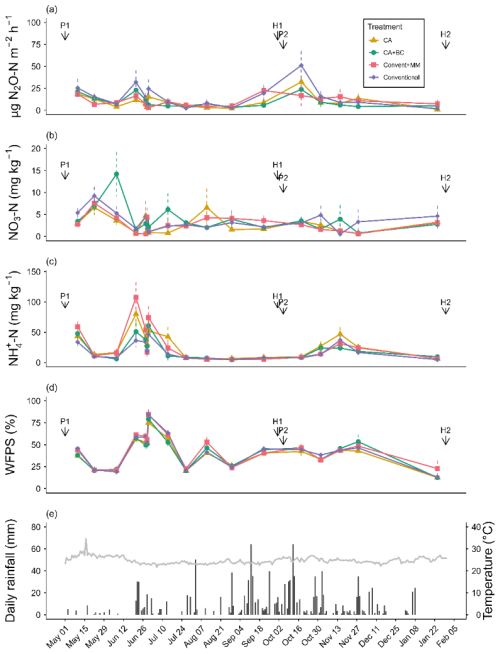
Figure 1Mean (± SE) of (a) N2O emission fluxes, (b) KCl-extractable NO and (c) , (d) WFPS, and (e) daily rainfall and air temperature (°C) during the two cropping seasons between May 2023–January 2024 in Gulu, Uganda. P1 and P2 indicate planting date for the 1st and 2nd second season (18 April 2023 and 5 October 2023, respectively). The data are based on 8 observations, average of 2 chamber positions and 4 blocks. H1 and H2 indicate harvesting dates (1 October 2023 and 30 January 2024, respectively).
3.3 N2O fluxes and mineral N dynamics
Treatment-averaged N2O fluxes ranged from 1.0–51.2 µg N m2 h−1 (Fig. 1a), and they were not affected by either season (p=0.06) and sampling position (0.35). There was a significant relationship between N2O fluxes and mineral N (NH and ), but with a small coefficient of determination (R2=0.04, p<0.001) (Fig. S3). ranged from 0–15 and 0–5 mg N kg−1 in the 1st and 2nd season, respectively (Fig. 1b), while ranged from 10–110 and 10–45 mg N kg−1 in the first and second season, respectively (Fig. 1c). Cropping season significantly affected NO (p=0.03) and (p=0.004) (Tables S3, S4), generally, both NO and were more variable in the long rain season than in the short rain season (Fig. 2). NO was not affected by treatments (p=0.192) and chamber position (p=0.871) (Table S3, Fig. 2a, b). Treatments significantly (p=0.02) affected , with more extractable NH in the ConventMM (31.22 mg N kg−1, 20–42.40 CI) than in the Conventional (19.20 mg kg−1, 8–30.50 CI), in the first season (Table S4, Fig. 2c).
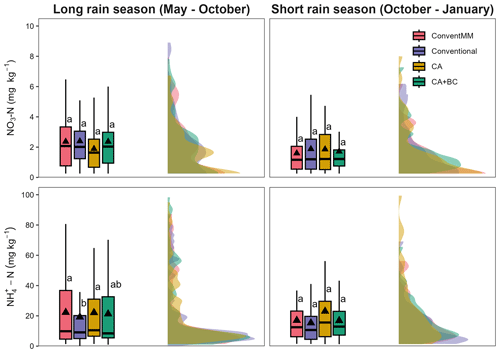
Figure 2Box-whisker and half violin plot showing the effect of cropping systems on NO and NH during the Long rain season and Short rain season in Gulu, Uganda. Upper and lower edges of boxes indicate 75th and 25th percentiles, horizontal lines within boxes indicate median, whiskers below and above the boxes indicate the 10th and 90th percentiles, and triangles indicate arithmetic mean. Differences between treatments were tested using the Tukey post-hoc test. Different letters represent significant differences (p<0.05).
3.4 Effect of rotations, tillage and biochar on cumulative N2O emissions
Cumulative N2O emissions and area-weighted N2O emissions ranged from 0.24–0.50 and 0.25–0.39 kg N ha−1, respectively, in the 1st season (long rain season); and from 0.19–0.61 and 0.20–0.58 kg N ha−1, respectively, in the 2nd season (short rain season). For the entire sampling period, May 2023–January 2024, cumulative N2O emissions and area-weighted N2O emissions ranged from 0.44–1.11 and 0.46–0.88 kg N ha−1, respectively. (Fig. 3). Cumulative N2O emissions were significantly affected by treatments (p<0.001) and growing season (p=0.01) (Table S7). In the 1st season, interrow emissions in the conventional treatment were 0.27±0.03 kg N ha−1, which was significantly higher than in all the other treatments. In the 2nd season, interrow emissions in the conventional treatment increased to 0.61±0.14 kg N ha−1, which was significantly higher than in the CA + BC treatment (0.19±0.14 kg N ha−1). Cumulative N2O emissions from the basins/inrows were not affected by treatments. Under pigeon pea maize rotation, CA + BC significantly reduced (p<0.05) seasonal and area-weighted cumulative N2O emissions, combared to the Conventional for both individual seasons and for the whole sampling period combined (Seasons 1 and 2). Whole-period emissions averaged 0.46±0.08 kg N ha−1 under CA + BC compared to 0.88±0.14 kg N ha−1 under the conventional treatment. There were no significant differences in N2O emissions between CA + BC and CA alone. Under conventional tillage, weighted cumulative N2O emissions in the conventionally tilled pigeon pea–maize rotation (Conventional) did not differ (p>0.05) from the conventionally tilled maize monocrop (ConventMM) in either seasons (Figs. 3, S3, Table S8).
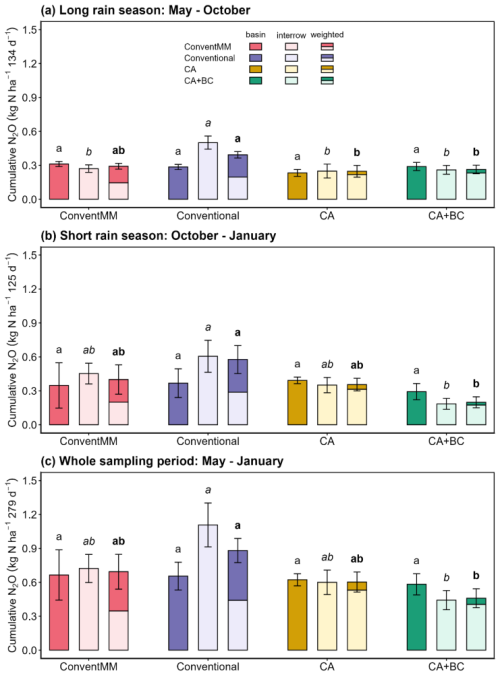
Figure 3Position-specific (basin/inrow and interrow) and area-weighted cumulative N2O emissions (kg N ha−1) for different cropping systems during the (a) Long rain season (May 2023–September 2023), (b) Short rain season (October 2023–January 2024) and (c) sum of both seasons (May 2023–January 2024), in Gulu, Uganda. Error bars represent standard errors (N=4). Different letters represent significant differences between treatments for each of the seasons or the cumulative of all the seasons (p<0.05) using the Tukey post-hoc test. Roman letters compare basins/in-rows while italicized and bold letters compare inter-rows and area-weighted N2O emissions, respectively.
Table 3Grain yield, grain N yield, area-weighted N2O, yield scaled N2O emissions and N yield scaled N2O emissions during the first and second rain season, in northern Uganda. When calculating yield and N scaled N2O emissions, a weighing factor of 0.12 in planting basins and 0.88 in inter-rows was used in CA treatments (CA and CA + BC), and a weighing factor of 0.50 was used for both inrows and interrows in conventional treatments (ConventMM and Conventional). Shown are arithmetic means with standard errors (N=4). Uppercase letters compare seasons specifically for a monocrop treatment (ConventMM), maize was grown in both seasons, for grain yield and grain N yield. Lowercase letters compare treatments within a season. Different letters represent significant differences (p<0.05), determined at 5 % level using Tukey test.
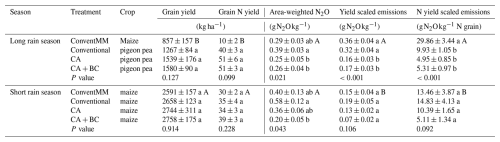
3.5 Treatment effects on grain yield and yield scaled N2O emissions
Different treatments did not affect grain yield in the 1st (p=0.127) and 2nd season. Pigeon pea grain yield ranged from 1.3–1.6 t ha−1 in the first season, and maize grain yield ranged from 2.6–2.8 t ha−1 in the second season. Grain N yield was also not affected by treatments and ranged from 10–51 kg N ha−1 in the first season, and from 30–39 kg N ha−1 in the second season (Table 3). Yield scaled emissions and N yield scaled emissions ranged from 0.16–0.32 g N2O-N kg−1 grain and 5.11–29.86 g N2O-N kg−1 grain N, respectively; with significantly lower values for CA and CA + BC than for Conventional in the first season. Conventional also had significantly high area-weighted N2O (p=0.021), yield scaled emissions (p<0.001) and N yield scaled emissions (p<0.001) in the 1st season. For the first season, ConventMM was not compared to other treatments as it was the only treatment with maize whilst other treatments had pigeon pea. Maize yield in ConventMM were significantly higher in the second season compared to the first season, while yield scaled emissions were greater in the first season.
4.1 Dynamics of N2O fluxes
Treatment-averaged N2O fluxes ranged from 1.0–51.2 µg N m2 h−1 (Fig. 1a), and the cumulative N2O emissions from May 2023–January 2024 were less than 1.2 kg N2O-N ha−1. These emission rates are in the same order of magnitude as those found by research carried out in fertilized tropical soils under conservation agriculture with biochar in Kenya and Zambia (Fungo et al., 2017; Munera-Echeverri et al., 2022). Low fluxes of <1 µg N m2 h−1 were also reported in unfertilized systems (control treatments) in Kenya and Zimbabwe (Hickman et al., 2015; Mapanda et al., 2011). Low N2O emissions can be attributed to low soil mineral N contents (Fig. 1b, c) (Chapuis-Lardy et al., 2009). Increased N2O emission rates were recorded in October 2023, after harvesting pigeon pea and immediate sowing for the second season (Fig. 1a). These emissions might have been associated with decomposition of pigeon pea residues, leaf litter and root turnover, which provided readily available C and N substrates that fuel N2O production. In the conventional treatment, soil disturbance during land preparation for second season, combined with pigeon pea residues from the first season might have further stimulated microbial activities and accelerated N2O fluxes. Additionally, consumption of labile C by heterotrophs may have created anaerobic microsites that promoted denitrification, especially since the period following harvest coincided with heavy rainfalls and high WFPS values (Fig. 2d). A similar, though smaller emission peak was observed in June when abundant rainfall terminated a dry spell. Rewetting of dry soil triggers N2O fluxes likely due to increased nitrification and denitrification fueled by release of readily available N and C from dead microbial biomass (Namoi et al., 2019), and it is an important N2O source in seasonally dry ecosystems (Hickman et al., 2011). Likewise, after harvest and sowing, plant N uptake is small, which might have supported elevated microbial C and N turnover. N2O flux peaks were short-lived lasting for only 2 weeks (Fig. 1a).
4.2 Mineral N and WFPS
Daily N2O fluxes were weakly correlated to mineral N. The Gulu site has a relatively low soil δ15N value of 4.64 and a soil N content of 0.11 % (Namatsheve et al., 2025), indicating a highly efficient and tight N cycling (Craine et al., 2015). This suggests that microbes in these soils compete effectively for mineral N, likely immobilizing N and thereby reducing its availability for microbial N transformations such as nitrification and denitrification. We anticipated that biological N2-fixation by pigeon pea in the first season would result in higher N2O emissions in the second season, especially in CA treatments where N2-fixation ranged 50–105 kg N ha−1 (Namatsheve et al., 2025). However, N2O emissions and mineral N did not appear to be driven by N2-fixation. Our results imply that N2-fixation and residue retention do not directly affect soil mineral N or N2O emissions in unfertilized soils with inherently low N. Rochette et al. (2004) also reported considerable uncertainty in N2O emission from soils under legumes, they noted that soil mineral N alone was a poor predictor of N2O emissions for two seasons in acidic soils in Canada. Biological N2-fixation was removed as a direct source of N2O because of the lack of evidence of significant emissions arising from the N fixation (IPCC, 2019).
Generally, NH and NO contents were more variable in the 1st season (May–October) than the 2nd season (October–January) Figs. 1b, c, 2). At the onset of the experiment, mineral N was most likely from mineralisation of mulched grasses having grown under fallow for 3 years prior to the experiment. Although residual glyphosate applied during land preparation may have influenced soil N dynamics in the first season (Kanissery et al., 2019), the relatively high temperatures at the site (mean 25 °C, range 23–34 °C) likely promoted rapid degradation, reducing the likelihood of persistent effects during the crop growth period. In the 1st season, ConventMM was planted with maize whereas other treatments were planted with pigeon pea, which may have contributed to differences in concentrations. Under conventional tillage, higher NH in maize (ConventMM) than pigeon pea (Conventional) in the first season might be attributed to differences in crop phenology. Pigeon pea is a slow starter; its nodulation and peak biological N2-fixation typically occur around 80 d after sowing (Kumar Rao and Dart, 1987). Legumes generally show a stronger affinity for due to lower energy cost for its assimilation, thus reducing soil levels compared to maize, which generally exhibits slower early-season N uptake and preference towards (Daryanto et al., 2019). However, during the first season, CA and CA + BC had also signficant concentrations although they were planted with pigeon pea. Reduced tillage, residue retention and biochar may have enhanced early-season mineralization, while simultaneously slowing nitrification, allowing to accumulate. The concentration of was low and not affected by treatments in any of the two seasons. Our result for mineral N aligns with findings of Mapanda et al. (2011) who also reported low mineral N in an unfertilized treatment on clay and sandy soils.
High N2O emission rates go often along with high WFPS values, increasing the anaerobic volume and hence denitrification in soils (Hao et al., 2025; Wang et al., 2023). We found a weak positive relationship between WFPS and N2O emissions (R2=0.02, p<0.001; Fig. S3). This relationship is only expected to be linear over a limited part of the range of potential WFPS values. High evaporation under tropical conditions due to high temperatures (mean 25 °C, range 23–34 °C) result in rapid water loss, which drastically reduced the time the soil was above 60 % WFPS, despite high rainfalls throughout the sampling period (Fig. 1d, e). Apart from June–July 2023, WFPS was <60 %, which is often considered a threshold for denitrification-driven N2O emissions. This may, in part, explain the weak correlation between WFPS and N2O fluxes in our study.
4.3 Crop rotation, tillage and biochar on N2O emissions
Area-weighted cumulative N2O emissions ranged from 0.2 to 0.6 kg ha−1 in the 1st and 2nd season, respectively. These results are consistent with Millar et al. (2004) who also converted a natural fallow to an improved system and reported low N2O emissions in an unfertilized control treatment. Similarly, Baggs et al. (2006), Bwana et al. (2021) and Hickman et al. (2015) reported low N2O emissions of 0.1–0.5 kg ha−1 in unfertilized conventional treatments; these comparisons indicate that N2O emissions from unfertilized soils remain relatively low. In fertilized systems in SSA, N2O emissions also remain relatively low. For instance, Mapanda et al. (2011) reported emissions of <0.29 kg N2O ha−1 following application of 120 kg N ha−1 of fertilizer. The authors speculated that low emissions at high N fertilizer rates were due to the experimental design which was not particularly good at fully identifying hotspots of N2O associated with the spot application of fertilizer used, and sporadic measurements.
Weighted cumulative N2O emissions in the conventionally tilled pigeon pea–maize rotation (Conventional) did not differ from the conventionally tilled maize monocrop (ConventMM) (Figs. 3, S3). The trials were established in a soil with low organic N content without inorganic N fertilization. However, after some seasons, inputs from crop residues and biological N2-fixation may enhance soil fertility. Effect of crop rotations appear to be relevant for N2O emissions only when inorganic fertilizers are applied in long term trials. Jeuffroy et al. (2013) reported a 75 %–80 % reduction in N2O emissions in a 4-year study under pea rotation without N input compared to a fertilized monocrop, illustrating the significance of N input for N2O emissions, rather than the effect of rotation itself.
In pigeon pea–maize rotations, area-weighted cumulative N2O emissions under CA (area weighting factors of 0.12 and 0.88 for basins (disturbed) and interrow (undisturbed) areas, respectively) were significantly lower than those under the conventional rotation (area weighting factors of 0.5 and 0.5 for inrow (disturbed) and interrow (undisturbed) areas, respectively) in the first season. This difference may be attributed to full soil inversion in the conventional system, as soil disturbance promotes rapid drying and rewetting cycles, thereby increasing area-weighted N2O emissions compared to reduced tillage. However, in the second season, and for the whole sampling period, CA alone did not affect weighted N2O emissions. These findings are partly consistent with Jantalia et al. (2008) and Ruan and Robertson (2013) who reported higher N2O emissions under conventional tillage compared with reduced tillage, which they attributed to soil disturbance, increased soil aeration and accelerated organic matter breakdown. Lack of consistent differences across seasons in our study suggests that the effect of tillage under rotations on N2O emissions may be context-dependent, particularly under varying environmental conditions. In the Conventional treatment, interrow and area-weighted N2O emissions exhibited persistently high levels of N2O across all seasons. We have shown that conventional treatment had the least root biomass compared to other treatments, and that roots were concentrated in the in-rows (Namatsheve et al., 2025). The inter-rows typically have lower root density which limits N uptake and leaves more mineral N for microbial processes thus creating localised hotspots that fuelled N2O emissions. We speculate that low root density in the inter-rows might reduce rhizospheric activity and O2 diffusion, which can favor denitrification.
Although there are no significant differences in weighted N2O emissions between reduced tillage treatments (CA and CA + BC) in pigeon pea–maize rotations, biochar applied at a rate of 5 Mg ha−1 to CA systems (CA + BC) reduced area-weighted N2O emissions by 33 % and 66 % compared to Conventional treatment in the 1st and 2nd season, respectively (Fig. 3). Biochar with a high C:N ratio of >60, as applied in this study, was shown to reduce the bioavailability of inorganic N through microbial immobilisation (Namoi et al., 2019) or sorption of NO due to unconventional H-bonding between ions and biochar surface functional groups (Kammann et al., 2017; Nguyen et al., 2017). In a fertilized Ultisol in western Kenya, Fungo et al. (2019) reported a 22 % reduction in emissions. Case et al. (2015) also reported that biochar suppressed N2O emissions in a sandy loam soil fertilized with 140 . Biochar applied in a calcareous soil also reduced N2O emissions by 50 %, and increased soil ratio, indicating that biochar impaired nitrification and N immobilisation processes (Lentz et al., 2014). Our results support the growing evidence that biochar can mitigate N2O emissions via various N cycling modifications (Liu et al., 2018; Zhang et al., 2021; Kammann et al., 2017). However, biochar did not significantly reduce N2O scaled emissions compared to conventMM, suggesting that these modifying effects were insufficient to offset the partly legume induced increase in N2O emissions (Fig. 3). In a short-term study, Munera-Echeverri et al. (2022) also reported that biochar amendments did not affect N2O emissions in Zambia despite increased gross nitrification rates in the biochar treatments. In a global meta-analysis, Shakoor et al. (2021) showed that biochar increased N2O emissions by 20 %. Discrepancies on response of N2O emissions to biochar might be explained by biochar type, soil parameters, climatic conditions and experimental duration.
4.4 Yield and yield scaled N2O emissions
In pigeon pea–maize treatments had no significant effect on grain and grain N yields in either season. Rusinamhodzi et al. (2011) highlighted that high inputs of especially N fertilizer are required to realize yield benefits of CA. When inorganic fertilizer is applied, positive effects of CA become more prominent in the long term. In Zambia, benefits of biochar and/or CA on grain yield were reported after several seasons (Martinsen et al., 2014, 2017; Munera-Echeverri et al., 2020). Yields in the first season under conventional tillage with maize monocropping (ConventMM) were low, <1.6 t ha−1 the average maize yield in Uganda without N fertilization (Kaizzi et al., 2012). Low rainfall received from early May to mid-June, during the critical growth stage for maize (tasselling and grain filling) drastically reduced maize grain yield in the first season.
Reducing yield-scaled N2O emission has been pointed out as one of the most promising strategies to increase crop yield while reducing N2O emissions. Assessing yield and N yield-scaled emissions is therefore important, as sustainable cropping systems must maintain or increase grain and protein yield while reducing greenhouse gas emissions per unit of agricultural output to reconcile food security with climate change mitigation (Yao et al., 2024). In this study, yield-scaled N2O emission were 0.16–0.32 g N kg−1 grain in the first season, and <0.20 g N kg−1 grain in the second season. During the first season, yield-scaled N2O emission in CA and CA + BC were significantly reduced by 50 % compared to conventional practices, indicating that N use efficiency was high. These practices were effective in minimising emissions without penalising pigeon pea yields. This supports CA and CA + BC as sustainable agricultural practices.
A key challenge for the sustainability of unfertilized agroecosystems is the management of soil nutrient balances. While biochar amendments in CA systems can effectively reduce N2O emissions and maintain crop productivity (Borchard et al., 2019), these systems gradually deplete soil nutrient reserves when mineral fertilizers are not applied (Falconnier et al., 2023). Although N2O is a powerful GHG, it represents a relatively small component of the total annual N budget, often less than 1 kg N ha−1. The primary source of nutrient removal is the export of grain, which removes a significant amount of N from the system. While biological N2-fixation from pigeon pea can fix up to 110 kg N ha−1 (Namatsheve et al., 2025), some of this input can as well be exported from the field in the exported grain and stalks, and the remaining N in the form of decaying roots, rhizodeposits and leaf fall is often insufficient to fully compensate for the N removed (Adu-Gyamfi et al., 2007). Furthermore, the biologically fixed N is prone to rapid mineralization and subsequent loss through leaching or other pathways before the next crop can fully utilize it. To achieve long term sustainability, an integrated approach is required, including targeted fertilization to prevent continuous nutrient mining and to ensure the long-term viability of the agroecosystem.
This study provides insight into N2O emissions from unfertilized low-input agroecosystems that dominate much of sub-Saharan Africa. We have shown that biochar amendments under reduced tillage mitigate N2O scaled emissions compared to conventional pigeon pea–maize rotations but not compared to conventional maize monocropping or CA alone. Pigeon pea rotation under conventional tillage did not affect the emissions compared to conventional maize monocropping, suggesting that incorporating legumes in rotations do not increase N2O emissions in unfertilized systems. N2O fluxes peaks occurred following rainfall events after dry spells and the incorporation of high-quality pigeon pea residues, these peaks were short lived, indicating that crop residue-driven pulses are temporary in these unfertilized systems. Yield scaled N2O emissions were substantially lower under conservation agriculture, and conservation agriculture with biochar, implying that N2O emissions can be reduced without penalising pigeon pea grain yield. While our study was carried out in two consecutive growing seasons within the first year of establishing the experiment, limiting our ability to generalise the findings across years; our observations on low N2O emissions, crop residue driven peaks on N2O fluxes, and the mitigating effect of biochar are consistent with the limited number of studies available from SSA. These patterns highlight the potential of biochar amendments in CA systems to reduced N2O emissions in subsistence farming systems. Long term studies on response of soils to CA and biochar amendments in both fertilized and unfertilized systems are needed to improve understanding of GHG emissions.
The code will be available from the corresponding author and data is available at NMBU dataverse repository https://doi.org/10.18710/CNP8TF (Namatsheve et al., 2026).
The supplement related to this article is available online at https://doi.org/10.5194/bg-23-2787-2026-supplement.
TN: Experimental design, Data collection, Lab analysis, Data curation, Writing – original draft and editing, Visualization, Methodology, Investigation, Formal analysis, Conceptualization. VM: Experimental design, writing – review & editing, Formal analysis, Supervision, Resources, Project administration, Funding acquisition, Conceptualization. JM: Experimental design, Writing – review & editing, Supervision, Conceptualization. AO: Experimental design, Supervision, Conceptualization. PD: Lab analysis, Data curation, Writing – review & editing, Supervision, Conceptualization.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
This study was funded by Norwegian University of Life Sciences PhD grant to the first author through the CLIMSMART project funded by Norwegian Research Council (NRC no. 302713); aiming at increasing food security, on-farm profitability, and entrepreneurship of smallholder farms in Uganda through training and implementation of climate smart practices. Additional support was obtained from the Climate Smart Agriculture in Sub-Saharan Africa project (NORHED II NMBU, 2021-2026, QZA-21/0182) under the Norwegian Programme for Capacity Development in Higher Education and Research for Development (NORHED II), funded by the Norwegian Agency for Development Cooperation (NORAD).
This study was funded by Norwegian University of Life Sciences PhD grant to the first author through the CLIMSMART project funded by Norwegian Research Council (NRC no. 302713); aiming at increasing food security, on-farm profitability, and entrepreneurship of smallholder farms in Uganda through training and implementation of climate smart practices. Additional support was obtained from the Climate Smart Agriculture in Sub-Saharan Africa project (NORHED II NMBU, 2021-2026, QZA-21/0182) under the Norwegian Programme for Capacity Development in Higher Education and Research for Development (NORHED II), funded by the Norwegian Agency for Development Cooperation (NORAD).
This paper was edited by Andreas Ibrom and reviewed by Kathrin Fuchs and one anonymous referee.
Abalos, D., Recous, S., Butterbach-Bahl, K., De Notaris, C., Rittl, T. F., Topp, C. F. E., Petersen, S. O., Hansen, S., Bleken, M. A., Rees, R. M., and Olesen, J. E.: A review and meta-analysis of mitigation measures for nitrous oxide emissions from crop residues, Sci. Total Environ., 828, https://doi.org/10.1016/j.scitotenv.2022.154388, 2022.
Adegbeye, M. J., Ravi Kanth Reddy, P., Obaisi, A. I., Elghandour, M. M. M. Y., Oyebamiji, K. J., Salem, A. Z. M., Morakinyo-Fasipe, O. T., Cipriano-Salazar, M., and Camacho-Díaz, L. M.: Sustainable agriculture options for production, greenhouse gasses and pollution alleviation, and nutrient recycling in emerging and transitional nations – An overview, J. Clean. Prod., 242, 118319, https://doi.org/10.1016/j.jclepro.2019.118319, 2020.
Adu-Gyamfi, J. J., Myaka, F. A., Sakala, W. D., Odgaard, R., Vesterager, J. M., and Høgh-Jensen, H.: Biological nitrogen fixation and nitrogen and phosphorus budgets in farmer-managed intercrops of maize-pigeonpea in semi-arid southern and eastern Africa, Plant Soil, 295, 127–136, https://doi.org/10.1007/s11104-007-9270-0, 2007.
Africa Union: Africa fertilizer and soil health summit, Nairobi, 1–53 pp., 2024.
Aho, K., Derryberry, D., and Peterson, T.: Model selection for ecologists: the worldviews of AIC and BIC, Ecology, 95, 631–636, 2014.
Baggs, E. M., Chebii, J., and Ndufa, J. K.: A short-term investigation of trace gas emissions following tillage and no-tillage of agroforestry residues in western Kenya, Soil Till. Res., 90, 69–76, https://doi.org/10.1016/j.still.2005.08.006, 2006.
Bates, D. M.: lme4: Mixed-effects modeling with R, http://lme4.r-forge.r-project.org/book/ (last access: 17 April 2025), 2010.
Boateng, K. K., Obeng, G. Y., and Mensah, E.: Agricultural Greenhouse Gases from Sub-Saharan Africa, in: Energy, Environment, and Sustainability, Springer Nature, 73–85, https://doi.org/10.1007/978-981-13-3272-2_6, 2019.
Borchard, N., Schirrmann, M., Cayuela, M. L., Kammann, C., Wrage-Mönnig, N., Estavillo, J. M., Fuertes-Mendizábal, T., Sigua, G., Spokas, K., Ippolito, J. A., and Novak, J.: Biochar, soil and land-use interactions that reduce nitrate leaching and N2O emissions: a meta-analysis, Sci. Total Environ., 651, 2354–2364, https://doi.org/10.1016/j.scitotenv.2018.10.060, 2019.
Bwana, T. N., Amuri, N. A., Semu, E., Elsgaard, L., Butterbach-Bahl, K., Pelster, D. E., and Olesen, J. E.: Soil N2O emission from organic and conventional cotton farming in Northern Tanzania, Sci. Total Environ., 785, 1–10, https://doi.org/10.1016/j.scitotenv.2021.147301, 2021.
Case, S. D. C., McNamara, N. P., Reay, D. S., Stott, A. W., Grant, H. K., and Whitaker, J.: Biochar suppresses N2O emissions while maintaining N availability in a sandy loam soil, Soil Biol. Biochem., 81, 178–185, https://doi.org/10.1016/j.soilbio.2014.11.012, 2015.
Chapuis-Lardy, L., Metay, A., Martinet, M., Rabenarivo, M., Toucet, J., Douzet, J. M., Razafimbelo, T., Rabeharisoa, L., and Rakotoarisoa, J.: Nitrous oxide fluxes from Malagasy agricultural soils, Geoderma, 148, 421–427, https://doi.org/10.1016/j.geoderma.2008.11.015, 2009.
Cornelissen, G., Pandit, N. R., Taylor, P., Pandit, B. H., Sparrevik, M., and Schmidt, H. P.: Emissions and char quality of flame-curtain “Kon Tiki” kilns for farmer-scale charcoal/biochar production, PLoS One, 11, https://doi.org/10.1371/journal.pone.0154617, 2016.
Craine, J. M., Brookshire, E. N. J., Cramer, M. D., Hasselquist, N. J., Koba, K., Marin-Spiotta, E., and Wang, L.: Ecological interpretations of nitrogen isotope ratios of terrestrial plants and soils, Plant Soil, 396, 1–26, https://doi.org/10.1007/s11104-015-2542-1, 2015.
Daryanto, S., Wang, L., Gilhooly, W. P., and Jacinthe, P.: Nitrogen preference across generations under changing ammonium nitrate ratios, J. Plant Ecol., 12, 235–244, https://doi.org/10.1093/jpe/rty014, 2019.
Dick, J., Kaya, B., Soutoura, M., Skiba, U., Smith, R., Niang, A., and Tabo, R.: The contribution of agricultural practices to nitrous oxide emissions in semi-arid Mali, Soil Use Manag., 24, 292–301, https://doi.org/10.1111/j.1475-2743.2008.00163.x, 2008.
Falconnier, G.N., Cardinael, R., Corbeels, M., Baudron, F., Chivenge, P., Couëdel, A., Ripoche, A., Affholder, F., Naudin, K., Benaillon, E., Rusinamhodzi, L., Leroux, L., Vanlauwe, B., and Giller, K. E.: The input reduction principle of agroecology is wrong when it comes to mineral fertilizer use in sub-Saharan Africa, Outlook Agric., 52, 311–326, https://doi.org/10.1177/00307270231199795, 2023.
Fang, H., Mo, J., Peng, S., Li, Z., and Wang, H.: Cumulative effects of nitrogen additions on litter decomposition in three tropical forests in southern China, Plant Soil, 297, 233–242, https://doi.org/10.1007/s11104-007-9339-9, 2007.
Fungo, B., Lehmann, J., Kalbitz, K., Tenywa, M., Thionìo, M., and Neufeldt, H.: Emissions intensity and carbon stocks of a tropical Ultisol after amendment with Tithonia green manure, urea and biochar, Field Crops Res., 209, 179–188, https://doi.org/10.1016/j.fcr.2017.05.013, 2017.
Fungo, B., Lehmann, J., Kalbitz, K., Thionìo, M., Tenywa, M., Okeyo, I., and Neufeldt, H.: Ammonia and nitrous oxide emissions from a field Ultisol amended with tithonia green manure, urea, and biochar, Biol. Fertil. Soils, 55, 135–148, https://doi.org/10.1007/s00374-018-01338-3, 2019.
Gee, G. W. and Bauder, J. W.: Particle size analysis, in: Methods of Soil Analysis, Part 1, edited by: Klute, A., Am. Soc. Agron., Madison, WI, 383–411 pp., 1986.
Giller, K. E., Andersson, J. A., Corbeels, M., Kirkegaard, J., Mortensen, D., Erenstein, O., and Vanlauwe, B.: Beyond conservation agriculture, Front. Plant Sci., 6, https://doi.org/10.3389/fpls.2015.00870, 2015.
Glaser, B., Haumaier, L., Guggenberger, G., and Zech, W.: The `Terra Preta' phenomenon: a model for sustainable agriculture in the humid tropics, Naturwissenschaften, 88, 37–41, https://doi.org/10.1007/s001140000193, 2001.
Guenet, B., Gabrielle, B., Chenu, C., Arrouays, D., Balesdent, J., Bernoux, M., Bruni, E., Caliman, J. P., Cardinael, R., Chen, S., Ciais, P., Desbois, D., Fouche, J., Frank, S., Henault, C., Lugato, E., Naipal, V., Nesme, T., Obersteiner, M., Pellerin, S., Powlson, D. S., Rasse, D. P., Rees, F., Soussana, J. F., Su, Y., Tian, H., Valin, H., and Zhou, F.: Can N2O emissions offset the benefits from soil organic carbon storage?, Glob. Change Biol., 27, 237–256, https://doi.org/10.1111/gcb.15342, 2021.
Hao, Y., Mao, J., Bachmann, C. M., Hoffman, F. M., Koren, G., Chen, H., Tian, H., Liu, J., Tao, J., Tang, J., Li, L., Liu, L., Apple, M., Shi, M., Jin, M., Zhu, Q., Kannenberg, S., Shi, X., Zhang, X., Wang, Y., Fang, Y., and Dai, Y.: Soil moisture controls over carbon sequestration and greenhouse gas emissions: a review, npj Climate and Atmospheric Science. 8, 16, https://doi.org/10.1038/s41612-024-00888-8, 2025.
Hickman, J. E., Havlikova, M., Kroeze, C., and Palm, C. A.: Current and future nitrous oxide emissions from African agriculture, Current Opinion in Environmental Sustainability, 3, 370–378, https://doi.org/10.1016/j.cosust.2011.08.001, 2011.
Hickman, J. E., Tully, K. L., Groffman, P. M., Diru, W., and Palm, C. A.: A potential tipping point in tropical agriculture: Avoiding rapid increases in nitrous oxide fluxes from agricultural intensification in Kenya, J. Geophys. Res.-Biogeo., 120, 938–951, https://doi.org/10.1002/2015JG002913, 2015.
Hobbs, P. R., Sayre, K., and Gupta, R.: The role of conservation agriculture in sustainable agriculture, Philos. Trans. R. Soc. Lond. B., 363, 543–555, https://doi.org/10.1098/rstb.2007.2169, 2008.
Hothorn, T., Bretz, F., and Westfall, P.: Simultaneous inference in general parametric models, Biometrical J., 50, 346–363, https://doi.org/10.1002/bimj.200810425, 2008.
IPCC: N2O emissions from managed soils, and CO2 emissions from lime and urea application, in: 2019 Refinement to the 2006 IPCC Guidelines for National Greenhouse Gas Inventories, edited by: Buendia, E. C., Tanabe, K., Kranjc, A., Jamsranjav, B., Fukuda, M., Ngarize, S., Osako, A., Pyrozhenko, Y., Shermanau, P., and Federici, S., IPCC, Switzerland, ISBN 978-4-88788-232-4, 2019.
Jantalia, C. P., Dos Santos, H. P., Urquiaga, S., Boddey, R. M., and Alves, B. J. R.: Fluxes of nitrous oxide from soil under different crop rotations and tillage systems in the South of Brazil, Nutr. Cycl. Agroecosyst., 82, 161–173, https://doi.org/10.1007/s10705-008-9178-y, 2008.
Jeffery, S., Bezemer, T. M., Cornelissen, G., Kuyper, T. W., Lehmann, J., Mommer, L., Sohi, S. P., van de Voorde, T. F. J., Wardle, D. A., and van Groenigen, J. W.: The way forward in biochar research: Targeting trade-offs between the potential wins, GCB Bioenergy, 7, 1–13, https://doi.org/10.1111/gcbb.12132, 2015.
Jensen, E. S., Peoples, M. B., Boddey, R. M., Gresshoff, P. M., Henrik, H. N., Alves, B. J. R., and Morrison, M. J.: Legumes for mitigation of climate change and the provision of feedstock for biofuels and biorefineries. A review, Agron. Sustain. Dev., 32, 329–364, https://doi.org/10.1007/s13593-011-0056-7, 2012.
Jeuffroy, M. H., Baranger, E., Carrouée, B., de Chezelles, E., Gosme, M., Hénault, C., Schneider, A., and Cellier, P.: Nitrous oxide emissions from crop rotations including wheat, oilseed rape and dry peas, Biogeosciences, 10, 1787–1797, https://doi.org/10.5194/bg-10-1787-2013, 2013.
Kaizzi, K. C., Byalebeka, J., Semalulu, O., Alou, I., Zimwanguyizza, W., Nansamba, A., Musinguzi, P., Ebanyat, P., Hyuha, T., and Wortmann, C. S.: Maize response to fertilizer and nitrogen use efficiency in Uganda, Agron. J., 104, 73–82, https://doi.org/10.2134/agronj2011.0181, 2012.
Kammann, C., Ippolito, J., Hagemann, N., Borchard, N., Cayuela, M. L., Estavillo, J. M., Fuertes-Mendizabal, T., Jeffery, S., Kern, J., Novak, J., Rasse, D., Saarnio, S., Schmidt, H. P., Spokas, K., and Wrage-Mönnig, N.: Biochar as a tool to reduce the agricultural greenhouse-gas burden–knowns, unknowns and future research needs, J. Environ. Eng. Landsc., 25, 114–139, https://doi.org/10.3846/16486897.2017.1319375, 2017.
Kanissery, R., Gairhe, B., Kadyampakeni, D., Batuman, O., and Alferez, F.: Glyphosate: Its environmental persistence and impact on crop health and nutrition, Plants, 8, 1–11, https://doi.org/10.3390/plants8110499, 2019.
Kim, D.-G., Thomas, A. D., Pelster, D., Rosenstock, T. S., and Sanz-Cobena, A.: Greenhouse gas emissions from natural ecosystems and agricultural lands in sub-Saharan Africa: synthesis of available data and suggestions for further research, Biogeosciences, 13, 4789–4809, https://doi.org/10.5194/bg-13-4789-2016, 2016.
Kumar Rao, J. V. D. K. and Dart, P. J.: Nodulation, nitrogen fixation and nitrogen uptake in pigeonpea (Cajanus cajan (L.) Millsp) of different maturity groups, Plant Soil, 99, 255–266, 1987.
Lehmann, J.: A handful of carbon, Nature, 143–144, https://doi.org/10.1038/447143a, 2007.
Lenth, R. V.: Least-Squares Means: The R Package lsmeans, J. Stat. Softw., 69, 1–33, https://doi.org/10.18637/jss.v069.i01, 2016.
Lentz, R. D., Ippolito, J. A., and Spokas, K. A.: Biochar and Manure Effects on Net Nitrogen Mineralization and Greenhouse Gas Emissions from Calcareous Soil under Corn, Soil Sci. Soc. Am. J., 78, 1641–1655, https://doi.org/10.2136/sssaj2014.05.0198, 2014.
Liu, Q., Zhang, Y., Liu, B., Amonette, J. E., Lin, Z., Liu, G., Ambus, P., and Xie, Z.: How does biochar influence soil N cycle? A meta-analysis, Plant Soil, 426, 211–225, https://doi.org/10.1007/s11104-018-3619-4, 2018.
Mapanda, F., Wuta, M., Nyamangara, J., and Rees, R. M.: Effects of organic and mineral fertilizer nitrogen on greenhouse gas emissions and plant-captured carbon during maize cropping in Zimbabwe, Plant Soil, 343, 67–81, https://doi.org/10.1007/s11104-011-0753-7, 2011.
Martinsen, V., Mulder, J., Shitumbanuma, V., Sparrevik, M., Børresen, T., and Cornelissen, G.: Farmer-led maize biochar trials: Effect on crop yield and soil nutrients under conservation farming, J. Plant Nutr. Soil Sci., 177, 681–695, https://doi.org/10.1002/jpln.201300590, 2014.
Martinsen, V., Shitumbanuma, V., Mulder, J., Ritz, C., and Cornelissen, G.: Effects of hand-hoe tilled conservation farming on soil quality and carbon stocks under on-farm conditions in Zambia, Agric. Ecosyst. Environ., 241, 168–178, https://doi.org/10.1016/j.agee.2017.03.010, 2017.
Meier, E. A., Thorburn, P. J., Bell, L. W., Harrison, M. T., and Biggs, J. S.: Greenhouse Gas Emissions From Cropping and Grazed Pastures Are Similar: A Simulation Analysis in Australia, Front. Sustain. Food Syst., 3, 1–18, https://doi.org/10.3389/fsufs.2019.00121, 2020.
Millar, N., Ndufa, J. K., Cadisch, G., and Baggs, E. M.: Nitrous oxide emissions following incorporation of improved fallow residues in the humid tropics, Global Biogeochem. Cycles, 18, 1–9, https://doi.org/10.1029/2003GB002114, 2004.
Mubiru, D. N., Komutunga, E., Agona, A., Apok, A., Ngara, T., and Mubiru, D.: Characterising agrometeorological climate risks and uncertainties: Crop production in Uganda, S. Afr. J. Sci., 108, https://doi.org/10.4102/sajs, 2012.
Munera-Echeverri, J. L., Martinsen, V., Strand, L. T., Cornelissen, G., and Mulder, J.: Effect of conservation farming and biochar addition on soil organic carbon quality, nitrogen mineralization, and crop productivity in a light textured Acrisol in the sub-humid tropics, PLoS One, 15, 1–17, https://doi.org/10.1371/journal.pone.0228717, 2020.
Munera-Echeverri, J. L., Martinsen, V., Dörsch, P., Obia, A., and Mulder, J.: Pigeon pea biochar addition in tropical Arenosol under maize increases gross nitrification rate without an effect on nitrous oxide emission, Plant Soil, 474, 195–212, https://doi.org/10.1007/s11104-022-05325-4, 2022.
Namatsheve, T.: Biological N2-fixation, grain yield and N2O emissions: the role of conservation agriculture and biochar in unfertilized tropical soils, PhD thesis, Norwegian University of Life Sciences, 2025.
Namatsheve, T., Chikowo, R., Corbeels, M., Mouquet-Rivier, C., Icard-Vernière, C., Cardinael, R., Icard-Verniere, C., and Cardinael, R.: Maize-cowpea intercropping as an ecological intensification option for low input systems in sub-humid Zimbabwe: Productivity, biological N2-fixation and grain mineral content, Field Crops Res., 263, 1–21, https://doi.org/10.1016/j.fcr.2020.108052, 2021.
Namatsheve, T., Martinsen, V., Obia, A., and Mulder, J.: Grain yield and nitrogen cycling under conservation agriculture and biochar amendment in agroecosystems of sub-Saharan Africa. A meta-analysis, Agric. Ecosyst. Environ., 376, https://doi.org/10.1016/j.agee.2024.109243, 2024.
Namatsheve, T., Mulder, J., Obia, A., and Martinsen, V.: Biological N2-fixation and grain yield of pigeon pea: The role of biochar and conservation agriculture in low-input systems, Field Crops Res., 328, 109923, https://doi.org/10.1016/j.fcr.2025.109923, 2025.
Namatsheve, T., Martinsen, V., Mulder, J, Obia, A., and Dörsch, P.: Data for: Nitrous oxide emissions from pigeon pea–maize rotation in response to conservation agriculture and biochar amendments in a Ferralsol, northern Uganda, NMBU dataverse [data set], https://doi.org/10.18710/CNP8TF, 2026.
Namoi, N., Pelster, D., Rosenstock, T. S., Mwangi, L., Kamau, S., Mutuo, P., and Barrios, E.: Earthworms regulate ability of biochar to mitigate CO2 and N2O emissions from a tropical soil, Appl. Soil Ecol., 140, 57–67, https://doi.org/10.1016/j.apsoil.2019.04.001, 2019.
Nguyen, T. T. N., Xu, C. Y., Tahmasbian, I., Che, R., Xu, Z., Zhou, X., Wallace, H. M., and Bai, S. H.: Effects of biochar on soil available inorganic nitrogen: A review and meta-analysis, Geoderma, 288, 79–96, https://doi.org/10.1016/j.geoderma.2016.11.004, 2017.
Oriangi, G., Mukwaya, P. I., Luwa, J. K., Menya, E., Malinga, G. M., and Bamutaze, Y.: Variability and Changes in Climate in Northern Uganda, African Journal of Climate Change and Resource Sustainability, 3, 81–97, https://doi.org/10.37284/ajccrs.3.1.1830, 2024.
Pittelkow, C. M., Liang, X., Linquist, B. A., Van Groenigen, L. J., Lee, J., Lundy, M. E., Van Gestel, N., Six, J., Venterea, R. T., and Van Kessel, C.: Productivity limits and potentials of the principles of conservation agriculture, Nature, 517, 365–368, https://doi.org/10.1038/nature13809, 2015.
Portmann, R. W., Daniel, J. S., and Ravishankara, A. R.: Stratospheric ozone depletion due to nitrous oxide: Influences of other gases, Philos. T. R. Soc. B, 367, 1256–1264, https://doi.org/10.1098/rstb.2011.0377, 2012.
Powlson, D. S., Whitmore, A. P., and Goulding, K. W. T.: Soil carbon sequestration to mitigate climate change: A critical re-examination to identify the true and the false, Eur. J. Soil Sci., 62, 42–55, https://doi.org/10.1111/j.1365-2389.2010.01342.x, 2011.
Raji, S. G. and Dörsch, P.: Effect of legume intercropping on N2O emissions and CH4 uptake during maize production in the Great Rift Valley, Ethiopia, Biogeosciences, 17, 345–359, https://doi.org/10.5194/bg-17-345-2020, 2020.
Rochette, P., Angers, D. A., Bélanger, G., Chantigny, M. H., Prévost, D., and Lévesque, G.: Emissions of N2O from Alfalfa and Soybean Crops in Eastern Canada, Soil Sci. Soc. Am. J., 68, 493–506, https://doi.org/10.2136/sssaj2004.4930, 2004.
Ruan, L. and Philip Robertson, G.: Initial nitrous oxide, carbon dioxide, and methane costs of converting conservation reserve program grassland to row crops under no-till vs. conventional tillage, Glob. Chang. Biol., 19, 2478–2489, https://doi.org/10.1111/gcb.12216, 2013.
Rusinamhodzi, L., Corbeels, M., Van Wijk, M. T., Rufino, M. C., Nyamangara, J., and Giller, K. E.: A meta-analysis of long-term effects of conservation agriculture on maize grain yield under rain-fed conditions, Agron. Sustain. Dev., 31, 657–673, https://doi.org/10.1007/s13593-011-0040-2, 2011.
Saggar, S., Jha, N., Deslippe, J., Bolan, N. S., Luo, J., Giltrap, D. L., Kim, D. G., Zaman, M., and Tillman, R. W.: Denitrification and N2O: N2 production in temperate grasslands: Processes, measurements, modelling and mitigating negative impacts, Sci. Total Environ., 465, 173–195, https://doi.org/10.1016/j.scitotenv.2012.11.050, 2013.
Scheer, C., Fuchs, K., Pelster, D. E., and Butterbach-Bahl, K.: Estimating global terrestrial denitrification from measured N2O:(N2O + N2) product ratios, Current Opinion in Environmental Sustainability, 47, 72–80, https://doi.org/10.1016/j.cosust.2020.07.005, 2020.
Schmidt, H. P., Kammann, C., Hagemann, N., Leifeld, J., Bucheli, T. D., Sánchez Monedero, M. A., and Cayuela, M. L.: Biochar in agriculture – A systematic review of 26 global meta-analyses, GCB Bioenergy, 13, 1708–1730, https://doi.org/10.1111/gcbb.12889, 2021.
Shakoor, A., Shahbaz, M., Farooq, T. H., Sahar, N. E., Shahzad, S. M., Altaf, M. M., and Ashraf, M.: A global meta-analysis of greenhouse gases emission and crop yield under no-tillage as compared to conventional tillage, Sci. Total Environ., 750, https://doi.org/10.1016/j.scitotenv.2020.142299, 2021.
Tian, H., Xu, R., Canadell, J. G., Thompson, R. L., Winiwarter, W., Suntharalingam, P., Davidson, E. A., Ciais, P., Jackson, R. B., Janssens-Maenhout, G., Prather, M. J., Regnier, P., Pan, N., Pan, S., Peters, G. P., Shi, H., Tubiello, F. N., Zaehle, S., Zhou, F., Arneth, A., Battaglia, G., Berthet, S., Bopp, L., Bouwman, A. F., Buitenhuis, E. T., Chang, J., Chipperfield, M. P., Dangal, S. R. S., Dlugokencky, E., Elkins, J. W., Eyre, B. D., Fu, B., Hall, B., Ito, A., Joos, F., Krummel, P. B., Landolfi, A., Laruelle, G. G., Lauerwald, R., Li, W., Lienert, S., Maavara, T., MacLeod, M., Millet, D. B., Olin, S., Patra, P. K., Prinn, R. G., Raymond, P. A., Ruiz, D. J., van der Werf, G. R., Vuichard, N., Wang, J., Weiss, R. F., Wells, K. C., Wilson, C., Yang, J., and Yao, Y.: A comprehensive quantification of global nitrous oxide sources and sinks, Nature, 586, 248–256, https://doi.org/10.1038/s41586-020-2780-0, 2020.
Turmel, M. S., Speratti, A., Baudron, F., Verhulst, N., and Govaerts, B.: Crop residue management and soil health: A systems analysis, Agric. Syst., 134, 6–16, https://doi.org/10.1016/j.agsy.2014.05.009, 2015.
Wang, H., Yan, Z., Ju, X., Song, X., Zhang, J., Li, S., and Zhu-Barker, X.: Quantifying nitrous oxide production rates from nitrification and denitrification under various moisture conditions in agricultural soils: Laboratory study and literature synthesis, Front. Microbiol., 13, https://doi.org/10.3389/fmicb.2022.1110151, 2023.
Wang, Y., Guo, J., Vogt, R. D., Mulder, J., Wang, J., and Zhang, X.: Soil pH as the chief modifier for regional nitrous oxide emissions: New evidence and implications for global estimates and mitigation, Glob. Chang. Biol., 24, e617–e626, https://doi.org/10.1111/gcb.13966, 2018.
Weldon, S., van der Veen, B., Farkas, E., Kocatürk-Schumacher, N. P., Dieguez-Alonso, A., Budai, A., and Rasse, D.: A re-analysis of NH4+ sorption on biochar: Have expectations been too high?, Chemosphere, 301, 134662, https://doi.org/10.1016/j.chemosphere.2022.134662, 2022.
Wickham, H.: ggplot2: Elegant Graphics for Data Analysis, Springer-Verlag, New York, https://doi.org/10.1007/978-0-387-98141-3, 2016.
Wortmann, C. S. and Eledu, C. A.: Uganda's Agroecological Zones: A guide for planners and policy makers, CIAT, https://hdl.handle.net/10568/54311, 1999.
Wrage-Mönnig, N., Horn, M. A., Well, R., Müller, C., Velthof, G., and Oenema, O.: The role of nitrifier denitrification in the production of nitrous oxide revisited, Soil Biol. Biochem., 123, A3–A16, https://doi.org/10.1016/j.soilbio.2018.03.020, 2018.
Yao, Z., Guo, H., Wang, Y., Zhan, Y., Zhang, T., Wang, Zheng, X., Butterbach-Bahl, K.: A global meta-analysis of yield-scaled N2O emissions and its mitigation efforts for maize, wheat, and rice, Glob. Change Biol., 30, e17177, https://doi.org/10.1111/gcb.17177, 2024.
Zhang, L., Jing, Y., Chen, C., Xiang, Y., Rezaei Rashti, M., Li, Y., Deng, Q., and Zhang, R.: Effects of biochar application on soil nitrogen transformation, microbial functional genes, enzyme activity, and plant nitrogen uptake: A meta-analysis of field studies, Glob. Change Biol., 13, 1859–1873, https://doi.org/10.1111/gcbb.12898, 2021.
Žurovec, O., Sitaula, B. K., Čustović, H., Žurovec, J., and Dörsch, P.: Effects of tillage practice on soil structure, N2O emissions and economics in cereal production under current socio-economic conditions in central Bosnia and Herzegovina, PLoS One, 12, https://doi.org/10.1371/journal.pone.0187681, 2017.
Zuur, A. F., Ieno, E. N., Walker, N., Saveliev, A. A., and Smith, G. M.: Mixed effects models and extensions in ecology with R, Springer New York, New York, NY, https://doi.org/10.1007/978-0-387-87458-6, 2009.





