the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Pb and Fe flow through the mire-lake complex of Skogaryd catchment – a system under anthropogenic influence
Signe Lett
Leif Klemedtsson
Delia Rösel
Louise Rütting
Katja Salomon Johansen
Tobias Rütting
Sphagnum-dominated peatlands store not only vast amounts of carbon (C) but also metals derived from bedrock and anthropogenic sources. Some metals are involved in peat C stability, and some are toxic to living organisms. However, the pools of such metals and their export in relation to the export of C in the form of dissolved organic C (DOC) in mire-lake complexes have received little attention. We characterized a hemiboreal mire in Sweden previously exposed to heavy metal pollution in terms of physiochemistry, age, total C pool, lead (Pb) and iron (Fe) content. We investigated export of Fe and Pb in relation to DOC in the mire-lake complex. We found strong and linear correlations of Fe and DOC export in streams and the export occurred in similar ratios from both the mire and the downstream lake. The export of Fe and DOC seemed to be controlled by hydrological connectivity, while the production of DOC and mobilisation of Fe and Pb most likely was controlled by drought conditions. We found the Pb content exceeded threshold values for toxicity in the top layer of the peat and in stream water leaving the mire. Stream water concentrations of Pb were, as for Fe and DOC, highest after drought periods. Lead isotope analysis revealed that Pb leaving the mire accumulated through anthropogenic contamination (most likely from gasoline), while Pb leaving the lake appeared to contain natural sources as well. In addition, the lake appeared to be a sink for the anthropogenic Pb exported from the mire through sedimentation. This study suggests that peat decomposition in peatlands affected by climate change may pose a risk to not only losing stored C, but also through release of heavy metals to the local environment.
- Article
(3522 KB) - Full-text XML
-
Supplement
(905 KB) - BibTeX
- EndNote
Peatlands hold vast amounts of organic carbon (C), equivalent to 20 %–30 % of the global pool of soil C (Gorham, 1991). Peat is formed over thousands of years and is preserved in the deep peat layers under waterlogged and anoxic conditions (Wilson et al., 2016). Large pools of metals from both atmospheric deposition and parent bedrock are bound in peat. Some metals like iron (Fe) are involved in peat stabilisation and degradation (Qin et al., 2022; Wang et al., 2017) and some metals like lead (Pb) are toxic to living organisms (Rothwell et al., 2005; Tipping et al., 2003). Climate change is expected to cause longer periods of drought in the boreal zone (Helbig et al., 2020) with a concurrent decrease in precipitation during summer and an increase during winter (Dore, 2005; IPCC, 2021). As a result, peatlands in the boreal zone will experience periodically lower water table levels, particularly in summers, and more extreme rain events (Zhong et al., 2020). These changes to peatland hydrology may expose the deeper peat to oxygen and promote C turnover (Chen et al., 2020; Fenner and Freeman, 2011; Zhang et al., 2025). This will lead to loss of C from peatlands in different forms, such as CO2 from microbial respiration, but also in the form of dissolved organic carbon (DOC) that is lost through water streams (Prijac et al., 2023). This loss of DOC can stimulate metal export from peatlands as metals tend to be co-exported with DOC. Changes in peatland hydrology and climate are known to affect the export of DOC and metals from peatlands (Aleshina et al., 2024; Broder and Biester, 2017; Prijac et al., 2023; Zhong et al., 2020), but whether the concentrations of heavy metals reach concentrations that require attention in relation to ecosystem function both in peatlands and downstream catchments is not well understood.
Sphagnum mosses dominate the vegetation in boreal peatlands and are the main contributors to peat formation. Acidic and phenolic compounds in the Sphagnum cell wall provide a high cation exchange capacity (CEC) and Sphagnum and Sphagnum-derived peat efficiently bind positively charged ions such as metals (Clymo, 1963; Verhoeven and Liefveld, 1997; Zhao et al., 2023). As such, peatlands are efficient sinks for natural and anthropogenic derived metals (Roux et al., 2004; Shotyk et al., 1996). In addition to the recalcitrant litter of Sphagnum, the environmental conditions of peatlands such as cold temperatures and waterlogged conditions contribute to peat accumulation and thus metal sequestration (Aerts et al., 1992; Moore and Basiliko, 2006; Thomsen et al., 2025). Distribution of metals in the vertical peat profile from the living top layer to the deep peat depends on the source of the metal. Iron is one of the most abundant metals in peatlands and is mainly derived from bedrock, with a relatively small contribution from wind-blown dust (Osborne et al., 2024; Steinnes et al., 2005). Therefore, Fe concentrations typically increase with peat depth.
During the last decade, Fe has been increasingly investigated because of two proposed mechanisms influencing peat stability. Firstly, the “iron gate” theory suggests that oxidation of Fe, which occurs during water-table decline, induces complexation between Fe and lignin-like compounds (Wang et al., 2017). This complexation seemingly leads to resistance against mineralization (Riedel et al., 2013; Wang et al., 2017). However, the degree to which Fe-bound peat is protected is not entirely clear and the protecting effect of Fe may be limited to static oxic conditions (Chen et al., 2020). At the same time, oxic conditions can also promote production of DOC and release of CO2 from OM through abiotic reactions in addition to microbial breakdown and mineralization (Brouns et al., 2014; Fenner and Freeman, 2011; Qin et al., 2022; Strack et al., 2008). Specifically, abiotic Fe-mediated reactions occur in the presence of oxygen and DOC (Miles and Brezonik, 1981) and have been shown to be relevant in peat (Thomsen et al., 2025) and industrial biorefineries (Peciulyte et al., 2018) leading to a substantial contribution of CO2 production through decarboxylation processes (Page et al., 2014; Trusiak et al., 2018). These reactions depend on Fe's ability to cycle between redox states (Koppenol and Hider, 2019) and primarily occur at the oxic-anoxic interface around the water table (Qin et al., 2022; Wang et al., 2022; Yu and Kuzyakov, 2021). Therefore, changing water-table level influences DOC concentrations and CO2 release from peat. In that regard, it is also important to mention that changes in redox conditions affect the solubility of Fe and DOC even without degradation taking place (Knorr, 2013). Understanding how fluctuating exposure to oxygen will affect DOC and Fe export from peatlands should therefore be considered in relation to future climate changes when assessing mire C dynamics.
Toxic heavy metals such as Pb are anthropogenically sourced through deposition and are generally found in the top peat layers. Lead has been one of the most prominent heavy metals in a variety of industries (Tchounwou et al., 2012) and Pb pollution can be traced in different depositional environments such as lake and offshore sediments as well as peat and peatland vegetation (Bindler et al., 1999; Bränvall et al., 2001; Kylander et al., 2013; Renberg et al., 2000). In addition, peatlands sequester a larger proportion of heavy metals compared to other ecosystems (McCarter et al., 2024). The primary recent anthropogenic sources of Pb in mires is from car fuel and ship paint that increased at the start of the industrial revolution and peaked in the 1970s (Marcantonio et al., 2000; Renberg et al., 2000). The isotopic composition of Pb can be used to fingerprint the source (anthropogenic versus natural) of Pb and even differentiate between contamination sources (Gmochowska et al., 2019; Jeong et al., 2023). Lead is toxic to microorganisms, plants and humans and accumulates within ecosystems (Collin et al., 2022). Identifying the source of Pb can indicate whether the problem is from persistent contamination from human activities and could pose an ecological risk. The maximum allowable concentration in waters for Pb as set by the European Union is 14 µg L−1, in sediments 163 mg kg−1 dry weight and in biota 110 µg kg−1 wet weight (EU directive, 2013). Due to the toxic nature of Pb, its mobility is important to understand, particularly in potentially Pb polluted peatlands from which Pb can be mobilised and exported to downstream catchments (Choudhury and Panda, 2005; Doelman and Haanstra, 1979; Shakya et al., 2008). A large proportion of pollution derived Pb deposited in Swedish peatlands will eventually end up in downstream ecosystems such as lake sediments (Brännvall et al., 2001), where it will accumulate and potentially reach harmful concentrations for benthic living organisms.
Several key processes exhibit control on metal export from peatlands, such as pH and the different metal's binding affinity to DOC, but hydrological connectivity that refers to the transport of water from peat to stream, and seasonality, which includes drought and precipitation events, seem to be most important (Broder and Biester, 2015, 2017; Prijac et al., 2023). In bogs, DOC is primarily produced from decomposition of OM, which is likely the main mobilisation process of Fe and Pb (Broder and Biester, 2017). Temperature is important for DOC production as it affects decomposition rates (Rosset et al., 2022), but the most important factor is likely oxygen availability that is linked to water table depth and enables degradation of accumulated lignin-like compounds in the peat (Fenner and Freeman, 2011; Freeman et al., 2001). Water-table decline primarily occurs during warm and dry periods during summer and early fall. Typically, at this time of year the highest export and stream water concentrations of DOC, Fe and Pb are observed and particularly in relation to high discharge events where hydrological connectivity between peatland and stream re-establishes (Broder and Biester, 2015, 2017; Clark et al., 2007; Prijac et al., 2023). Lead is very immobile in peat and is therefore primarily bound in the top parts of peat (Kharanzhevskaya et al., 2023; Mariussen et al., 2017; Okkenhaug et al., 2018), while redox active metals such as Fe will change mobility depending on redox conditions that occur from water-table fluctuations (Knorr, 2013; Strack et al., 2008). Nevertheless, both metals have a high binding affinity for OM and are commonly found to correlate with DOC in water streams leaving peatlands, a relationship that is partly explained by high binding affinity (Aleshina et al., 2024; Neubauer et al., 2013; Okkenhaug et al., 2018; Vile et al., 1999). Dissolved organic matter from peatlands might even have a higher binding affinity to metals due to high contents of polyphenolic compounds derived from Sphagnum moss, compared to DOC supplied from other types of catchments (Kaal et al., 2017). The strong correlation between Pb and DOC is likely important for the total export of Pb from peatlands, but it could also have implications for the retention of Pb in downstream systems. Therefore, the expected increase in drought periods and extreme precipitation events in light of climate change, might lead to higher exports of DOC, Fe and Pb from peatlands (Broder and Biester, 2015; IPCC, 2021; Zhong et al., 2020).
The overall aim of this study was to investigate the relationship between export and stream water concentrations of DOC, Fe and Pb in a mire-lake complex and assess if Fe and Pb occurred in concentrations high enough to have potential effects on ecosystem functioning. In addition, we characterized the mire in terms of physiochemistry, age and total C content. The studied mire-lake complex is located inland from the west coast of Sweden and is subjected to recent climate change and exposed to heavy metal pollution. The mire-lake complex is part of Swedish research infrastructure for ecosystem research that provides continuous data on soil, air and water status. Based on previous findings (Aleshina et al., 2024; Broder and Biester, 2015, 2017), we expected to find strong correlations between Fe, Pb and DOC in stream water from the mire. In addition, we expected the concentrations would be highest following drought periods when hydrological connectivity between mire and stream would re-establish (Broder and Biester, 2015, 2017). Also, specifically for the investigated mire-lake complex, we wanted to investigate if Pb would amount to any concentration of ecological risk in relation to EU guidelines for freshwater threshold values and in that regard determine the source of Pb pollution. In relation to Fe, we wanted to assess if the proposed mechanism of peat stability and peat destabilisation could be relevant in the studied mire-lake complex. In comparison to most previous studies, this study presents a 4 year period of export and stream water concentration data including one year with unusual drought (2018). This long-term dataset can help identify patterns and give a more stable view of export trends in the studied system and the mechanisms behind.
2.1 Site description
This study was conducted for the period 2017–2021 in Mycklemossen mire and Erssjön lake (58°21′ N, 12°10′ E; 80 m a.s.l.), which are part of the Skogaryd research catchment in the hemiboreal forest zone in southwestern Sweden. The Skogaryd research catchment is part of the Swedish Infrastructure for Ecosystem Science (SITES). Air temperatures range between −0.8 °C in February and 16.8 °C in July and precipitation is highest in July to December with approximately 75 mm per month and driest January to March with approximately 54 mm per month (Swedish Meterological and Hydrological Institute normal means for the period 1991–2020, Fig. S1 in the Supplement). During the survey period, the highest annual precipitation was in 2019 and 2020 with 1150 and 1100 mm annually, respectively. The lowest annual precipitation was 580 mm in 2017 (https://meta.fieldsites.se/resources/stations/Skogaryd, last access: 22 August 2025). Due to climate change, summers (June, July and August) at the west coast of Sweden are predicted to receive 7 % less rain in June–August, 13 %–18 % more rain in December to February and annually be 2.2–3.1 °C warmer by 2041–2060 in reference to preindustrial levels (IPCC, 2021).
Mycklemossen mire is a nutrient-poor fen with a surface topography composed of a mosaic of hummocks and hollows, referring to parts of the vegetation found above and below the water table respectively, and transition zones called “intermediate” in our study, with hummocks as the dominating topography (Rinne et al., 2022). The hummocks are mainly dominated by Eriophorum vaginatum, Calluna vulgaris and Erica tetralix and the hollows consist of different Sphagnum species, mainly S. rubellum, S. fallax and S. austinii as well as Rhyncospora alba, and the peat samples were sampled approximately within a square-meter plots (Kelly et al., 2021). Two streams lead away from Mycklemossen. One runs from the north into the lake Erssjön (this stream is hereafter referred to as S1) and the other runs into a neighbouring catchment. Erssjön is 0.061 km2, 4.5 m deep in the deepest part and has characteristics of a mesotrophic lake. Erssjön is sourced from Mycklemossen and from a neighbouring mostly forested catchment and drains through a stream (hereafter referred to as S6). The total catchment area of Mycklemossen (S1) is 0.595 km2 and the catchment of Erssjön (S6) is 1.337 km2 (Table S1 in the Supplement). The sizes of the catchments have been generated based on detailed topographic maps in relation to the location of the measuring stations (out-flow). The Skogaryd Research Catchment is located in one of the densest populated areas in Sweden and east of a highway and a former industrial ship wharf. In Europe, gasoline contained Pb from 1922 until it was banned in 1998. Mycklemossen is downwind from a potential source of industrial pollution, Uddevalla ship wharf. The ship wharf actively produced ships in the years between 1946 and 1985 and used Pb-containing antifouling paint. The area can therefore be expected to have been subjected to considerable Pb pollution during a period of 70 years.
2.2 Peat sampling
On 3 March 2021, one peat core of 4.5 m depth was sampled at five locations in three topography types: hummock, intermediate and hollow with 1–2 m apart. This resulted in 15 peat cores in total. The five locations were chosen to represent Mycklemossen (Fig. 1). The top 50 cm of the peat was sampled with a hand saw. The deeper peat was collected with a stainless-steel, cylinder-shaped Russian peat corer (inner dimensions: 5 cm in diameter). Just after sampling, the peat cores were sliced into 5 cm segments and frozen.
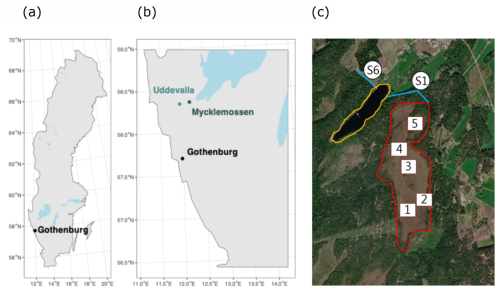
Figure 1(a) Map of Sweden, (b) Mycklemossen mire located east of Uddevalla where a ship wharf was active until 1985, (c) the mire-lake complex comprising of Mycklemossen mire (encircled in red) and lake Erssjön (encircled in yellow). Sampling points in Mycklemossen are marked as 1–5 and the stream sampling location from Mycklemossen are marked as S1 and the stream from Erssjön as S6. Satellite photo from https://meta.fieldsites.se/resources/stations/Skogaryd (last access: 28 April 2025).
2.3 Radiocarbon dating of peat
Peat age was determined by radiocarbon dating in samples from 20–25, 120–125, and 420–425 cm depth from hummock, intermediate and hollow from sampling location 3 (Fig. 1c). Samples were freeze-dried and homogenized prior to 14C analysis at the Tandem laboratory (Uppsala, Sweden), using a MICADAS accurate accelerator mass spectrometer, following routine sample preparation by the tandem laboratory (Salehpour et al., 2013).
2.4 Physical and chemical characterization of peat
For all 15 peat cores (locations 1–5, Fig. 1c) the top 5 cm of each 100 cm increment from 0–5 cm below surface to 400 cm (0–5, 100–105, 200–205, 300–305, and 400–405 cm) were analysed for physical and chemical properties. pH was analysed in filtered peat pore water (Metrodam 691 pH Meter; Switzerland). Subsamples of peat were oven-dried at 40 °C until constant mass. Dry matter (DM) content was used to determine peat bulk density. Soil organic matter content (SOM) was determined from loss on ignition at 550 °C for 8 h in a muffle furnace (Heraeus Instruments). For total C, total N, δ13C and δ15N, dried samples were ground and weighed (5 mg) into tin capsules and analysed with a GSL elemental analyzer coupled to an isotope ratio mass spectrometer (IRMS, Sercon 20–22, Sercon Ltd., UK).
2.5 Total pools of C and N in mire
To map the three-dimensional layout of the mire, bathymetry, which is based on ground-penetrating radar was performed. The radar measurements were carried out with Malå Geoscience Ramac ground-penetrating radar with a shielded 250 MHz antenna. Measurements were collected at a sampling frequency of 2600 MHz and radar depth scans were made every 0.1 m in the different transects. For the radar measurements, the mire was divided with 4 lines from south to the north, and 10 lines east to west, forming a grid of 27 intersections, which were surveyed. The radar measurements were calibrated against 5 soil cores taken in grid intersections using a “Russian peat corer”. Bathymetry data was illustrated with QGIS 3.18 and the data is available on the SITES data portal (https://hdl.handle.net/11676.1/lmj-WqszXOi5T_XafhHU75Ps, last access: 27 February 2025). The bathymetry data were combined with peat bulk density and C and N content to calculate total C and N in increments of 50 cm depth. Specifically, total soil volume layer (m3) at each 50 cm increment was multiplied by depth-specific peat bulk density (g m−3) and the average fraction of C or N.
2.6 Heavy metals in peat cores
The concentration of Pb that is considered one of the highest ranked heavy metals to consider for public health (Tchounwou et al., 2012) and Fe were quantified in technical triplicates of the peat samples at 10–15, 15–20, 20–25, 120–125, and 420–425 cm depth with inductively coupled plasma-mass spectrometry (ICP-MS) (iCAP, Thermoscientific) (Table S2). Prior to analysis, peat samples were freeze-dried and homogenized with a mortar and pestle. A subsample of 0.3 g was weighed into a Teflon bomb and 10 mL of concentrated nitric acid (70 %) was added. The samples were heated in a microwave with a ramping phase of 20 min and a hold phase of 25 min at 180 °C. After cooling, the solutions were first diluted to 50 mL with MilliQ water and further diluted 10-fold before analysis.
2.7 Concentrations and export of DOC and metals in stream water
Manual grab samples from the streams were taken fortnightly during the ice-free period for chemical analysis. Water samples were filtered through 0.7 µm pore size filters prior to analysis. DOC was measured via spectrometry Shimadzu TOC-VCPH. Lead and Fe concentrations were analysed using an Agilent 8800 Triple Quadrupole ICP-MS at the Microgeochemistry Laboratory, University of Gothenburg. Samples, reference waters, and blanks were prepared in 2 % HNO3 with internal standards (Re, Rh, Ge) added for drift and matrix correction. The ICP-MS operated in MS/MS mode with N2O as the reaction gas. Each analyte was analyzed in four replicates of 25 sweeps at three points per peak (0.02 a.m.u. spacing). Pb was measured on mass (206Pb) and Fe as oxide (56→72Fe), with dwell times of 0.5 and 0.3 s respectively. Quantification was done in the MassHunter Workstation Software v4.4. using a six-point calibration curve (0–1000 µg L−1) from the Periodic Table Mix 1 (TraceCERT®, Sigma Aldrich).
Discharge calculations were based on continuous stream level measurements using an ISCO 2110 ultrasonic flow module (Mire north, S1) and Mjk 1400 0–1 m (Erssjön outlet, S6), respectively. Location-specific stream level discharge relation curves have been established. The data were gap-filled when necessary either via linear interpolation for shorter gaps, or by cross-correlations with nearby measurement locations within the Skogaryd catchment for longer gaps. Longer gaps usually occurred during low-flow conditions. The export of DOC and metals were calculated for each manual water sampling as the product of discharge and concentration. Linear interpolations between sampling dates were done to estimate annual export rates.
2.8 Pb isotope ratios in stream water
To determine the source of Pb in the mire catchment the composition of Pb isotopes were determined in stream water from both the mire and lake from the manual grab samples collected between February 2022 and May 2024 (N=25) for the mire, and between May 2022 and May 2024 (N=21) for the lake, respectively. The Pb isotopic composition was analysed in the Microgeochemical Laboratory at the University of Gothenburg using an Agilent 8800 ICP-MS/MS coupled to an ASX-500 autosampler, a peristaltic pump, a concentric glass nebulizer (MicroMist) and a Scott double-pass quartz glass spray chamber. The autosampler was placed in an ISO 5 clean room and was connected to the ICP-MS in the adjacent laboratory with an approximately 1 m long tubing. All sample preparations were performed in the ISO 5 clean room using Milli-Q water and ultrapure nitric acid (NORMATOM®, VWR chemicals). The ICP-MS was run in MS/MS mode with N2O as reaction gas. A comprehensive description of Pb isotopic analysis is given in the supplementary information together with a summary of ICP-MS/MS settings, acquisition parameters and reference materials used (Table S3–S5). Uncertainties of the Pb isotopic composition are reported at 2 s and include quadratic addition of the standard error for the 10 replicates, the excess scatter of the primary reference solution NIST SRM 981 for the respective ratio from the measurement session and the uncertainty in the 204Hg correction. The uncertainties from the published ratios for the primary reference material (approximately 0.04 %; NIST SRM 981; Cantanzaro et al., 1968) would need to be propagated to add systematic uncertainties. These uncertainties are considerably smaller than the random uncertainties (approximately 0.30 %) and thus do not significantly contribute to the total uncertainties when added in quadrature.
To determine the fate of anthropogenic Pb in the mire-lake complex, we applied an isotope mixing model (Eq. 1). Assuming Erssjön receives Pb from two different sources, water from Mycklemossen and other sources from the surrounding forest with distinct isotopic value the mixing model used is:
With R being the isotope ratio of Pb and f being the relative contribution of the two sources, with f1+f2 equal to 1. The model was applied both with the and ratios, using the IsoError tool (Phillips and Gregg, 2001) that provides mean contributions in addition to an uncertainty estimate. 11 samples from a forest stream within the Skogaryd catchment were analysed in 2023.
2.9 Data handling and statistics
Data were analysed and visualized in R version 4.3.3 (R Core Team, 2024). For parameters measured in peat, we tested the effects of depth and topography type using mixed-effects models with the nlme package (Pinheiro et al., 2023). Depth, topography type and their interaction were included as fixed, independent predictors. For all dependent variables, except pH, depth was included as an orthogonal quadratic polynomial term. Sampling location was included as a random factor to take the nested sampling design into account. The relationships between annual export of DOC and annual export of Fe and Pb were tested with linear regression including DOC and stream identity as independent variables. For the regression model with Pb as dependent variable, there was a significant interaction, and the model was run separately for the two streams.
3.1 Age, physical and chemical parameters of Mycklemossen mire
The maximum depth of Mycklemossen was about 600 cm (Fig. 2a) and the age of the mire centre was determined to be at least 3900 years by 14C dating at 420–425 cm depth (Table S6). The total surface area of the mire was 0.23 km2 in 2021. For the deepest part, the 550–600 cm depth interval, the area was only 0.0069 km2 which is 0.03 % of the total area. The total C pool of Mycklemossen was 34 806±1633.23 t of which 18 % (6340±4.76 t) was in the top 50 cm of the mire (Fig. 2b). The C and N pool declined to 1570 t±2.28 and 35 t±48 at 550–600 cm depth, respectively.
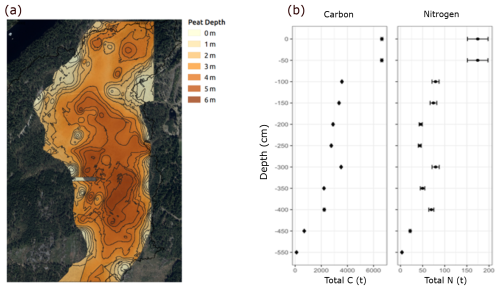
Figure 2Spatial distribution of peat depth based on radar bathymetry (a) and total C and N pools in 50 cm depth increments (b) in Mycklemossen.
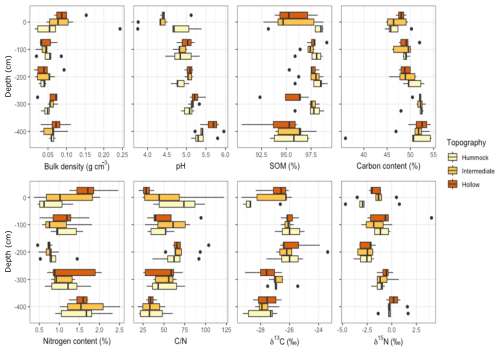
Physicochemical parameters were measured to a depth of 400 cm. Peat bulk density (g cm−3) significantly decreased with depth from 0.085±0.023 at the surface to 0.04±0.009 at 200 cm depth and then increased to 0.064±0.011 to 400 cm (p=0.001, Fig. 3 and Table S7). The SOM content was above 90 % at all depths and locations and was highest in hummock. The SOM content for intermediate and hollow was lowest at the mire surface and at 400 cm depth, and highest at 200 cm depth (p=0.04, Table S7). Peat pH was lowest at the mire surface and increased with depth from the surface to 400 cm depth, spanning from a pH of 4.5±0.18 to 5.5±0.11 (Fig. 3). The increase in pH with depth was most pronounced for the hollow topography (p=0.04, Fig. 3 and Table S7). Peat C content increased slightly with depth from around 47±0.8 % at the top to 51±1.9 % at 400 cm, while N content decreased from 1.27±0.23 % at the surface of the mire to 0.78±0.01 % in 200 cm depth and increased to 1.59±0.21 % at 400 cm depth (p=0.001, Table S7). The change in N with depth was less extreme for hummock compared to intermediate and hollow (p=0.04, Fig. 3 and Table S7). Peat δ13C increased from ‰ at the mire surface to ‰ at 100 cm depth followed by a decrease to ‰ at 400 cm depth (p=0.001, Fig. 3 and Table S7). Peat δ15N decreased from ‰ at the surface to ‰ at 200 cm depth and then increased to 0.06±1.18 ‰ at 400 cm depth (p=0.001, Fig. 3 and Table S7).
3.2 Spatial distribution of heavy metals and Fe in Mycklemossen
Peat Pb content was highest in the top of the mire and was essentially absent in peat samples at 420–425 cm depth (Fig. 4). The highest Pb content was generally measured in intermediate, which was more than twice as high as in hollows, while the single highest measurement of Pb content was measured in hummock at 25–50 cm depth. Specifically in hummocks, the Pb content increased from 28.26 mg kg−1 in 15–20 cm depth to 91.8 mg kg−1 at 25–50 cm depth (Fig. 4 and Table S2). In intermediate and hollow topographies Pb content was highest at 15–20 cm: 64.25 and 32.21 mg kg−1 and decreased to 58.5 and 14.89 mg kg−1 at 25–50 cm depth, respectively (p=0.02, Fig. 4 and Table S8). Peat Fe concentrations at the top of the mire were between 606 and 1237 mg kg−1 and were measured to between 1434 and 1474 mg kg−1 at 120 cm depth (Fig. 4, Table S2). At 420–425 cm depth, Fe content was between 7147 and 8481 mg kg−1 and contents were similar between topography types.
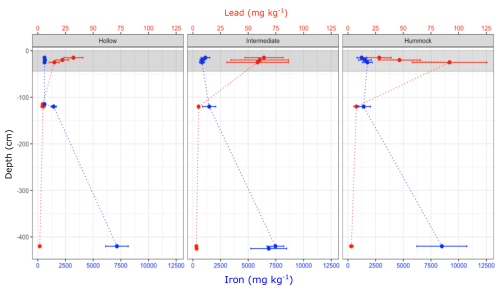
Figure 4Pb (red) and Fe (blue) concentrations (mg kg−1 DM) in peat at various depths of Mycklemossen. Points are means of 5 sampling locations and error bars are SE. Dotted lines are included for illustration only. Grey bands indicate the zone affected by water-table fluctuations measured as absolute minimum and maximum water-table depth during the period 2017–2021. For statistics see Table S8.
3.3 Pb, Fe and DOC concentrations in export water from Mycklemossen
The discharge from Mycklemossen (S1) was lowest during summer and highest in winter (Fig. 5). The water table concurrently decreased during summer and caused periods without discharge from S1. Stream water concentration of Pb, Fe and DOC in water from S1 were highest during summer at low discharge and low water table level (Fig. 5). The highest concentrations were in general measured during summer after a drought period when discharge became measurable again. Stream water pH increased at low water table depth during summer and decreased again in winter (Fig. S2). The seasonal tendencies in Fe, Pb and DOC concentrations seemed alike, but did not follow the same pattern all the way through the study period. Lead concentrations in S1 were highest in 2018 at the end of summer and after a long drought period and the Pb concentration measured was almost twice as high as in 2017 and 2019. During the study period, Pb concentrations varied between 0.4 and 14.3 µg L−1 (Fig. 5). Iron also peaked in 2018 at 4.6 mg L−1, which was clearly higher than the other years, whereas DOC peaked in the summer of 2019.
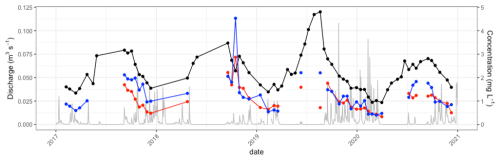
Figure 5Stream discharge (grey), and water concentration of Fe (blue) and Pb×200 (red) and C in DOC×0.05 (black) from S1. Discharge shows daily averages of measurements made every 15 min, whereas concentrations in water were analysed from grab samples usually every 2–3 weeks during high flow and throughout 2019. Dots mark timing of sampling. Data points for Fe and Pb were removed when discharge <0.0001 (m3 s−1), which occurred during summer when the water level was too low to measure discharge, making calculation of export impossible.
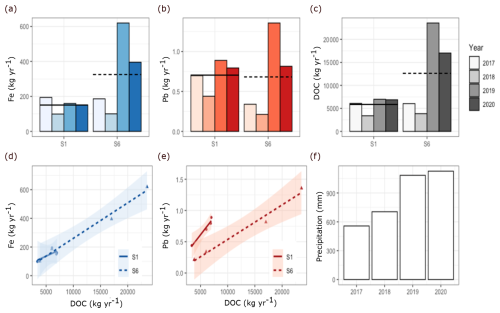
Figure 6Annual export of DOC, Fe and Pb from Mycklemossen to Erssjön lake (S1) and from Erssjön (S6) in 2017 to 2020 (a–c) and the linear relationship between export of DOC and export of Fe and Pb (d, e). Horizontal lines in bar chart are means of the years 2017–2020 in S1 (solid line) and S6 (hatched line). Note differences in scale between plots. Annual precipitation for the years 2017–2020 (f).
3.4 Export of DOC, Fe, and Pb from Mycklemossen and Erssjön Lake
During the period 2017–2020 the average annual DOC export was 5834 kg from the north part of Mycklemossen (S1) and 12 616 kg leaving Erssjön (S6) (Table S1). The DOC export varied considerably between years and especially from Erssjön (Fig. 6). The highest DOC export from both Mycklemossen and Erssjön was in 2019, while the lowest export was in 2018. On average, annual DOC export from Erssjön in 2017–2020 was almost double of what it received from Mycklemossen, although this pattern was not consistent for individual years. In 2017 and 2018 DOC export from Mycklemossen and Erssjön was very similar, whereas in 2019 and 2020 export from S6 was around 3-fold higher than from S1. In 2017–2020, the average annual Fe export was 150 kg from S1 and 325 kg from Erssjön (S6) (Table S1). The annual Fe export from Erssjön was therefore 215 % higher than the input from Mycklemossen. The average annual export of Pb was 0.705 kg from Mycklemossen and 0.681 kg from Erssjön (Table S1).
Within streams, the inter-annual variation in Pb export mirrored the patterns observed for Fe and DOC. However, opposite to DOC and Fe, the export of Pb was only slightly higher from Mycklemossen than Erssjön. The annual export of DOC linearly correlated with annual Fe export in a similar way across the two streams (p<0.001, R2=0.96). DOC was also linearly correlated with Pb, but the correlation slopes differed between streams (p<0.001). Specifically, the annual DOC export in S6 was more than twice as high as the export from S1.
3.5 Pb isotopic composition
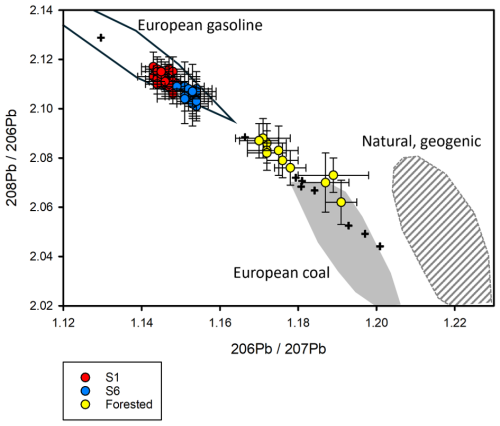
The Pb isotopic composition from the 57 water samples was analysed to determine whether the Pb exported from Mycklemossen and Erssjön was of anthropogenic origin (Table S9). The and composition from Mycklemossen (S1, n=25), Erssjön (S6, n=21) and forested catchment (s12, n=11) is plotted in Fig. 7, and for water leaving Mycklemossen, the ratio ranged between 2.106±0.004 (2se) and 2.117±0.006 (2se) and has a weighted mean ratio of 2.11175±0.00095 (n=25, MSWD=1.4). The ratio ranged between 1.143±0.004 (2Se) and 1.150±0.003 (2Se) and a weighted mean ratio of 1.1460±0.0008 (n=25, MSWD=1.5). For the water leaving Erssjön, the ratio ranged between 2.101±0.005 (2Se) and 2.109±0.010 (2Se) with a weighted mean ratio of 2.10690±0.0014 (n=21, MSWD=0.6). The ratio was between 1.149±0.004 (2Se) and 1.154±0.004 (2Se) with a weighted mean ratio of 1.1527±0.0009 (n=21, MSWD=0.4). For the forested neighbouring catchment, the ratio was between 2.062±0.009 (2Se) and 2.088±0.008 (2Se) with a weighted men ratio of 2.08010±0.0046 (n=11, MSWD=3.8). For the ratio, it ranged between 1.170±0.005 (2Se) and 1.191±0.004 (2Se) with a weighted mean of 1.778±0.0052 (n=11, MSWD=9). The isotope mixing model revealed that Mycklemossen contributed by 84.2±2.6 % of the total Pb load to Erssjön (S6), despite the much lower total annual discharge (Table S1).
Peatlands are crucial global C stores but are subjected to climate change that threatens the future stability of the peat (Zhong et al., 2020). Stored metals from natural and anthropogenic sources could become mobilised when climate change promotes peat degradation. Here we aimed to document the size, age and total C content of a northern European mire representing mires in the temperate and boreal transition zone and investigate the pools and export of DOC, Fe and Pb. Specifically, we wanted to investigate the export of Fe as an important metal influencing peat stability and to document the Pb pollution status in the mire and a downstream lake. We found clear indications that warm and dry periods with higher peat degradation followed by precipitation that re-established hydrological connectivity between mire and lake, were most important for release and export of Fe, Pb and DOC. Below, we discuss the context, magnitude and consequences of these findings.
4.1 Assessment of decomposition and C stability in Mycklemossen
The peat in Mycklemossen was dated to 3900 years in 4 m depth and is thus likely closer to 6000 years in the mire centre at almost 6 m deep. This is within the age range of mires in Sweden (Ehnvall et al., 2024). However, Mycklemossen is in the younger end of the range and seems to have accumulated peat at a faster rate compared to other Swedish mires (Andersson and Schoning, 2010; Hansson et al., 2024). The difference in dominant vegetation of hollow and hummock likely controls the difference in C to N ratio of the top layer, as hummock was mostly dominated by vascular plants (Eriophorum vaginatum, Calluna vulgaris and Erica tetralix) and the hollows mainly consist of different Sphagnum species as S. rubellum, S. fallax and S. austinii as well as Rhyncospora alba (Kelly et al., 2021). The higher C to N ratio in hummocks is likely due to higher lignin content from the vascular plants, as Sphagnum does not contain lignin (Biester et al., 2014; Weng and Chapple, 2010; Zeh et al., 2020). This effect was also reflected in the δ13C in the top layer, with slightly more 13C depletion at the hummock with Calluna vulgaris than hollow with Sphagnum dominance (Biester et al., 2014). Furthermore, plant-sourced C near the surface is more 13C depleted nowadays than before the beginning of the industrial age (“the Suess effect”) (Drollinger et al., 2019; Esmeijer-Liu et al., 2012). Below the top layer and the water table (8–25 cm depth; Rinne et al., 2022) in the permanently anaerobic zone, the slightly higher 13C enrichment may also reflect methane production activity as methanogens discriminate against 13C, which consequently enriches the peat in 13C (Keane et al., 2021; Krull and Retallack, 2000). Further down, the decreasing C to N ratio likely reflects increasing degree of degradation with depth as C is removed (Biester et al., 2014; Smeds et al., 2025). While the 15N enrichment increased with depth, reflecting discrimination during peat decomposition (Biester et al., 2014), the 13C on the other hand decreased with depth. This latter can be explained by climatic changes/variation during the Holocene, in particular changes in soil moisture which in turn affects photosynthetic 13C discrimination and consequently peat 13C (Kumar Das et al., 2024; Nykänen et al., 2020).
The Fe content in the peat increased about 10-fold from the top of the mire to 400 cm depth, supporting that Fe in the Mycklemossen is primarily sourced from bedrock and groundwater and to a small extent from deposition (Steinnes et al., 2005). 18 % of Mycklemossen C pool was in the top 50 cm, partly because of the large mire area at this depth interval and a higher peat bulk density in the top one meter of the mire. This part of the mire was exposed to oxic conditions during summers, which makes a large part of the peat susceptible to decay (Fenner and Freeman, 2011). However, static oxic conditions during summer could also stabilise peat and DOC through adsorption and complexation interactions with Fe (Riedel et al., 2013; Song et al., 2022; Wang et al., 2017), though most Fe in Mycklemossen is placed in deep anoxic peat layers. Nevertheless, Fe-OM complexes are formed in both oxic and anoxic peat (Bhattacharyya et al., 2018). The stabilising effect of Fe might therefore not be limited to the oxic layer. However, in the presence of oxygen, oxidative reactions catalysed by Fe can lead to production of hydroxyl radicals that can promote degradation of peat (Qin et al., 2022; Trusiak et al., 2018). Such reactions might be driven by small concentrations of Fe (between 280–2.300 mg kg−1 peat; Curtinrich et al., 2024), which is within the range of Fe measured in the top part of Mycklemossen. In the top part of Mycklemossen mire there might therefore be both stabilising and destabilising reactions with Fe taking place, but it is unclear how important the destabilising reactions compared to the stabilising reactions is for peat stability and mire C dynamics.
4.2 Export of Fe and DOC downstream of Mycklemossen
Iron exhibited strong and linear correlations with DOC in the streams from Mycklemossen (S1) and Erssjön (S6). The average ratio between DOC and Fe of 0.03 in both S1 and S6 is the same as the median ratio value reported for Fe and DOC ratios in Swedish lakes from a national investigation (Weyhenmeyer et al., 2014). The Fe-DOC export relationship in the mire-lake complex of Mycklemossen and Erssjön therefore likely represents similar ecosystems in Sweden. In general, Fe in boreal rivers is primarily transported in colloidal form (between 1 kDa and 0.22 µm), for instance as Fe-DOC complexes with humic substances, and Fe as Fe2+ or Fe(oxy)hydroxides and to a much lesser extent found as “truly dissolved” (<1 kDa) (Aleshina et al., 2024; Heikkinen et al., 2022).
In addition, water that leaves Sphagnum-dominated bogs is typically rich in polyphenolic DOM that is seemingly derived from Sphagnum and binds strongly to metals (Kaal et al., 2017). The mobility of the different forms of Fe is primarily affected by redox conditions (oxic/anoxic) and pH, whereas oxic conditions favor co-precipitation of DOC and Fe (Riedel et al., 2013) and acidic pH (4–5) favors soluble Fe-DOC complexes, while a pH of 6 and higher favors precipitation of Fe-DOC complexes (Neubauer et al., 2013). The Fe-DOC relationship reported in this study is probably best explained by colloidal transport of Fe-DOC complexes, with essentially all Fe bound to an organic ligand and a small proportion as Fe(oxy)hydroxides. Björnerås et al. (2021) investigated Fe inflow to a lake from a Mire in the south of Sweden. Here, Fe was found bound mainly in colloidal form, but also a noteworthy proportion as Fe(oxy)hydroxides, but the inflow water had a pH of 6.7 and above, which favors precipitation of Fe-DOC complexes (Björnerås et al., 2021). At the pH we measured (4–5), a higher fraction of soluble Fe-DOC complexes would be expected.
DOC production is highest under oxic conditions when the water table is low (Fenner and Freeman, 2011; Strack et al., 2008) and at high temperatures (Rosset et al., 2022). Thus, the notably warm and dry year of 2018, where the mire was exposed to oxic conditions for the longest period (Fig. S2) could therefore be expected to yield the highest DOC production also leading to the highest export of DOC together with Fe and Pb considering the strong correlations presented (Fig. 6), but that was not the case. The asynchronous timing of Fe (2018) and DOC (2019) peaks was probably due to a higher soil respiration of DOC in 2018. Both Fe and Pb concentrations were highest in 2018, so the peat degradation was likely highest that year. The year of 2018 was an extreme warm and dry summer in Scandinavia, and the soil respiration in the study site was 15 % higher compared to “normal” years (Keane et al., 2021) making it likely that the higher soil respiration was due to mineralization of DOC, leading the lower DOC peaks in 2018 compared to 2019. A higher peat degradation in 2018, which is likely the main mobilisation process of Fe and Pb in the top part of bogs (Broder and Biester, 2017), would also explain the higher release of Pb and Fe in 2018. The large DOC peak in 2019 is harder to explain. One possible explanation is that the stream was supplemented by DOC rich water from the catchment, as supported by Fig. S2, that shows the pH in the stream water in 2019 (pH 5–6.5) was notably higher than the stream water pH in 2018 in the summer period. The high pH is not what we expect from water coming from the mire. Furthermore, there was a much higher precipitation in 2019 compared to 2018 (Fig. 6f), that could lead to a higher runoff, rich in DOC, from the forested catchment. One point this study could not address is whether longer periods of drought, despite not increasing DOC export from Mycklemossen, increased the overall C loss through increased mineralization under oxic conditions (Fenner and Freeman, 2011).
4.3 Pb in Mycklemossen
In agreement with other studies, we observed highest Pb content in the top 50 cm which then dropped and reached a non-detectable content below 120 cm depth in all topographies (Klaminder et al., 2006; Nieminen et al., 2002). This indicates that Pb was deposited onto Mycklemossen via wet and dry deposition. Hummock forming species have higher biomass production rates (Malmer and Wallén, 1999), but typically degrade slower compared to hollow species, which is generally due to a higher content of recalcitrant components as lignin-like compounds and secondary metabolites in hummock (Limpens et al., 2017; Mäkilä et al., 2018; Turetsky et al., 2008). This is supported by the higher SOM and C content in the top of the mire compared to hollow (Fig. 3). Under the assumption that the hummock and hollow topographies have remained stable at least since the start of anthropogenic pollution (Nungesser, 2003), a slower decomposition of hummock material could be what led to a higher accumulation of Pb in the top part of Mycklemossen in hummock compared to hollow. Peat in hummock was too young to date with 14C at 20–25 cm and was likely younger than the ban of lead. Thus, it is likely that new hummock material formed on top of the polluted layer during the last few decades since Pb was banned from gasoline and antifouling paint, explaining why the highest concentration of Pb was measured in 25–50 cm depth in hummocks and not in 15–20 cm depth. The observed local differences in Pb content might lead to differences in peat decomposition depending on topography.
Lead is toxic for mosses (Choudhury and Panda, 2005) and hampers the growth of microbial communities associated with Sphagnum fallax (Nguyen-Viet et al., 2007). Lead contents of 80–300 mg kg−1 soil can affect microbial decomposition of C and nutrient turnover (Bååth, 1989; Zheng et al., 2017). The Pb content of 92 mg kg−1 in hummocks at 25–50 cm depth is thus likely to affect the microbial community and the biomass turnover rate. The studies by Nguyen-Viet et al. (2007); Bååth (1989), and Zheng et al. (2017) report effects of Pb based on contents (mg kg−1) and not bioavailable Pb. Therefore, the high Pb content measured in hummock in this study, could have a potentially harmful effect on microbes. The bioavailability of Pb can be assessed in different ways, but requires some form of bioassay (Fleming et al., 2013; Hoang et al., 2025). In peatlands, Pb is mostly found strongly bound to organic matter and minerals with a low available fraction and is generally very immobile due to high binding affinity for OM (Hou et al., 2019; Lu et al., 2025; Novak et al., 2011; Vile et al., 1999). The bioavailable Pb in hummock is therefore certainly lower than the 92 mg kg−1, but Pb can be made available by microbial decomposition and accumulate in organisms over time. Most studies that report toxic effects of Pb on microbial decomposition perform experiments in periods of weeks or months, but toxicity of Pb increases with exposure time (Connell et al., 2016). Therefore, the toxic effect of Pb could be more severe in long term in situ decomposition studies. In that regard, Sphagnum species associated with hummock commonly decompose slower than hollow species under the same environmental conditions, but to which extent contents of Pb, or other heavy metals, could affect decomposability has not previously been considered (Bengtsson et al., 2016; Johnson et al., 1990; Johnson and Damman, 1991). In Mycklemossen, we only detected contents of Pb exceeding the toxicity threshold in hummocks. Decomposition experiments with peat were not tested in this study, but different pollution loads between topographies and local degradation dynamics might be necessary to understand Pb cycling in mires exposed to Pb pollution.
4.4 Pb pollution status in stream water
The concentration of Pb in stream water from Mycklemossen was up to 10-fold higher compared to streams from pristine Siberian bogs located 200 km from anthropogenic sources of heavy metals (Kharanzhevskaya et al., 2023), and slightly lower compared to a bog in Germany exposed to pollution from a nearby shooting range (Broder and Biester, 2015). The average Pb concentrations in stream water from Mycklemossen in the period of 2017 to 2020 were 4-fold higher than the long-term accepted values of 1.2 µg L−1 and suggests that this stream, according to European standards, is of poor environmental quality (EU directive, 2013). In stream water from Erssjön, the concentration of Pb was drastically lower (), and can be considered good quality for long term exposure (EU directive, 2013). The highest concentration of Pb in stream water from Mycklemossen was measured in late 2018 and reached 14 µg L−1, which is the short-term maximum limit value for fresh water (EU directive, 2013). As already pointed out, the high Pb concentration in S1 (14 µg L−1) in 2018 was likely due to a higher degree of Pb mobilisation (Broder and Biester, 2017). Beside the European standard for Pb thresholds, a comprehensive analysis taking bioavailability into account and including 7 European surface water scenarios and 25 freshwater species, found Pb toxicity thresholds within 6.1 and 31 µg L−1 (Van Sprang et al., 2016). Therefore, Pb in S1 reached concentrations on short-term basis that can be considered potentially harmful for freshwater fauna. Longer periods of drought in a future climate could lead to higher mobilisation of Pb and thus stream water concentrations of Pb exceeding toxicity thresholds (Szkokan-Emilson et al., 2013).
4.5 Export and fate of Pb in the mire-lake complex
While twice as much Fe and DOC was exported from Erssjön through S6 compared to S1 leaving Mycklemossen, which roughly follows differences in catchment size, the export of Pb was similar between the two streams in the period 2017 to 2020. At the same time, about 2-fold more Pb per amount of DOC left S1 compared to S6. The main explanation is probably found in how Pb in the two streams was sourced. The expected ratio in the area around Skogaryd is in the range of 1.245–1.326 (Reimann et al., 2012), which is higher than the data from Mycklemossen of 1.146. The value at Mycklemossen is at the lowest end for Northern Europe (Reimann et al., 2012) and is indicative for anthropogenic pollution (Fig. 7). For the ratio the average value of 2.112 for Mycklemossen is close to the median for Northern Europe (Reimann et al., 2012). Generally, the Pb isotope ratios revealed that Pb in S1 and S6 primarily was sourced from gasoline (Fig. 7), but the Pb isotopic signature in S6 was slightly shifted towards the signature of forest soil which overlaps with European coal, marine antifouling paint and natural geogenic sources. As isotope signature of marine antifouling paint falls on the mixing line of geological and gasoline Pb, its contribution is unclear. Erssjön also receives water from other land-uses (forest) within its catchment, which could be dominated by natural Pb isotopes, which supplies Pb to S6 at lower concentrations than S1. Except for one sampling, the ratio is always lower in Erssjön than Mycklemossen, indicating that the forest land almost always contributes to the lead in Erssjön. No previous estimates of the water residence time have been made but given the size of the Erssjön (6.2 ha) and the mean depth of 1.7 m (Milberg et al., 2017) the volume would be ∼100 000 m3, which gives a mean residence time of about 3 months. Conducting mixing model on the individual sampling dates (N=10), Mycklemossen contributed by 68–100 % to the lead in Erssjön outflow. Furthermore, the mixing model showed that around 19 % of the Pb coming from Mycklemossen was retained in Erssjön, indicating that Erssjön acts as a sink for the Pb from Mycklemossen. Thus, the similar Pb export from S1 and S6 is likely explained by an additional Pb input from the forested catchment to S6 of a lower concentration than what Mycklemossen supplies, and sedimentation/retention of Pb in Erssjön, which we estimated to be around 0.132 kg yr−1.
Here, we investigated a mire-lake complex inland from the west coast of Sweden subjected to recent climate change and exposed to heavy-metal pollution. We wanted to characterize the mire in terms of physiochemistry, age and total C content and evaluated the export of Fe and Pb in relation to DOC in the light of future climate. We showed that export of Fe, Pb and DOC from a nutrient poor mire located in the temperate and boreal transition zone is sensitive to climate. The mobilisation of Fe, Pb and DOC from the mire was seemingly affected by drought and thus decomposition, while the export mainly was controlled by hydrological connectivity. The ratio between Fe and DOC demonstrated strong linear correlations in stream water from both the mire and lake, which indicated that transport of Fe and DOC was tightly connected. Our investigation of Pb in the mire-lake complex showed that Pb pollution from gasoline had resulted in potentially toxic concentrations, specifically in the hummock topography and in downstream water ways. In a future climate with longer periods of drought and higher occurrence of heavy rain events, export of DOC and metals will likely increase from Mycklemossen and similar mire ecosystems. This study highlights that mires in anthropogenically exposed areas not only store C, which upon release will cause positive feedback to the climate, but also that this decomposition is linked to an environmental risk of the local environment in the form of heavy-metal pollution.
Data on water, soil and peat chemistry underlying this study can be found at https://meta.fieldsites.se/resources/sites/mycklemossen-mire (last access: 10 March 2026). Remaining underlying data is available from the appendices.
The supplement related to this article is available online at https://doi.org/10.5194/bg-23-2413-2026-supplement.
JT: Conceptualization, Investigation, Methodology, Writing – original draft, Writing – review and editing. SL: Conceptualization, Investigation, Formal analysis, Methodology, Visualization, Writing – original draft, Writing – review and editing, Supervision. LK: Conceptualization, Investigation, Formal analysis, Methodology, Resources, Writing – review and editing. DR: Methodology, Formal analysis, Writing – review and editing. LR: Conceptualization, Investigation, Methodology, Writing – review and editing. KSJ: Conceptualization, Supervision, Funding acquisition, Writing – review and editing. TR: Conceptualization, Investigation, Formal analysis, Funding acquisition, Resources, Writing – review and editing.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
Maria Shmarina, Haldor Lorimer-Olsson, David Allbrand, Josefina Carlberg is thanked for field work, peat sampling and analysis at IRMS. Martin Person is thanked for the georadar investigation.
This paper is a contribution to the Strategic Research Area “Biodiversity and Ecosystem Services in a Changing Climate” (BECC) funded by the Swedish government.
This work was partly funded by the Oxymist Challenge grant from the NNF (NNF20OC0059697) to KSJ and was made possible with data provided by the Swedish Infrastructure for Ecosystem Science (SITES), funded by the Swedish Research Council (VR).
This paper was edited by Robert Rhew and reviewed by three anonymous referees.
Aerts, R., Wallen, B., and Malmer, N.: Growth-limiting nutrients in Sphagnum-dominated bogs subject to low and high atmospheric nitrogen supply, J. Ecol., 80, 131–140, https://doi.org/10.2307/2261070, 1992.
Aleshina, A., Rusakova, M.-A., Drozdova, O. Y., Pokrovsky, O. S., and Lapitskiy, S. A.: Dissolved Iron and Organic Matter in Boreal Rivers across a South–North Transect, Environments, 11, 65, https://doi.org/10.3390/environments11040065, 2024.
Andersson, S. and Schoning, K.: Surface wetness and mire development during the late Holocene in central Sweden, Boreas, 39, 749–760, https://doi.org/10.1111/j.1502-3885.2010.00157.x, 2010.
Bååth, E.: Effects of heavy metals in soil on microbial processes and populations (a review), Water Air Soil Poll., 47, 335–379, https://doi.org/10.1007/BF00279331, 1989.
Bengtsson, F., Granath, G., and Rydin, H.: Photosynthesis, growth, and decay traits in Sphagnum – a multispecies comparison, Ecol. Evol., 6, 3325–3341, https://doi.org/10.1002/ece3.2119, 2016.
Bhattacharyya, A., Schmidt, M. P., Stavitski, E., and Martínez, C. E.: Iron speciation in peats: Chemical and spectroscopic evidence for the co-occurrence of ferric and ferrous iron in organic complexes and mineral precipitates, Org. Geochem., 115, 124–137, https://doi.org/10.1016/j.orggeochem.2017.10.012, 2018.
Biester, H., Knorr, K.-H., Schellekens, J., Basler, A., and Hermanns, Y.-M.: Comparison of different methods to determine the degree of peat decomposition in peat bogs, Biogeosciences, 11, 2691–2707, https://doi.org/10.5194/bg-11-2691-2014, 2014.
Bindler, R., Brännvall, M.-L., Renberg, I., Emteryd, O., and Grip, H.: Natural Lead Concentrations in Pristine Boreal Forest Soils and Past Pollution Trends: A Reference for Critical Load Models, Environ. Sci. Technol., 33, 3362–3367, https://doi.org/10.1021/es9809307, 1999.
Björnerås, C., Persson, P., Weyhenmeyer, G. A., Hammarlund, D., and Kritzberg, E. S.: The lake as an iron sink – new insights on the role of iron speciation, Chem. Geol., 584, 120529, https://doi.org/10.1016/j.chemgeo.2021.120529, 2021.
Brännvall, M.-L., Kurkkio, H., Bindler, R., Emteryd, O., and Renberg, I.: The role of pollution versus natural geological sources for lead enrichment in recent lake sediments and surface forest soils, Environ. Geol., 40, 1057–1065, https://doi.org/10.1007/s002540100260, 2001.
Bränvall, M.-L., Bindler, R., Emteryd, O., and Renberg, I.: Four thousand years of atmospheric lead pollution in northern Europe: a summary from Swedish lake sediments, J. Paleolimnol., 25, 421–435, https://doi.org/10.1023/A:1011186100081, 2001.
Broder, T. and Biester, H.: Hydrologic controls on DOC, As and Pb export from a polluted peatland – the importance of heavy rain events, antecedent moisture conditions and hydrological connectivity, Biogeosciences, 12, 4651–4664, https://doi.org/10.5194/bg-12-4651-2015, 2015.
Broder, T. and Biester, H.: Linking major and trace element concentrations in a headwater stream to DOC release and hydrologic conditions in a bog and peaty riparian zone, Appl. Geochem., 87, 188–201, https://doi.org/10.1016/j.apgeochem.2017.11.003, 2017.
Brouns, K., Verhoeven, J. T. A., and Hefting, M. M.: Short period of oxygenation releases latch on peat decomposition, Sci. Total Environ., 481, 61–68, https://doi.org/10.1016/j.scitotenv.2014.02.030, 2014.
Catanzaro, E. J., Murphy, T. J., Shields, W. R., and Garner, E. L.: Absolute Isotopic Abundance Ratios of Common, Equal-Atom, and Radiogenic Lead Isotopic Standards, J. Res. Natl. Bur. Stand. A Phys. Chem., 72A, 261–267, https://doi.org/10.6028/jres.072A.025, 1968.
Chen, C., Hall, S. J., Coward, E., and Thompson, A.: Iron-mediated organic matter decomposition in humid soils can counteract protection, Nat. Commun., 11, 2255, https://doi.org/10.1038/s41467-020-16071-5, 2020.
Choudhury, S. and Panda, S. K.: Toxic Effects, Oxidative Stress and Ultrastructural Changes in Moss Taxithelium Nepalense (Schwaegr.) Broth. Under Chromium and Lead Phytotoxicity, Water Air Soil Poll., 167, 73–90, https://doi.org/10.1007/s11270-005-8682-9, 2005.
Clark, J. M., Lane, S. N., Chapman, P. J., and Adamson, J. K.: Export of dissolved organic carbon from an upland peatland during storm events: Implications for flux estimates, J. Hydrol., 347, 438–447, https://doi.org/10.1016/j.jhydrol.2007.09.030, 2007.
Clymo, R. S.: Ion Exchange in Sphagnum and its Relation to Bog Ecology, Ann. Bot.-London, 27, 309–324, https://doi.org/10.1093/oxfordjournals.aob.a083847, 1963.
Collin, M. S., Venkatraman, S. K., Vijayakumar, N., Kanimozhi, V., Arbaaz, S. M., Stacey, R. G. S., Anusha, J., Choudhary, R., Lvov, V., Tovar, G. I., Senatov, F., Koppala, S., and Swamiappan, S.: Bioaccumulation of lead (Pb) and its effects on human: A review, Journal of Hazardous Materials Advances, 7, 100094, https://doi.org/10.1016/j.hazadv.2022.100094, 2022.
Connell, D. W., Yu, Q. J., and Verma, V.: Influence of exposure time on toxicity – An overview, Toxicology, 355–356, 49–53, https://doi.org/10.1016/j.tox.2016.05.015, 2016.
Curtinrich, H. J., Sebestyen, S. D., and Hall, S. J.: Metal-bound carbon and nutrients across hydrologically diverse boreal peatlands, Biogeochemistry, 168, 7, https://doi.org/10.1007/s10533-024-01199-z, 2024.
Doelman, P. and Haanstra, L.: Effects of lead on the decomposition of organic matter, Soil Biol. Biochem., 11, 481–485, https://doi.org/10.1016/0038-0717(79)90006-3, 1979.
Dore, M. H. I.: Climate change and changes in global precipitation patterns: What do we know?, Environ. Int., 31, 1167–1181, https://doi.org/10.1016/j.envint.2005.03.004, 2005.
Drollinger, S., Kuzyakov, Y., and Glatzel, S.: Effects of peat decomposition on δ13C and δ15N depth profiles of Alpine bogs, Catena, 178, 1–10, https://doi.org/10.1016/j.catena.2019.02.027, 2019.
Ehnvall, B., Ratcliffe, J. L., Nilsson, M. B., Öquist, M. G., Sponseller, R. A., and Grabs, T.: Topography and Time Shape Mire Morphometry and Large-Scale Mire Distribution Patterns in the Northern Boreal Landscape, J. Geophys. Res.-Earth, 129, e2023JF007324, https://doi.org/10.1029/2023JF007324, 2024.
Esmeijer-Liu, A. J., Kürschner, W. M., Lotter, A. F., Verhoeven, J. T. A., and Goslar, T.: Stable Carbon and Nitrogen Isotopes in a Peat Profile Are Influenced by Early Stage Diagenesis and Changes in Atmospheric CO2 and N Deposition, Water Air Soil Poll., 223, 2007–2022, https://doi.org/10.1007/s11270-011-1001-8, 2012.
EU directive: Directive 2013/39/EU of the European Parliament and of the Council of 12 August 2013 amending Directives 2000/60/EC and 2008/105/EC as regards priority substances in the field of water policy Text with EEA relevance, https://eur-lex.europa.eu/eli/dir/2013/39/oj/eng (last access: 9 April 2026), 2013.
Fenner, N. and Freeman, C.: Drought-induced carbon loss in peatlands, Nat. Geosci., 4, 895–900, https://doi.org/10.1038/ngeo1323, 2011.
Fleming, M., Tai, Y., Zhuang, P., and McBride, M. B.: Extractability and bioavailability of Pb and As in historically contaminated orchard soil: Effects of compost amendments, Environ. Pollut., 177, 90–97, https://doi.org/10.1016/j.envpol.2013.02.013, 2013.
Freeman, C., Ostle, N., and Kang, H.: An enzymic “latch” on a global carbon store, Nature, 409, 149–149, https://doi.org/10.1038/35051650, 2001.
Gmochowska, W., Pietranik, A., Tyszka, R., Ettler, V., Mihaljevič, M., Długosz, M., and Walenczak, K.: Sources of pollution and distribution of Pb, Cd and Hg in Wrocław soils: Insight from chemical and Pb isotope composition, Geochemistry, 79, 434–445, https://doi.org/10.1016/j.chemer.2019.07.002, 2019.
Gorham, E.: Northern Peatlands: Role in the Carbon Cycle and Probable Responses to Climatic Warming, Ecol. Appl., 1, 182–195, https://doi.org/10.2307/1941811, 1991.
Hansson, A., Kjällquist, M., and Boethius, A.: Caring for preservation – coring for prehistoric life. Revisiting 15 000 years of sedimentation at the Ageröd peatland, Southern Sweden, Quaternary Environments and Humans, 2, 100039, https://doi.org/10.1016/j.qeh.2024.100039, 2024.
Heikkinen, K., Saari, M., Heino, J., Ronkanen, A.-K., Kortelainen, P., Joensuu, S., Vilmi, A., Karjalainen, S.-M., Hellsten, S., Visuri, M., and Marttila, H.: Iron in boreal river catchments: Biogeochemical, ecological and management implications, Sci. Total Environ., 805, 150256, https://doi.org/10.1016/j.scitotenv.2021.150256, 2022.
Helbig, M., Waddington, J. M., Alekseychik, P., Amiro, B. D., Aurela, M., Barr, A. G., Black, T. A., Blanken, P. D., Carey, S. K., Chen, J., Chi, J., Desai, A. R., Dunn, A., Euskirchen, E. S., Flanagan, L. B., Forbrich, I., Friborg, T., Grelle, A., Harder, S., Heliasz, M., Humphreys, E. R., Ikawa, H., Isabelle, P.-E., Iwata, H., Jassal, R., Korkiakoski, M., Kurbatova, J., Kutzbach, L., Lindroth, A., Löfvenius, M. O., Lohila, A., Mammarella, I., Marsh, P., Maximov, T., Melton, J. R., Moore, P. A., Nadeau, D. F., Nicholls, E. M., Nilsson, M. B., Ohta, T., Peichl, M., Petrone, R. M., Petrov, R., Prokushkin, A., Quinton, W. L., Reed, D. E., Roulet, N. T., Runkle, B. R. K., Sonnentag, O., Strachan, I. B., Taillardat, P., Tuittila, E.-S., Tuovinen, J.-P., Turner, J., Ueyama, M., Varlagin, A., Wilmking, M., Wofsy, S. C., and Zyrianov, V.: Increasing contribution of peatlands to boreal evapotranspiration in a warming climate, Nat. Clim. Change, 10, 555–560, https://doi.org/10.1038/s41558-020-0763-7, 2020.
Hoang, H. G., Hadi, M., Nguyen, M. K., Hai Nguyen, N. S., Huy Le, P. Q., Nguyen, K. N., Tran, H.-T., and Mishra, U.: Assessing heavy metal pollution levels and associated ecological risks in peatland areas in the Mekong Delta region, Environ. Res., 274, 121319, https://doi.org/10.1016/j.envres.2025.121319, 2025.
Hou, S., Zheng, N., Tang, L., Ji, X., and Li, Y.: Effect of soil pH and organic matter content on heavy metals availability in maize (Zea mays L.) rhizospheric soil of non-ferrous metals smelting area, Environ. Monit. Assess., 191, 634, https://doi.org/10.1007/s10661-019-7793-5, 2019.
IPCC: Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change, edited by: Masson-Delmotte, V., Zhai, P., Pirani, A., Connors, S. L., Péan, C., Berger, S., Caud, N., Chen, Y., Goldfarb, L., Gomis, M. I., Huang, M., Leitzell, K., Lonnoy, E., Matthews, J. B. R., Maycock, T. K., Waterfield, T., Yelekçi, O., R. Yu, and Zhou, B., Cambridge, United Kingdom and New York, NY, USA, 2391 pp., https://doi.org/10.1017/9781009157896, 2021.
Jeong, H., Araújo, D. F., Knœry, J., Briant, N., and Ra, K.: Isotopic (Cu, Zn, and Pb) and elemental fingerprints of antifouling paints and their potential use for environmental forensic investigations, Environ. Pollut., 322, 121176, https://doi.org/10.1016/j.envpol.2023.121176, 2023.
Johnson, L. C. and Damman, A. W. H.: Species-Controlled Sphagnum Decay on a South Swedish Raised Bog, Oikos, 61, 234–242, https://doi.org/10.2307/3545341, 1991.
Johnson, L. C., Damman, A. W. H., and Malmer, N.: Sphagnum Macrostructure as an Indicator of Decay and Compaction in Peat Cores from an Ombrotrophic South Swedish Peat-Bog, J. Ecol., 78, 633–647, https://doi.org/10.2307/2260889, 1990.
Kaal, J., Cortizas, A. M., and Biester, H.: Downstream changes in molecular composition of DOM along a headwater stream in the Harz mountains (Central Germany) as determined by FTIR, Pyrolysis-GC–MS and THM-GC–MS, J. Anal. Appl. Pyrol., 126, 50–61, https://doi.org/10.1016/j.jaap.2017.06.025, 2017.
Keane, J. B., Toet, S., Ineson, P., Weslien, P., Stockdale, J. E., and Klemedtsson, L.: Carbon Dioxide and Methane Flux Response and Recovery From Drought in a Hemiboreal Ombrotrophic Fen, Front. Earth Sci., 8, https://doi.org/10.3389/feart.2020.562401, 2021.
Kelly, J., Kljun, N., Eklundh, L., Klemedtsson, L., Liljebladh, B., Olsson, P.-O., Weslien, P., and Xie, X.: Modelling and upscaling ecosystem respiration using thermal cameras and UAVs: Application to a peatland during and after a hot drought, Agr. Forest Meteorol., 300, 108330, https://doi.org/10.1016/j.agrformet.2021.108330, 2021.
Kharanzhevskaya, Y., Gashkova, L., Sinyutkina, A., and Kvasnikova, Z.: Assessment of Present-Day Heavy Metals Pollution and Factors Controlling Surface Water Chemistry of Three Western Siberian Sphagnum-Dominated Raised Bogs, Water, 15, 1869, https://doi.org/10.3390/w15101869, 2023.
Klaminder, J., Bindler, R., Laudon, H., Bishop, K., Emteryd, O., and Renberg, I.: Flux Rates of Atmospheric Lead Pollution within Soils of a Small Catchment in Northern Sweden and Their Implications for Future Stream Water Quality, Environ. Sci. Technol., 40, 4639–4645, https://doi.org/10.1021/es0520666, 2006.
Knorr, K.-H.: DOC-dynamics in a small headwater catchment as driven by redox fluctuations and hydrological flow paths – are DOC exports mediated by iron reduction/oxidation cycles?, Biogeosciences, 10, 891–904, https://doi.org/10.5194/bg-10-891-2013, 2013.
Komárek, M., Ettler, V., Chrastný, V., and Mihaljevič, M.: Lead isotopes in environmental sciences: A review, Environ. Int., 34, 562–577, https://doi.org/10.1016/j.envint.2007.10.005, 2008.
Koppenol, W. H. and Hider, R. H.: Iron and redox cycling. Do's and don'ts, Free Radical Bio. Med., 133, 3–10, https://doi.org/10.1016/j.freeradbiomed.2018.09.022, 2019.
Krull, E. S. and Retallack, G. J.: δ13C depth profiles from paleosols across the Permian-Triassic boundary: Evidence for methane release, GSA Bulletin, 112, 1459–1472, https://doi.org/10.1130/0016-7606(2000)112%3C1459:CDPFPA%3E2.0.CO;2, 2000.
Kumar Das, S., Ekblad, A., Stojanović, K., Mikusinska, A., Gaillard, M.-J., Mitrović, D., and Cui, Q.-Y.: Holocene Stable Isotope (δ13C and δ15N) record of peatland development in Stavsåkra, southern Sweden, Catena, 247, 108510, https://doi.org/10.1016/j.catena.2024.108510, 2024.
Kylander, M. E., Bindler, R., Cortizas, A. M., Gallagher, K., Mörth, C.-M., and Rauch, S.: A novel geochemical approach to paleorecords of dust deposition and effective humidity: 8500 years of peat accumulation at Store Mosse (the “Great Bog”), Sweden, Quaternary Sci. Rev., 69, 69–82, https://doi.org/10.1016/j.quascirev.2013.02.010, 2013.
Limpens, J., Bohlin, E., and Nilsson, M. B.: Phylogenetic or environmental control on the elemental and organo-chemical composition of Sphagnum mosses?, Plant Soil, 417, 69–85, https://doi.org/10.1007/s11104-017-3239-4, 2017.
Lu, Z., Ning, Y., Liu, C., Song, X., Pang, Y., Li, Q., Yang, M., and Zeng, L.: Geochemical Regulation of Heavy Metal Speciation in Subtropical Peatlands: A Case Study in Dajiuhu Peatland, Land, 14, 1256, https://doi.org/10.3390/land14061256, 2025.
Mäkilä, M., Säävuori, H., Grundström, A., and Suomi, T.: Sphagnum decay patterns and bog microtopography in south-eastern Finland, Mires Peat, 1–12, https://doi.org/10.19189/MaP.2017.OMB.283, 2018.
Malmer, N. and Wallén, B.: The dynamics of peat accumulation on bogs: mass balance of hummocks and hollows and its variation throughout a millennium, Ecography, 22, 736–750, https://doi.org/10.1111/j.1600-0587.1999.tb00523.x, 1999.
Marcantonio, F., Flowers, G. C., and Templin, N.: Lead contamination in a wetland watershed: isotopes as fingerprints of pollution, Environ. Geol., 39, 1070–1076, https://doi.org/10.1007/s002549900093, 2000.
Mariussen, E., Johnsen, I. V., and Strømseng, A. E.: Distribution and mobility of lead (Pb), copper (Cu), zinc (Zn), and antimony (Sb) from ammunition residues on shooting ranges for small arms located on mires, Environ. Sci. Pollut. R., 24, 10182–10196, https://doi.org/10.1007/s11356-017-8647-8, 2017.
McCarter, C. P. R., Clay, G. D., Wilkinson, S. L., Sigmund, G., Davidson, S. J., Taufik, M., Page, S., Shuttleworth, E. L., McLagan, D., Chenier, G., Clark, A., and Waddington, J. M.: Peat fires and legacy toxic metal release: An integrative biogeochemical and ecohydrological conceptual framework, Earth-Sci. Rev., 256, 104867, https://doi.org/10.1016/j.earscirev.2024.104867, 2024.
Milberg, P., Törnqvist, L., Westerberg, L. M., and Bastviken, D.: Temporal variations in methane emissions from emergent aquatic macrophytes in two boreonemoral lakes, AoB Plants, 9, plx029, https://doi.org/10.1093/aobpla/plx029, 2017.
Miles, C. J. and Brezonik, P. L.: Oxygen consumption in humic-colored waters by a photochemical ferrous-ferric catalytic cycle, Environ. Sci. Technol., 15, 1089–1095, https://doi.org/10.1021/es00091a010, 1981.
Moore, T. and Basiliko, N.: Decomposition in Boreal Peatlands, in: Boreal Peatland Ecosystems, vol. 188, edited by: Wieder, R. K. and Vitt, D. H., Springer, Berlin, Heidelberg, 125–143, https://doi.org/10.1007/978-3-540-31913-9_7, 2006.
Neubauer, E., Köhler, S. J., von der Kammer, F., Laudon, H., and Hofmann, T.: Effect of pH and stream order on iron and arsenic speciation in boreal catchments, Environ. Sci. Technol., 47, 7120–7128, https://doi.org/10.1021/es401193j, 2013.
Nguyen-Viet, H., Gilbert, D., Mitchell, E. A. D., Badot, P.-M., and Bernard, N.: Effects of Experimental Lead Pollution on the Microbial Communities Associated with Sphagnum fallax (Bryophyta), Microb. Ecol., 54, 232–241, https://doi.org/10.1007/s00248-006-9192-z, 2007.
Nieminen, T. M., Ukonmaanaho, L., and Shotyk, W.: Enrichment of Cu, Ni, Zn, Pb and As in an ombrotrophic peat bog near a Cu-Ni smelter in Southwest Finland, Sci. Total Environ., 292, 81–89, https://doi.org/10.1016/S0048-9697(02)00028-1, 2002.
Novak, M., Zemanova, L., Voldrichiva, P., Stepanova, M., Adamova, M., Pacherova, P., Komarek, A., Krachler, M., and Prechova, E.: Experimental Evidence for Mobility/Immobility of Metals in Peat, Environ. Sci. Technol., https://doi.org/10.1021/es201086v, 2011.
Nungesser, M. K.: Modelling microtopography in boreal peatlands: hummocks and hollows, Ecol. Model., 165, 175–207, https://doi.org/10.1016/S0304-3800(03)00067-X, 2003.
Nykänen, H., Rissanen, A. J., Turunen, J., Tahvanainen, T., and Simola, H.: Carbon storage change and δ13C transitions of peat columns in a partially forestry-drained boreal bog, Plant Soil, 447, 365–378, https://doi.org/10.1007/s11104-019-04375-5, 2020.
Okkenhaug, G., Smebye, A. B., Pabst, T., Amundsen, C. E., Sævarsson, H., and Breedveld, G. D.: Shooting range contamination: mobility and transport of lead (Pb), copper (Cu) and antimony (Sb) in contaminated peatland, J. Soils Sediments, 18, 3310–3323, https://doi.org/10.1007/s11368-017-1739-8, 2018.
Osborne, C., Gilbert-Parkes, S., Spiers, G., Lamit, L. J., Lilleskov, E. A., Basiliko, N., Watmough, S., Andersen, R., Artz, R. E., Benscoter, B. W., Bragazza, L., Bräuer, S. L., Carson, M. A., Chen, X., Chimner, R. A., Clarkson, B. R., Enriquez, A. S., Grover, S. P., Harris, L. I., Hazard, C., Hribljan, J., Juutinen, S., Kane, E. S., Knorr, K.-H., Kolka, R., Laine, A. M., Larmola, T., McCalley, C. K., McLaughlin, J., Moore, T. R., Mykytczuk, N., Normand, A. E., Olefeldt, D., Rich, V., Roulet, N., Rupp, D. L., Rutherford, J., Schadt, C. W., Sonnentag, O., Tedersoo, L., Trettin, C. C., Tuittila, E.-S., Turetsky, M., Urbanová, Z., Varner, R. K., Waldrop, M. P., Wang, M., Wang, Z., Wiedermann, M. M., Williams, S. T., Yavitt, J. B., Yu, Z.-G., and Global Peatland Microbiome Project: Global Patterns of Metal and Other Element Enrichment in Bog and Fen Peatlands, Arch. Environ. Con. Tox., 86, 125–139, https://doi.org/10.1007/s00244-024-01051-3, 2024.
Page, S. E., Logan, J. R., Cory, R. M., and McNeill, K.: Evidence for dissolved organic matter as the primary source and sink of photochemically produced hydroxyl radical in arctic surface waters, Environ. Sci.-Proc. Imp., 16, 807–822, https://doi.org/10.1039/C3EM00596H, 2014.
Peciulyte, A., Samuelsson, L., Olsson, L., McFarland, K. C., Frickmann, J., Østergård, L., Halvorsen, R., Scott, B. R., and Johansen, K. S.: Redox processes acidify and decarboxylate steam-pretreated lignocellulosic biomass and are modulated by LPMO and catalase, Biotechnol. Biofuels, 11, 165, https://doi.org/10.1186/s13068-018-1159-z, 2018.
Phillips, D. L. and Gregg, J. W.: Uncertainty in source partitioning using stable isotopes, Oecologia, 127, 171–179, https://doi.org/10.1007/s004420000578, 2001.
Pinheiro, J., Bates, D., and R Core Team: Linear and Nonlinear Mixed Effects Models, https://doi.org/10.32614/CRAN.package.nlme, 2023.
Prijac, A., Gandois, L., Taillardat, P., Bourgault, M.-A., Riahi, K., Ponçot, A., Tremblay, A., and Garneau, M.: Hydrological connectivity controls dissolved organic carbon exports in a peatland-dominated boreal catchment stream, Hydrol. Earth Syst. Sci., 27, 3935–3955, https://doi.org/10.5194/hess-27-3935-2023, 2023.
Qin, L., Freeman, C., Zou, Y., Wang, G., Fenner, N., Yang, L., Otte, M. L., and Jiang, M.: An iron-reduction-mediated cascade mechanism increases the risk of carbon loss from mineral-rich peatlands, Appl. Soil Ecol., 172, 104361, https://doi.org/10.1016/j.apsoil.2021.104361, 2022.
R Core Team: R: A Language and Environment for Statistical Computing, 2024.
Reimann, C., Flem, B., Fabian, K., Birke, M., Ladenberger, A., Négrel, P., Demetriades, A., and Hoogewerff, J.: Lead and lead isotopes in agricultural soils of Europe – The continental perspective, Appl. Geochem., 27, 532–542, https://doi.org/10.1016/j.apgeochem.2011.12.012, 2012.
Renberg, I., Brännvall, M.-L., Bindler, R., and Emteryd, O.: Atmospheric Lead Pollution History during Four Millennia (2000 BC to 2000 AD) in Sweden, ambi, 29, 150–156, https://doi.org/10.1579/0044-7447-29.3.150, 2000.
Riedel, T., Zak, D., Biester, H., and Dittmar, T.: Iron traps terrestrially derived dissolved organic matter at redox interfaces, P. Natl. Acad. Sci. USA, 110, 10101–10105, https://doi.org/10.1073/pnas.1221487110, 2013.
Rinne, J., Łakomiec, P., Vestin, P., White, J. D., Weslien, P., Kelly, J., Kljun, N., Ström, L., and Klemedtsson, L.: Spatial and temporal variation in δ13C values of methane emitted from a hemiboreal mire: methanogenesis, methanotrophy, and hysteresis, Biogeosciences, 19, 4331–4349, https://doi.org/10.5194/bg-19-4331-2022, 2022.
Rosset, T., Binet, S., Rigal, F., and Gandois, L.: Peatland Dissolved Organic Carbon Export to Surface Waters: Global Significance and Effects of Anthropogenic Disturbance, Geophys. Res. Lett., 49, e2021GL096616, https://doi.org/10.1029/2021GL096616, 2022.
Rothwell, J. J., Robinson, S. G., Evans, M. G., Yang, J., and Allott, T. E. H.: Heavy metal release by peat erosion in the Peak District, southern Pennines, UK, Hydrol. Process., 19, 2973–2989, https://doi.org/10.1002/hyp.5811, 2005.
Roux, G. L., Weiss, D., Grattan, J., Givelet, N., Krachler, M., Cheburkin, A., Rausch, N., Kober, B., and Shotyk, W.: Identifying the sources and timing of ancient and medieval atmospheric lead pollution in England using a peat profile from Lindow bog, Manchester, J. Environ. Monitor., 6, 502–510, https://doi.org/10.1039/B401500B, 2004.
Salehpour, M., Håkansson, K., Westermark, P., Antoni, G., Wikström, G., and Possnert, G.: Life Science Applications Utilizing Radiocarbon Tracing, Radiocarbon, 55, 865–873, https://doi.org/10.1017/S0033822200058021, 2013.
Shakya, K., Chettri, M. K., and Sawidis, T.: Impact of Heavy Metals (Copper, Zinc, and Lead) on the Chlorophyll Content of Some Mosses, Arch. Environ. Con. Tox., 54, 412–421, https://doi.org/10.1007/s00244-007-9060-y, 2008.
Shotyk, W., Cheburkin, A. K., Appleby, P. G., Fankhauser, A., and Kramers, J. D.: Two thousand years of atmospheric arsenic, antimony, and lead deposition recorded in an ombrotrophic peat bog profile, Jura Mountains, Switzerland, Earth Planet. Sc. Lett., 145, E1–E7, https://doi.org/10.1016/S0012-821X(96)00197-5, 1996.
Smeds, J., Ehnvall, B., Liu, T., Bertilsson, S., Björn, E., Nilsson, M. B., Bishop, K., Skyllberg, U., and Öquist, M.: Effect of Restoration on Physical and Chemical Peat Properties in Previously Drained Boreal Peatlands, Ecosystems, 28, 45, https://doi.org/10.1007/s10021-025-00991-8, 2025.
Song, X., Wang, P., Van Zwieten, L., Bolan, N., Wang, H., Li, X., Cheng, K., Yang, Y., Wang, M., Liu, T., and Li, F.: Towards a better understanding of the role of Fe cycling in soil for carbon stabilization and degradation, Carbon Res., 1, 5, https://doi.org/10.1007/s44246-022-00008-2, 2022.
Steinnes, E., Hvatum, O. Ø., Bølviken, B., and Varskog, P.: Atmospheric supply of trace elements studied by peat samples from ombrotrophic bogs, J. Environ. Qual., 34, 192–197, https://doi.org/10.2134/jeq2005.0192, 2005.
Strack, M., Waddington, J. M., Bourbonniere, R. A., Buckton, E. L., Shaw, K., Whittington, P., and Price, J. S.: Effect of water table drawdown on peatland dissolved organic carbon export and dynamics, Hydrol. Process., 22, 3373–3385, https://doi.org/10.1002/hyp.6931, 2008.
Szkokan-Emilson, E. J., Kielstra, B., Watmough, S., and Gunn, J.: Drought-induced release of metals from peatlands in watersheds recovering from historical metal and sulphur deposition, Biogeochemistry, 116, 131–145, https://doi.org/10.1007/s10533-013-9919-0, 2013.
Tchounwou, P. B., Yedjou, C. G., Patlolla, A. K., and Sutton, D. J.: Heavy Metals Toxicity and the Environment, EXS, 101, 133–164, https://doi.org/10.1007/978-3-7643-8340-4_6, 2012.
Thomsen, J., Lett, S., Martens, H. J., Sørensen, H., Kelleher, D., Tryfona, T., Dupree, P., and Johansen, K. S.: Enzymatic saccharification of peat polysaccharides is limited by accessibility, PLoS One, 20, e0312219, https://doi.org/10.1371/journal.pone.0312219, 2025.
Tipping, E., Smith, E. J., Lawlor, A. J., Hughes, S., and Stevens, P. A.: Predicting the release of metals from ombrotrophic peat due to drought-induced acidification, Environ. Pollut., 123, 239–253, https://doi.org/10.1016/S0269-7491(02)00375-5, 2003.
Trusiak, A., Treibergs, L. A., Kling, G. W., and Cory, R. M.: The role of iron and reactive oxygen species in the production of CO2 in arctic soil waters, Geochim. Cosmochim. Ac., 224, 80–95, https://doi.org/10.1016/j.gca.2017.12.022, 2018.
Turetsky, M. R., Crow, S. E., Evans, R. J., Vitt, D. H., and Wieder, R. K.: Trade-Offs in Resource Allocation among Moss Species Control Decomposition in Boreal Peatlands, J. Ecol., 96, 1297–1305, 2008.
Van Sprang, P. A., Nys, C., Blust, R. J. P., Chowdhury, J., Gustafsson, J. P., Janssen, C. J., and De Schamphelaere, K. A. C.: The derivation of effects threshold concentrations of lead for European freshwater ecosystems, Environ. Toxicol. Chem., 35, 1310–1320, https://doi.org/10.1002/etc.3262, 2016.
Verhoeven, J. T. A. and Liefveld, W. M.: The ecological significance of organochemical compounds in Sphagnum, Acta Bot. Neerl., 46, 117–130, 1997.
Vile, M. A., Wieder, R. K., and Novák, M.: Mobility of Pb in Sphagnum-derived peat, Biogeochemistry, 45, 35–52, https://doi.org/10.1023/A:1006085410886, 1999.
Wang, Y., Wang, H., He, J.-S., and Feng, X.: Iron-mediated soil carbon response to water-table decline in an alpine wetland, Nat. Commun., 8, 15972, https://doi.org/10.1038/ncomms15972, 2017.
Wang, Y., Liu, X., Zhang, X., Dai, G., Wang, Z., and Feng, X.: Evaluating wetland soil carbon stability related to iron transformation during redox oscillations, Geoderma, 428, 116222, https://doi.org/10.1016/j.geoderma.2022.116222, 2022.
Weng, J.-K. and Chapple, C.: The origin and evolution of lignin biosynthesis, New Phytol., 187, 273–285, https://doi.org/10.1111/j.1469-8137.2010.03327.x, 2010.
Weyhenmeyer, G. A., Prairie, Y. T., and Tranvik, L. J.: Browning of boreal freshwaters coupled to carbon-iron interactions along the aquatic continuum, PLoS One, 9, e88104, https://doi.org/10.1371/journal.pone.0088104, 2014.
Wilson, R., Hopple, A., Tfaily, M., Sebestyen, S., Schadt, C., Pfeifer-Meister, L., Zalman, C., Mcfarlane, K., Kostka, J., Kolton, M., Kolka, R., Kluber, L., Keller, J., Guilderson, T., Griffiths, N., Chanton, J., Bridgham, S., and Hanson, P.: Stability of Peatland Carbon to Rising Temperatures, Nat. Commun., 7, https://doi.org/10.1038/ncomms13723, 2016.
Yu, G.-H. and Kuzyakov, Y.: Fenton chemistry and reactive oxygen species in soil: Abiotic mechanisms of biotic processes, controls and consequences for carbon and nutrient cycling, Earth-Sci. Rev., 214, 103525, https://doi.org/10.1016/j.earscirev.2021.103525, 2021.
Zeh, L., Igel, M. T., Schellekens, J., Limpens, J., Bragazza, L., and Kalbitz, K.: Vascular plants affect properties and decomposition of moss-dominated peat, particularly at elevated temperatures, Biogeosciences, 17, 4797–4813, https://doi.org/10.5194/bg-17-4797-2020, 2020.
Zhang, H., Piilo, S., Aquino-López, M. A., Guo, Z., Zhao, Y., Laine, A. M., Korrensalo, A., Tuittila, E.-S., and Väliranta, M.: Drivers on Carbon Accumulation Vary Along the Hydrological Gradient of a Subarctic Patterned Peatland, J. Geophys. Res.-Biogeo., 130, e2024JG008677, https://doi.org/10.1029/2024JG008677, 2025.
Zhao, Y., Liu, C., Li, X., Ma, L., Zhai, G., and Feng, X.: Sphagnum increases soil's sequestration capacity of mineral-associated organic carbon via activating metal oxides, Nat. Commun., 14, 5052, https://doi.org/10.1038/s41467-023-40863-0, 2023.
Zheng, H., Chen, L., Li, N., Liu, B., Meng, N., Wang, M., and Chen, S.: Toxicity threshold of lead (Pb) to nitrifying microorganisms in soils determined by substrate-induced nitrification assay and prediction model, J. Integr. Agr., 16, 1832–1840, https://doi.org/10.1016/S2095-3119(16)61586-1, 2017.
Zhong, Y., Jiang, M., and Middleton, B. A.: Effects of water level alteration on carbon cycling in peatlands, Ecosystem Health and Sustainability, 6, 1806113, https://doi.org/10.1080/20964129.2020.1806113, 2020.