the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Ideas and perspectives: Mineralizing fluid control on foreign elements in biogenic CaCO3: insights from otoliths
Athina Kekelou
Gerald Langer
Patrizia Ziveri
The foreign element composition of calcium carbonate (CaCO3) biominerals from marine calcifying organisms leaving a sedimentary record has been used for decades to reconstruct various biogeochemical parameters. Advancing geochemical proxies and understanding their underlying mechanisms is essential for climate reconstructions, environmental research, and investigations of biomineralization processes. Despite considerable success of proxy applications, limited mechanistic understanding still restricts their full potential. The problem is often summarized by the term “vital effect”, i.e. foreign element partitioning due to biological activity. The element partitioning from the calcifying fluid into the biomineral, however, is usually described in terms of inorganic precipitation of a mineral from an aqueous solution of inorganic ions. Although this assumption is central to many partitioning models it has not been tested because the calcifying fluid of classic proxy archives such as foraminifera, molluscs, and coccolithophores has not been successfully sampled for element analysis. The calcifying fluid of fish otolith formation (endolymph), by contrast, was sampled and chemically analysed accompanied by corresponding otolith data. However, previous datasets have not been compared to inorganic partitioning coefficients to test this assumption. In this study, we address this gap using published data from four fish species and six elements. Our results indicate that the final stage of otolith foreign element incorporation is influenced by organic matter in the endolymph fluid and therefore cannot be considered purely inorganic. Our conclusion questions a central assumption of many foreign element partitioning models. This does not imply that existing models are questionable, but that they share a common oversimplification. By removing this oversimplification all kinds of different models can be improved. Our study contributes broadly to the understanding of biogenic CaCO3 geochemistry, and it is relevant to the majority of existing models.
- Article
(1621 KB) - Full-text XML
-
Supplement
(543 KB) - BibTeX
- EndNote
The foreign element (Me) and isotopic composition of marine calcium carbonate (CaCO3) (mostly aragonite and calcite) biominerals from the sedimentary record has been used as a proxy for the reconstruction of specific environmental parameters such as seawater temperature since the 1950s (Katz et al., 2010; Urey et al., 1951). These geochemical proxies are instrumental in e.g. detecting effects of anthropogenic climate change on marine calcifying organisms (calcifiers) (Druffel, 1997; Katz et al., 2010; Pallacks et al., 2023). A geochemical paleo-proxy application requires a correlation of the proxy with the target environmental parameter, and this is traditionally achieved by various calibration methods (Allen et al., 2016; Elderfield and Ganssen, 2000). The calibration of a geochemical proxy alone, however, does convey little knowledge about the processes underlying proxy signals and accuracy. This knowledge is, however, essential for developing a mechanistic understanding of the proxy and eventually will enable us to predict proxy signals using conceptual biomineralization models (Nehrke and Langer, 2023). Biomineralization models, as opposed to calculations premised on precipitation of the mineral from seawater, are required because marine calcifiers used as proxy archives do not precipitate their hard parts from seawater but from a special calcification fluid thereby introducing the problem of the vital effect (Nehrke and Langer, 2023; Urey et al., 1951). This calcification fluid is localised in the so-called site of calcification (SOC). Different proxy-archive forming calcifiers have SOCs formed by different structures such as pseudopodia (foraminifera, single-celled calcifying organisms), mantle epithelium (molluscs, invertebrate animals that form a calcified shell), or intracellular vesicles (coccolithophores, single-celled calcifying algae) (Angell, 1967; Crenshaw, 1972; Langer et al., 2021; Wilbur and Watabe, 1963).
In all cases, however, the proxy signal will be influenced by the transport of ions from seawater into the SOC (Nehrke and Langer, 2023). This transport can introduce partitioning steps that render the overall partitioning different from what would be expected based on inorganic precipitation from seawater. A striking example is the signature in diverse calcifiers (Fig. 1). The data selected for Fig. 1 from the aragonite literature illustrate that the Sr partitioning coefficient (DSr= ()biomineral ()seawater) in some cases falls within the range of inorganic precipitation, in others it does not. We selected Sr incorporation in aragonitic biominerals here, but a well-known riddle is the Mg-problem, as it is often informally referred to, in calcitic biominerals. Specifically, the fact that does not simply reflect inorganic temperature-modulated partitioning but is strongly affected by biological (vital) effects. Organisms can actively regulate Mg transport, potentially resulting in calcifying fluids with varying ratios. Additionally, Mg might partly reside in the organics of a biomineral (Schöne et al., 2010), as opposed to the mineral phase (Branson et al., 2013) exhibiting different partitioning behaviour in organic and mineral phase respectively. These factors might cause biomineral to deviate from what would be expected from inorganic precipitation from seawater (Bentov and Erez, 2006; Nehrke et al., 2013).
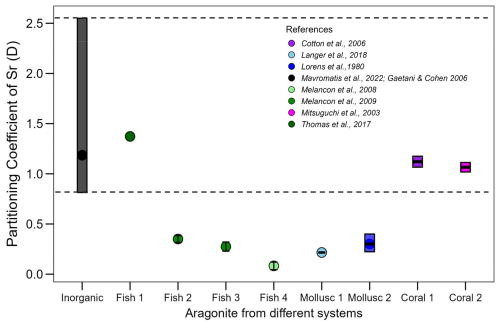
Figure 1Sr partitioning coefficient range (DSr= ()biomineral ()seawater) observed in different organisms with aragonite biominerals (shown in various colors) compared with the range for inorganic aragonite (black bar, with the full range indicated by the dashed black line). The references of the data are listed in the figure legend.
An intuitive, and often used, assumption is that knowledge of the ionic composition of the calcifying fluid would solve this problem. In other words, it is expected that a partitioning coefficient calculated using the calcifying fluid foreign element (Me) to Ca ratio () will fall within the range of values determined in inorganic precipitation experiments (Elderfield et al., 1996; Langer et al., 2006, 2016, 2018; Stoll et al., 2012). Unfortunately, the SOCs of most classic proxy-archive forming calcifiers are too small to be sampled for element analysis (Checa, 2018; Kadan et al., 2021; Nomaki et al., 2018). Therefore, various model approaches have been developed to calculate foreign element partitioning into biominerals (D'Olivo and McCulloch, 2017; Elderfield et al., 1996; Hohn and Merico, 2015; Langer et al., 2006, 2016; Nehrke and Langer, 2023; Ziveri et al., 2003, 2012). These models have provided new insights into the relationship between conceptual biomineralization models and foreign element partitioning, but they have, yet, failed to predict partitioning patterns based solely on independent constraints (Nehrke and Langer, 2023). Therefore, these models rely on assumptions, many of which do not account for the complexity of foreign element partitioning during biomineral formation. It is, for example, by no means self-evident that a partitioning coefficient calculated using the calcifying fluid composition will fall within the range of inorganic values.
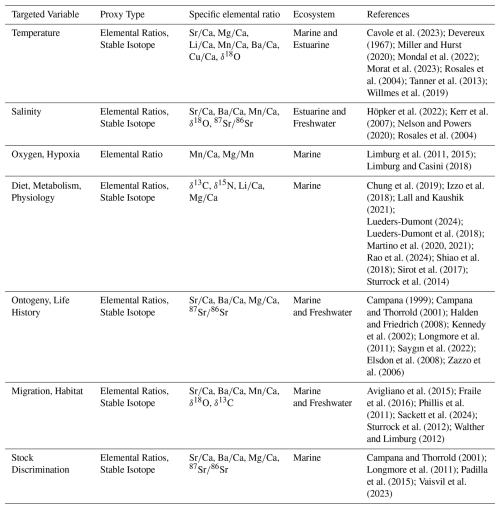
In this paper, we focus on fish otoliths, mostly aragonitic biominerals in the inner ears of bony fish, that are an understudied and underappreciated model system to address element partitioning patterns (Hüssy et al., 2021; Melancon et al., 2005). Otoliths can be found as fish remains in the sedimentary record (Elder et al., 1996; Mellars et al., 1980) and they have been used in many ways in fisheries research, ecology, and the reconstruction of fish stock environments (Reis-Santos et al., 2023). The foreign element compositions of otoliths can serve as proxies for e.g. migration patterns, salinity, and temperature (Albertsen et al., 2021; Bath Martin and Thorrold, 2005; Shiao et al., 2006). The otolith isotopic composition is also used as a proxy, e.g. habitat/migration is inferred from Sr and C isotopes, and dietary history from N and C isotopes, while O isotopes provide information about temperature and salinity (see Table 1 for an overview of otolith-based geochemical proxies). Otoliths serve as valuable proxy archives for several reasons: (a) unlike coccolithophores (which have coccoliths ranging from 2 to 20 µm) and require complex species-specific separation, otoliths allow monospecific analyses; (b) unlike planktonic foraminifera that live and calcify exclusively in marine environments and benthic foraminifera that are found also in brackish environments, otoliths biomineralize in teleost fishes across marine, brackish, and freshwater habitats, providing a potential archive of environmental conditions across diverse aquatic systems; (c) element-to-calcium ratios in individual otoliths can be spatially mapped, offering insights into the fish life history traits and seasonal patterns.
Table 1Summary of commonly studied geochemical elements in fish otoliths, their targeted environmental or biological variables, proxy types, specific elemental ratio, relevant ecosystems, and key references.
As with any other proxy archive, otolith-based proxies are subject to secondary influences. For example, and are influenced by their correspondent concentrations in ambient water, but also salinity and temperature (Hüssy et al., 2021). It has also been noted that, besides environmental parameters, physiology influences foreign element and isotope composition (Bareille et al., 2024; Izzo et al., 2018; Sturrock et al., 2015). The value of otoliths as geochemical proxy archives has been highlighted but is also, unsurprisingly, critically discussed (Hüssy et al., 2021; Thomas and Swearer, 2019; Walther, 2019). The latter authors emphasize that future steps towards improving otolith proxy applications critically include an understanding of the processes bringing about the proxy signal. Hüssy et al. (2021) effectively summarize the fundamental processes governing elemental and isotope fractionation into otoliths. They distinguish ion transport into the endolymph from “biomineralization” by which they mean the formation of the otolith within the endolymph. Note that often the term “biomineralization” covers both ion transport and formation of the biomineral within the SOC (Nehrke and Langer, 2023). As for foraminifera, there has been an increasing interest in the relationship between partitioning (usually called fractionation when referring to isotopes) patterns and biomineralization concepts in otoliths (Campana, 1999; Hüssy et al., 2021). To understand even the most straightforward and useful proxies, such as in foraminifera, both biological and inorganic processes need to be considered (Langer et al., 2016). Otoliths offer the unique opportunity to study the fractionation processes within the SOC in greater detail than is possible in classic proxy archives such as foraminifera.
An outstanding feature of otolith formation is the fact that the calcifying fluid, i.e. the endolymph, has such a large volume that it can be sampled for element analysis (Kalish, 1989). This offers the unique opportunity to measure foreign element composition of both the biomineral and its parent solution (Allemand et al., 2007; Edeyer et al., 2000; Kalish 1989, 1991; Melancon et al., 2005, 2008, 2009; Payan et al., 1997, 1998, 1999, 2002, 2004; Thomas et al., 2017). Although some of the latter studies provide the relevant data and discuss the relationship between partitioning and biomineralization processes, no study has addressed the following question: is the foreign element partitioning coefficient from endolymph into otolith (in the following called De) numerically equivalent to the one from an aqueous solution of inorganic ions into aragonite? Here we therefore use the relevant datasets in the literature to address this question. We look at six different foreign elements in four different species, one marine and three freshwater ones. We compare foreign element partitioning coefficient from endolymph to otolith with partitioning coefficient from inorganic aragonite precipitation. This study provides a deeper mechanistic understanding of the vital effect by identifying one, so far neglected, locus of the vital effect in the calcifying organism. The aim is to test the commonly made assumption that biogenic partitioning coefficients should be indistinguishable from inorganic ones if the of the actual parent solution (the calcifying fluid) of biomineral formation is used as denominator (e.g. Langer et al., 2006). Our results suggest that partitioning of foreign elements from endolymph into otolith cannot be modelled solely in terms of aragonite precipitation from an aqueous solution of inorganic ions. Our conclusion not only has implications for proxy understanding but also for biomineralization concepts because the latter centrally feature ideas about the composition of the calcifying fluid and its effect on biomineral formation.
2.1 Data collection and data handling
Literature data on ratios in endolymph and otolith were used to calculate partitioning coefficient. The latter were compared to partitioning coefficient determined in inorganic precipitation experiments. The literature data used are summarized in Table 2.
The literature on inorganic system provides many measurements of the partitioning coefficient (DIn) from different experimental designs. We selected the full range of values to get a realistic overall picture of the inorganic system. For the otolith-endolymph system the literature was limited. Only three papers gave us enough data to estimate the partitioning coefficient of the elements into the otolith. In the study of Thomas et al. (2017) they used a marine species, Acanthopagrus butcheri (Sp1), and the number of individuals measured was N=3. Otoliths were diluted and measured with an inductively coupled plasma mass spectrometer (ICP-MS) as the endolymph fluid. They provide the concentration of foreign elements (Me) normalized to the concentration calcium of (Ca) in the otolith and the endolymph ratio, with the ± SD and the range (R) = min(R) − max(R), as well the partitioning coefficient (D×100). We estimate the range of the D using a simple formula of range ratio (Eq. 1):
In the other two available studies with data meeting the criteria required for these estimates, were in a form of “concentration of foreign elements” in the otolith and in the endolymph (Melancon et al., 2008, 2009). The species that were used in these studies were the freshwater species burbot Lota lota (Sp2), lake trout Salvelinus namaycush (Sp3) and a walleye Sander vitreus (Sp4), the number of the individuals (N) that were used were Nburbot=18 and Ntrout=11 and Nwalley=8, respectively. In these studies, the concentrations of the elements in the otoliths were quantified by laser ablation (LA-ICP-MS) and they performed a series of ablations at the growing otolith edges that were in contact with the endolymph and represent the last 30 to 60 d of growth. They provide the average concentration of the foreign elements av [Me] ± SD (ppm) in the otolith and in the endolymph, which we converted to mmol mol−1. The ratio in the otolith and in the endolymph and the av (D) ± SD (D) was estimated using Eqs. (2) and (3)
2.2 Partitioning coefficient estimations
Then the ratio and the partitioning coefficient, in the different parts of the ion transport pathway that the elements need to cross to precipitate in the otolith, were estimated. Only one paper provided sufficient data for this purpose (Melancon et al., 2009). The ion transport pathway starts from the ambient water to blood, then from blood to endolymph and the final step is from endolymph to otolith (key figure). The partitioning coefficient that describes the last step of endolymph to otolith was estimated (Eq. 4). Some extra variables were also estimated. The first was the D commonly used in biomineralization studies. This Dw is the partitioning coefficient using as parent solution the ambient water (Eq. 5). The other two were estimated based on the idea that the last step of the precipitation is purely inorganic (Eqs. 7 and 8). The first of those (Eq. 7), is the ratio that the otolith would have, if the last precipitation step was completely inorganic and the parent solution was the endolymph (Rotolith1). The second (Eq. 8) is the theoretical ratio if the parent solution was water (Rotolith2).
We address the following question. Does the numerical value of the foreign element partitioning coefficient from endolymph into otolith equal that of the partitioning coefficient from an aqueous solution into pure aragonite? As mentioned in the introduction, Fig. 1 illustrates the range of the Sr partitioning coefficient (D) in different calcifying marine organisms, we used as parent solution seawater and the ratios of the elements in each organism from the literature (Broecker and Peng, 1982; Cohen et al., 2006; Langer et al., 2018; Lorens and Bender, 1980; Mitsuguchi et al., 2003).
The partitioning coefficient of various foreign elements from four fish species was comparatively analyzed with the inorganic partitioning coefficient of the same element (Fig. 2). To elaborate further, the distinct environments have a different impact on Ba, which exhibit divergent patterns among marine and freshwater species. We observe that for Ba in the Sp1 (marine) their D is within the range of inorganic values while the Sp2, Sp3, Sp4 are below the inorganic range. This phenomenon stands in contrast to the behavior observed in Mg that all fish otoliths D are in the range of the inorganic DIn of Mg. The De of the elements Na and Sr, with the borderline case of Sr in one fish (Sp2), fall within the inorganic range when the Dw does not. Zn represents a distinctive case; while Dw appears to be within the range of the inorganic system, De is not. A comprehensive analysis of the available evidence suggests that the “vital effect” manifests in some elements while remaining invisible in others. The supplementary material contains the partitioning coefficient De of Li in only for the Sp1 which is above the DIn of Li.
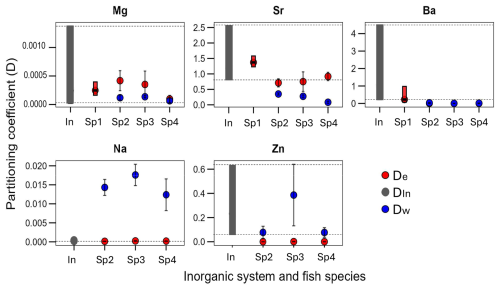
Figure 2The partitioning coefficient (D) per element (Mg, Sr, Ba, Na, Zn), the range of the inorganic system (In) and the mean ± SD of the D in three different freshwater fish burbot Lota lota (Sp2), lake trout Salvelinus namaycush (Sp3)and a walleye Sander vitreus (Sp4) and the range of the marine fish the Acanthopagrus butcheri (Sp1). Colour code: red is the De (endolymph as parent solution) and blue the Dw (water as parent solution) for fish and black is the DIn of the inorganic aragonite.
The ion transport pathways of five distinct elements of the Sp2 otolith are demonstrated (Fig. 3) and for the Sp3 (Fig. S2). The ratios of elements normalized to calcium (Ca) were measured in different solutions to trace their transport pathway into the otolith. Foreign elements of the ambient water pass to the blood and then to the endolymph where the biomineral is precipitated. The largest partitioning steps occur during the transition from water to blood and from endolymph to otolith. Despite the different patterns in the outcomes, it has been demonstrated that, in certain instances, the occurrence of the vital effect is visible as evidenced by the observation of a discrepancy in otolith1 relative to otolith (Fig. 3c, e). Conversely, in other instances, the vital effect remains invisible (Fig. 3a, b, d).
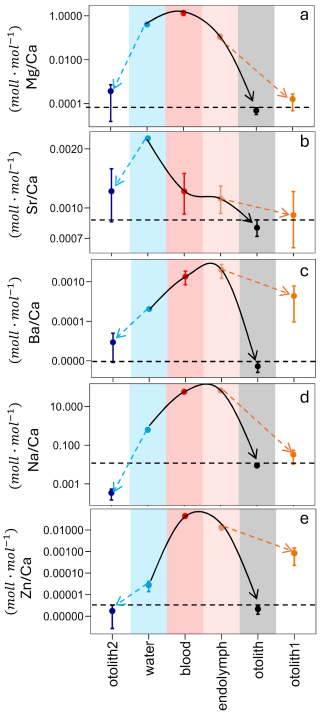
Figure 3 of burbot Lota lota (Sp2) in different reservoirs indicated by colored areas. The white areas (otolith 1 and otolith 2) do not represent measured values but are calculated according to otolith1 = DIn * ()endo, and otolith2 = DIn * ()water. For DIn we used either the minimum or the maximum value depending on which one would minimize the offset between () otolith-measured and () otolith-calculated. The error bar represents the range of the values that the system can reach. (a) is the pathway of Mg, (b) the pathway of Sr, (c) the pathway of Ba, (d) the pathway of Na and (e) the pathway of Zn. All the data are displayed on a log10-scaled y-axis.
Here we challenge the assumption that biomineral formation from the calcifying fluid is fully describable in terms of the formation of synthetic monocrystals from an aqueous solution of inorganic ions. Although intuitive, this idea might underestimate the complexity of biomineral formation for, inter alia, the following reasons. Firstly, biominerals are not monocrystals but organo-mineral composite structures, implying the possibility that foreign elements reside in the organic material (Cuif et al., 2010; Hüssy et al., 2021; Miller et al., 2006; Walker and Langer, 2021) Secondly, the calcifying fluid usually contains organic molecules, which could interact with inorganic ions thereby decreasing their activity ratios in solution and hence in the biomineral since mostly free ions are incorporated in the crystal (e.g. Borelli et al., 2001; Hüssy et al., 2021; Meyer et al., 2020; Moura et al., 2000; Thomas and Swearer, 2019). In the following we show that both processes do indeed influence foreign element distribution into otoliths which is, therefore, not reducible to inorganic aragonite co-precipitation.
4.1 Foreign element partitioning from endolymph to otolith cannot be modelled in terms of inorganic aragonite precipitation
We looked at the partitioning coefficient of six elements (Sr, Ba, Mg, Na, Zn, and Li) in four different species, Sp1–Sp4 (Figs. 2, S1 in the Supplement). For our question, it is helpful to consider several elements, as opposed to just one, because results from a single element might be misleading (Langer et al., 2018). A species comparison will further strengthen the conclusions because the question concerns the endolymph-otolith system in general. The partitioning coefficients De of Na, Mg (mostly), Sr and Ba (in Sp1) fall within the range of inorganic values (Figs. 2, S1). For the remaining elements, Zn and Li, otoliths show a partitioning behaviour different from inorganic aragonite. Taken together these results clearly show that the endolymph-otolith system produces foreign element partitioning coefficients different from the ones determined in synthetic aragonite precipitation. Therefore, we conclude that foreign element partitioning during otolith formation in the endolymph involves processes that do not occur during inorganic aragonite precipitation. An obvious further question is why the partitioning behaviour is both element and species specific. In general, the answer will likely involve specific organic material both in the endolymph and the otolith. In the following we will concretize this somewhat vague hypothesis.
4.2 Element and species specificity of partitioning behaviour
Otolith partitioning coefficients De of Ba and Zn (and Sr in Sp2 and partly Sp3) in freshwater species are lower than those in the inorganic system (Fig. 2). In the case of Sr and Ba incorporation into the organic part of the otolith seems negligible, ruling out a significant influence of otolith organics on partitioning (Izzo et al., 2016). This strongly suggests that endolymph organic material (Borelli et al., 2001; Thomas et al., 2019) forms complexes with divalent cations, fractionating for Sr and Ba. The remaining free ions in solution are incorporated into the growing otolith aragonite, with foreign element partitioning depending on crystal growth rate, in turn depending on various factors such as supersaturation, stoichiometry, and surface topography (Nehrke et al., 2007; Wolthers et al., 2013).
An example of organic material fractionating for Sr and Ba are polysaccharides such as alginates (Yuryev et al., 1979). The situation might be different for Mg which is fractionated against when forming complexes with organics thereby weakening fractionation against Mg into calcite (Mavromatis et al., 2017; Takeuchi et al., 2008). Complexation of foreign elements with inorganic ligands can also affect partitioning into calcium carbonate. In the case of Mg, sulfate complexes lead to an apparently harder fractionation against Mg into calcite (Goetschl et al., 2019; Mucci et al., 1989). Since for Mg, organic and inorganic complexes influence partitioning behaviour differently the overall change in the partitioning coefficient will partly depend on the relative concentrations of these different ligands. Inorganic ligands such as sulfate, phosphate, and carbonate might play a considerable role in modifying partitioning behaviour in calcifying fluids. The modification of the partitioning behaviour will be foreign element specific too. While alkali metal (e.g. Na and Li) complexes are of foreign importance, Zn for example has a high affinity to form inorganic complexes with e.g. sulfate and carbonate (Krȩżel and Maret, 2016; Lewis and Randall, 1921; Olsher et al., 1991; Stanley and Byrne, 1990). However, Li partitioning into calcite is pH dependent (Füger et al., 2019). Since calcifying fluids are likely to feature high pH, Li partitioning into calcitic biominerals, and maybe aragonitic ones too, might display a “high pH signal”.
Why does Ba partitioning in the marine species Sp1 differ so strikingly from the one in the freshwater species (Fig. 2)? Rather than being a species effect, this might be a methodological effect. Otoliths from the freshwater species were analyzed by LA-ICP-MS, where only the edge of the otolith was targeted to achieve a better match with the endolymph analysis (see Material and Methods). Otoliths from the marine species were dissolved whole for solution analysis by ICP-MS. The of otoliths can vary substantially within single otoliths, often with high values near the otolith core (Hermann et al., 2016). This could explain both the higher De in the marine species and the larger range reaching both below and above the inorganic range (Fig. 2).
The situation for Zn is different from the one for Sr and Ba because 40 %–60 % of the Zn reside in otolith organics (McFadden et al., 2016; Miller et al., 2006). Although an effect of endolymph organics cannot be ruled out for Zn, it is equally possible that partitioning into otolith organics is different from partitioning into otolith aragonite. Differential partitioning between the organic and the mineral part of mollusc shells has been reported, supporting this possibility (Schöne et al., 2010). Additionally, organic complexes with Zn can comprise the majority of total Zn, for example in surface seawater down to 500 m (Bruland, 1989). These naturally occurring organic ligands in seawater will be important in calcifiers using seawater as substrate supply for calcification, e.g. foraminifera (Elderfield et al., 1996). The influence of organic material in the calcifying fluid on foreign element partitioning shows that the localization of the foreign element in the mineral part of the biomineral does not justify the conclusion that the partitioning process is inorganic. This reasoning has nevertheless been applied to Mg partitioning into foraminiferal calcite (Branson et al., 2013). The latter authors show that Mg resides in foraminiferal calcite and from this observation conclude that its partitioning behaviour of Mg is inorganic. Since Mg is an important temperature proxy (Elderfield and Ganssen, 2000), this example illustrates the usefulness of the endolymph-otolith system for the development of a process-based understanding of proxy signal formation more generally.
Please note that foraminiferal shells are calcitic while otoliths are aragonitic. This difference in the calcium carbonate polymorph used by different organisms has implications for numerical values of partitioning coefficients (Langer et al., 2018), but has no bearing on the argument made above. We claim that the fact that Mg resides in the mineral phase of a biomineral, as opposed to the organic phase, is not sufficient to support the inference that the partitioning process is a case of inorganic co-precipitation. This claim holds regardless of the calcium carbonate mineral into which Mg is incorporated, and regardless of the of the respective mineral. Foraminifera, for example, comprise low-Mg (e.g. Ammonia), intermediate-Mg (e.g. Amphistegina), and high-Mg (e.g. Heterostegina) species (Mewes et al., 2014, 2015; Raitzsch et al., 2010). In the example mentioned above (Branson et al., 2013), both a low-Mg species (Orbulina) and an intermediate-Mg species (Amphistegina) are discussed. The author's argument, as well as our findings, apply equally to both species; and would do so for any other species.
To sum up, foreign elements might reside either in the mineral (e.g. aragonite in otoliths) or the organic part of the biomineral. Partitioning of foreign elements into the organic part is most likely different from partitioning into the mineral part. Hence partitioning is not homogeneous across a biomineral. The calcifying fluid often contains organic and, potentially, inorganic ligands that form complexes with foreign elements thereby influencing partitioning into the biomineral.
4.3 Biogenic and inorganic partitioning coefficient numerically identical: Mg, Na and Zn
The partitioning behaviour of Mg and Na seems to suggest that these elements are coprecipitated into aragonite in a manner akin to synthetic aragonite formation (Fig. 2). If this was indeed so this would nevertheless not contradict our conclusion (see above), namely foreign element partitioning into otoliths involves processes other than aragonite precipitation from an aqueous solution of inorganic ions. The latter conclusion rests on the behaviour of the other elements as discussed above and is not invalidated by a putatively different behaviour of Mg and Na. However, the behaviour of Mg and Na might as well merely appear inorganic numerically (in terms of De) but the processes underlying De might involve organic material, i.e. the overall process of partitioning might be very different from inorganic precipitation. This phenomenon has been described for different calcifiers and is known by the term “invisible vital effect” (Nehrke and Langer, 2023, and references therein).
The likelihood of an invisible vital effect in De is nevertheless much smaller than in the D calculated traditionally, i.e. using the external water (seawater or freshwater) as denominator. We added the traditional partitioning coefficient (Dw) to our dataset (Fig. 2). For Mg and Zn, Dw falls within the range of inorganic values, but from this we cannot conclude that Mg and Zn partitioning proceeds via inorganic precipitation from external water. We know that for ions to enter the endolymph they need to be transported via the blood (Hüssy et al., 2021; McCormick and McKinlay, 2000; Sturrock et al., 2015) so that there are at least two partitioning steps operative before the endolymph-otolith step. We look at these partitioning steps in the following section.
4.4 The pathway of foreign elements from water to otolith
In the freshwater species Sp2 (Fig. 3) and Sp3 (Fig. S2) were the only dataset that allows us to trace the pathway of foreign elements from external water into the otolith. In Fig. 3 we use (R) in different reservoirs along the ion transport pathway as given in the literature, and we additionally calculate two further values: (1) the Rotolith2 that results from multiplying Rw by DIn; (2) the Rotolith1 that results from multiplying Re by DIn. The DIn that are used are selected to minimize the offset between otolith (measured) and otolith (calculated). The aim of the figures is to illustrate the resulting (R) of different partitioning steps as they actually occur (coloured reservoirs), as opposed to the ones that would theoretically occur if DIn were applied.
Several things can be gleaned from this figure. The first concerns the match/mismatch of otolith and otolith1. A match indicates that partitioning from endolymph to otolith could be inorganic. This is the case for Sr, Mg, and Na. Note that the same conclusion can be drawn from Fig. 2 with the exception of DSr which does not fall within the inorganic range but is close to it. The reason for this discrepancy is that in Fig. 2a mean and standard deviation is given whereas in Fig. 3 the minimum value of DIn is used. The latter choice represents a conservative approach aiming at a match between otolith and otolith1. The case of Sr is therefore borderline, but its behaviour could still be considered inorganic. In stark contrast, the behaviour of Ba and Zn is clearly not inorganic.
The traditional way of calculating partitioning coefficients is from the ambient water to the biomineral because the composition of the calcifying fluid is unknown (e.g. Langer et al., 2006). This poses the central problem of the vital effect. The main question we are asking here is: can the problem of the vital effect be solved by knowledge of the composition of the calcifying fluid. The answer is yes for Sr and Na, and no for Ba, Zn, and Mg. Note that Dw of Zn and Mg show an invisible vital effect, so that using the correct parent solution can confer no numerical advantage. There is nevertheless knowledge to be gained. Knowing the values of otolith2 (Zn and Mg) merely tells us that there will be partitioning steps along the way from water into otolith, but the localization of partitioning along this pathway remains the classic “black box” (Nehrke and Langer, 2023). Here we can take a look into the black box in unprecedented detail. The step from water into blood fractionates weakly for Mg but strongly for Zn, while the following step into the endolymph fractionates weakly against both Mg and Zn. The last step from endolymph to otolith fractionates strongly against both Zn and Mg. While this step could be inorganic for Mg, it is more complex for Zn, i.e. the interaction of Zn with organics (as discussed above) contributes to this partitioning step. Biological partitioning steps are hard to predict in general, and in particular if the foreign element and Ca are transported by separate transport systems.
4.5 Essential versus non-essential elements
While Ca, Na, Mg, and Zn are essential elements, i.e. needed in physiological processes, there is no known physiological role for Sr and Ba, which are therefore considered non-essential (Lall and Kaushik, 2021; Marshall, 2002; Pors Nielsen, 2004; Salisbury and Ross, 1992). When considering foreign element partitioning into biominerals the distinction between essential and non-essential elements is of great importance because essential elements have their own transport systems while non-essential elements are thought to pass through the transport systems of essential elements (Langer et al., 2006, 2009). This means that partitioning from one reservoir into another (e.g. from water into blood) can be conceptualized easier for non-essential elements, because only the partitioning of individual transport systems has to be known. If one transport system transports the foreign element and another transports Ca the situation is more complicated because the two systems can be regulated independently. In the case of the non-essential elements Sr and Ba it is usually expected that they partition similarly if not with identical partitioning coefficient (Allen and Sanders, 1994; Langer et al., 2006, 2009; Nachshen and Blaustein, 1982). It is therefore surprising that the step from water to blood fractionates for Ba but against Sr, whereas the step from blood to endolymph does not fractionate at all (or only minimally) for both elements (Fig. 3). The partitioning from endolymph to otolith is against both Sr and Ba, i.e. according to expectation. The fact that Ba fractionation is harder than Sr fractionation could be explained by differential Sr and Ba partitioning of cellular transporters as well as organic polymers (e.g. Nachshen and Blaustein, 1982; Yuryev et al., 1979).
In this study we used literature data on foreign element composition of the endolymph-otolith system to calculate partitioning coefficient and analyse the partitioning behaviour of six elements in four species of fish. The endolymph-otolith system is outstanding because the parent solution (endolymph) of biomineral (otolith) formation can be sampled, and its elemental composition be determined. Our approach is novel since up to now the focus on traditional geochemical proxy archives (foraminifers, molluscs, and coccolithophores) has precluded such an analysis. Our data suggests that:
-
Otolith mineralization in the endolymph shows a vital effect. Partitioning from endolymph into otolith is influenced by organic material present in both endolymph and otolith and therefore cannot be reduced to aragonite precipitation from an aqueous solution of inorganic ions.
-
Differential partitioning patterns are more complex than generally assumed, as illustrated by the easy-to-conceptualize “model elements” Sr and Ba, which behave counter to expectation.
-
Future research should be specifically designed to address elemental partitioning within the endolymph, as clearly warranted by the findings of this study.
The data will be published in the “Mendeley data” platform with the reference as Kekelou (2026, https://doi.org/10.17632/8ysgz5nb82.3). But also they have been submitted as a Supplement.
Supplementary Figs. S1 and S2 are provided in the separate supplement file. Figure S1 illustrates the partitioning coefficient (D) for Li, comparing the range of the inorganic system and the marine fish (Acanthopagrus butcheri – Sp1). And Fig. S2 illustrates the Ion transport pathway of the elements Mg, Ba, Na, Sr and Zn in the Salvelinus namaycush (Sp3). Data Excel file: inorganic vs otolith-endolymph The supplement related to this article is available online at https://doi.org/10.5194/bg-23-2831-2026-supplement.
AK: Investigation, Formal analysis, Methodology, Data Curation, Writing – Original Draft, Visualization. GL: Conceptualization, Investigation, Writing – Original Draft and Review and Editing, Validation, Visualization, Project administration PZ: Supervision, Visualization, Writing – Review and Editing.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
We acknowledge support by the Open Access publication fund of Alfred-Wegener- Institute Helmholtz-Zentrum für Polar- und Meeresforschung. And we also acknowledge the reviewers and the editor for their constructive comments, which helped improve the clarity and quality of the manuscript.
This article processing charges for this open-access publication were covered by the Alfred-Wegener-Institut Helmholtz-Zentrum für Polar- und Meeresforschung. AK acknowledges funding from the Spanish Ministry of Science and Innovation through the FPI fellowship (PRE2022-102298). GL acknowledges funding from the Spanish Ministry of Universities through a Maria Zambrano grant and the Generalitat de Catalunya (MERS, 2021 SGR 00640). This work contributes to ICTA-UAB “María de Maeztu” Programmes for Units of Excellence of the Spanish Ministry of Science and Innovation (CEX2019-000940-M; MICIU/AEI/10.13039/501100011033), the BIOCAL Project (PID2020-113526RB-I00), and the AEI-DFG project BONITOS (PCI2025-163190, 541693727).
This paper was edited by Niels de Winter and reviewed by Nina Keul and Karin Limburg.
Albertsen, C. M., Hüssy, K., Serre, S. H., Hemmer-Hansen J., and Thomsen, T. B.: Estimating migration patterns of fish from otolith chemical composition time series, Can. J. Fish. Aquat. Sci., 78, 1512–1523, https://doi.org/10.1139/cjfas-2020-0356, 2021.
Allemand, D., Mayer-Gostan, N., De Pontual, H., Boeuf, G., and Payan, P.: Fish Otolith Calcification in Relation to Endolymph Chemistry, in: Handbook of Biomineralization, 291–308, https://doi.org/10.1002/9783527619443.ch17, 2007.
Allen, G. J. and Sanders, D.: Two Voltage-Gated, Calcium Release Channels Coreside in the Vacuolar Membrane of Broad Bean Guard Cells, The Plant Cell, 685–694, https://doi.org/10.1105/tpc.6.5.685, 1994.
Allen, K. A., Hönisch, B., Eggins, S. M., Haynes, L. L., Rosenthal, Y., and Yu, J.: Trace element proxies for surface ocean conditions: a synthesis of culture calibrations with planktic foraminifera, Geochim. Cosmochim. Ac., 193, 197–221, https://doi.org/10.1016/j.gca.2016.08.015, 2016.
Angell, R. W.: The process of chamber formation in the foraminifer Rosalina floridana (Cushman), J. Protozool., 14, 566–574, https://doi.org/10.1111/j.1550-7408.1967.tb02043.x, 1967.
Avigliano, E., Callicó-Fortunato, R., Buitrago, J., and Volpedo, A. V.: A microquímica do otólito é um indicador do habitat de Mugil curema no sudeste do Mar Caribenho?, Braz. J. Biol., 75, S45–S51, https://doi.org/10.1590/1519-6984.01014, 2015.
Bareille, G., Vignon, M., Chappaz, A., Fontaine, A., Tabouret, H., Morat, F., Martin, J., Aymes, J. C., Daverat, F., Pécheyran, C., and Donard, O.: Freshwater fish otoliths record signals from both water and physiological processes: new insights from and ratios, Can. J. Fish. Aquat. Sci., 81, 223–240, https://doi.org/10.1139/cjfas-2022-0030, 2024.
Bath Martin, G. and Thorrold, S.: Temperature and salinity effects on magnesium, manganese, and barium incorporation in otoliths of larval and early juvenile spot Leiostomus xanthurus, Mar. Ecol. Prog. Ser., 293, 223–232, https://doi.org/10.3354/meps293223, 2005.
Bentov, S. and Erez, J.: Impact of biomineralization processes on the Mg content of foraminiferal shells: a biological perspective, Geochem. Geophy. Geosy., 7, https://doi.org/10.1029/2005GC001015, 2006.
Borelli, G., Mayer-Gostan, N., De Pontual, H., Boeuf, G., and Payan, P.: Biochemical relationships between endolymph and otolith matrix in the trout (Oncorhynchus mykiss) and turbot (Psetta maxima), Calcif. Tissue Int., 69, 356–364, https://doi.org/10.1007/s00223-001-2016-8, 2001.
Branson, O., Redfern, S. A. T., Tyliszczak, T., Sadekov, A., Langer, G., Kimoto, K., and Elderfield, H.: The coordination of Mg in foraminiferal calcite, Earth Planet. Sc. Lett., 383, 134–141, https://doi.org/10.1016/j.epsl.2013.09.037, 2013.
Brazier, J.-M., Blanchard, M., Méheut, M., Schmitt, A.-D., Schott, J., and Mavromatis, V.: Experimental and theoretical investigations of stable Sr isotope fractionation during its incorporation in aragonite, Geochim. Cosmochim. Ac., 358, 134–147, https://doi.org/10.1016/j.gca.2023.08.013, 2023.
Brazier, J. M., Goetschl, K. E., Dietzel, M., and Mavromatis, V.: Effect of mineral growth rate on zinc incorporation into calcite and aragonite, Chem. Geol., 643, https://doi.org/10.1016/j.chemgeo.2023.121821, 2024a.
Brazier, J. M., Harrison, A. L., Rollion-Bard, C., and Mavromatis, V.: Controls of temperature and mineral growth rate on lithium and sodium incorporation in abiotic aragonite, Chem. Geol., 654, https://doi.org/10.1016/j.chemgeo.2024.122057, 2024b.
Broecker, W. S. and Peng, T.-H.: Tracers in the sea, Eldigio Press, Lamont-Doherty Geological Observatory, Columbia University, 1982.
Bruland, K. W.: Complexation of zinc by natural organic ligands in the central North Pacific, Limnol. Oceanogr., 34, 269–285, https://doi.org/10.4319/lo.1989.34.2.0269, 1989.
Campana, S.: Chemistry and composition of fish otoliths: pathways, mechanisms and applications, Mar. Ecol. Prog. Ser., 188, 263–297, https://doi.org/10.3354/meps188263, 1999.
Campana, S. E. and Thorrold, S. R.: Otoliths, increments, and elements: keys to a comprehensive understanding of fish populations?, Can. J. Fish. Aquat. Sci., 58, 30–38, https://doi.org/10.1139/f00-177, 2001.
Cavole, L. M., Limburg, K. E., Gallo, N. D., Vea Salvanes, A. G., Ramírez-Valdez, A., Levin, L. A., Oropeza, O. A., Hertwig, A., Liu, M. C., and McKeegan, K. D.: Otoliths of marine fishes record evidence of low oxygen, temperature and pH conditions of deep oxygen minimum zones, Deep-Sea Res. Pt. I, 191, https://doi.org/10.1016/j.dsr.2022.103941, 2023.
Checa, A. G.: Physical and biological determinants of the fabrication of molluscan shell microstructures, Front. Mar. Sci., 5, https://doi.org/10.3389/fmars.2018.00353, 2018.
Chung, M.-T., Trueman, C. N., Godiksen, J. A., and Grønkjær, P.: Otolith δ13C values as a metabolic proxy: approaches and mechanical underpinnings, Mar. Freshwater Res., 70, 1747, https://doi.org/10.1071/MF18317, 2019.
Cohen, A. L., Gaetani, G. A., Lundälv, T., Corliss, B. H., and George, R. Y.: Compositional variability in a cold-water scleractinian, Lophelia pertusa: new insights into “vital effects”, Geochem. Geophy. Geosy., 7, https://doi.org/10.1029/2006GC001354, 2006.
Crenshaw, M. A.: The inorganic composition of molluscan extrapallial fluid, Biol. Bull., 143, 506–512, https://doi.org/10.2307/1540180, 1972.
Cuif, J.-P., Dauphin, Y., and Sorauf, J. E.: Biominerals and fossils through time, Cambridge University Press, https://doi.org/10.1017/CBO9780511781070, 2010.
Devereux, I.: Temperature measurements from oxygen isotope ratios of fish otoliths, Science, 155, 1684–1685, https://doi.org/10.1126/science.155.3770.1684, 1967.
Dietzel, M., Gussone, N., and Eisenhauer, A.: Co-precipitation of Sr2+ and Ba2+ with aragonite by membrane diffusion of CO2 between 10 and 50 °C, Chem. Geol., 203, 139–151, https://doi.org/10.1016/j.chemgeo.2003.09.008, 2004.
D'Olivo, J. P. and McCulloch, M. T.: Response of coral calcification and calcifying fluid composition to thermally induced bleaching stress, Sci. Rep., 7, 2207, https://doi.org/10.1038/s41598-017-02306-x, 2017.
Druffel, E. R. M.: Geochemistry of corals: proxies of past ocean chemistry, ocean circulation, and climate, P. Natl. Acad. Sci. USA, 94, 8354–8361, https://doi.org/10.1073/pnas.94.16.8354, 1997.
Edeyer, A., De Pontual, H., Payan, P., Troadec, H., Sévère, A., and Mayer-Gostan, N.: Daily variations of the saccular endolymph and plasma compositions in the turbot Psetta maxima: relationship with the diurnal rhythm in otolith formation, Mar. Ecol. Prog. Ser., 192, 287–294, https://doi.org/10.3354/meps192287, 2000.
Elder, K. L., Jones, G. A., and Bolz, G.: Distribution of otoliths in surficial sediments of the U.S. Atlantic continental shelf and slope and potential for reconstructing Holocene fish stocks, Paleoceanography, 11, 359–367, https://doi.org/10.1029/96PA00042, 1996.
Elderfield, H. and Ganssen, G.: Past temperature and δ18O of surface ocean waters inferred from foraminiferal ratios, Nature, 405, 442–445, https://doi.org/10.1038/35013033, 2000.
Elderfield, H., Bertram, C. J., and Erez, J.: A biomineralization model for the incorporation of trace elements into foraminiferal calcium carbonate, Earth Planet. Sc. Lett., 142, 409–423, https://doi.org/10.1016/0012-821X(96)00105-7, 1996.
Fraile, I., Arrizabalaga, H., Santiago, J., Goñi, N., Arregi, I., Madinabeitia, S., Wells, R. J. D., and Rooker, J. R.: Otolith chemistry as an indicator of movements of albacore (Thunnus alalunga) in the North Atlantic Ocean, Mar. Freshwater Res., 67, 1002, https://doi.org/10.1071/MF15097, 2016.
Füger, A., Konrad, F., Leis, A., Dietzel, M., and Mavromatis, V.: Effect of growth rate and pH on lithium incorporation in calcite, Geochim. Cosmochim. Ac., 248, 14–24, https://doi.org/10.1016/j.gca.2018.12.040, 2019.
Gaetani, G. A. and Cohen, A. L.: Element partitioning during precipitation of aragonite from seawater: a framework for understanding paleo proxies, Geochim. Cosmochim. Ac., 70, 4617–4634, https://doi.org/10.1016/j.gca.2006.07.008, 2006.
Goetschl, K. E., Purgstaller, B., Dietzel, M., and Mavromatis, V.: Effect of sulfate on magnesium incorporation in low-magnesium calcite, Geochim. Cosmochim. Ac., 265, 505–519, https://doi.org/10.1016/j.gca.2019.07.024, 2019.
Halden, N. M. and Friedrich, L. A.: Trace-element distributions in fish otoliths: natural markers of life histories, environmental conditions and exposure to tailings effluence, Mineral. Mag., 72, 593–605, https://doi.org/10.1180/minmag.2008.072.2.593, 2008.
Hermann, T. W., Stewart, D. J., Limburg, K. E., and Castello, L.: Unravelling the life history of Amazonian fishes through otolith microchemistry, R. Soc. Open Sci., 3, 160206, https://doi.org/10.1098/rsos.160206, 2016.
Hohn, S. and Merico, A.: Quantifying the relative importance of transcellular and paracellular ion transports to coral polyp calcification, Front. Earth Sci., 2, https://doi.org/10.3389/feart.2014.00037, 2015.
Höpker, S. N., Wu, H. C., Lucassen, F., Sadio, O., Brochier, T., Nuworkpor, I. Y., Kasemann, S. A., Merschel, P., and Westphal, H.: Sr isotope ratios () in water and fish otoliths as estuarine salinity tracers: case studies from three NW African rivers, Estuar. Coast., 45, 1780–1802, https://doi.org/10.1007/s12237-021-01041-x, 2022.
Hüssy, K., Limburg, K. E., de Pontual, H., Thomas, O. R. B., Cook, P. K., Heimbrand, Y., Blass, M., and Sturrock, A. M.: Trace element patterns in otoliths: the role of biomineralization, Rev. Fish. Sci. Aquac., 29, 445–477, https://doi.org/10.1080/23308249.2020.1760204, 2021.
Izzo, C., Doubleday, Z. A., and Gillanders, B. M.: Where do elements bind within the otoliths of fish?, Mar. Freshwater Res., 67, 1072, https://doi.org/10.1071/MF15064, 2016.
Izzo, C., Reis-Santos, P., and Gillanders, B. M.: Otolith chemistry does not just reflect environmental conditions: a meta-analytic evaluation, Fish Fish., 19, 441–454, https://doi.org/10.1111/faf.12264, 2018.
Kadan, Y., Tollervey, F., Varsano, N., Mahamid, J., and Gal, A.: Intracellular nanoscale architecture as a master regulator of calcium carbonate crystallization in marine microalgae, P. Natl. Acad. Sci. USA, 118, https://doi.org/10.1073/pnas.2025670118, 2021.
Kalish, J. M.: Otolith microchemistry: validation of the effects of physiology, age and environment on otolith composition, J. Exp. Mar. Biol. Ecol., 132, 151–178, https://doi.org/10.1016/0022-0981(89)90126-3, 1989.
Kalish, J. M.: Determinants of otolith chemistry: seasonal variation in the composition of blood plasma, endolymph and otoliths of bearded rock cod Pseudophycis barbatus, Mar. Ecol. Prog. Ser., 74, 137–159, https://doi.org/10.3354/meps074137, 1991.
Katz, M. E., Cramer, B. S., Franzese, A., Hönisch, B., Miller, K. G., Rosenthal, Y., and Wright, J. D.: Traditional and emerging geochemical proxies in foraminifera, J. Foraminiferal Res., 40, 165–192, https://doi.org/10.2113/gsjfr.40.2.165, 2010.
Kawabata, T., Takeda, Y., Hori, M., Kandori, K., and Yaji, T.: Partitioning of sodium into calcium carbonates synthesized at 10–40 °C: influence of organic ligands and temperature, Chem. Geol., 559, https://doi.org/10.1016/j.chemgeo.2020.119904, 2021.
Kekelou, A.: Mineralizing Fluid Control on Foreign Elements in Biogenic CaCO3: Insights from Otoliths, V3, Mendeley Data [data set], https://doi.org/10.17632/8ysgz5nb82.3, 2026.
Kennedy, B. P., Klaue, A., Blum, J. D., Folt, C. L., and Nislow, K. H.: Reconstructing the lives of fish using Sr isotopes in otoliths, Can. J. Fish. Aquat. Sci., 59, 925–929, https://doi.org/10.1139/f02-070, 2002.
Kerr, L. A., Secor, D. H., and Kraus, R. T.: Stable isotope (δ13C and δ18O) and composition of otoliths as proxies for environmental salinity experienced by an estuarine fish, Mar. Ecol. Prog. Ser., 349, 245–253, https://doi.org/10.3354/meps07064, 2007.
Krȩżel, A. and Maret, W.: The biological inorganic chemistry of zinc ions, Arch. Biochem. Biophys., 611, 3–19, https://doi.org/10.1016/j.abb.2016.04.010, 2016.
Lall, S. P. and Kaushik, S. J.: Nutrition and metabolism of minerals in fish, Animals, 11, 2711, https://doi.org/10.3390/ani11092711, 2021.
Langer, G., Gussone, N., Nehrke, G., Riebesell, U., Eisenhauer, A., Kuhnert, H., Rost, B., Trimborn, S., and Thoms, S.: Coccolith strontium to calcium ratios in Emiliania huxleyi: the dependence on seawater strontium and calcium concentrations, Limnol. Oceanogr., 51, 310–320, https://doi.org/10.4319/lo.2006.51.1.0310, 2006.
Langer, G., Nehrke, G., Thoms, S., and Stoll, H.: Barium partitioning in coccoliths of Emiliania huxleyi, Geochim. Cosmochim. Ac., 73, 2899–2906, https://doi.org/10.1016/j.gca.2009.02.025, 2009.
Langer, G., Sadekov, A., Thoms, S., Keul, N., Nehrke, G., Mewes, A., Greaves, M., Misra, S., Reichart, G.-J., de Nooijer, L. J., Bijma, J., and Elderfield, H.: Sr partitioning in the benthic foraminifera Ammonia aomoriensis and Amphistegina lessonii, Chem. Geol., 440, 306–312, https://doi.org/10.1016/j.chemgeo.2016.07.018, 2016.
Langer, G., Sadekov, A., Nehrke, G., Baggini, C., Rodolfo-Metalpa, R., Hall-Spencer, J. M., Cuoco, E., Bijma, J., and Elderfield, H.: Relationship between mineralogy and minor element partitioning in limpets from an Ischia CO2 vent site provides new insights into their biomineralization pathway, Geochim. Cosmochim. Ac., 236, 218–229, https://doi.org/10.1016/j.gca.2018.02.044, 2018.
Langer, G., Taylor, A. R., Walker, C. E., Meyer, E. M., Ben Joseph, O., Gal, A., Harper, G. M., Probert, I., Brownlee, C., and Wheeler, G. L.: Role of silicon in the development of complex crystal shapes in coccolithophores, New Phytol., 231, 1845–1857, https://doi.org/10.1111/nph.17230, 2021.
Lewis, G. N. and Randall, M.: The activity coefficient of strong electrolytes, J. Am. Chem. Soc., 43, 1112–1154, 1921.
Limburg, K. E. and Casini, M.: Effect of marine hypoxia on Baltic Sea cod Gadus morhua: evidence from otolith chemical proxies, Front. Mar. Sci., 5, 482, https://doi.org/10.3389/fmars.2018.00482, 2018.
Limburg, K. E., Olson, C., Walther, Y., Dale, D., Slomp, C. P., and Høie, H.: Tracking Baltic hypoxia and cod migration over millennia with natural tags, P. Natl. Acad. Sci. USA, 108, https://doi.org/10.1073/pnas.1100684108, 2011.
Limburg, K. E., Walther, B. D., Lu, Z., Jackman, G., Mohan, J., Walther, Y., Nissling, A., Weber, P. K., and Schmitt, A. K.: In search of the dead zone: use of otoliths for tracking fish exposure to hypoxia, J. Mar. Syst., 141, 167–178, https://doi.org/10.1016/j.jmarsys.2014.02.014, 2015.
Longmore, C., Trueman, C. N., Neat, F., O'Gorman, E. J., Milton, J. A., and Mariani, S.: Otolith geochemistry indicates life-long spatial population structuring in a deep-sea fish, Coryphaenoides rupestris, Mar. Ecol. Prog. Ser., 435, 209–224, https://doi.org/10.3354/meps09248, 2011.
Lorens, R. B. and Bender, M. L.: The impact of solution chemistry on Mytilus edulis calcite and aragonite, Geochim. Cosmochim. Ac., 44, 1265–1278, https://doi.org/10.1016/0016-7037(80)90087-3, 1980.
Lueders-Dumont, J.: Postcards from prehistoric marine food webs: nitrogen isotopes in fish otoliths as a paleoecological archive, Geol. Soc. Am. Abstr., 56, 403326, https://doi.org/10.1130/abs/2024AM-403326, 2024.
Lueders-Dumont, J. A., Wang, X. T., Jensen, O. P., Sigman, D. M., and Ward, B. B.: Nitrogen isotopic analysis of carbonate-bound organic matter in modern and fossil fish otoliths, Geochim. Cosmochim. Ac., 224, 200–222, https://doi.org/10.1016/j.gca.2018.01.001, 2018.
Marriott, C. S., Henderson, G. M., Crompton, R., Staubwasser, M., and Shaw, S.: Effect of mineralogy, salinity, and temperature on and Li isotope composition of calcium carbonate, Chem. Geol., 212, 5–15, https://doi.org/10.1016/j.chemgeo.2004.08.002, 2004.
Marshall, W. S.: Na+, Cl−, Ca2+ and Zn2+ transport by fish gills: retrospective review and prospective synthesis, J. Exp. Zool., 293, 264–283, https://doi.org/10.1002/jez.10127, 2002.
Martino, J. C., Doubleday, Z. A., Chung, M.-T., and Gillanders, B. M.: Experimental support towards a metabolic proxy in fish using otolith carbon isotopes, J. Exp. Biol., 223, jeb217091, https://doi.org/10.1242/jeb.217091, 2020.
Martino, J. C., Doubleday, Z. A., Fowler, A. J., and Gillanders, B. M.: Corrigendum to: Identifying physiological and environmental influences on otolith chemistry in a coastal fishery species, Mar. Freshw. Res., 72, 922, https://doi.org/10.1071/MF20196_CO, 2021.
Mavromatis, V., Immenhauser, A., Buhl, D., Purgstaller, B., Baldermann, A., and Dietzel, M.: Effect of organic ligands on Mg partitioning and Mg isotope fractionation during low-temperature precipitation of calcite in the absence of growth rate effects, Geochim. Cosmochim. Ac., 207, 139–153, https://doi.org/10.1016/j.gca.2017.03.020, 2017.
Mavromatis, V., Goetschl, K. E., Grengg, C., Konrad, F., Purgstaller, B., and Dietzel, M.: Barium partitioning in calcite and aragonite as a function of growth rate, Geochim. Cosmochim. Ac., 237, 65–78, https://doi.org/10.1016/j.gca.2018.06.018, 2018.
Mavromatis, V., Brazier, J. M., and Goetschl, K. E.: Controls of temperature and mineral growth rate on Mg incorporation in aragonite, Geochim. Cosmochim. Ac., 317, 53–64, https://doi.org/10.1016/j.gca.2021.10.015, 2022.
McCormick, S. and McKinlay, D. M.: Ion regulation in fish, symposium proceedings of the International Congress on the Biology of Fish, University of British Columbia, Vancouver, Canada, Am. Fish. Soc., ISBN: 1-894337-34-4, 2002.
McFadden, A., Wade, B., Izzo, C., Gillanders, B. M., Lenehan, C. E., and Pring, A.: Quantitative electron microprobe mapping of otoliths suggests elemental incorporation is affected by organic matrices: implications for the interpretation of otolith chemistry, Mar. Freshw. Res., 67, 889–898, https://doi.org/10.1071/MF15074, 2016.
Melancon, S., Fryer, B. J., Ludsin, S. A., Gagnon, J. E., and Yang, Z.: Effects of crystal structure on the uptake of metals by lake trout (Salvelinus namaycush) otoliths, Can. J. Fish. Aquat. Sci., 62, 2609–2619, https://doi.org/10.1139/f05-161, 2005.
Melancon, S., Fryer, B. J., Gagnon, J. E., and Ludsin, S. A.: Mineralogical approaches to the study of biomineralization in fish otoliths, Mineral. Mag., 72, 627–637, https://doi.org/10.1180/minmag.2008.072.2.627, 2008.
Melancon, S., Fryer, B. J., and Markham, J. L.: Chemical analysis of endolymph and the growing otolith: fractionation of metals in freshwater fish species, Environ. Toxicol. Chem., 28, 1279–1287, https://doi.org/10.1897/08-358.1, 2009.
Mellars, P. A., Wilkinson, M. R., and Fieller, N. R. J.: Fish otoliths as indicators of seasonality in prehistoric shell middens: the evidence from Oronsay (Inner Hebrides), Proc. Prehist. Soc., 46, 19–44, https://doi.org/10.1017/S0079497X00009300, 1980.
Mewes, A., Langer, G., de Nooijer, L. J., Bijma, J., and Reichart, G. J.: Effect of different seawater Mg2+ concentrations on calcification in two benthic foraminifers, Mar. Micropaleontol., 113, 56–64, https://doi.org/10.1016/j.marmicro.2014.09.003, 2014.
Mewes, A., Langer, G., de Nooijer, L. J., Reichart, G. J., and Bijma, J.: Effect of seawater chemistry on calcification in benthic foraminifera, Chem. Geol., 2015, https://doi.org/10.1016/j.chemgeo.2015.06.026, 2015.
Meyer, E. M., Langer, G., Brownlee, C., Wheeler, G. L., and Taylor, A. R.: Sr in coccoliths of Scyphosphaera apsteinii: partitioning behavior and role in coccolith morphogenesis, Geochim. Cosmochim. Ac., 285, 41–54, https://doi.org/10.1016/j.gca.2020.06.023, 2020.
Miller, J. A. and Hurst, T. P.: Growth rate, ration, and temperature effects on otolith elemental incorporation, Front. Mar. Sci., 7, 320, https://doi.org/10.3389/fmars.2020.00320, 2020.
Miller, M. B., Clough, A. M., Batson, J. N., and Vachet, R. W.: Transition metal binding to cod otolith proteins, J. Exp. Mar. Biol. Ecol., 329, 135–143, https://doi.org/10.1016/j.jembe.2005.08.016, 2006.
Mitsuguchi, T., Matsumoto, E., and Uchida, T.: and ratios of Porites coral skeleton: evaluation of the effect of skeletal growth rate, Coral Reefs, 22, 381–388, https://doi.org/10.1007/s00338-003-0326-1, 2003.
Mondal, S., Chakrabarti, R., and Ghosh, P.: A multi-proxy (Ca, , and Δ47) study of fish otoliths for determination of seawater temperature, Chem. Geol., 605, 120950, https://doi.org/10.1016/j.chemgeo.2022.120950, 2022.
Morat, F., Blamart, D., Bounket, B., Argillier, C., Carrel, G., and Maire, A.: Reconstructing the thermal history of fish juveniles using stable oxygen isotope analysis of otoliths, Front. Environ. Sci., 11, 1213239, https://doi.org/10.3389/fenvs.2023.1213239, 2023.
Moura, G., Vilarinho, L., Santos, A. C., and Machado, J.: Organic compounds in the extrapallial fluid and haemolymph of Anodonta cygnea with emphasis on biomineralization, Comp. Biochem. Physiol. B., 125, 293–306, https://doi.org/10.1016/S0305-0491(99)00192-3, 2000.
Mucci, A., Canuel, R., and Zhong, S.: Solubility of calcite and aragonite in sulfate-free seawater and seeded growth kinetics at 25 °C, Chem. Geol., 74, 309–320, https://doi.org/10.1016/0009-2541(89)90040-5, 1989.
Nachshen, D. A. and Blaustein, M. P.: Influx of calcium, strontium, and barium in presynaptic nerve endings, J. Gen. Physiol., 79, 1065–1087, https://doi.org/10.1085/jgp.79.6.1065, 1982.
Nehrke, G. and Langer, G.: Proxy archives based on marine calcifying organisms and the role of process-based biomineralization concepts, Minerals, 13, 561, https://doi.org/10.3390/min13040561, 2023.
Nehrke, G., Reichart, G. J., Van Cappellen, P., Meile, C., and Bijma, J.: Dependence of calcite growth rate and Sr partitioning on solution stoichiometry: Non-Kossel crystal growth, Geochim. Cosmochim. Ac., 71, 2240–2249, https://doi.org/10.1016/j.gca.2007.02.002, 2007.
Nehrke, G., Keul, N., Langer, G., de Nooijer, L. J., Bijma, J., and Meibom, A.: A new model for biomineralization and trace-element signatures of Foraminifera tests, Biogeosciences, 10, 6759–6767, https://doi.org/10.5194/bg-10-6759-2013, 2013.
Nelson, T. R. and Powers, S. P.: Elemental concentrations of water and otoliths as salinity proxies in a northern Gulf of Mexico estuary, Estuar. Coast., 43, 843–864, https://doi.org/10.1007/s12237-019-00686-z, 2020.
Nomaki, H., LeKieffre, C., Escrig, S., Meibom, A., Yagyu, S., Richardson, E. A., Matsuzaki, T., Murayama, M., Geslin, E., and Bernhard, J. M.: Innovative TEM-coupled approaches to study foraminiferal cells, Mar. Micropaleontol., 138, 90–104, https://doi.org/10.1016/j.marmicro.2017.10.002, 2018.
Olsher, U., Izatt, R. M., Bradshaw, J. S., and Dalley, N. K.: Coordination chemistry of lithium ion: a crystal and molecular structure review, Chem. Rev., 91, 137–164, https://doi.org/10.1021/cr00002a003, 1991.
Padilla, A. J., Brown, R. J., and Wooller, M. J.: Strontium isotope analyses () of otoliths from anadromous Bering cisco (Coregonus laurettae) to determine stock composition, ICES J. Mar. Sci., 72, 2110–2117, https://doi.org/10.1093/icesjms/fsv096, 2015.
Pallacks, S., Ziveri, P., Schiebel, R., Vonhof, H., Rae, J. W. B., Littley, E., Garcia-Orellana, J., Langer, G., Grelaud, M., and Martrat, B.: Anthropogenic acidification of surface waters drives decreased biogenic calcification in the Mediterranean Sea, Commun. Earth Environ., 4, 301, https://doi.org/10.1038/s43247-023-00947-7, 2023.
Payan, P., Kossmann, H., Watrin, A., Mayer-Gostan, N., and Bœuf, G.: Ionic composition of endolymph in teleosts: origin and importance of endolymph alkalinity, J. Exp. Biol., 200, 1905–1912, https://doi.org/10.1242/jeb.200.13.1905, 1997.
Payan, P., Borelli, G., Boeuf, G., and Mayer-Gostan, N.: Relationship between otolith and somatic growth: consequence of starvation on acid-base balance in plasma and endolymph in the rainbow trout Oncorhynchus mykiss, in: Fish Physiol. Biochem., 19, 35–41, https://doi.org/10.1023/A:1016064813517, 1998.
Payan, P., Edeyer, A., De Pontual, N., Borelli, G., Bœuf, G., and Mayer-Gostan, N.: Chemical composition of saccular endolymph and otolith in fish inner ear: lack of spatial uniformity, Am. J. Physiol. Regul. Integr. Comp. Physiol., 277, R123–R131, https://doi.org/10.1152/ajpregu.1999.277.1.R123, 1999.
Payan, P., Borelli, G., Priouzeau, F., De Pontual, H., Bœuf, G., and Mayer-Gostan, N.: Otolith growth in trout Oncorhynchus mykiss: supply of Ca2+ and Sr2+ to the saccular endolymph, J. Exp. Biol., 205, 2687–2695, https://doi.org/10.1242/jeb.205.17.2687, 2002.
Payan, P., De Pontual, H., Bœuf, G., and Mayer-Gostan, N.: Endolymph chemistry and otolith growth in fish, C. R. Palevol., 3, 535–547, https://doi.org/10.1016/j.crpv.2004.07.013, 2004.
Phillis, C. C., Ostrach, D. J., Ingram, B. L., and Weber, P. K.: Evaluating otolith as a tool for reconstructing estuarine habitat use, Can. J. Fish. Aquat. Sci., 68, 360–373, https://doi.org/10.1139/F10-152, 2011.
Pors Nielsen, S.: The biological role of strontium, Bone, 35, 583–588, https://doi.org/10.1016/j.bone.2004.04.026, 2004.
Raitzsch, M., Dueñas-Bohórquez, A., Reichart, G.-J., de Nooijer, L. J., and Bickert, T.: Incorporation of Mg and Sr in calcite of cultured benthic foraminifera: impact of calcium concentration and associated calcite saturation state, Biogeosciences, 7, 869–881, https://doi.org/10.5194/bg-7-869-2010, 2010.
Rao, Z. C., Lueders-Dumont, J. A., Stringer, G. L., Ryu, Y., Zhao, K., Myneni, S. C., Oleynik, S., Haug, G. H., Martinez-Garcia, A., and Sigman, D. M.: A nitrogen isotopic shift in fish otolith–bound organic matter during the Late Cretaceous, P. Natl. Acad. Sci. USA, 121, 32, https://doi.org/10.1073/pnas.2322863121, 2024.
Reis-Santos, P., Gillanders, B. M., Sturrock, A. M., Izzo, C., Oxman, D. S., Lueders-Dumont, J. A., Hüssy, K., Tanner, S. E., Rogers, T., Doubleday, Z. A., Andrews, A. H., Trueman, C., Brophy, D., Thiem, J. D., Baumgartner, L. J., Willmes, M., Chung, M. T., Charapata, P., Johnson, R. C., and Walther, B. D.: Reading the biomineralized book of life: expanding otolith biogeochemical research and applications for fisheries and ecosystem-based management, Rev. Fish Biol. Fish., 33, 411–449, https://doi.org/10.1007/s11160-022-09720-z, 2023.
Rosales, I., Robles, S., and Quesada, S.: Elemental and oxygen isotope composition of Early Jurassic belemnites: salinity vs. temperature signals, J. Sediment. Res., 74, 342–354, https://doi.org/10.1306/112603740342, 2004.
Sackett, D. K., Chrisp, J. K., and Farmer, T. M.: Isotopes and otolith chemistry provide insight into the biogeochemical history of mercury in southern flounder across a salinity gradient, Environ. Sci.-Proc. Imp., 26, 233–246, https://doi.org/10.1039/D3EM00482A, 2024.
Salisbury, F. B. and Ross, C. W.: Plant Physiology, Wadsworth Publishing Company, ISBN: 978-0534151621, 1992.
Saygın, S., Polat, N., Willmes, M., Lewis, L. S., Hobbs, J. A., Atıcı, A. A., and Elp, M.: Strožntium isotopes in otoliths reveal a diversity of natal origins for Tarek (Alburnus tarichi) in Lake Van, Turkey, Fish. Res., 255, 106441, https://doi.org/10.1016/j.fishres.2022.106441, 2022.
Schöne, B. R., Zhang, Z., Jacob, D., Gillikin, D. P., Tütken, T., Garbe-Schönberg, D., McConnaughey, T., and Soldati, A.: Effect of organic matrices on the determination of the trace element chemistry (Mg, Sr, , ) of aragonitic bivalve shells (Arctica islandica): comparison of ICP-OES and LA-ICP-MS data, Geochem. J., 44, 23–37, https://doi.org/10.2343/geochemj.1.0045, 2010.
Shiao, J. C., Loys, L., Iizuka, Y., and Tzeng, W. N.: Migratory patterns and contribution of stocking to the population of European eel in Lithuanian waters as indicated by otolith Sr:Ca ratios, J. Fish Biol., 69, 749–769, https://doi.org/10.1111/j.1095-8649.2006.01147.x, 2006.
Shiao, J. C., Shirai, K., Tanaka, K., Takahata, N., Sano, Y., Hsiao, S.-Y., Lee, D.-C., and Tseng, Y.-C.: Assimilation of nitrogen and carbon isotopes from fish diets to otoliths as measured by nanoscale secondary ion mass spectrometry, Rapid Commun. Mass Spectrom., 32, 1250–1256, https://doi.org/10.1002/rcm.8171, 2018.
Sirot, C., Grønkjær, P., Pedersen, J., Panfili, J., Zetina-Rejon, M., Tripp-Valdez, A., Ramos-Miranda, J., Flores-Hernández, D., Sosa-López, A., and Darnaude, A.: Using otolith organic matter to detect diet shifts in Bardiella chrysoura during a period of environmental changes, Mar. Ecol. Prog. Ser., 575, 137–152, https://doi.org/10.3354/meps12166, 2017.
Stanley, J. K. and Byrne, R. H.: Inorganic complexation of zinc (II) in seawater, Geochim. Cosmochim. Ac., 54, 753–760, https://doi.org/10.1016/0016-7037(90)90370-Z, 1990.
Stoll, H., Langer, G., Shimizu, N., and Kanamaru, K.: B/Ca in coccoliths and relationship to calcification vesicle pH and dissolved inorganic carbon concentrations, Geochim. Cosmochim. Ac., 80, 143–157, https://doi.org/10.1016/j.gca.2011.12.003, 2012.
Sturrock, A., Trueman, C., Milton, J., Waring, C., Cooper, M., and Hunter, E.: Physiological influences can outweigh environmental signals in otolith microchemistry research, Mar. Ecol. Prog. Ser., 500, 245–264, https://doi.org/10.3354/meps10699, 2014.
Sturrock, A. M., Trueman, C. N., Darnaude, A. M., and Hunter, E.: Can otolith elemental chemistry retrospectively track migrations in fully marine fishes?, J. Fish Biol., 81, 766–795, https://doi.org/10.1111/j.1095-8649.2012.03372.x, 2012.
Sturrock, A. M., Hunter, E., Milton, J. A., Johnson, R. C., Waring, C. P., and Trueman, C. N.: Quantifying physiological influences on otolith microchemistry, Methods Ecol. Evol., 6, 806–816, https://doi.org/10.1111/2041-210X.12381, 2015.
Takeuchi, T., Sarashina, I., Iijima, M., and Endo, K.: In vitro regulation of CaCO3 crystal polymorphism by the highly acidic molluscan shell protein Aspein, FEBS Lett., 582, 591–596, https://doi.org/10.1016/j.febslet.2008.01.026, 2008.
Tanner, S. E., Reis-Santos, P., Vasconcelos, R. P., Fonseca, V. F., França, S., Cabral, H. N., and Thorrold, S. R.: Does otolith geochemistry record ambient environmental conditions in a temperate tidal estuary?, J. Exp. Mar. Biol. Ecol., 441, 7–15, https://doi.org/10.1016/j.jembe.2013.01.009, 2013.
Thomas, O. R. B. and Swearer, S. E.: Otolith biochemistry – a review, in: Rev. Fish. Sci. Aquacult., 27, Taylor and Francis Inc., 458–489, https://doi.org/10.1080/23308249.2019.1627285, 2019.
Thomas, O. R. B., Ganio, K., Roberts, B. R., and Swearer, S. E.: Trace element–protein interactions in endolymph from the inner ear of fish: implications for environmental reconstructions using fish otolith chemistry, Metallomics, 9, 239–249, https://doi.org/10.1039/c6mt00189k, 2017.
Thomas, O. R. B., Swearer, S. E., Kapp, E. A., Peng, P., Tonkin-Hill, G. Q., Papenfuss, A., Roberts, A., Bernard, P., and Roberts, B. R.: The inner ear proteome of fish, FEBS J., 286, 66–81, https://doi.org/10.1111/febs.14715, 2019.
Urey, H. C., Lowenstam, H. A., Epstein, S., and McKinney, C. R.: Measurement of paleotemperatures and temperatures of the Upper Cretaceous of England, Denmark, and the southeastern United States, Geol. Soc. Am. Bull., 62, 399–416, https://doi.org/10.1130/0016-7606(1951)62[399:MOPATO]2.0.CO;2, 1951.
Vaisvil, A., Willmes, M., Enriquez, E. J., Klein, Z. B., and Caldwell, C. A.: A needle in a haystack: strontium isotopes () in otoliths identify origin of largemouth bass from a large Southwest reservoir, Can. J. Fish. Aquat. Sci., 80, 1857–1868, https://doi.org/10.1139/cjfas-2023-0148, 2023.
Walker, J. M. and Langer, G.: Coccolith crystals: pure calcite or organic–mineral composite structures?, Acta Biomater., 125, 83–89, https://doi.org/10.1016/j.actbio.2021.02.025, 2021.
Walther, B. D.: The art of otolith chemistry: interpreting patterns by integrating perspectives, Mar. Freshw. Res., 70, 1643, https://doi.org/10.1071/MF18270, 2019.
Walther, B. D. and Limburg, K. E.: The use of otolith chemistry to characterize diadromous migrations, J. Fish Biol., 81, 796–825, https://doi.org/10.1111/j.1095-8649.2012.03371.x, 2012.
Elsdon, T. S., Wells, B. K., Campana, S. E., Gillanders, B. M., Jones, C. M., Limburg, K. E., Secor, D. H., Thorrold, S. R., and Walther, B. D.: Otolith chemistry to describe movements and life-history parameters of fishes: hypotheses, assumptions, limitations and inferences, in Oceanography and Marine Biology: An Annual Review, Vol. 46, CRC Press, ISBN: 978-1-4200-6574-9, 2008.
Wilbur, K. M. and Watabe, N.: Experimental studies on calcification in molluscs and the alga Coccolithus huxleyi, Ann. N. Y. Acad. Sci., 109, 82–112, https://doi.org/10.1111/j.1749-6632.1963.tb13463.x, 1963.
Willmes, M., Lewis, L. S., Davis, B. E., Loiselle, L., James, H. F., Denny, C., Baxter, R., Conrad, J. L., Fangue, N. A., Hung, T. C., Armstrong, R. A., Williams, I. S., Holden, P., and Hobbs, J. A.: Calibrating temperature reconstructions from fish otolith oxygen isotope analysis for California's critically endangered Delta Smelt, Rapid Commun. Mass Spectrom., 33, 1207–1220, https://doi.org/10.1002/rcm.8464, 2019.
Wolthers, M., Di Tommaso, D., Du, Z., and de Leeuw, N. H.: Variations in calcite growth kinetics with surface topography: molecular dynamics simulations and process-based growth kinetics modelling, CrystEngComm, 15, 5506, https://doi.org/10.1039/c3ce40249e, 2013.
Yuryev, V. P., Grinberg, N. V., Braudo, E. E., and Tolstoguzov, V. B.: A study of the boundary conditions for the gel formation of alginates of polyvalent metals, Starch/Stärke, 31, 121–124, https://doi.org/10.1002/star.19790310406, 1979.
Zazzo, A., Smith, G. R., Patterson, W. P., and Dufour, E.: Life history reconstruction of modern and fossil sockeye salmon (Oncorhynchus nerka) by oxygen isotopic analysis of otoliths, vertebrae, and teeth: implication for paleoenvironmental reconstructions, Earth Planet. Sc. Lett., 249, 200–215, https://doi.org/10.1016/j.epsl.2006.07.003, 2006.
Zhong, S. and Mucci, A.: Calcite and aragonite precipitation from seawater solutions of various salinities: precipitation rates and overgrowth compositions, Chem. Geol., 78, 283–299, https://doi.org/10.1016/0009-2541(89)90064-8, 1989.
Ziveri, P., Stoll, H., Probert, I., Klaas, C., Geisen, M., Ganssen, G., and Young, J.: Stable isotope `vital effects' in coccolith calcite, Earth Planet. Sc. Lett., 210, 137–149, https://doi.org/10.1016/S0012-821X(03)00101-8, 2003.
Ziveri, P., Thoms, S., Probert, I., Geisen, M., and Langer, G.: A universal carbonate ion effect on stable oxygen isotope ratios in unicellular planktonic calcifying organisms, Biogeosciences, 9, 1025–1032, https://doi.org/10.5194/bg-9-1025-2012, 2012.