the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Environmental and habitat controls on non-marine ostracod distribution in Greenlandic Arctic lakes
Lucy R. Roberts
Suzanne McGowan
Amanda Burson
Jonathan A. Holmes
David J. Horne
The Arctic is warming almost four times faster than the global average. Lakes in the Arctic are a prominent feature of the landscape and are consequently undergoing limnological and ecological change such as shifts in algal productivity, water column mixing depths, and ice persistence. Most recently, the nutrient-colour paradigm has been associated with extensive loss of benthic habitat. Ostracods (small aquatic crustaceans) are a significant contributor to the benthic biomass of shallow to mid-depth lakes (< 20 m) and there is great potential to use fossil ostracods to reconstruct past environmental change and predict future ecosystem states in these lake-rich regions. However, relative to mid-latitude regions, little is known of the ecological traits of ostracods in the Arctic. Here we present the first systematic survey of ostracod species and ecological preferences for the Kangerlussuaq region of southwest Greenland, the largest ice-free margin of Greenland. Twenty-four lakes (< 16 m deep) were surveyed in July 2021 in a SW-NE gradient from the Greenland Ice Sheet. Electrical conductivity in the lakes ranged from 0.01 to 4.1 mS cm−1. All lakes were ultra-oligotrophic to mesotrophic; soluble reactive phosphorus ranged from 1.9 to 49.7 µg L−1 and nitrate concentrations from below detection limit to 12.3 µg L−1. In total, thirteen species of ostracods were recorded across the study lakes. Candona candida is a generalist species in the Kangerlussuaq region, being present in deeper lakes and at the higher end of the bioavailable phosphorus and nitrate gradients. These traits suggest that C. candida will become abundant in the Greenlandic ostracod fauna, and potentially across the Arctic. For some species, particularly Cypris pubera, bioavailable nutrient concentrations are a dominant control on distribution. Nutrient status of water appears to be a significant control on ostracod presence and abundance and should be included in future ecological studies globally.
- Article
(4240 KB) - Full-text XML
-
Supplement
(441 KB) - BibTeX
- EndNote
Since 1979 CE, the Arctic has warmed almost four times faster than the global average (Rantanen et al., 2022). Changes to the physical environment are marked and ongoing, including permafrost thaw, retreating glaciers, reduced seasonal snow cover, increased river run off, longer ice-free periods both at sea and on lakes, and altered nutrient availability (Post et al., 2009; Box et al., 2019). Globally, in areas where temperature and solar radiation are increasing, and cloud cover is decreasing, seasonally ice-covered lakes are warming at an average summer surface temperature rate of 0.72 °C per decade (compared to an average lake warming of 0.34 °C per decade; O'Reilly et al., 2015).
Lakes in the Arctic are, therefore, particularly at risk of ecological and limnological transformations (Woolway et al., 2022). With >3.5 million lakes in the Arctic, they are prominent features in the landscape (Paltan et al., 2015). There are still significant knowledge gaps for many aspects of Arctic freshwater biodiversity (Saros et al., 2022) with much previous work focused on shifts in algal productivity related to increased and altered growing seasons owing to longer ice-free periods (e.g. Smol et al., 2005; Burpee and Saros, 2020). Longer periods of light penetration rapidly alter habitat structure, increasing primary productivity and lake mixing depths as heat is transferred deeper into the water column (Olsen et al., 2012). With greater mixing depths, there is a consequent expansion of the lake littoral zone to deeper areas of the lake and “benthification”, i.e the increase of benthic productivity (Saros et al., 2019). Recent studies, however, have demonstrated that lakes have shifted from blue to brown (under the nutrient-colour paradigm) with higher nutrient concentrations (nitrogen (N) and phosphorus (P)) and a consequent extensive loss of benthic habitat due to light reduction (Saros et al., 2025).
Viable benthic habitat in lakes has important implications for whole lake productivity and functioning. Benthic primary productivity is often equal to or greater than phytoplankton productivity in the pelagic zone, particularly in low productivity Arctic lakes, and zoobenthos productivity accounts for 42 % of whole-lake secondary productivity (Vadeboncoeur et al., 2002). In addition, the sublittoral zone, where there is high benthic biodiversity, is particularly vulnerable to changes in temperature, oxygen availability and light penetration (McGoff et al., 2013). Consequently, this might ultimately favour organisms with wide ecological and environmental tolerances. More pertinent, however, is that shifts in diversity of benthic organisms are most likely to record and characterise environmental changes.
In shallow and mid-depth lakes (<20 m), ostracods (small benthic or nektobenthic aquatic crustaceans) can be a significant contributor to the benthic biomass (e.g. Geiger, 1998; Rodríguez-Pérez and Baltanás, 2008). As both consumers and secondary producers, ostracods are an important component of the aquatic food web (Mesquita-Joanes et al., 2012). In addition to playing a vital role in the lake food web, non-marine ostracods are sensitive to a range of climatic and environmental conditions, including temperature (e.g. Horne, 2007), salinity (e.g. McCormack et al., 2019), pH (e.g. Wang et al., 2025), substrate (e.g. Higuti et al., 2010) and aquatic plant diversity and abundance (e.g. Frenzel et al., 2005). Consequently, fossil ostracod shells in lacustrine sediments are a commonly used indicator of climatic and environmental change in the Quaternary. However, insufficient knowledge of ecological preferences of species can lead to speculative or poorly constrained paleolimnological reconstructions (Greenway et al., 2024). This is of particular concern for studies in the Arctic since much of the ecological information known about ostracod species is based on present and past occurrence in mid-latitude regions. The difference in seasonality, ice persistence and occurrence, and daylight hours between the regions raises questions about the viability of transferring ecological traits. In Arctic environments the ecological niches and diversity of ostracods remain largely unknown (Schneider et al., 2016), particularly in Greenland (Smith and Horne, 2016). However, the Arctic, and particularly the benthic environment (e.g. Saros et al., 2025), is entering a new ecosystem state, which will not return to the previous state(s) within the next 100 years (AMAP, 2017). There is great potential to use palaeolimnology, and particularly ostracods, to reconstruct past environmental change and predict future ecosystem states in these lake-rich regions. There is, therefore, a pressing need to understand and characterise current environmental and habitat preferences of Arctic ostracod species to understand and interpret current, future and past change.
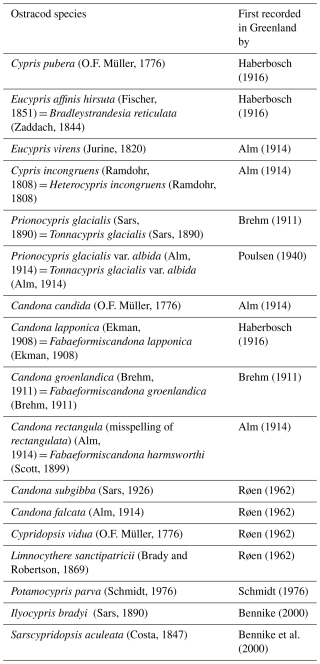
Previous surveys of freshwater cladoceran, copepod and ostracod crustaceans in Greenland have sampled around 300 waterbodies (Poulsen, 1940; Røen, 1962, 1968, 1970, 1981) recording 13 ostracod species and one variety (Table 1). Palaeolimnological studies (Bennike, 2000) and Bennike et al. (2000, 2010) have recorded a further three species (Table 1), giving 16 species recorded in total. Here, we provide the first systematic survey of ostracod species and ecological preferences for the Kangerlussuaq region of southwest Greenland, the largest ice-free margin of Greenland with a landscape comprising ∼20 000 glacially-derived lakes, accounting for ∼15 % of the land area (Anderson et al., 2009). This density of lakes extending a range of environmental conditions allows a space-for-time approach in determining the ecological preferences of ostracod species.
2.1 Study area
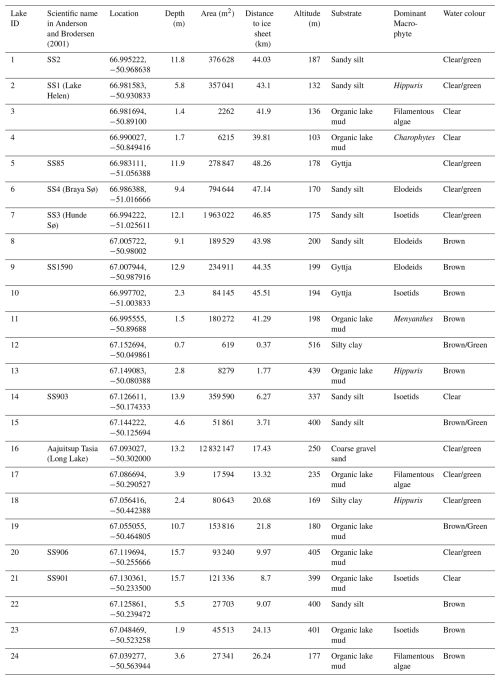
Twenty-four lakes (Table 2) <16 m deep in the Kangerlussuaq (67° 00′ N, 50° 43′20′′ W) region located 0.4 to 48.3 km from the Greenland ice sheet (GrIS; Fig. 1) were sampled in July 2021. The transect characterised expected spatial trends in environmental and limnological conditions (e.g. glacial inflow versus aridity, depth, size) in the region. The bedrock of the area is Precambrian gneiss. The study lakes are predominantly hydrologically isolated with no inflows and negligible input from groundwater or snowmelt (Anderson and Brodersen, 2001; Johansson et al., 2015), except those close to the GrIS, which are, in some circumstances, glacially fed (Burpee et al., 2018). The lakes furthest from the GrIS in Kellyville (Fig. 1) are oligohaline due to low precipitation, high rates of evaporation and locally derived salts (primarily an aeolian input). Annual precipitation is low (<250 mm y−1; Mernild et al., 2018; Box et al., 2023) and the mean annual temperature between 1961 to 1990 was −5.7 °C (DMI, 2026). For the period July 2020 to July 2021, mean annual temperature was −2.2 °C with a maximum of 21 °C and a minimum of −34.6 °C. Mean January air temperatures in 2021 were −16.5 °C and mean July temperatures were 11.1 °C (DMI, 2026). Conditions close to the ice sheet, at higher elevations, are on average 2–3 °C cooler and with at least 10 mm more precipitation per month (Fowler et al., 2020).
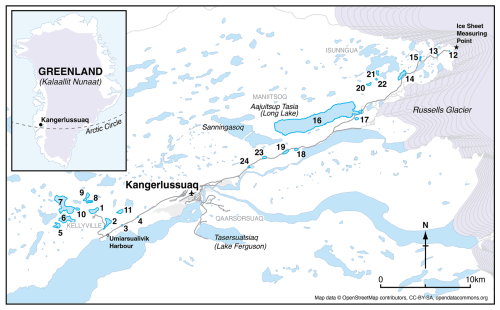
Figure 1Location of the study lakes in Kangerlussuaq, Greenland, showing the 24 study lakes in a SW-NE gradient. Coordinates of each lake are given in Table 2. The numbers indicate lake ID and distance from the ice sheet has been calculated from the location denoted with a star (OpenStreetMap).
2.2 Field methods
Water column profiles of chlorophyll-a, dissolved oxygen (DO), pH, temperature and electrical conductivity (EC) at the deepest point of the lakes were measured using a YSI EXO2 multiparameter probe, which was set to log at 2 s intervals. The presence of an oxycline was assessed by subtracting the DO of bottom waters from the DO of surface waters. For pH, temperature, EC and chlorophyll-a, an average value for the top 2 m was calculated to represent values for the surface mixed layer and where ostracods were sampled. Secchi depth to determine water clarity was measured using a Secchi disk. Water samples for the determination of nutrient concentrations were collected from the same location using HCl-washed bottles. Samples were filtered with Whatman GF/F 0.7 µm filters, or with 0.4 µm polypropylene filters for silicate. Ostracods were collected in a 250 µm mesh zooplankton net from the littoral zone by sampling the top ∼1 cm of sediment. Where submerged macrophytes were present, samples were collected with the net from amongst the vegetation and included sampling the top ∼1 cm of sediment. Water colour, substrate and the dominant vegetation were noted from the littoral zone. Benthic images and videos were taken using an underwater camera and used to identify if submerged macrophyte cover was evident across the littoral zone. These videos were also used to visually determine water colour as clear, clear/green, green/brown and brown. According to the Nutrient Colour Paradigm, nutrient concentrations in the lakes are too low for colour to be classified as green. However, to visually distinguish between low nutrient clear water and more turbid water of “green” colour, these lakes have been visually classified as “clear/green”. Lake area was calculated at a later date using the polygon function in Google Earth.
2.3 Laboratory methods
2.3.1 Water chemistry
Samples for dissolved nutrients were passed through 0.7 µm GF/F filters and analysed colorimetrically by the molybdate blue method for soluble reactive phosphorus (SRP), azo dye method for nitrite (NO2-N), which included a cadmium reduction step for nitrate (NO3-N), the indophenol blue method for ammonium (NH4-N), and the molybdenum yellow method for silicate (Mackereth et al., 1989). The dissolved inorganic nitrogen forms were summed to give total dissolved inorganic nitrogen (TDIN). Bicarbonate and carbonate alkalinity (summed to total alkalinity) were determined through titrations with 0.1N hydrochloric acid (Mackereth et al., 1989). Chlorophyll a (Chl-a) was measured by filtering a known volume of water through a GF/F filter, extraction of the filtered residue and trichromatic analysis on a spectrophotometer (Jeffrey and Humphrey, 1975).
2.3.2 Ostracod identification
Sediment samples were wet-sieved at 250 µm, to remove any remaining fine sediment. Sample residue was dried in an oven at 40 °C and weighed to calculate dry weight. Ostracod shells (i.e. carapaces and valves) were picked under low-power stereo microscope using a 0000-paintbrush. A total of 100 ostracods were picked or the whole sample, depending on which limit was reached first. Using the dry weight, or, where 100 ostracods were picked, the picked fraction, the number of individuals per gram dry weight was calculated.
Although ostracods comprise two valves, it is not possible to determine paired valves once they become disarticulated, which can occur naturally or during sample processing. Therefore carapaces, single valves and fragments > half a valve (allowing identification but also ensuring fragments of the same valve were not duplicated in the count) were treated as one individual. The number of carapaces with soft parts was noted and used as an indicator of individuals that were living at the time of collection.
Ostracod specimens were sorted and mounted on standard micropalaeontological slides, and identified using Meisch (2000) and Fuhrmann (2012) together with other literature as appropriate. Selected specimens were imaged using a Jeol JSM-6480LV Scanning Electron Microscope at University College London. Ostracods were mounted on double-sided carbon tape on aluminium stubs and coated with gold palladium before examination. Figured specimens are to be deposited in the Natural History Museum (catalogue numbers will be allocated once the paper has been accepted for publication).
2.4 Statistical methods
Spatial patterns in species composition were determined by grouping the 24 lakes into clusters according to their similarity in ostracod assemblages based on Ward's method hierarchical clustering using the Pheatmap package in R version 4.4.1 (Kolde, 2025). The Corrplot package in R version 4.4.1 (Wei and Simko, 2024) was used to test for any statistically significant correlations between environmental variables. Any variables that were significantly correlated were excluded from further analysis. Multivariate correlations between ostracod species and environmental variables were examined using Redundancy Analysis (RDA). Variance partitioning analysis (VPA) was undertaken using the Vegan package in R version 4.4.1 (Oksanen et al., 2025). For inclusion in statistical tests, the dominant submerged macrophytes, presence of an oxycline, water colour and substrate were converted from categorical data to numeric.
3.1 Limnology
For the 24 lakes, the dominant substrate was organic lake mud with low growing isoetid form macrophytes being the most common macrophytes. In seven lakes, no aquatic plants were observed. Water colour was categorised through observations as clear in four lakes, clear/green in nine lakes, brown in seven lakes and brown/green in three lakes (Table 2).
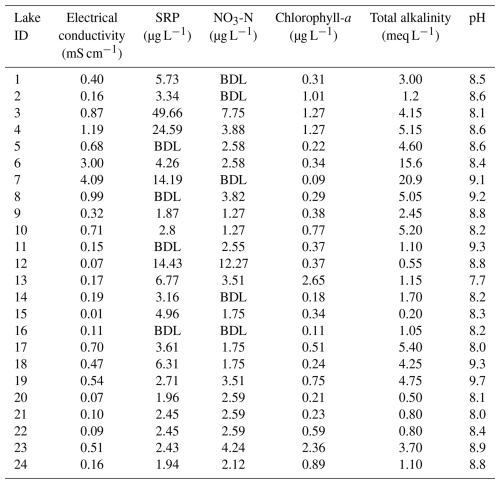
The electrical conductivity (EC) of the lakes ranged from 0.01 to 4.1 mS cm−1, with the highest EC in lake 7 and lowest in lake 15 (Table 3). Water temperature ranged from 9.5 to 10.5 °C at lake 1 and lake 24. Total Alkalinity ranged from 0.2 meq L−1 in lake 15 to 20.9 meq L−1 in lake 7. pH ranged from 7.7 in lake 13 to 9.7 in lake 19. All lakes were ultra-oligotrophic to mesotrophic; soluble reactive phosphorus (SRP) ranged 1.9 to 49.7 µg L−1 from lake 9 to lake 3 and nitrate (NO3-N) concentrations were highest in lake 12 at 12.3 µg L−1. The concentrations were below detection limit in lakes 1, 2, 7, 14 and 16. Chlorophyll-a ranged from 0.06 µg L−1 in lake 22 to 2.65 µg L−1 in lake 13.
Secchi depth, altitude, silicate, total dissolved inorganic nitrogen, nitrite, ammonium, water temperature and the macrophyte cover were all significantly correlated (at p< 0.001 or p< 0.01 and an r> 0.42) with at least two other variables (Table S1 and Fig. S1 in the Supplement).
3.2 Ostracod fauna
Thirteen species of ostracods were recorded across the study lakes (Figs. 2, 3). Other than Limnocythere friabilis (Benson and MacDonald, 1963) and species of Ilyocypris, some individuals with soft parts were collected (Table S2) and so were assumed to be living at the time of collection. The most abundant species was Cypris pubera (O.F. Müller, 1776), with a maximum abundance of 87 valves per gram in lake 17. Candona candida (O.F. Müller, 1776) was present in the most sites (n=20) with Ilyocypris gibba (Ramdohr, 1808) present in the fewest (n=1). Shells (carapaces and valves) of Candona juveniles were present in ten sites, all of which also had adult C. candida individuals present. Cypridopsis vidua (O.F. Müller, 1776), Ilyocypris bradyi (Sars, 1890), Ilyocypris gibba, Sarscypridopsis aculeata (Costa, 1847) and Potamocypris parva (Schmidt, 1976) were rarely recorded, occurring in one to six sites at an abundance of no more than 3, 2, 2 and 2 valves per gram, respectively. Arctocypris sp., Heterocypris incongruens (Ramdohr, 1808), L. sanctipatricii (Brady and Robertson, 1869) and L. inopinata (Baird, 1843) were somewhat abundant with maximum abundances of 7, 15, 15 and 11 valves per gram, and occurring in four to seven sites. Ostracods were not recorded in three sites (lake 1, 4 and 16). The highest diversity of eight species was recorded in lake 6. Lowest diversity of one species was recorded in lakes 13, 14, 20 and 24. At each of these lakes, the only species recorded was C. candida.
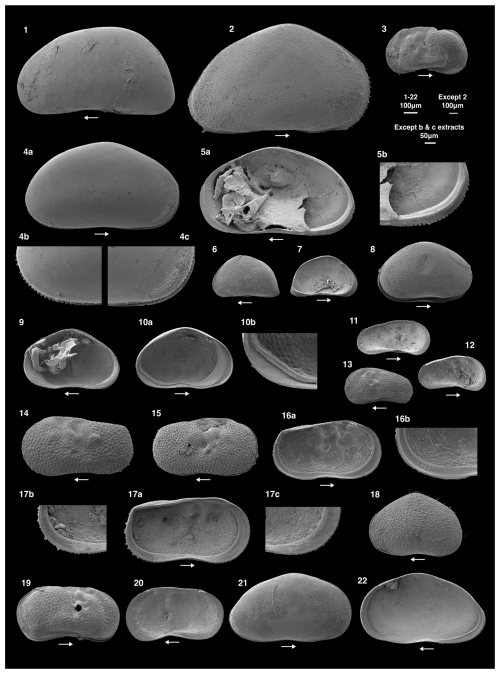
Figure 2Scanning electron microscope images of ostracod species collected from the 24 study lakes. The 100 µm scale bar applies to images 1–20, the 200 µm scale bar to image 2 and the 50 µm scale bar to the b and c extracts. The lake in brackets represents where the specimen was collected. LV refers to left valve and RV to right valve. 1. Candona candida, female, LV (Lake 7); 2. Cypris pubera, female, RV (Lake 17); 3. Limnocythere inopinata, female, RV (Lake 7); 4a. Heterocypris incongruens, female, RV external; 4b. extract of the posterior margin of 4a; 4c. extract of the anterior margin of 4a (Lake 17); 5a. Heterocypris incongruens with evidence of soft parts, female, RV internal; 5b. extract of the posterior margin of 5a. The flange on the anterior margin is visible in 5a (Lake 17). Images 4a, b, c, and 5a, b are of the same individual; 6. Potamocypris parva, assumed female since males are not known, LV (Lake 7); 7. Potamocypris parva, assumed female since males are not known, LV internal (Lake 7). Images 6 and 7 are of the same individual; 8. Cypridopsis vidua, female, carapace (Lake 18); 9. Cypridopsis vidua, female, RV (Lake 18); 10a. Cypridopsis vidua, female, LV (Lake 18); 10b. extract of the posterior margin of 10a. Images 8, 9, 10a and 10b are of the same individual; 11. Limnocythere friabilis, female, LV internal (Lake 7); 12. Limnocythere friabilis, female, juvenile, LV internal (Lake 7); 13. Limnocythere friabilis, female, juvenile, LV (Lake 7). Images 12 and 13 are of the same individual; 14. Ilyocypris bradyi, female, LV (Lake 6); 15. Ilyocypris gibba, female, LV (Lake 6); 16a. Ilyocypris bradyi, female, LV internal (Lake 7); 16b. extract of the posterior margin of 16a with no ripplets on the inner lamella; 16c. extract of the anterior margin of 16a. Images 14 and 16a, b, c are of the same individual; 17a. Ilyocypris gibba, female, LV internal (Lake 7); 17b. extract of the posterior margin of 16a with six visible ripplets on the inner lamella. Images 15 and 17a, b are of the same individual; 18. Sarcypridopsis aculeata, female, adult, LV (Lake 6; N.B this individual was not collected during the July 2023 survey). 19. Limnocythere sanctipatricii, female, carapace (Lake 9); 20. Limnocythere sanctipatricii, female, RV (Lake 9); 21. Arctocypris sp., A-1, RV (Lake 7); 22. Arctocypris sp., A-1, RV internal (Lake 7). Images 21 and 22 are of the same individual.
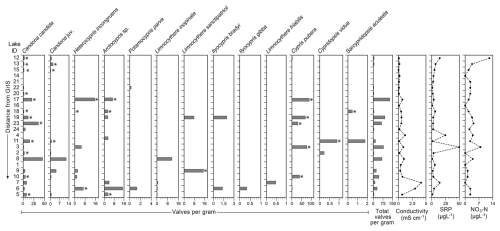
Figure 3Ostracod species abundance as valves per gram in each of the 24 study lakes alongside selected recorded environmental variables (electrical conductivity, soluble reactive phosphorus (SRP) and nitrate (NO3-N)). The stars indicate the occurrence of individuals with soft parts, suggesting that they were collected alive. Table S2 provides a detailed breakdown of valves, carapaces and individuals with soft parts. Lakes are ordered in the distance from the Greenland Ice Sheet.
3.3 Abundance of ostracod species along environmental gradients
All ostracod species except Arctocypris sp. were most abundant at low total alkalinity (≤5.4 meq L−1; Fig. 4a). Cypridopsis sp. and S. aculeata were not present at total alkalinity values above this. Cypris pubera and C. candida were the only ostracod species abundant above chlorophyll-a concentrations of 2 µg L−1, with only Candona juveniles also present (Fig. 4b). Heterocypris incongruens and Candona juveniles were abundant between 0.2 and 0.8 µg Chl-a L−1. Heterocypris incongruens, Cypridopsis vidua, Cypris pubera, S. aculeata, I. bradyi, I. gibba, P. parva and Candona juveniles were not present below Chlorophyll-a concentrations of 0.2 µg L−1. Candona candida and C. pubera were most abundant at NO3-N concentration between 1.3 and 4.2 µg L−1 (Fig. 4c). Only C. pubera, H. incongruens, C. candida and Candona juveniles were present at concentrations above this. Other than L. friabilis, which was most abundant at concentrations of 14.2 µg L−1, all species were most abundant at SRP concentrations of <6.8 µg L−1 (Fig. 4d). Only three species were present at the highest SRP concentration of 49.7 µg L−1 namely C. pubera, which was very abundant (at 75 valves per gram), C. candida and H. incongruens. Potamocypris parva, L. friabilis, L. inopinata L. sanctipatricii, I. bradyi, I. gibba., C. candida and Candona juveniles are most abundant at depths >9 m (Fig. 4e). Cypridopsis vidua was only present at depths <6 m and C. pubera was most abundant at depths <4 m. All ostracod species were most abundant at EC <1 mS cm−1, other than Arctocypris sp. (Fig. 4f). Cypris pubera, Cypridopsis vidua and S. aculeata were not present at ECs above this. Cypridopsis vidua was only present at ECs between 0.15 and 0.16 mS cm−1. Lakes 5, 6, 7, 8, 9 and 10 are oligohaline and nine species were present at EC >3 mS cm−1; of these L. friabilis L. sanctipatricii, L. inopinata, I. bradyi, I. gibba, C. candida and Arctocypris sp. were present at the EC of 4.09 mS cm−1. At the lowest EC of 0.01 mS cm−1, only C. candida, Candona juveniles and Arctocypris sp. were present. Both species of Ilyocypris were only present at ECs above 0.54 mS cm−1 and L. inopinata and L. sanctipatricii at concentrations >0.32 mS cm−1 (Fig. 4f).
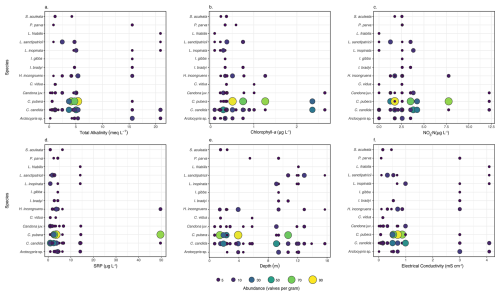
Figure 4Abundance of ostracod species along gradients of (a) total alkalinity, (b) chlorophyll-a concentration, (c) nitrate (NO3-N) concentration, (d) soluble reactive phosphorus (SRP) concentration, (e) water depth and (f) electrical conductivity. Ostracod abundance is given in valves per gram and includes all specimens collected. Table S2 provides a detailed breakdown of valves, carapaces and individuals with soft parts.
3.4 Lake clusters
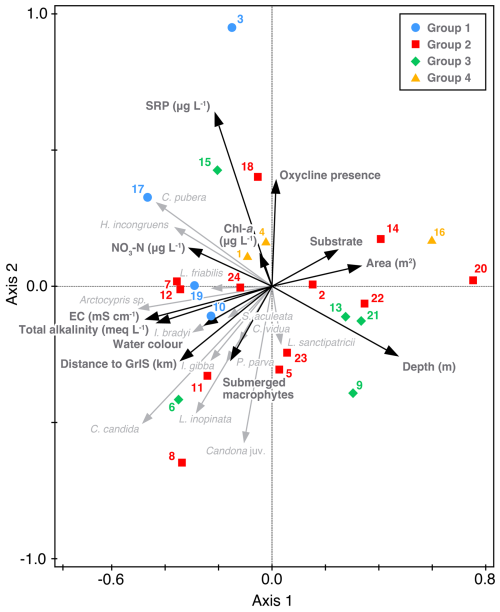
Based on ostracod assemblages, three clusters of lakes were identified (Fig. 5). Cluster 1 (4 lakes – 17, 19, 3, 10) is characterised by high abundance of C. pubera. Cluster 2 (twelve lakes) is characterised by high abundance of C. candida. Cluster 3 (five lakes – 13, 15, 21, 6, 9) is characterised by a more diverse ostracod fauna with high abundances of Candona juv, H. incongruens, Arctocypris sp., L. inopinata, L. sanctipatricii and P. parva with intermediate abundance of C. candida. Cluster 3 lakes also have the lowest abundances of C. pubera and Cypridopsis vidua. A fourth cluster (three lakes – 1, 4 and 16), not depicted on Fig. 5, is characterised by the absence of ostracods.
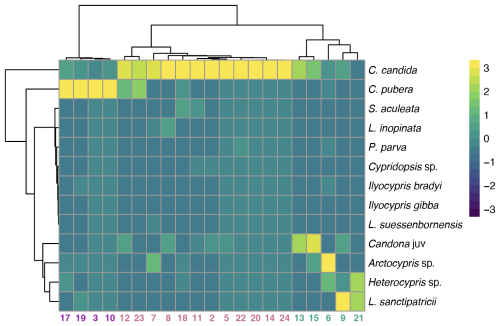
Figure 5Identification of three clusters (cluster 1 N=4, cluster 2 N=12, cluster 3 N=5) based on ostracod assemblages Ward's method hierarchal clustering. A fourth cluster (N=3) is not depicted as no ostracod species were recorded in these lakes. Clusters are colour coded on the x-axis (Group 1 in blue, Group 2 in red, Group 3 in green and Group 4 in yellow). These colours correspond to grouping in Fig. 6. The analysis includes all specimens collected. Table S2 provides a detailed breakdown of valves, carapaces and individuals with soft parts.
RDA axis 1 is negatively correlated with EC and positively correlated with lake area and substrate and explains 16.23 % of variation (Fig. 6). Axis 2 is positively correlated with presence of an oxycline and nutrients and is negatively correlated with the presence of submerged macrophytes and depth. Axes 1 and 2 together explain 28.94 % of variation. Cluster 1 lakes are characterised by the lowest depths (mean 4.6 m, ranging from 1.4 to 10.7 m), higher SRP (mean 14.7 µg L−1, ranging from 2.7 to 49.7 µg L−1), higher NO3-N (mean 3.6 µg L−1, ranging from 1.3 to 7.8 µg L−1) and higher chlorophyll-a (mean 0.82 µg L−1, ranging from 0.51 to 1.27 µg L−1). Cluster 2 lakes, on the other hand, are characterised by lowest mean chlorophyll-a (0.50 µg L−1, ranging from 0.09 to 2.36 µg L−1) and, although still high for lakes, the lowest pH (8.7, ranging from 8.0 to 9.3). Cluster 3 lakes are the deepest (mean 9.1 m, ranging from 2.8 to 15.7 m), encompass the oligohaline lakes 6 and 9 so have the highest EC (0.72 mS cm−1, ranging from 0.01 to 3.00 mS cm−1), lowest SRP (4.1 µg L−1, ranging from 1.9 to 5.0 µg L−1) and the majority of lakes (3 of 5 lakes) are within 10 km of the GrIS. In contrast, the majority of lakes in cluster 4 (2 of 3) are ≤40 km from the GrIS and have the largest average area (4.4 km2, ranging from 0.01 to 12.8 km2). Lakes in cluster 4 also have the lowest average NO3-N (1.29 µg L−1, ranging from BDL to 3.88 µg L−1) and lowest total alkalinity (3.1 meq L−1, ranging from 1.1 to 3.0 meq L−1). There were also no macrophytes present in cluster 4 lakes other than the presence of filamentous algae in lake 3.
Over recent decades, mean June air temperatures have increased by 2.2 °C and mean winter precipitation has doubled with continued predicted increased precipitation in SW Greenland (Saros et al., 2019; Huai et al., 2025). Reponses to recent warming have been non-linear but include increasing ice sheet discharge (Van As et al., 2018), increasing dust deposition (Bullard and Mockford, 2018), and earlier ice out (Hazuková et al., 2024). It is, therefore, expected for lakes to become more nutrient rich due to wind-driven P in dust (Prater et al., 2022) and snowmelt-derived N (Whiteford et al., 2016), water colour to be more brown from increased dissolved organic material, mixing regimes and growing seasons to be altered from longer ice-out periods, and benthic productivity to decline (Saros et al., 2025). Spatial controls on water chemistry, particularly distance from the GrIS, have previously been documented and include increases in EC with distance from the GrIS, due to increases in aridity (Aebly and Fritz, 2009; Fig. 3), and higher nutrient concentrations in lakes close to the GrIS (Prater et al., 2022), particularly in those lakes that are glacially fed (Grider et al., 2025).
4.1 Controls on ostracod species distribution in Kangerlussuaq
Our results indicate a more complex pattern of nutrient distribution with lakes close to the GrIS and a spatial cluster of lakes (3 and 4) having higher concentrations of SRP and NO3-N. Total phosphorus (TP) and NO3-N concentrations in glacially fed lakes have been shown to be three times higher than in snowmelt fed lakes (Grider et al., 2025). Lower bioavailable nutrient concentrations in Kellyville lakes (those located furthest from the GrIS; Fig. 1) could be related to being further from the source of dust and to the reduced wind speed at >10 km from the GrIS (Heinemann, 1999) therefore decreasing dust derived P (Burpee et al., 2016; Prater et al., 2022). As expected, therefore, highest NO3-N concentrations (12.27 µg L−1) were in lake 12, which is glacially fed and located 0.37 km from the GrIS. After lakes 3 and 4, lake 12 had the third highest SRP concentrations of 14.43 µg L−1. Lake 15 (GL6 in Grider et al., 2025) is located 3.71 km from the GrIS but has relatively low NO3-N concentrations (1.75 µg L−1) with high SRP concentrations (4.96 µg L−1). Meltwater is therefore likely a dominant source of nutrients with N concentrations derived from atmospheric deposition on the ice sheet and P derived from geological weathering of the glacial bed (Hawkings et al., 2016). However, previous work has suggested that most of this mineralogically-derived P is not biologically available (Burpee et al., 2018).
Consequently, cluster 1 lakes, which have high SRP (mean 14.7 µg L−1, ranging from 2.7 to 49.7 µg L−1), NO3-N (mean 3.57 µg L−1, ranging from 1.3 to 7.8 µg L−1), and chlorophyll-a (mean 0.82 µg L−1, ranging from 0.51 to 1.27 µg L−1) are likely to become more dominant in the Kangerlussuaq landscape in the future. Higher nitrate concentration in lakes is associated with visual water colour (brown and brown/green) and higher chlorophyll-a concentrations (0.51 to 1.27 µg L−1; Fig. 6). Cluster 1 lakes are characterised by a high abundance of C. pubera (Fig. 5), which is most abundant at depths <4 m but present and still relatively abundant between 8 and 12 m. Here the species is only present in lakes with an EC <1 mS cm−1 but reportedly found at salinity up to 4 ‰ (∼7.3 mS cm−1; Stephanides, 1948). In paleolimnological records of higher salinity lakes in the region (SS6, Lille Saltsø and Store Saltsø), C. pubera has been considered rare, being only recorded in two lake basins across Greenland (Bennike, 2000; Bennike et al., 2000, 2010). Whilst C. pubera is considered abundant in our study, C. pubera was not present in the Kellyville “salt” lakes, suggesting high salinity significantly limits distribution in the Kangerlussuaq region.
In general, information on nutrient status of lakes is often not included when documenting ostracod species presence and abundance. In a study of three ponds in Patagonia, however, C. pubera was most abundant in the pond with highest TP of 121.4 µg L−1 (Coviaga et al., 2018), suggesting an ecological preference for higher nutrient availability. A preference for waters with higher nutrient concentrations may be related to food source. Cypris pubera is omnivorous, feeding on algae, bacteria and Daphnia (Meisch, 2000; Coviaga et al., 2018). In lakes 3, 10 and 19, C. pubera was present in very high numbers (75, 38 and 67 valves per gram respectively). These lakes have high coverage of filamentous algae or large Nostoc cyanobacteria balls, colloquially named sea tomatoes. It is likely, therefore, that C. pubera is present in large numbers in these lakes due to a dietary preference.
Cluster 1 lakes are typically shallower (<4.6 m) than those belonging to other clusters. Most ostracod species that are abundant in deeper lakes are not present above NO3-N concentrations of 4.24 µg L−1 (Fig. 4c, e). Cluster 3 lakes are on average the deepest and are characterised by a diverse ostracod fauna including L. inopinata, H. incongruens, Arctocypris sp., P. parva, L. sanctipatricii, I. bradyi and C. candida. Limnocythere inopinata and C. candida are known to inhabit deep lakes, but both are also present in shallow lakes across Europe (Meisch, 2000). Both are also found in lakes across a range of salinities. Depth and EC are therefore likely not the controls on distribution for L. inopinata in this region. Total alkalinity has also been suggested as a control on L. inopinata abundance (Löffler, 1959). Indeed lakes 6 and 7 have the highest alkalinities of 20.9 and 15.6 meq L−1. As suggested by Jungwirth (1979), L. inopinata is also not present in lakes with clay or gravel substrates. In the Canadian Arctic, the abundance of L. inopinata is negatively correlated with chlorophyll-a concentrations (Viehberg and Pienitz, 2017). Our results suggest L. inopinata is not present at concentrations above 0.8 µg L−1 and is most abundant in Cluster 2 lake 8.
Cluster 2 lakes are characterised by the lowest mean chlorophyll-a concentrations (0.50 µg L−1, ranging from 0.09 to 2.36 µg L−1) and an abundance of C. candida. Candona candida is considered to be oligothermophilic (Vesper, 1975), preferring low nutrient concentrations and adults are present throughout the year in waters where the water temperature does not exceed 18 °C in the summer (Hartmann and Hiller, 1977), suggesting an upper temperature limit on adult life stage persistence. The species has a known Holarctic distribution and its life cycle preference for cooler summers would suggest abundance of the species in the Arctic; indeed, it has been shown to be abundant at higher latitudes (Alkalaj et al., 2019). Increasing temperatures are, however, likely to affect the life cycle and abundance, but not presence of this species.
Nutrient concentration has also been suggested as a control on L. sanctipatricii, which shows a preference for oligotrophic habitats (Scharf, 1981). The species is documented to have disappeared from Lake Mondsee, Austria, following anthropogenically-derived eutrophication (Danielopol et al., 1993). It is considered to be a cold-water indicator and has been found previously in Greenland (Table 1) as well as in Arctic Siberia (Wetterich et al., 2008). The presence of L. sanctipatricii is characteristic of Cluster 3 lakes 9 and 21 (Fig. 5), which are relatively deep, large lakes with relatively low nutrient concentrations (Fig. 6). Future increased water temperatures and nutrient concentrations in the region, may therefore limit the abundance and distribution of L. sanctipatricii.
It may be considered surprising that variance partitioning analysis (VPA) of nutrients (SRP, NO3-N, Chl-a), EC, and habitat (the dominant submerged macrophytes and macrophyte cover), explained little of the overall variation in ostracod species composition (∼2.5 %). EC contributed the largest unique contribution (adjusted R2=0.035) but, overall, the variation is not explained by these three categories of variables (residuals =1.01; Fig. S2). However, for some species that are nektobenthic and large (e.g. C. pubera), provision of food and protection from predation offered by macrophyte cover may be a larger contributor to its presence and abundance than can be determined from this dataset. Due to the sampling strategy, it is also likely that variables such as pH, Chl-a, macrophyte cover and bioavailable nutrients vary within and between seasons, particularly in the late summer with longer ice-free periods (McGowan et al., 2018). Our results also provide a time-averaged “present day” living ostracod fauna, which is not an unreasonable approach in these remote environments, but it is likely that different ostracod species will be abundant in different seasons and that the measured parameters do not reflect the full range of habitat and environmental preferences.
Our record of Limnocythere friabilis is the only published occurrence of recent to living individuals outside North America. The North American species Limnocythere friabilis, considered to be a senior synonym of the extinct European species Limnocythere suessenbornensis (Horne et al., 2023), is unique to cluster 3 and only recorded in lake 7. Limnocythere friabilis is common in the Great Lakes region, which shares limnological features, which may favour L. friabilis, with the Kangerlussuaq region such as seasonal ice cover, increasing anthropogenic N and P enrichment since the 1970s CE (Nelligan et al., 2021) and are deep with an average depth of 19 m in Lake Erie. Candona candida, L. inopinata, and L. suessenbornensis occur together in interglacial records in Europe (e.g. Benardout, 2015; Marchegiano et al., 2020; Horne et al., 2023), with L. suessenbornensis regarded as a cold-water species. Limnocythere suessenbornensis is present during warm interglacial periods when, at least, UK summer temperatures are suggested to be similar or slightly warmer than today, but winter temperatures were up to 10 °C cooler (Benardout, 2015; Horne et al., 2023). There is, therefore, likely a significant winter temperature control on its life cycle and hence distribution. Our record would corroborate the requirement for significantly cooler average winter temperatures. Increased temperature and earlier ice out in western Greenland may therefore have an adverse impact on the distribution and abundance of L. friabilis.
Potamocypris parva is, to our knowledge, only recorded in the Kangerlussuaq area. It is considered to be endemic to Greenland, and specifically the oligohaline lakes within the Kangerlussuaq region. The species was first described from lakes close to Kellyville (Schmidt, 1976) and since then, it has been recorded in other saline lakes including Store Saltsø and SS6 (Bennike, 2000; Bennike et al., 2010). Our results suggest that P. parva is also present in lakes with lower EC but is most abundant (40 valves per gram) at EC up to 4.09 mS cm−1. In carapace morphology it is closely similar to an African species, Potamocypris paludum (Gauthier, 1939), which has been found in European Pleistocene ostracod assemblages (Fuhrmann, 2012; Marchegiano et al., 2018). However, while P. parva has long antennal swimming setae, those of P. paludum are relatively short, suggesting that these are two distinct species (Claude Meisch, Musée national d'histoire naturelle, Luxembourg, personal communication, 22 September 2025); we have not found any specimens with preserved antennae in our material.
Sarscypridopsis aculeata is considered an indicator of saline waters. Here, the species is not present in the higher conductivity waters but is found in lakes with an EC of <0.47 mS cm−1. However, it has previously been collected from lake 6 with an EC of 3 mS cm−1 (L. Roberts, unpublished data) and is therefore likely still an indicator of more saline waters in this region. Previously, the species was not thought to be extant in Greenland (Bennike, 2000) with the only previous record in isolation basins formed during the Early Holocene. Ilyocypris bradyi was also not considered to be extant in Greenland with the last recorded occurrence in Store Saltsø in Kangerlussuaq during the Holocene warm period (∼7000 years BP) before going extinct in the region (Bennike, 2000). The recording of these species in the modern fauna suggests that increasing average temperature are now placing the region within the temperature tolerance of these species and they will continue to thrive.
4.2 Implications for future distribution of ostracod species
Predictions for the Arctic are for temperature and precipitation to continue to increase in the 21st century (Hu et al., 2021; McCrystall et al., 2021). Higher temperatures in the Arctic will increase P and N loading into lakes by reactivating hydrological flows transporting soil- and dust-borne nutrients into lakes. For the limited number of glacially-fed lakes (e.g lakes 12 and 15) meltwater discharge into lakes will likely increase P and N loading associated with the release of ice-locked atmospheric deposition of N and P from glacial bed erosion (Hawkings et al., 2016). Predicted increases in precipitation will also increase N in lakes. Previous δ15N of NO3-N in lakes from the Kangerlussuaq region suggest an addition from direct atmospheric N deposition (Anderson et al., 2017). In ice core records this has been shown to have increased over the last 50–100 years (Hastings et al., 2009). Future trends of N deposition, however, are reliant on policies to control emissions. Conversely, increased precipitation and meltwater may reduce P derived from dust by increasing the fluvial area, consequently reducing the effectiveness of aeolian erosion and transportation.
Increased NO3-N concentrations in lakes is likely to favour C. pubera, C. candida, and H. incongruens occurrence, abundance and distribution (Figs. 4c, 6). With warmer temperatures, earlier ice out will alter the timing of, and support longer, growing seasons for phytoplankton and macrophytes, which may reduce bioavailable N due to N-uptake and denitrification (McGowan et al., 2018). During this process, P concentrations may increase due to internal loading. Increases in SRP concentrations would favour the same ostracod species (C. pubera, C. candida, and H. incongruens; Fig. 4d). The results presented here do not suggest a strong association between dominant macrophyte taxa and ostracod species distribution or abundance (Fig. 6) despite previous studies linking macrophyte cover and ostracod species occurrences (e.g. Roca and Danielopol, 1991; Roca et al., 1993; Frenzel et al., 2005). Perhaps the macrophyte species present, including sparse low form isoetids, do not offer the same habitat structure as species with leaves distributed throughout the water column (e.g. Potamogeton). However, a more systematic macrophyte survey would be needed to verify this; notwithstanding a lower diversity and abundance in the oligohaline lakes, a more diverse flora, including Potamogeton has been described in the freshwater lakes in Kangerlussuaq (Reuss et al., 2014). The implications of macrophyte persistence and diversity for future ostracod distribution are, therefore, currently uncertain.
With increased temperatures, precipitation is more likely to occur as rainfall, rather than snow. Coupled Model Intercomparison Project Phase 6 (CMIP6) experiments suggest that, across most of the Arctic, precipitation in winter will continue to have snowfall as the dominant type but with some rainfall and increasing in amount. However, in summer and autumn the dominant precipitation will be rainfall (McCrystall et al., 2021). By 2100, relative to the year 2000, there is a 422 % increase in CMIP6 predicted rainfall in winter, 261 % in spring, 71 % in summer, and 268 % in autumn. Greenland is predicted to have rainfall-dominated precipitation with 1.5 °C warming in CMIP6 (McCrystall et al., 2021). Currently, precipitation-evapotranspiration (P-E) patterns in the region are paced by ice and snowmelt-derived freshwater pulses. Increased rainfall will alter this seasonal pattern and could result in lower evaporation from lakes in Kellyville, with increased seasonal outflow already reported since 2023 from lake 6. Potamocypris parva and L. friabilis are most abundant at high EC (Fig. 4f), and therefore alterations to the P-E balance may affect the future distribution of these species, potentially restricting their distribution to coastal lakes.
Sixteen species had previously been documented from Greenland of which only five have been recorded living or in recent sediments from Kangerlussuaq (Bennike et al., 2000). Eight (C. candida, C. vidua, L. sanctipatricii, I. bradyi, C. pubera, P. parva, H. incongruens, and S. aculeata) of the sixteen species are present in this study, the other species presented here are new records for Greenland. Furthermore, two species (I. bradyi and S. aculeata) were considered to be extinct in Greenland, neither being recorded since the early Holocene. Candona candida has previously been recorded in the Siberian and Canadian Arctic (Wetterich et al., 2008; Viehberg and Pienitz, 2017) with C. pubera, and L. inopinata also recorded in Canada (Viehberg and Pienitz, 2017). Candona candida is a generalist species in the Kangerlussuaq region, being present in deeper lakes, higher SRP concentrations and higher nitrate concentrations. These traits suggest that C. candida will become abundant in the Greenlandic ostracod fauna, and potentially across the Arctic. For some species, particularly C. pubera, nutrient concentrations are a dominant control on distribution. As Arctic warming increases, nutrient sources are predicted to increase. However, currently there is little understanding of the direct and indirect nutrient controls on ostracod fauna. Nutrient status of water appears, however, to be a significant control on ostracod presence and abundance and should be included in future ecological studies globally.
All data is available in the Supplement.
The supplement related to this article is available online at https://doi.org/10.5194/bg-23-2847-2026-supplement.
LR: Conceptualization, Methodology, Validation, Formal analysis, Investigation, Resources, Writing – original draft, Visualization. SMcG: Methodology, Validation, Investigation, Resources, Writing – original draft. AB: Investigation, Resources, Writing – Review & Editing. JH: Investigation, Writing – Review & Editing. DH: Investigation, Writing – Review & Editing.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
The authors thank: Chris Sørensen at Kangerlussuaq International Science Support (KISS); Mathi Woolford for assistance with ostracod picking; Miles Irving for help with editing figures; and Jim Davy for SEM operation. Travel to Greenland was funded by NWO Rubicon grant 019.183EN.018 awarded to AB.
This paper was edited by Pierre Amato and reviewed by Claude Meisch and Steffen Mischke.
Aebly, F. A. and Fritz, S. C.: Palaeohydrology of Kangerlussuaq (Søndre Strømfjord), west Greenland during the last ∼8000 years, The Holocene, 19, 91–104, https://doi.org/10.1177/0959683608096601, 2009.
Alkalaj, J., Hrafnsdottir, T. K., Ingimarsson, F., Smith, R. J., Kreiling, A.-K., and Mischke, S.: Distribution of Recent non-marine ostracods in Icelandic lakes, springs, and cave pools, J. Crustacean Biol., 39, 202–212, https://doi.org/10.1093/jcbiol/ruz008, 2019.
Alm, G.: Beiträge zur Kenntniss der nördlichen und arktischen Ostracoden Fauna, Ark. Zool., 9, 1914.
AMAP: Snow, Water, Ice and Permafrost. Summary for Policy-makers, Arctic Monitoring and Assessment Programme (AMAP), Oslo, Norway, 2017.
Anderson, N. J. and Brodersen, K. P.: Determining the date of ice-melt for low Arctic lakes along Søndre Strømfjord, southern West Greenland, Geol. Greenland Surv. Bull., 189, 54–59, 2001.
Anderson, N. J., D'andrea, W., and Fritz, S. C.: Holocene carbon burial by lakes in SW Greenland, Glob. Change Biol., 15, 2590–2598, https://doi.org/10.1111/j.1365-2486.2009.01942.x, 2009.
Anderson, N. J., Saros, J. E., Bullard, J. E., Cahoon, S. M., McGowan, S., Bagshaw, E. A., Barry, C. D., Bindler, R., Burpee, B. T., Carrivick, J. L., and Fowler, R. A.: The Arctic in the twenty-first century: Changing biogeochemical linkages across a paraglacial landscape of Greenland, BioScience, 67, 118–133, https://doi.org/10.1093/biosci/biw158, 2017.
Baird, W.: Notes on British Entomostraca, The Zoologist – A Popular Miscellany of Natural History, 1, 193–197, 1843.
Benardout, G.: Ostracod-based palaeotemperature reconstructions for MIS 11 human occupation at Beeches Pit, West Stow, Suffolk, UK, J. Archaeol. Sci., 54, 421–425, https://doi.org/10.1016/j.jas.2014.07.027, 2015.
Bennike, O.: Palaeoecological studies of Holocene lake sediments from west Greenland, Palaeogeogr. Palaeocl., 155, 285-304, https://doi.org/10.1016/S0031-0182(99)00121-2, 2000.
Bennike, O., Björck, S., Böcher, J., and Walker, I. R.: The Quaternary arthropod fauna of Greenland: a review with new data, Bull. Geol. Soc. Den., 47, 111–134, 2000.
Bennike, O., Anderson, N. J., and McGowan, S.: Holocene palaeoecology of southwest Greenland inferred from macrofossils in sediments of an oligosaline lake, J. Paleolimnol., 43, 787–798, https://doi.org/10.1007/s10933-009-9368-x, 2010.
Benson, R. H. and MacDonald, H. C.: Postglacial (Holocene) Ostracodes from Lake Erie. Arthropoda, University of Kansas Publications, Paleont. Contr., 1–34, 1963.
Box, J. E., Colgan, W. T., Christensen, T. R., Schmidt, N. M., Lund, M., Parmentier, F. J. W., Brown, R., Bhatt, U. S., Euskirchen, E. S., Romanovsky, V. E., and Walsh, J. E.: Key indicators of Arctic climate change: 1971–2017, Environ. Res. Lett., 14, 045010, https://doi.org/10.1088/1748-9326/aafc1b, 2019.
Box, J. E., Nielsen, K. P., Yang, X., Niwano, M., Wehrlé, A., Van As, D., Fettweis, X., Køltzow, M. A., Palmason, B., Fausto, R. S., and van den Broeke, M. R.: Greenland ice sheet rainfall climatology, extremes and atmospheric river rapids, Meteorol. Appl., 30, e2134, https://doi.org/10.1002/met.2134, 2023.
Brady, G. S. and Robertson, D.: Notes of a week's dredging in the west of Ireland, Annals and Magazine of Natural History Series 4, 3, 353–374, 1869.
Brehm, V.: Die Entomostraken der Danmark Expedition, Meddelelser om Grønland, 45, 12, 1911.
Bullard, J. E. and Mockford, T.: Seasonal and decadal variability of dust observations in the Kangerlussuaq area, west Greenland, Arct. Antarct. Alp. Res., 50, S100011, https://doi.org/10.1080/15230430.2017.1415854, 2018
Burpee, B., Saros, J. E., Northington, R. M., and Simon, K. S.: Microbial nutrient limitation in Arctic lakes in a permafrost landscape of southwest Greenland, Biogeosciences, 13, 365–374, https://doi.org/10.5194/bg-13-365-2016, 2016.
Burpee, B. T. and Saros, J. E.: Cross-ecosystem nutrient subsidies in Arctic and alpine lakes: implications of global change for remote lakes, Environ. Sci.-Proc. Imp., 22, 1166–1189, https://doi.org/10.1039/C9EM00528E, 2020.
Burpee, B. T., Anderson, D., and Saros, J. E.: Assessing ecological effects of glacial meltwater on lakes fed by the Greenland Ice Sheet: The role of nutrient subsidies and turbidity, Arct. Antarct. Alp. Res., 50, S100019, https://doi.org/10.1080/15230430.2017.1420953, 2018.
Costa, O. G.: Entomostraci, ostracodi, Fauna del regno di Napoli, 1, 7–12, 1847.
Coviaga, C. A., Perez, A. P., Ramos, L. Y., Alvear, P., and Cusminsky, G. C.: On two species of Riocypris (Crustacea, Ostracoda) from northern Patagonia and their relation to Eucypris fontana: implications in paleoenvironmental reconstructions, Can. J. Zool., 96, 801–817, https://doi.org/10.1139/cjz-2017-0233, 2018.
Danielopol, D. L., Handl, M., and Yin, Y.: Benthic ostracods in the pre-alpine deep lake Mondsee: Notes on the origin and distribution, in: Ostracoda in the Earth and Life Sciences, Proceedings of the 11th International Symposium on Ostracoda, edited by: McKenzie, K. G. and Jones, P. J., Warrnambool, Victoria, Australia, A. A. Balkema, Rotterdam, 465–480, 1993.
Danish Meteorological Institute (DMI): Mittarfik Kangerlussuaq vejrdata, https://www.dmi.dk/friedata/observationer, last access: 20 March 2026.
Ekman, S.: Ostracoden aus den nordschwedischen Hochgebirgen, Naturwissenschaftliche Untersuchungen des Sarekgebirges in Schwedisch-Lappland, 4, 169–198, 1908.
Fischer, S.: Abhandlung über das Genus Cypris und dessen in der Umgebung von St. Petersburg und von Fall bei Reval vorkommenden Arten, Mémoires de l'Académie Impériale des Sciences de St. Pétersbourg, 7, 127–167, 1851.
Fowler, R. A., Osburn, C. L., and Saros, J. E.: Climate-driven changes in dissolved organic carbon and water clarity in Arctic lakes of West Greenland, J. Geophys. Res.-Biogeo., 125, e2019JG005170, https://doi.org/10.1029/2019JG005170, 2020.
Frenzel, P., Henkel, D., Siccha, M., and Tschendel, L.: Do ostracod associations reflect macrophyte communities? A case study from the brackish water of the southern Baltic Sea coast, Aquat. Sci., 67, 142–155, https://doi.org/10.1007/s00027-004-0756-z, 2005.
Fuhrmann, R.: Atlas quartärer und rezenter Ostrakoden Mitteldeutschlands, Altenburger Naturwissenschaftliche Forschungen, 15, 1–320, 2012.
Gauthier, H.: Sur la structure de la coquille chez quelques Cypridopsinés et sur la validité du genre Cyprilla (Ostracodes), Bulletin de la Société zoologique de France, 64, 203–228, 1939.
Geiger, W.: Population dynamics, life histories and reproductive modes, in: Sex and Parthenogenesis – Evolutionary Ecology of Reproductive Modes in Non-Marine Ostracods, Backhuys, Leiden, 228, 1998.
Greenway, H., Holmes, J., and Burn, M.: The response of ostracod faunal assemblages to hydrology, lake level, and carbon cycling in a Jamaican marl lake: a palaeolimnological investigation, J. Micropalaeontol., 43, 81–91, https://doi.org/10.5194/jm-43-81-2024, 2024.
Grider, A., Saros, J., Northington, R., and Yde, J. C.: Glacially-fed lakes of West Greenland have elevated metal and nutrient concentrations and serve as regional repositories of these materials, Sci. Total Environ., 967, 178744, https://doi.org/10.1016/j.scitotenv.2025.178744, 2025.
Haberbosch, P.: Uber Arktischen Süsswassercrustaceen, Zool. Anz., Leipzig, 47, 1916.
Hartmann, G. and Hiller, D.: Beitrag zur Kenntnis der Ostracodenfauna des Harzes und seines nördlichen Vorlandes (unter besonderer Berücksichtigung des Männchens von Candona candida), 125 Jahre Naturwissenschaftlicher Verein Goslar, 99–116, 1977.
Hastings, M. G., Jarvis, J. C., and Steig, E. J.: Anthropogenic impacts on nitrogen isotopes of ice-core nitrate, Science, 324, 1288–1288, https://doi.org/10.1126/science.1170510, 2009.
Hawkings, J., Wadham, J., Tranter, M., Telling, J., Bagshaw, E., Beaton, A., Simmons, S. L., Chandler, D., Tedstone, A., and Nienow, P.: The Greenland Ice Sheet as a hot spot of phosphorus weathering and export in the Arctic, Global Biogeochem. Cy., 30, 191–210, https://doi.org/10.1002/2015GB005237, 2016.
Hazuková, V., Burpee, B. T., Northington, R. M., Anderson, N. J., and Saros, J. E.: Earlier ice melt increases hypolimnetic oxygen despite regional warming in small Arctic lakes, Limnol. Oceanogr.-Lett., 9, 258–267, https://doi.org/10.1002/lol2.10386, 2024.
Heinemann, G.: The KABEG'97 field experiment: An aircraft-based study of katabatic wind dynamics over the Greenland ice sheet, Bound.-Lay. Meteorol., 93, 75–116, 1999.
Higuti, J., Declerck, S. A., Lansac-Tôha, F. A., Velho, L. F. M., and Martens, K.: Variation in ostracod (Crustacea, Ostracoda) communities in the alluvial valley of the upper Paraná River (Brazil) in relation to substrate, Hydrobiologia, 644, 261–278, https://doi.org/10.1007/s10750-010-0122-1, 2010.
Horne, D. J.: A mutual temperature range method for Quaternary palaeoclimatic analysis using European nonmarine Ostracoda, Quaternary Sci. Rev., 26, 1398–1415, https://doi.org/10.1016/j.quascirev.2007.03.006, 2007.
Horne, D. J., Ashton, N., Benardout, G., Brooks, S. J., Coope, G. R., Holmes, J. A., Lewis, S. G., Parfitt, S. A., White, T. S., Whitehouse, N. J., and Whittaker, J. E.: A terrestrial record of climate variation during MIS 11 through multiproxy palaeotemperature reconstructions from Hoxne, UK, Quaternary Res., 111, 21–52, https://doi.org/10.1017/qua.2022.20, 2023.
Hu, X. M., Ma, J. R., Ying, J., Cai, M., and Kong, Y. Q.: Inferring future warming in the Arctic from the observed global warming trend and CMIP6 simulations, Adv. Clim. Chang. Res., 12, 499–507, https://doi.org/10.1016/j.accre.2021.04.002, 2021.
Huai, B., Ding, M., van den Broeke, M. R., Reijmer, C. H., Noël, B., Sun, W., and Wang, Y.: Future large-scale atmospheric circulation changes and Greenland precipitation, npj Clim. Atmos. Sci., 8, 10, https://doi.org/10.1038/s41612-025-00899-z, 2025.
Jeffrey, S. W. and Humphrey, G. F.: New spectrophotometric equations for determining chlorophyll a, b, c1 and c2 in higher plants, algae and natural phytoplankton, Biochem. Physiol. Pfl., 167, 191–194, https://doi.org/10.1016/S0015-3796(17)30778-3, 1975.
Johansson, E., Berglund, S., Lindborg, T., Petrone, J., van As, D., Gustafsson, L.-G., Näslund, J.-O., and Laudon, H.: Hydrological and meteorological investigations in a periglacial lake catchment near Kangerlussuaq, west Greenland – presentation of a new multi-parameter data set, Earth Syst. Sci. Data, 7, 93–108, https://doi.org/10.5194/essd-7-93-2015, 2015.
Jungwirth, W.: Limnocythere inopinata (Baird) (Cytheridae, Ostracoda): Its distribution pattern and relation to the superficial sediments of Neusiedlersee, in: Neusiedlersee: The limnology of a shallow lake in central Europe, (Monographiae Biologicae), edited by: Löffler, H., Dr. W. Junk Publishers, The Hague, 37, 385–388, 1979.
Jurine, L.: Histoire des Monocles, qui se trouvent aux environs de Genève, Genève & Paris, 1–22, https://doi.org/10.5962/bhl.title.10137, 1820.
Kolde, R.: pheatmap: Pretty Heatmaps, R package version 1.0.13 [code], https://github.com/raivokolde/pheatmap (last access: 22 April 2026), 2025.
Löffler, H.: Zur Limnologie, Entomostraken- und Rotatorienfauna des Seewinkel-Gebietes, Sitzungsberichte der Osterreichischen Akademie der Wissenschaften, mathematischnaturwissenschaftliche Klasse, 1168, 315–362, 1959.
Mackereth, F. J. H., Heron, J., and Talling, J. F.: Water analysis: some revised methods for limnologists (Freshwater Biological Association Scientific Publication No. 36), 2nd edn., Titus Wilson, Kendal, 1989.
Marchegiano, M., Francke, A., Gliozzi, E., and Ariztegui, D.: Arid and humid phases in central Italy during the Late Pleistocene revealed by the Lake Trasimeno ostracod record, Palaeogeogr. Palaeoecl. 490, 55–69, https://doi.org/10.1016/j.palaeo.2017.09.033, 2018.
Marchegiano, M., Horne, D. J., Gliozzi, E., Francke, A., Wagner, B., and Ariztegui, D.: Rapid Late Pleistocene climate change reconstructed from a lacustrine ostracod record in central Italy (Lake Trasimeno, Umbria), Boreas, 49, 739–750, https://doi.org/10.1111/bor.12450, 2020.
McCormack, J., Viehberg, F., Akdemir, D., Immenhauser, A., and Kwiecien, O.: Ostracods as ecological and isotopic indicators of lake water salinity changes: the Lake Van example, Biogeosciences, 16, 2095–2114, https://doi.org/10.5194/bg-16-2095-2019, 2019.
McCrystall, M. R., Stroeve, J., Serreze, M., Forbes, B. C., and Screen, J. A.: New climate models reveal faster and larger increases in Arctic precipitation than previously projected, Nat. Commun., 12, 6765, https://doi.org/10.1038/s41467-021-27031-y, 2021.
McGoff, E., Aroviita, J., Pilotto, F., Miler, O., Solimini, A. G., Porst, G., Jurca, T., Donohue, L., and Sandin, L.: Assessing the relationship between the Lake Habitat Survey and littoral macroinvertebrate communities in European lakes, Ecol. Indic., 25, 205–214, https://doi.org/10.1016/j.ecolind.2012.09.018, 2013.
McGowan, S., Gunn, H. V., Whiteford, E. J., John Anderson, N., Jones, V. J., and Law, A. C.: Functional attributes of epilithic diatoms for palaeoenvironmental interpretations in South-West Greenland lakes, J. Paleolimnol., 60, 273–298, https://doi.org/10.1007/s10933-017-9968-9, 2018.
Meisch, C.: Freshwater Ostracoda of western and central Europe, Gustav Fischer, Spektrum Akademischer Verlag, Stuttgart, ISBN 9783827410016, 2000.
Meisch, C., Smith, R. J., and Martens, K.: An updated subjective global checklist of the extant non-marine Ostracoda (Crustacea), Eur. J. Taxon., 974, 1–144, https://doi.org/10.5852/ejt.2024.974.2767, 2024.
Mernild, S. H., Liston, G. E., van As, D., Hasholt, B., and Yde, J. C.: High-resolution ice sheet surface mass-balance and spatiotemporal runoff simulations: Kangerlussuaq, west Greenland, Arct. Antarct. Alp. Res., 50, S100008, https://doi.org/10.1080/15230430.2017.1415856, 2018.
Mesquita-Joanes, F., Smith, A. J., and Viehberg, F. A: The Ecology of Ostracoda across Levels of Biological Organization from individual to Ecosystem: A Review of Recent Developments and Future Potential, in: Ostracoda as Proxies for Quaternary Climate Change, edited by: Horne, D. J., Holmes, J., Rodriguez-Lazaro, J., and Viehberg, F. A., Elsevier Science BV, London, 15–35, https://doi.org/10.1016/B978-0-444-53636-5.00002-0, 2012.
Müller, O. F.: Zoologiae Danicae Prodromus, seu Animalium Daniae et Norvegiae Indigenarum characters, nomina, et synonyma imprimis popularium, Typis Hallageriis, Havniae, 32, 1–282, 1776.
Nelligan, C., Sorichetti, R. J., Yousif, M., Thomas, J. L., Wellen, C. C., Parsons, C. T., and Mohamed, M. N.: Then and now: revisiting nutrient export in agricultural watersheds within southern Ontario's lower Great Lakes basin, J. Great Lakes Res., 47, 1689–1701, https://doi.org/10.1016/j.jglr.2021.08.010, 2021.
Olsen, J., Anderson, N. J., and Knudsen, M. F.: Variability of the North Atlantic Oscillation over the past 5,200 years, Nat. Geosci., 5, 808–812, https://doi.org/10.1038/ngeo1589, 2012.
Oksanen, J., Simpson, G., Blanchet, F., Kindt, R., Legendre, P., Minchin, P., O'Hara, R., Solymos, P., Stevens, M., Szoecs, E., Wagner, H., Barbour, M., Bedward, M., Bolker, B., Borcard, D., Borman, T., Carvalho, G., Chirico, M., De Caceres, M., Durand, S., Evangelista, H., Fitz, J. R., Friendly, M., Furneaux, B., Hannigan, G., Hill, M., Lahti, L., Martino, C., McGlinn, D., Ouellette, M., Ribeiro, C. E., Smith, T., Stier, A., Ter Braak, C., and Weedon, J.: Vegan: Community Ecology Package, R package version 2.8-0 [data set], https://vegandevs.github.io/vegan/ (last access: 22 April 2026), 2025.
O'Reilly, C. M., Sharma, S., Gray, D. K., Hampton, S. E., Read, J. S., Rowley, R. J., Schneider, P., Lenters, J. D., McIntyre, P. B., Kraemer, B. M., and Weyhenmeyer, G. A.: Rapid and highly variable warming of lake surface waters around the globe, Geophys. Res. Lett., 42, 10–773, https://doi.org/10.1002/2015GL066235, 2015.
Paltan, H., Dash, J., and Edwards, M.: A refined mapping of Arctic lakes using Landsat imagery, Int. J. Remote Sens., 36, 5970–5982, https://doi.org/10.1080/01431161.2015.1110263, 2015.
Post, E., Forchhammer, M. C., Bret-Harte, M. S., Callaghan, T. V., Christensen, T. R., Elberling, B., Fox, A. D., Gilg, O., Hik, D. S., Høye, T. T., and Ims, R. A.: Ecological dynamics across the Arctic associated with recent climate change, Science, 325, 1355–1358, https://doi.org/10.1126/science.1173113, 2009.
Poulsen, E. M.: The Zoology of East Greenland, Freshwater Entomostraca, Meddelelser om Grønland, 121, 335, 1940.
Prater, C., Bullard, J. E., Osburn, C. L., Martin, S. L., Watts, M. J., and Anderson, N. J.: Landscape controls on nutrient stoichiometry regulate lake primary production at the margin of the Greenland Ice Sheet, Ecosystems, 25, 931–947, https://doi.org/10.1007/s10021-021-00693-x, 2022.
Ramdohr, K. A.: Über die Gattung Cypris Müll. und drei zu derselben gehörige neue Arten, Magazin für die neuesten Entdeckungen in der gesammten Naturkunde der Gesellschaft naturforschender Freunde zu Berlin, 2, 83–93, 1808.
Rantanen, M., Karpechko, A. Y., Lipponen, A., Nordling, K., Hyvärinen, O., Ruosteenoja, K., Vihma, T., and Laaksonen, A.: The Arctic has warmed nearly four times faster than the globe since 1979, Commun. Earth Environ., 3, 168, https://doi.org/10.1038/s43247-022-00498-3, 2022.
Reuss, N. S., Hamerlík, L., Velle, G., Michelsen, A., Pedersen, O., and Brodersen, K. P.: Microhabitat influence on chironomid community structure and stable isotope signatures in West Greenland lakes, Hydrobiologia, 730, 59–77, https://doi.org/10.1007/s10750-014-1821-9, 2014.
Roca, J. R. and Danielopol, D. L.: Exploration of interstitial habitats by the phytophilous Ostracod Cypridopsis vidua (OF Müller): experimental evidence, Annales de Limnologie-International Journal of Limnology, 27, 243–252, 1991.
Roca, J. R., Baltanás, A., and Uiblein, F.: Adaptive responses in Cypridopsis vidua (Crustacea: Ostracoda) to food and shelter offered by a macrophyte (Chara fragilis), Hydrobiologia, 262, 127–131, 1993.
Rodríguez-Pérez, H. and Baltanás, A.: Ecology and production of Heterocypris exigua and Plesiocypridopsis newtoni (Crustacea, Ostracoda) in an oligohaline hypertrophic shallow lake, Fund. Appl. Limnol./Arch. Hydrobiol., 172, 13–26, 2008.
Røen, U.: Studies on freshwater Entomostraca in Greenland II. Localities, ecology, and geographical distribution of the species, Meddelelser om Grønland, 170, 1–249, 1962.
Røen, U.: Studies on freshwater Entomostraca in Greenland III. Entomostraca from Peary Land with notes on their biology, Meddelelser om Grønland, 184, 1–59, 1968.
Røen, U.: Studies on freshwater Entomostraca in Greenland IV. A collection from Angmagssalik, East Greenland, Meddelelser om Grønland, 184, 1–18, 1970.
Røen, U.: Studies on the freshwater Entomostraca in Greenland V. The fauna of the Hazen camp study area, Ellesmere Island, N.WT., Canada compared to that of the Thule area, Greenland, Steenstrupia 7, 321–335, 1981.
Saros, J. E., Anderson, N. J., Juggins, S., McGowan, S., Yde, J. C., Telling, J., Bullard, J. E., Yallop, M. L., Heathcote, A. J., Burpee, B. T., and Fowler, R. A.: Arctic climate shifts drive rapid ecosystem responses across the West Greenland landscape, Environ. Res. Lett., 14, 074027, https://doi.org/10.1088/1748-9326/ab2928, 2019.
Saros, J. E., Arp, C. D., Bouchard, F., Comte, J., Couture, R. M., Dean, J. F., Lafrenière, M., MacIntyre, S., McGowan, S., Rautio, M., and Prater, C.: Sentinel responses of Arctic freshwater systems to climate: linkages, evidence, and a roadmap for future research, Arct. Sci., 9, 356–392, 2022.
Saros, J. E., Hazuková, V., Northington, R. M., Huston, G. P., Lamb, A., Birkel, S., Pereira, R., Bourdin, G., Jiang, B., and McGowan, S.: Abrupt transformation of West Greenland lakes following compound climate extremes associated with atmospheric rivers, P. Natl. Acad. Sci., 122, e2413855122, https://doi.org/10.1073/pnas.2413855122, 2025.
Sars, G. O.: Oversigt at Norges Crustaceer, medforelöbige Bemaerkninger over de nye eller mindre bekjendte Arter. II. (Branchiopoda – Ostracoda – Cirripedia), Forhl. Christiania Vidensk-selsk, 1890, 1–80, 1890.
Sars, G. O: Freshwater Ostracoda from Canada and Alaska. Report of the Canadian Arctic Expedition 1913–18, Vol. VII (Part I: Ostracoda), 1–23, 1926.
Scharf, B. W.: Zur rezenten Muschelkrebsfauna der Eifelmaare (Crustacea: Ostracoda), Mitteilungen der Pollichia, 69, 185–204, 1981.
Schmidt, P. P.: Recent and subfossil finds of a new species of ostracod, Potamocypris parva, in Greenland (Crustacea, Ostracoda, Cyprididae), Astarte, 9, 13–17, 1976.
Schneider, A., Wetterich, S., Schirrmeister, L., Herzschuh, U., Meyer, H., and Pestryakova, L. A.: Freshwater ostracods (Crustacea) and environmental variability of polygon ponds in the tundra of the Indigirka Lowland, north-east Siberia, Polar Res., 35, 25225, https://doi.org/10.3402/polar.v35.25225, 2016.
Scott, T.: Report on the marine and freshwater Crustacea from Franz-Josef Land, collected by Mr William Brice, of the Jackson-Harmsworth Expedition, Journal of the Linnean Society of London (Zool.), 6, 1–161, 1899.
Smith, A. J. and Horne, D. J.: Class Ostracoda, in: Ecology and General Biology: Thorp and Covich's Freshwater Invertebrates, edited by: Thorp, J. H. and Covich, A. P., Volume 2, Academic Press (Elsevier), Burlington, https://doi.org/10.1016/C2015-0-01546-5, 2016.
Smol, J. P., Wolfe, A. P., Birks, H. J. B., Douglas, M. S., Jones, V. J., Korhola, A., Pienitz, R., Rühland, K., Sorvari, S., Antoniades, D., and Brooks, S. J.: Climate-driven regime shifts in the biological communities of arctic lakes, P. Natl. Acad. Sci., 102, 4397–4402, https://doi.org/10.1073/pnas.0500245102, 2005.
Stephanides, T.: A survey of the fresh-water biology of Corfu and of certain other regions of Greece, Praktika of the Hellenic Hydrobiologicalinstitute, 2 (part 2), 1–263, 1948.
Vadeboncoeur, Y., Vander Zanden, M. J., and Lodge, D. M.: Putting the Lake Back Together: Reintegrating Benthic Pathways into Lake Food Web Models: Lake ecologists tend to focus their research on pelagic energy pathways, but, from algae to fish, benthic organisms form an integral part of lake food webs, Bioscience, 52, 44–54, https://doi.org/10.1641/0006-3568(2002)052[0044:PTLBTR]2.0.CO;2, 2002.
Van As, D., Hasholt, B., Ahlstrøm, A. P., Box, J. E., Cappelen, J., Colgan, W., Fausto, R. S., Mernild, S. H., Mikkelsen, A. B., Noël, B. P., and Petersen, D.: Reconstructing Greenland ice sheet meltwater discharge through the Watson River (1949–2017), Arct. Antarct. Alp. Res., 50, S100010, https://doi.org/10.1080/15230430.2018.1433799, 2018.
Vesper, B.: Ein Beitrag zur Ostracodenfauna Schleswig-Holsteins, Mitteilungen aus dem hamburgischen zoologischen Museum und Institut, 72 97–108, 1975.
Viehberg, F. A. and Pienitz, R.: Trends in Ostracoda and Cladocera distribution and water chemistry in subarctic Canada: Churchill (Manitoba) lakes and ponds revisited, J. Limnol., 76, https://doi.org/10.4081/jlimnol.2017.1578, 2017.
Wang, C., Zhao, D., Zhou, Z., and Yuan, C.: Ostracod-based transfer function shifting to a broad prospect in palaeolimnology and palaeoclimate, Sci. Total Environ., 958, 177894, https://doi.org/10.1016/j.scitotenv.2024.177894, 2025.
Wei, T. and Simko, V.: R package “corrplot”: Visualization of a Correlation Matrix (Version 0.95), GitHub, https://github.com/taiyun/corrplot (last access: 22 April 2026), 2024.
Wetterich, S., Schirrmeister, L., Meyer, H., Viehberg, F. A., and Mackensen, A.: Arctic freshwater ostracods from modern periglacial environments in the Lena River Delta (Siberian Arctic, Russia): geochemical applications for palaeoenvironmental reconstructions, J. Paleolimnol., 39, 427–449, https://doi.org/10.1007/s10933-007-9122-1, 2008.
Whiteford, E. J., McGowan, S., Barry, C. D., and Anderson, N. J.: Seasonal and regional controls of phytoplankton production along a climate gradient in South-West Greenland during ice-cover and ice-free conditions, Arct. Antarct. Alp. Res., 48, 139–159, https://doi.org/10.1657/AAAR0015-003, 2016.
Woolway, R. I., Sharma, S., and Smol, J. P.: Lakes in hot water: the impacts of a changing climate on aquatic ecosystems, Bioscience, 72, 1050–1061, https://doi.org/10.1093/biosci/biac052, 2022.
Zaddach, E. G.: Bradleystrandesia reticulata, in Backlund M (2026), Dyntaxa. Svensk taxonomisk databas, SLU Artdatabanken, Checklist dataset, https://doi.org/10.15468/j43wfc, 1844.