the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
An unpredictable body size response to the Permo-Triassic climate crisis
William J. Foster
Herwig Prinoth
Evelyn Kustatscher
Michael Hautmann
A predictive ecological response to both the present and past climate crises is that marine ectotherm species will become smaller before going extinct or fluctuate in abundance and size with environmental conditions. The problem with studying past climate events with high rates and magnitude of warming, which may serve as analogues for projected climate change, is that very few species, or even genera, survived such events. Here, we utilized one of the few records of marine bivalves that spans the Permian-Triassic climate crisis with specimen-level data and at a high resolution. These measurements come from the Bellerophon and Werfen formations of the Dolomites in Italy, representing relatively shallow marine environments. At the species-level, there is almost a complete turnover, and the newly evolved species are typically significantly smaller, but not unusually small, whereas the three surviving species do not show a significant body size change. Our results indicate that the observed temporary size reduction at the genus-level is primarily driven by the preferential evolution of smaller species after the extinction, rather than, as often assumed, by a size decrease within existing species; this challenges the universal validity of the “Lilliput effect” in the sense of direct intra-species dwarfing, but confirms it as a consequence of faunal turnover. Subsequently, there are two pulses of genus-level body size recovery caused by different mechanisms. The first phase (late Griesbachian) is driven by the size-increase of the existing species, whereas the second phase (early Spathian) is also due to the evolution of larger species. The effects of abiotic and biotic factors in controlling these body size dynamics are superimposed during the Early Triassic. These results suggest a mechanism to explain size reductions during climate crises, but does not find a species-level body size reduction to be a forecastable response to extreme climate warming.
- Article
(4406 KB) - Full-text XML
-
Supplement
(1386 KB) - BibTeX
- EndNote
If scientists want to use past climate crises to forecast the potential ecological response of marine ecosystems to different projections of a modern-day climate and biodiversity crisis, then they must uncover predictive or forecastable responses of marine ecosystems to the main threats to biodiversity. One way to do this is to use past climate change events to test hypotheses related to the forecasted responses. It has, however, been suggested that the evolution of marine ecosystems has altered how climate change manifests, meaning it is difficult to uncover consistent ecological responses to climate crises (Payne et al., 2016; Foster et al., 2023a). For example, the evolution of siliceous and calcareous plankton, as well as past collapses in their productivity, would have affected how the oceans could buffer increased influxes of carbon dioxide (Kump et al., 2009; Isson et al., 2020; Rauzi et al., 2024). On the other hand, there are some consistent ecological responses observed at multiple hyperthermal events that may be considered forecastable during a projected climate and biodiversity crisis, with a key predictive response being the reduction in body size of marine ectotherms (Rita et al., 2019; Monarrez et al., 2020; Pörtner et al., 2023; Nätscher et al., 2025).
The consistent body size response, a temporary reduction in the size of ectotherms, in evolutionary studies of hyperthermals has been coined the “Lilliput effect” (Urbanek, 1993; Twitchett, 2007). This means that a predictive response to projected climate change would be an expected decrease in body size, but this finding is not unanimous and there has been an increasing number of studies suggesting that a body size decrease for past hyperthermal events is not always going to be observed (Brayard et al., 2010, 2015; Payne et al., 2016; Nätscher et al., 2023). In addition, the mechanism(s) that explains a body size decrease is often debated, e.g., is it the size-reduction at the species-level or genus-level that is being observed (Brayard et al., 2010, 2015), is it the preferential loss of larger species (Nützel et al., 2010), or is no body size reduction actually observed at all (Nätscher et al., 2023). There is also a clear lack of data when trying to understand the body-size response of marine ectotherms during and between different climate crises. The magnitude of extinction events also makes it challenging to investigate body size dynamics as regional studies show high turnover rates and organisms may show additional ecological responses, such as latitudinal migrations, overprinting the predicted body size response (De Baets et al., 2021). This means that some studies have only been able to look at the body size response at a coarse temporal or coarse taxonomical scale (e.g., Rego et al., 2012; Payne et al., 2016), which can lead to some conflicting results (Monarrez et al., 2020), because they do not capture species size dynamics during the event. Despite the increasing number of studies that have investigated body-size changes with bed-scale analyses for extinction events (e.g., Morten and Twitchett, 2009; Rita et al., 2019; De Baets et al., 2021; Nätscher et al., 2021, 2025; Piazza et al., 2019, 2020), the number of regions and events that have been investigated remains limited. It is, therefore, critical that if we want to better understand a predictable ecological response to climate change, then we need high-resolution datasets that span these past climate crises from multiple regions.
The debate surrounding the body size response for marine ectotherms could not be greater than for the Permian-Triassic climate crisis, a 10–12 °C hyperthermal event (Gliwa et al., 2022) resulting in the highest extinction rates of marine taxa of the Phanerozoic (Kocsis et al., 2018). This high turnover took place not only at the species-level, but also at higher taxonomic levels, particularly affecting phyla that dominated marine ecosystems prior to the extinction event (e.g., brachiopods, crinoids and bryozoans). This is, however, a general global trend; in shallow marine ecosystems bivalves, foraminifera, ostracods and gastropods were already dominant aspects of the marine ecosystem prior to the extinction, e.g., in the Dolomites (Italy) (Prinoth and Posenato, 2023; Mette and Roozabani, 2012; Gómez Correa et al., 2025), Antalya and Aladağ Nappes (Türkiye) (Karapunar et al., 2025a), and Spitzbergen (Svalbard) (Nakazawa, 1999; Bond et al., 2015). High-resolution studies from those regions and groups could, therefore, provide key insights into the response of ectotherms during a high magnitude climate crisis. Here, we have utilized a novel bivalve record of the Dolomites (Italy) to understand the bivalve body size response, if any, to the Permian-Triassic climate crisis. The Dolomites and neighbouring Carnic Alps are often regarded as one of the best-studied regions in terms of palaeontological and geochemical records for the Permian-Triassic crisis, owing to the numerous exposures of a Permian-Triassic succession representing a range of water depths (e.g., Holser et al., 1991; Broglio Loriga et al., 1986; Twitchett and Wignall, 1996; Farabegoli et al., 2007; Horacek et al., 2007; Posenato, 2019; Kustatscher et al., 2019) and the evolution of the Triassic carbonate platforms (e.g., Stefani et al., 2004). Yet, it is only recently that an extensive bivalve collection spanning the Permian-Triassic event has been produced (Prinoth and Posenato, 2023) and there is a relative dearth of robust proxy evidence for environmental changes, even at the best studied sections.
The Dolomites are a mountain area, classically considered bound by the Puster/Pusteria Valley to the North, Valsugana Valley to the South, River Eisack/Isarco and Etsch/Adige to the West and Piave Valley to the East. The UNESCO World Heritage Dolomites includes also the Brenta Group to the West and the Friulian Dolomites to the East. The Dolomites are part of the Southern Alps, where the sedimentological and ecological evolution of the Dolomites extends in other tectonic blocks from the same palaeogeographic region, e.g., Gemer-Bükk-Zagorje Unit (Kovács et al., 2011). During the Permian-Triassic transition, the western Palaeotethys was being subducted to the north and the Neotethys was opening to the south, separated by the Cimmerian microcontinent. The Mesozoic sediments of the Dolomites were deposited on the Adria tectonic block, in the western Palaeotethys around 15° N, which was drifting north as it became increasingly detached from the northern Africa plate (Dercourt et al., 2000). Contemporaneously, the Adria block is one of many tectonic blocks in the western Palaeotethys and northwestern Neotethys, which record extensive shallow water carbonate deposition during the late Permian and the Triassic, from the Arabian shield across the northern Africa plate, and from the Iberian massif to Dobrogea (Dercourt et al., 2000; Muttoni et al., 2009; Pérez-Valera et al., 2023). The Adria block thus represents part of a broad epicontinental shelf and substantial habitable area for shallow marine ecosystems. Tectonically, the Dolomites experienced limited deformation during the Permian–Triassic, allowing for exceptional preservation of coastal to platform-basin transitions (Brandner et al., 2009).
The depositional setting during the Changhsingian-Early Triassic of the Dolomites was a homoclinal, mixed carbonate-siliciclastic ramp (Broglio Loriga et al., 1983). Locally, the palaeocoastline was situated to the west and outcrops of the Permian-Triassic successions towards the east of the Dolomites and into the Carnic Alps generally show a deepening (Kustatscher et al., 2019) (Fig. 1). The Lopingian sedimentary succession is represented by the terrestrial Gröden Formation (aka., Val Gardena Sandstone) and shallow marine Bellerophon Formation, which overlays Permian Porphyry volcanics, basal conglomerates and a Brixen quartzite basement (Bosellini et al., 2003).
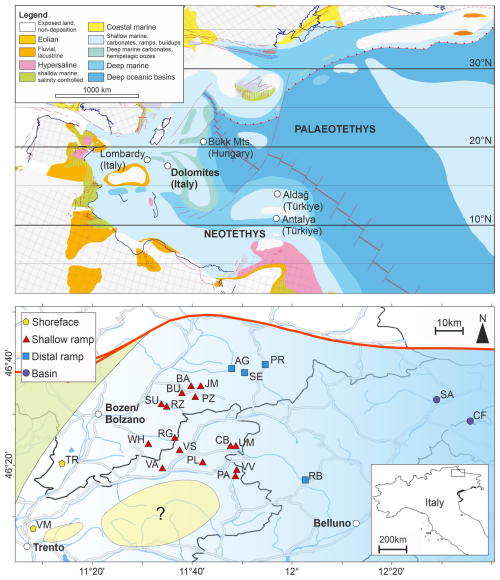
Figure 1Palaeogeographic setting of the Dolomites, Italy. Upper panel: Palaeogeographic map of the western Palaeotethys after the Ladinian reconstruction of Dercourt et al. (2000). The location of the Dolomites and other mentioned regions are indicated. Lower panel: The locations of the investigated sections from the Dolomites (Italy), overlain by a palaeodepositional map for the Permian/Triassic boundary emphasizing the onshore-offshore water depth gradient. Abbreviations: AG – Alferer Geisler, BA – Balest, BU – Pufels/Bulla, CB – Costabella, CF – Casera Federata, JM – Jmueia, PA – Punta Rolle, PL – , PR – Preroman, PZ – Ruf da Piz, RB – Rova-Baches, RG – Rosengarten/Catinaccio, RZ – Ratzes, SA – San Antonio, SE – Seres, SU – Seis/Siusi, TR – Tramin/Termeno, UM – Uomo/l'Om Picol, WH – Weißhorn/Corno Bianco, VA – Val Averta, VM – Vigo Meano, VS – Val Sorda, VV – Val Venegia. Note: the palaeocoastline and water-depths were dynamic during the Permian-Triassic transition and these figures are snapshots demonstrating the relative differences between the different localities.
The extinction interval and Early Triassic is found within the Werfen Formation. The timing of the mass extinction event is yet to be quantitatively constrained, with some studies recognizing multiple events starting with the Bellerophon/Werfen formation boundary (e.g., Farabegoli et al., 2007), and others recognizing a pulse within the Tesero Member (Werfen Formation) (e.g., Groves et al., 2007). Lithologically and palaeogeographically, the Dolomites succession is similar to the Antalya and Aladağ Nappes (Turkey), where the extinction has been quantitatively interpreted as an interval spanning the onset of ooid deposition up to the Permian/Triassic boundary (Karapunar et al., 2025a). At the Bulla section, the parastratotype for the Dolomites, this crisis interval corresponds to Beds BU7B–BU12B and coincides with a pronounced negative carbon-isotope excursion (Farabegoli et al., 2007), whereas the main biotic extinction horizon lies slightly below the boundary, in BU12A (Farabegoli et al., 2007; Farabegoli and Perri, 2012). This interval also corresponds to a species decline and mix of autochthonous and allochthonous Permian microfossils and sedimentological changes associated with the extinction drivers.
The bivalves that were measured in this study were primarily from Prinoth and Posenato (2023) with additional specimens from Broglio Loriga and Mirabella (1986), Hofmann et al. (2015) and Foster et al. (2020). Additional specimens were collected during targeted fieldwork on the Mazzin Member of the Werfen Formation, and measurements from figured specimens in Neri and Posenato (1985) and Posenato (2009) were additionally included. Some species which were found in Permian rocks of the Dolomites could not be found in the post-extinction succession, but are known from other European locations, in particular the Bükk Mountains in Hungary. Therefore, the body size data from the basal beds of the Gerennavár Limestone Formation at the Bálvány-North section reported by Posenato et al. (2005) were also included in the dataset. The species identifications were standardised to ensure taxonomic consistency and avoid synonymy. For example, Claraia orbicularis, C. wangi-griesbachi, and C. aurita from the Mazzin Member were recombined as Claraia sp., as the different species names represent different taxonomic opinions rather than different species (see also Foster et al., 2020 for a discussion). For each specimen, the height and the width were measured to the nearest 0.1 mm using digital callipers. The geometric mean of each specimen was calculated where: the square root of the product of shell height and shell length, and these were then log transformed so that proportional deviations are represented consistently.
Combined, bivalve measurements come from 27 different localities across the Dolomites (Fig. 1) and correlating the sections is difficult due to the lack of lithological marker beds. Therefore, the specimens were pooled based on the sequence stratigraphic profile of the Permian-Triassic succession (following Posenato, 2008; Foster et al., 2017; Prinoth and Posenato, 2023), where: three stratigraphic sequences (Lo3–Lo5) are identified from the Lopingian Bellerophon Formation; two sequences from the Induan (In1–In2); and three sequences from the Olenekian (O1–O3). The stratigraphic sequence In1 starts with the uppermost Bulla Member of the Bellerophon Formation, prior to the extinction event (sensu Farabegoli et al., 2007), but no bivalve measurements were recorded from the Bulla Member, and the body sizes of In1 represent exclusively post-extinction body sizes. All raw taxonomic, stratigraphic, locality and measurement data can be found in the supplementary dataset.
In the analysis, bivalve specimens in open nomenclature were included. All specimens were disarticulated and given the variability in left and right valve measurements, it is unlikely that measurements from a left and right valve both originated from the same individual. Therefore, all specimen measurements were included in the analysis. To determine if differences between the time intervals were significant, we used the non-parametric Kruskal-Wallis test and Mann-Whitney U pairwise tests with a significance level of 0.05.
To investigate the drivers of body size changes, we utilised the within- and among-taxa approach by Rego et al. (2012) using the modified version of Rita et al. (2019). This method allows division of the assemblage body-size shift into three components: a disappearance of taxa effect, within-lineage effect and appearance of new taxa effect (see Rita et al., 2019). The analysis by Rego et al. (2012), Rita et al. (2019) and Nätscher et al. (2023) re-classifies all taxa into the different assemblage components and then calculates the median body sizes for each component. That means that changes in the components are sensitive to changes in the relative abundance of different species within the time interval. I.e., an assemblage component can record significant changes in size, even if all the taxa stay the same size, due to changes in the taxon abundances. Here, to avoid the impact of taxonomic composition dynamics in understanding body-size dynamics, the median size of each species was first calculated before being categorized into the different assemblage components. A separate analysis was also done following Rego et al. (2012)'s approach to observe if this leads to major differences in the results. All statistical analyses were made in R (R Core Team, 2019) and the code and data is available at Zenodo, https://doi.org/10.5281/zenodo.18380698 (Foster, 2026).
4833 body sizes of 72 species from 25 genera were investigated from the Dolomites, Italy (Figs. S2–S11 in the Supplement). In this region, only Bakevellia ceratophaga, survived the extinction event. B. ceratophaga shows an increase in median size from the middle Changhsingian into the Triassic, despite only two measurements being available from the early Griesbachian, but no size data from the upper Changhsingian are available for this species. It is also possible that Stutchburia tschernyschewi also survived the extinction, where Stutchburia sp. B from the Triassic could be reassigned to S. tschernyschewi. If this taxonomic reassignment is correct, still only seven Changhsingian and one Dienerian specimen of Stutchburia tschernyschewi were measured in this study (Fig. 2A), making a fair comparison across the extinction event not possible. This single post-extinction specimen of S. tschernschewi would be a small representative, but within the size range of the pre-extinction measurements. In addition, this specimen was not recorded from the Mazzin Mb. immediately after the mass extinction event and a significant size decrease may have occurred, but was not observed. A third species that survived the initial extinctions is Eumorphotis lorigae, which is recorded from the upper Changhsingian of the Dolomites (Posenato, 2009; Prinoth and Posenato, 2023) and from the deeper water setting of the Bükk Mountains, Hungary (Posenato et al., 2005). During the extinction event and when the pre-extinction sizes are compared, this species also shows an insignificant (p=0.32) size increase during the extinction event (Fig. 2A). At the genus-level, however, six (50 %) genera survived, with three genera showing a significant body size decrease (Pernopecten (p<0.01), Towapteria (p<0.01) and “Unionites” (p<0.01)) and Eumorphotis with an insignificant body size decrease (p=0.16). Only Bakevellia shows a significant (p=0.01) body size increase, owing to an earlier size decrease in the upper Changhsingian (Fig. 2B). In addition, when the measurements of the closely related genera Schizodus and Neoschizodus (which may be part of an anagenetic lineage; Newell and Boyd, 1975) are combined, they show a significant size decrease across the extinction event (Fig. 2B; p<0.01).
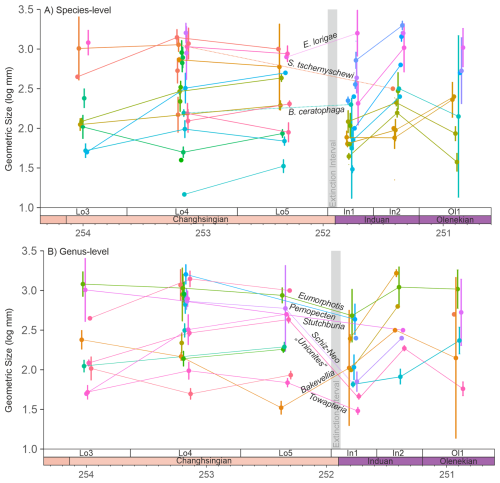
Figure 2Changes in the mean geometric size of bivalves from the Permian-Triassic transition in the Dolomites, Italy, with 95 % confidence intervals. (A) Body size changes at the species-level. The samples are grouped based on their sequence stratigraphic framework to reduce lithological biases on body sizes. Note: S. tschernyschewi is only considered an extinction survivor if the taxonomic reassignment of Stutchburia sp. B is accepted, see also the main text. (B) Body size changes at the genus-level. The closely related genera Schizodus and Neoschizodus are connected with a dotted line across the Permian/Triassic boundary. Schiz-Neo = Schizodus and Neoschizodus. Mann-Whitney U-tests of the significance in the body size changes are shown in Table S1 in the Supplement. The legend is shown in Fig. S1.
When the body size changes are broken down into their three different components, i.e., the effects of extinction/extirpation, within-lineage changes, and origination/immigration, four major signals appear: (1) The assemblage body size decreases, by 0.25 log units, which is a consequence of the origination of smaller-sized species (Fig. 3A), leading to a within-genus body size decrease (Fig. 3B); (2) smaller-sized species appear to have been selected against during the extinction event (Fig. 3A); (3) the species that evolved after the extinction show a 0.4 log unit size increase in the late Griesbachian (Fig. 3); and (4) a similar body size increase in the early Spathian is conversely driven by the origination of larger species and the increased size of existing species (Fig. 3A). The largest decline in assemblage body sizes is recorded in the late Spathian, but this can be attributed to a smaller number of body size measurements and because the stratigraphic sequence O3 does not record outer ramp lithofacies that typically has the larger body sizes (see also Foster et al., 2020). When comparing the three components of body size change across the extinction interval (Fig. 4), it turns out that, even though the median size of newly originated taxa is significantly smaller than pre-extinction data, the body sizes are not unusually different, i.e., the maximum and minimum body sizes are relatively similar (Fig. 4). The changes are driven by the increase in the number of smaller-sized species and the abundance of smaller specimens.
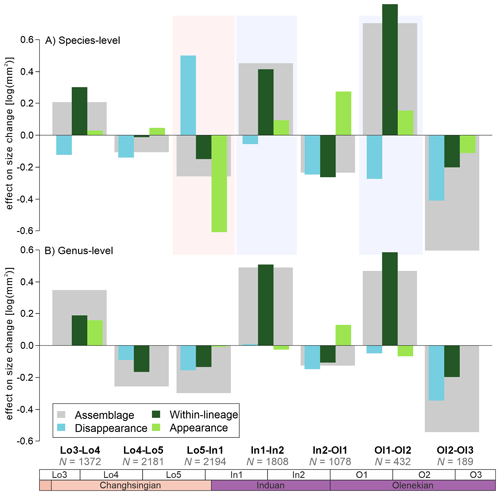
Figure 3The effect on body-size change on bivalve assemblages and the three components of body size change from the Dolomites, Italy. (A) The effects of body size changes at the species-level. (B) The effects of body size change at the genus-level. The number of observations based on each comparison are shown below each comparison. The effects of the Permian-Triassic mass extinction are shown between Lo5-In1.
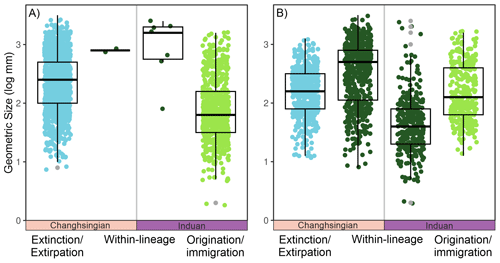
Figure 4The geometric body sizes of bivalves between Lo5 and In1 stratigraphic sequences, i.e., the impacts of the Permian-Triassic hyperthermal event, split into the three components of size changes from the Dolomites, Italy. The jitter plot shows the individual sizes of each measured bivalve specimen, with a box plot overlain. (A) At the species-level and (B) at the genus-level.
A comparison between the approach that the uses the median value for each species to calculate the three different components of body size change with the approach that creates a median value for all of the taxa within a time interval shows mostly similar patterns, with some exceptions, and the median value for the each species approach records fewer extreme fluctuations (Fig. S12). The clear difference in the results of the two different approaches, is that the origination of new species leading to an overall assemblage body size increase in the late Griesbachian and early Spathian is not observed when a median value for all the specimens is used (Fig. S12).
It is also noticeable that within the main genera and families that survived the extinction there are differences in the body size response depending on the ecology of the bivalves. The epifaunal and semi-infaunal species of Eumorphotis and the Bakevellidae (Fig. 5) do not show a major size decrease across the extinction interval. The shallow-infaunal, suspension feeding genera, “Unionites” and a comparison between the closely related Schizodus and Neoschizodus, do, however, show a significant size decrease across the extinction interval. However, no species belonging to “Unionites” nor Neoschizodus occurred both before and after extinction (Fig. 6). Furthermore, the recovery of body size differs between “Unionites” and Neoschizodus, with “Unionites” recovering to pre-extinction body sizes by the late Griesbachian (Fig. 6A), but Neoschizodus not recording pre-extinction Schizodus body sizes until the early Spathian (Fig. 6B). In terms of functional morphology, Neoschizodus and “Unionites” should be ecologically similar, and it may be expected that they would mirror the same trends. The different timing in recovery, however, suggests different ecological niches between the two genera, such as optimum thermal temperatures or temperature range, may explain why “Unionites” typically dominates pre-Spathian communities and Neoschizodus dominates Spathian communities (sensu Hofmann et al., 2015; Foster et al., 2017).
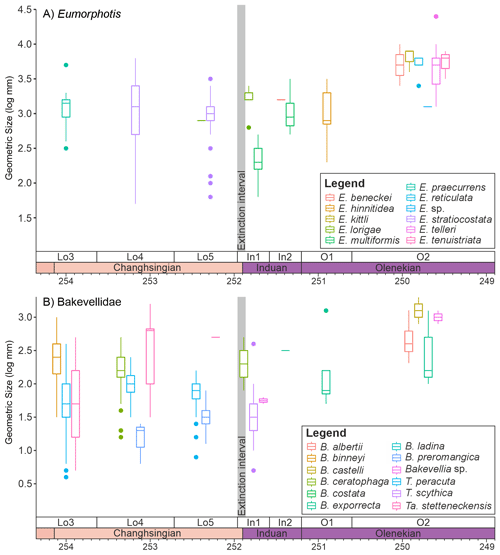
Figure 5Changes in the sizes of epifaunal/semi-infaunal bivalve genera that survive the mass extinction event. (A) Eumorphotis and (B) the Bakevellidae divided into the different species that were sampled from the Dolomites, Italy. The Bakevellidae include the genera Bakevellia (B), Towapteria (T) and Tambanella (Ta).
5.1 Nature of size change
The mechanism of a body size change is a highly contested topic. Twitchett (2007) proposed four models to explain the body size reduction following mass extinction events: (1) selective loss of large taxa, (2) temporary disappearance of large taxa from the fossil record, (3) origination of small taxa, and (4) decrease within taxa. The lack of an observed decline at the species-level, the reduction in the size at the genus-level being driven by the evolution of new species, and the lack of unusually small bivalves during the climate crisis are not consistent with the proposed Lilliput effect in the strict sense (sensu Twitchett, 2007), nor the predictive response of marine ectotherms to climate warming (Pörtner et al., 2023). This then leads to two very different interpretations: at the genus-level there is a clear Lilliput effect, which is consistent with the interpretation of previous studies that were limited to genus-level dynamics (e.g., Twitchett, 2007; Metcalfe et al., 2011), but at the species-level the Lilliput effect is not observed.
The recorded survivors, Bakevellia ceratophaga, Eumorphotis lorigae and potentially Stutchburia tschernyschewi, retained or slightly increased their body sizes. Even though they persist only with low abundances, these species were likely able to cope to the harsh conditions surrounding the Permian/Triassic boundary, possessing physiological traits that allowed persistence without substantial size reduction. For instance, E. lorigae thrived in the extreme conditions of the Bellerophon Formation, which was characterised by high temperatures, low oxygen levels, and variable salinity (Prinoth and Posenato, 2023). It is also striking that the species that survive the extinction are relatively rare in the extinction aftermath, often making it difficult to collect enough specimens for testing the significance of the lack of body size changes.
Our results are also consistent with previous studies that show a body size decrease in many different groups of marine ectotherms, but again this body size decrease is driven by the evolution of newly evolved, smaller-sized species (Song et al., 2011; Chu et al., 2015; Chen et al., 2019; Feng et al., 2020; 2024; Huang et al., 2023b). This trend has also been observed for brachiopods and bivalves from South China during the Permian-Triassic crisis (Chen et al., 2019; Huang et al., 2023b), bivalves at the Triassic-Jurassic climate crisis (Atkinson et al., 2019; Opazo and Twitchett, 2022), belemnites during the Toarcian climate crisis (Rita et al., 2019), molluscs during the end-Cretaceous event (Lockwood, 2005; Jarrett, 2016) and marine organisms during extinction events in general (Monarrez et al., 2020). This suggests a consistent (i.e., predictive) response to a climate crisis is the preferential evolution of small-sized species. One hypothesis is that newly evolved species tend to be smaller (Stanley, 1973), and the vacated ecospace following the mass extinction promoted the evolution of smaller-sized species (Twitchett, 2007). The preferential evolution of smaller-sized species after mass extinctions as a general evolutionary rule is, however, inconsistent in the Dolomites record for two reasons: (1) the newly evolved species become larger later in the Griesbachian, and (2) newly originating species in the Spathian are preferentially large, not small. Explanations for changes in body size include, growth rates (Metcalfe et al., 2011), maturation size, life expectancy and changes in population structure (Daufresne et al., 2009; Rita et al., 2019). The poor quality of the preservation of the bivalves from the Dolomites, means it is not possible to investigate changes in growth rates, ontogenetic allometry or changes in the larval ecology making it difficult to understand which of these factors explains the reduction in size at the genus-level after the mass extinction event. This means it is also difficult to investigate if there is an increase in the proportion of juvenile specimens driving the body size dynamics, although the sizes of the fossils investigated here and the morphology of the measured specimens do not suggest major changes in the abundance of juvenile individuals.
There has been considerable debate about the duration of a body size reduction following the Permian-Triassic climate crisis (Nützel et al., 2010; Brayard et al., 2010, 2015; Fraiser et al., 2011), and even if there is a body size reduction for all marine groups at all (Forel et al., 2015; Nätscher et al., 2023). Our results suggest that there was only a short-duration decrease in body sizes, limited to the early Griesbachian for most genera (Figs. 2 and 5), but body sizes also increased again later in the Spathian. These body size data are, however, consistent with Nützel et al. (2010), who highlighted that whilst generally small, Early Triassic, or even early Griesbachian, molluscs were not unusually small as previously suggested by Twitchett (2001). The increase in the size (so-called Brobdingnag effect; Atkinson et al., 2019) of these newly evolved species during the late Griesbachian suggests that some biotic or abiotic stressor was prevalent during the Permian-Triassic transition. A sudden within-species size increase can only be explained by a shift to more favourable conditions. A within-species size increase is also consistent with bivalves from South China (Huang et al., 2023b) and bivalves from the UK following the end-Triassic mass extinction (Atkinson et al., 2019).
Pre-extinction body size reductions have often been considered a precursor signal of a mass extinction event, and one expectation is that species would have reduced their size prior to their extinction. This has been observed for podocopid ostraocds from Iran (Nätscher et al., 2023) brachiopods from South China (He et al., 2010; Zhang et al., 2016), and at the family-level for ammonoids from Iran (Kiessling et al., 2018) prior to the Permian-Triassic mass extinction. Pre-extinction body size changes have also been recorded at other hyperthermals (e.g., Piazza et al., 2019). Pre-extinction changes in temperature, albeit at a slower rate and smaller magnitude, have also been recorded from South China and Iran (Wu et al., 2023; Gliwa et al., 2022). A decrease in body size prior to extinction may also, therefore, be a predictive response and warning sign for modern-day marine ecosystems. The pre-extinction body size decline is typically only visible at a very high-resolution, and for the Dolomites the lack of bivalves from the uppermost Changhsingian Bulla Member (Bellerophon Fm.), means that an immediate pre-extinction body size decline cannot be tested here. The measurements of E. lorigae from the boundary clay of the Gerennavár Limestone Fm. (Posenato et al., 2005) and the presence of E. lorigae reported from the Werfen Fm. in the Dolomites (Posenato 2009) correspond to the extinction interval. This species, therefore, did not show a body size decline prior to its extinction, as would be expected. Counterintuitively, warm-adapted taxa are also vulnerable to extreme climate warming because they have a narrower thermal tolerance (e.g., Payne and Smith, 2017; Sandoval-Castillo et al., 2020). The increase in body size of E. lorigae prior to its extinction, but during the initial stages of climate warming, likely reflects limited tolerance to extreme temperature fluctuations.
Summarised, our data reject, or at least question, the “Lilliput effect” in the sense of a temporal within-species size reduction during an extinction/hyperthermal event (Urbanek, 1993; Twitchett 2007). Rather, small post-extinction body size of bivalves in the wake of the end-Permian mass extinction in the Dolomites was the effect of the preferential evolution of small-sized species after the crisis. This evolution of small-sized species caused a decline of body size at the genus-level, which has previously been reported but now turns out to be an artefact of the taxonomic level. However, the question remains whether the preferential evolution of small-sized species was controlled by biotic controls, abiotic factors, or a combination of both.
5.2 Abiotic determinants of body size
The aerobic scope and temperature tolerance of species is paramount to an organism's growth and reproduction, with maximum growth at the Toptimum, but superimposed changes in oxygen availability would alter the aerobic scope of ectotherms at the same temperatures (Pörtner et al., 2023). The aerobic scope of marine invertebrates is thought to be mostly governed by temperature and oxygen availability, with each species having different Toptimum, Tpejus and Tcritical temperatures (Penn et al., 2018; Pörtner et al., 2023). There is no robust evidence to suggest that the Dolomites (Italy) experienced anoxic conditions during the Permian-Triassic climate crisis (Brand et al., 2012; Hofmann et al., 2015; Frank et al., 2025), but the shallow marine settings may have experienced slight deoxygenation owing to a reduction in oxygen solubility as a consequence of increased temperatures and increased uptake of oxygen due to the increased metabolic activity of microbes. Increased sea surface temperatures of ∼5–10 °C, based on δ18Ocalcite and clumped isotopes are reported for the Dolomites associated with the extinction event (Kearsey et al., 2009; Brand et al., 2012), and are likely to have had a greater impact on the aerobic scope of marine organisms than the changes in oxygen availability. The problem is that these estimates are only based on a single data point from post-extinction sediments. Despite the rich history of Permian-Triassic investigations of the Dolomites, there is a clear lack of environmental proxy data for making ecological comparisons. When compared to the δ18Ocalcite records from South China, Iran and Pakistan (Sun et al., 2012; Romano et al., 2012; Schobben et al., 2014; Chen et al., 2016; Gliwa et al., 2022), the preferential evolution of smaller-sized species coincides with rapid temperature increases, and the preferential evolution of larger body sizes in the early Spathian coinciding with cooler temperatures. In South China, the body sizes of two foraminifera species also appear to be closely related to the δ18Oapatite record (Foster et al., 2024), suggesting a link between body size and temperature for marine organisms.
An additional response to thermal stress is not just body size changes within a region, but also typically poleward migrations as organisms track their thermal niche during climate warming events (e.g., Daufresne et al., 2009; Yasuhara et al., 2020; Albano et al., 2024). This poleward migration or latitudinal thermal refugia have been recorded as a response to the Permian-Triassic hyperthermal for both terrestrial and marine species (e.g., Bomfleur et al., 2018; Foster et al., 2023b), as well as in response to smaller magnitude events, e.g., Paleocene-Eocene Thermal Maximum (Spiejer et al., 2012). It is also possible that the examples of extinction survivors that record poleward migrations also show a body size response to the climate events. To better understand the body size responses of marine ectotherms to climate warming, high resolution studies are not only required from single regions, but also high-resolution studies between regions.
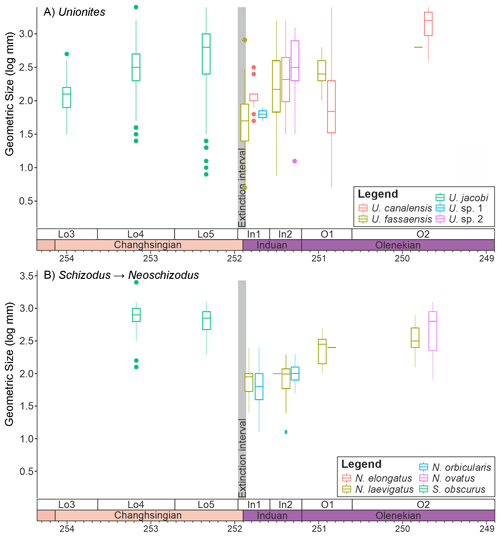
Figure 6Changes in the sizes of infaunal bivalve genera that survive the mass extinction event. (A) ”Unionites” and (B) the closely related genera Schizodus and Neoschizodus, divided into the different species that were sampled from the Dolomites, Italy.
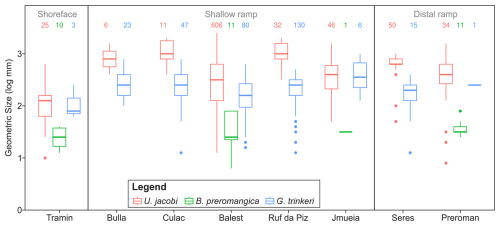
Figure 7Differences in the body sizes of three bivalves (Unionites jacobi, Bakevellia preromangica and Gardenapecten trinkeri) that appear in the Permian (pre-extinction) succession in all major environmental settings. The locations are organized according to their proximity to the coastline at the time of the Permian/Triassic boundary, see also Fig. 1.
Similarly, migrations of marine ectotherms to deeper, cooler waters may also be an expected response that has also been recorded at the Permian/Triassic boundary (Godbold et al., 2017). Water depth has also been shown to affect the size of post-extinction bivalves, with outer ramp lithofacies, typically recording larger body sizes (Foster et al., 2020). When the Permian body sizes are compared between the different localities, it is notable that the species recorded from the nearshore locality of Tramin/Termeno are smaller than the shallow ramp and distal ramp localities (Fig. 7), agreeing with the observations for the Werfen Fm. (Foster et al., 2020), where larger species are found at deeper water depths. It is possible, therefore, that the lack of a significant body size decrease recorded in E. lorigae, B. ceratophaga and Stutchburia tschernyschewi. is because they are all recorded from deeper water settings of the Werfen and Gerennavár Limestone formations. Individual sections also show major lithological changes associated during hyperthermal events and lithology can also be an important factor affecting the body sizes observed from the fossil record (De Baets et al., 2022), but not always (Rita et al., 2019; Nätscher et al., 2023). Similarly, in the Dolomites individual localities show substantial lithological changes associated with the Permian-Triassic transition. However, at the basin-scale of our analysis, bivalves are being recorded in the same lithologies from both the Bellerophon and Werfen formations, from lithofacies representing similar water depths, and with the same style of preservation.
A meta-analysis of what environmental stressors and combination of environmental stressors determine the growth rate of bivalves demonstrates that temperature, oxygen, pH and salinity all play a vital role (Kruft Welton et al., 2024). Given the normal marine setting of the post-extinction ramp and the presence of lithofacies consistent with saturated seawater with respect to carbonate (Foster et al., 2020), it is unlikely pH played a key role in limiting the growth of bivalves. Even though there is geochemical proxy evidence for sustained ocean acidification into the Induan (Jurikova et al., 2020), this record directly conflicts with the widespread deposition of ooids and microbialites that require normal to super-saturated, with respect to calcium carbonate, conditions. In addition, the absence of echinoderms and ammonoids from the Dolomites succession in the Griesbachian (Posenato, 1992; Foster et al., 2017) could be a consequence of the changes in salinity and cannot yet be completely ruled out in the absence of robust proxy data.
Another important factor for determining growth and life expectancy is nutrient availability, which has been hypothesised to be a key factor in shifting benthic communities in other regions (Foster et al., 2024). However, for the Dolomites there is no proxy data; in other regions, changes in primary productivity are highly debated with conflicting hypotheses (Shen et al., 2015; Zhang et al., 2018; Qiu et al., 2019). Regional variability in the degradation products of a lipid biomarkers that becomes abundant after the Permian-Triassic mass extinction and is interpreted to come from phytoplankton would, however, suggest low productivity in the Dolomites (Buchwald et al., 2025). On the other hand, what is clear from the record of the Dolomites is that there is an increased influx of sediment onto the ramp (Algeo and Twitchett, 2010; Brand et al., 2012), low amounts of TOC (Siegert et al., 2011) and the turbid conditions after the onset of extinction, which would have interfered with feeding for the filter-feeding species and availability of detritus for the deposit-feeding species, respectively, both negatively affecting growth. Therefore, whilst many environmental changes are hypothesised to have affected marine ecosystems during the Permian-Triassic climate crisis (Clapham and Renne, 2019), only limited temperature data exist for the Dolomites, and is considered here the most likely abiotic factor to explain the observed body size reduction.
5.3 Biotic determinants of body size
Biotic factors also play an important role in determining body sizes on evolutionary timescales that are superimposed on environmental effects (Nakazawa et al., 2007). Mass extinctions lead to a profound restructuring of marine ecosystems, primarily driven by extreme abiotic stressors. These environmental changes imposed intense selective pressures that eliminated most stenotopic species, while favouring opportunistic, r-strategist taxa characterised by high reproductive rates, short generation times, and broad ecological tolerances (Tong et al., 2007). Consequently, small-bodied bivalves became ecologically dominant due to their rapid growth, early maturity, and morphological plasticity, which enabled them to quickly colonise disturbed environments (Metcalfe et al., 2011).
Although all Early Triassic bivalve species must ultimately be derived from Permian ancestors, not all exhibit straightforward lineage continuity, and the connection to body size evolution is variable. Some Early Triassic repopulating taxa may have evolved from morphologically plastic ancestors that were predisposed to smaller body sizes under environmental stress (Prinoth and Posenato, 2023). For example, Towapteria scythica shows morphological similarity to the Upper Permian T. peracuta, and Eumorphotis lorigae likely represents a direct lineage from pre-existing species that retained or even slightly increased body size after the extinction. In contrast, Claraia, the most abundant and widespread taxon in the Early Triassic, and Neoschizodus, immigrated from the eastern Tethys and have morphologically distinct late Permian precursors (He et al., 2014; Nakazawa and Newell, 1968), making it difficult to assess whether their small size was inherited or evolved during the extinction crisis. Claraia sp. from the Mazzin Member lacks the radial ribs typical of Permian species, although Claraia liuqiaoensis, a smooth-shelled species, already occurred in the latest Permian and extended into the Griesbachian (He et al., 2014; Foster et al., 2019). Neoschizodus orbicularis and “Unionites” fassaensis also show substantial morphological divergence from their Permian counterparts. This morphological divergence could indicate either (1) the emergence of entirely new taxa adapted to Early Triassic environmental conditions, or (2) ecological and phenotypic plasticity within surviving lineages that enabled rapid morphological shifts in response to post-extinction stressors. Distinguishing between these scenarios requires further phylogenetic analysis and detailed stratigraphic correlation to assess lineage continuity. This variability indicates that size reduction may have resulted from a combination of inherited traits and evolutionary responses to post-extinction conditions of the abiotic and biotic environment.
One key biotic factor for body size reduction may have been the breakdown and simplification of trophic structures (Huang et al., 2023a). The collapse of complex ecosystems with specialised predators and prey, including durophagous gastropods, crustaceans, and fish, led to ecological release and the temporary dominance of low-level consumers and generalists (Schubert and Bottjer, 1995; McRoberts, 2001; Fraiser and Bottjer, 2007). In the absence of strong predatory pressure, smaller and thin-shelled bivalves could proliferate without investing in energetically costly defensive traits. This relaxation of size-selective predation, together with the general collapse of food webs (Chen and Benton, 2012), created conditions in which small size was selectively advantageous. However, the shift in functional traits to thicker shells and increased body size in the late Griesbachian and into the Spathian, could indicate that selective pressures gradually shifted, as predator guilds began to recover (Scheyer et al., 2014; Karapunar et al., 2025b), driving within-lineage size increases in some taxa.
Furthermore, this recovery interval was likely characterised by what Hautmann et al. (2015) described as “competition in slow motion”: a state of ecological simplicity in which interactions were weakly differentiated and resource partitioning was minimal. Communities were dominated by generalists with overlapping niches, and the dominant form of competition may have been intraspecific or density-dependent rather than shaped by complex interspecific interactions (Foster et al., 2019). In some Early Triassic assemblages of the Mazzin Member, a relatively high genus diversity suggests that multiple opportunistic taxa coexisted in crowded populations, where smaller body sizes may have conferred an energetic advantage through more efficient resource use (Hofmann et al., 2015). However, the dominance of certain highly dispersive generalists such as Claraia may have also suppressed ecological differentiation and delayed the establishment of larger-bodied species. This reflects a form of biotic monopolization, where early colonisers may create priority effects that inhibit niche access for later-arriving taxa, even in the absence of direct competition. This dynamic could have added inertia to the recovery process and reinforced the trend toward small body sizes by precluding competitive escalation (Hautmann et al., 2015), effectively layering on a form of trophic suppression.
Shallow marine bivalve communities from the Dolomites in Italy show a significant reduction in body-size at the genus-level, but conversely at the species-level body sizes stay the same or slightly increase, which is inconsistent with the Lilliput effect hypothesis. The reduction of bivalve body-size assemblages, including at the genus-level, is driven by the preferential origination and dominance of small-sized species. However, these new species are not unusually small, as has been previously suggested. Two of the species surviving the extinction that do not show a body size decrease, were already adapted to stressful conditions prior to the climate crisis. Due to the lack of proxy data available from the Permian-Triassic succession of the Dolomites, it is difficult to understand and distinguish the role of specific abiotic factors in causing the observed body size dynamics, but an effect of high seawater temperature and nutrient availability appears likely. The impact of the mass extinction event on biotic interactions also likely played a major role in the preferential origination of small-sized species, and these biotic impacts are superimposed on the abiotic controls during the Early Triassic.
All statistical analyses were made in R (R Core Team, 2019) and the code and data are available at https://doi.org/10.5281/zenodo.18380698 (Foster, 2026).
The supplement related to this article is available online at https://doi.org/10.5194/bg-23-1181-2026-supplement.
WJF, HP and EK conceptualised the project. WJF, HP and MH collected the data. WJF carried out the analysis and data visualisation. All authors contributed to writing the manuscript draft.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
We would like to express our sincerest gratitude to the Nature Park Office in South Tyrol for permission to collect the fossils and work on the outcrops in the Dolomites, Italy. WJF would also like to thank (in no specific order) Richard J. Twitchett, Stella Z. Buchwald, Autumn Pugh, Jana Gliwa, Dieter Korn, Mónica Alejandra Gómez Correa, Francesca Galasso, Baran Karapunar, Alberto Riva and Anja B. Frank for enjoyable field excursions to the Dolomites, which indirectly supported this research. Finally, we would like to thank Renato Posenato, Kenneth de Baets and the handling editor Niels de Winter for their constructive reviews of the manuscript.
This research has been supported by the Deutsche Forschungsgemeinschaft (grant no. FO 1297/1).
This paper was edited by Niels de Winter and reviewed by Kenneth De Baets and Renato Posenato.
Albano, P. G., Schultz, L., Wessely, J., Taviani, M., Dullinger, S., and Danise, S.: The dawn of the tropical Atlantic invasion into the Mediterranean Sea, P. Natl. Acad. Sci. USA, 121, e2320687121, https://doi.org/10.1073/pnas.2320687121, 2024.
Algeo, T. J. and Twitchett, R. J.: Anomalous Early Triassic sediment fluxes due to elevated weathering rates and their biological consequences, Geology, 38, 1023–1026, 2010.
Atkinson, J. W., Wignall, P. B., Morton, J. D., and Aze, T.: Body size changes in bivalves of the family Limidae in the aftermath of the end-Triassic mass extinction: the Brobdingnag effect, Palaeontology, 62, 561–582, 2019.
Bomfleur, B., Blomenkemper, P., Kerp, H., and McLoughlin, S.: Polar regions of the Mesozoice-Paleogene Greenhouse World as Refugia for Relict Plant Groups, in: Transformative Paleobotany, edited by: Krings, M., Harper, C. J., Rubén Cúneo, N., and Rothwell, G. W., 593–611, ISBN: 978-0-12-813012-4, 2018.
Bond, D. P. G., Wignall, P. B., Joachimski, M. M., Sun, Y., Savov, I., Grasby, S. E., Beauchamp, B., and Blomeier, D. P. G.: An abrupt extinction in the Middle Permian (Capitanian) of the Boreal Realm (Spitsbergen) and its link to anoxia and acidification, GSA Bulletin, 127, 1411–1421, 2015.
Bosellini, A., Gianolla, P., and Stefani, M.: Geology of the Dolomites, Episodes, 26, 181–185, 2003.
Brand, U., Posenato, R., Came, R., Affek, H., Angiolini L., Azmy, K., and Farabegoli, E.: The end-Permian mass extinction: A rapid volcanic CO2 and CH4-climatic catastrophe, Chemical Geology, 322–323, 121–144, 2012.
Brandner, R., Horacek, M., Keim, L., and Scholger, R.: The Pufels/Bulla road section: deciphering environmental changes across the Permian-Triassic boundary to the Olenekian by integrated litho-, magneto-and isotope stratigraphy. A field trip guide, Geo. Alp, 6, 116–132, 2009.
Brayard, A., Nützel, A., Stephem, D. A., Bylund, K. G., Jenks, J., and Bucher, H.: Gastropod evidence against the Early Triassic Lilliput effect, Geology, 38, 147–150, 2010.
Brayard, A., Meier, M., Escarguel, G., Fara, E., Nützel, A., Olivier, N., Bylund, K., Jenks, J., Stephen, D., Hautmann, M., Vennin, E., and Bucher, H.: Early Triassic Gulliver gastropods: spatio-temporal distribution and significance for the biotic recovery after the end-Permian mass extinction, Earth-Science Reviews, 146, 31–64, 2015.
Broglio Loriga, C. and Mirabella, S.: Il genere Eumorphotis Bittner 1901 nella biostratigrafia dello Scitico, Formazione di Werfen (Dolomiti), Memorie di Scienze Geologiche 38, 245–281, 1986.
Broglio Loriga, C., Masetti, D., and Neri, C.: La Formazione di Werfen (Scitico) delle Dolomiti occidentali. Rivista Italiana di Paleontologia e Stratigrafia, 88, 501–598, 1983.
Broglio Loriga, C., Neri, C., and Posenato R.: The early Macrofaunas of the Werfen Formation and the Permian-Triassic boundary in the Dolomites (Southern Alps, Italy), Studi Trentini di Scienze Naturali, 62, 3–18, 1986.
Buchwald, S. Z., Birgel, D., Senger, K., Mosociova, T., Pei, Y., Frank, A. B., Galasso, F., Gómez Correa, M. A., Koşun, E., Karapunar, B., Wang, X., Kustatscher, E., Prinoth, H., Steinkrauss, R., Peckmann, J., and Foster, W. J.: Primary productivity blooms on the Barents Shelf, Svalbard, associated with the Permian–Triassic mass extinction, ESS Open Archive [preprint], https://doi.org/10.22541/essoar.174558961.14311943/v1, 2025.
Chen, Z.-Q. and Benton, M. J.: The timing and pattern of biotic recovery following the end-Permian mass extinction, Nature Geoscience, 5, 375–383, 2012.
Chen, J., Shen, S.-Z., Li, X.-H., Xu, Y.-G., Joachimski, M. M., Bowring, S. A., Erwin, D. H., Yuan, D.-X., Chen, B., Zhang, H., and Wang, Y.: High-resolution SIMS oxygen isotope analysis on conodont apatite from South China and implications for the end-Permian mass extinction, Palaeogeogr. Palaeocl., 448, 26–38, 2016.
Chen, J., Song, H., He, W., Tong, J., Wang, F., and Wu, S.: Size variation of brachiopods from the Late Permian through the Middle Triassic in South China: evidence for the Lilliput effect following the Permian-Triassic extinction, Palaeogeogr. Palaeocl., 519, 248–257, 2019.
Chu, D., Tong, J., Song, H., Benton, M. J., Song, H., Yu, J., Qiu, X., Huang, Y., and Tian, L.: Lilliput effect in freshwater ostracods during the Permian–Triassic extinction, Palaeogeogr. Palaeocl., 435, 38–52, 2015.
Clapham, M. E. and Renne P. R.: Flood basalts and mass extinctions, Annual Review of Earth and Planetary Sciences, 47, 275–303, 2019.
Daufresne, M., Lengfellner, K., and Sommer, U.: Global warming benefits the small in aquatic ecosystems, P. Natl. Acad. Sci. USA, 106, 12788–12793, 2009.
De Baets, K., Nätscher, P. S., Rita, P., Fara, E., Neige, P., Bardin, J., Dera, G., Duarte, l. V., Hughes, Z., Laschinger, P., García-Ramos, J. C., Piñuela, L., Übelacker, C., and Weis, R.: The impact of the Pliensbachian–Toarcian crisis on belemnite assemblages and size distribution, Swiss Journal of Palaeontology, 140, 25, https://doi.org/10.1186/s13358-021-00242-y, 2021.
De Baets, K., Jarochowska, E., Buchwald, S. Z., Klug, C., and Korn, D.: Lithology controls ammonoid size distributions, PALAIOS, 37, 1–11, 2022.
Dercourt, J., Gaetani, M., Vrielynck, B., Barrier, E., Biju-Duval, B., Bru-net, M. F., Cadet, J. P., Crasquin, S., and Sandulescu, M.: Atlas Peri-Tethys, Palaeogeographical Maps, Maps and explanatory notes, 1–269, 2000.
Farabegoli, E. and Perri, M. C.: Millennial physical events and the end-Permian mass mortality in the western Palaeotethys: Timing and primary causes, in: Earth and Life: Global Biodiversity, Extinction Intervals and Biogeographic Perturbations Through Time, Springer Netherlands, Dordrecht, 719–758, https://doi.org/10.1007/978-90-481-3428-1_24, 2012.
Farabegoli, E., Perri, M., and Posenato, R.: Environmental and biotic changes across the Permian–Triassic boundary in western Tethys: the Bulla parastratotype, Italy, Glob. Planet. Chang., 55, 109–135, 2007.
Feng, Y., Song, H., and Bond, D. P. G.: Size variations in foraminifers from the early Permian to the Late Triassic: implications for the Guadalupian–Lopingian and the Permian–Triassic mass extinctions, Paleobiology, 46, 511–532, 2020.
Forel, M.-B., Crasquin, S., Chitnarin, A., Angiolini, L., and Gaetani, M.: Precocious sexual dimorphism and the Lilliput effect in Neo-Tethyan Ostracoda (Crustacea) through the Permian-Triassic boundary, Palaeontology, 58, 409–454, 2015.
Foster, W.: An unpredictable body size response to the Permo-Triassic climate crisis, Zenodo [code and data set], https://doi.org/10.5281/zenodo.18380698, 2026.
Foster, W. J., Danise, S., Price, G. D., and Twitchett, R. J.: Subsequent biotic crises delayed marine recovery following the late Permian mass extinction event in northern Italy, PLoS One, 12, e0172321, https://doi.org/10.1371/journal.pone.0172321, 2017.
Foster, W. J., Lehrmann, D. J., Hirtz, J. A., White, M., Yu, M., Li, J., and Martindale, R. C.: Early Triassic benthic invertebrates from the Great Bank of Guizhou, South China: systematic palaeontology and palaeobiology, Papers in Palaeontology 5, 613–656, 2019.
Foster, W. J., Gliwa, J., Lembke, C., Pugh, A., Hofmann, R., Tietje, M., Varela, S., Korn, D., and Aberhan, M.: Evolutionary and ecophenotypic controls on bivalve body size distributions following the end-Permian mass extinction, Global and Planetary Change, 185, 103088, https://doi.org/10.1016/j.gloplacha.2019.103088, 2020.
Foster, W. J., Allen, B. J., Kitzmann, N. H., Münchmeyer, J., Rettelbach, T., Witts, J. D., Whittle, R. J., Larina, E., Clapham, M. E., and Dunhill, A. M.: How predictable are mass extinction events?, Royal Society Open Science, 10, 221507, https://doi.org/10.1098/rsos.221507, 2023a.
Foster, W. J., Asatryan, G., Rauzi, S., Botting, J. P., Buchwald. S. Z., Lazarus, D. B., Isson, T., Renaudie, J., and Kiessling, W.: Response of siliceous marine organisms to the Permian-Triassic climate crisis based on new findings from central Spitsbergen, Svalbard, Paleoceanography and Paleoclimatology, 38, e2023PA004766, https://doi.org/10.1029/2023PA004766, 2023b.
Foster, W. J., Frank, A. B., Li, Q., Danise, S., Wang, X., and Peckmann, J.: Thermal and nutrient stress drove Permian–Triassic shallow marine extinctions, Cambridge Prisms: Extinction, 2, e9, https://doi.org/10.1017/ext.2024.9, 2024.
Fraiser, M. L. and Bottjer, D. J.: When bivalves took over the world, Paleobiology, 33, 397–413, 2007.
Fraiser, M. L., Twitchett, R. J., Frederickson, J. A., Metcalfe, B., and Bottjer, D. J.: Gastropod evidence against the Early Triassic Lilliput effect: comment, Geology 39, e232–e232, 2011.
Frank, A. B., Warncke-Rüting, E., Grasby, S. E., Kustatscher, E., Prinoth, H., Poulton, S. W., Xiong, Y., Korn, D., Gliwa, J., and Foster, W. J.: Oxic conditions in shallow marine settings during the Permian-Triassic Mass Extinction, Earth ArXiv [preprint], https://doi.org/10.31223/X5K135, 2025.
Gliwa, J., Wiedenbeck, M., Schobben, M., Ullmann, C. V., Kiessling, W., Ghaderi, A., Struck, U., and Korn, D.: Gradual warming prior to the end-Permian mass extinction, Palaeontology, 65, e12621, https://doi.org/10.1111/pala.12621, 2022.
Godbold, A., Schoepfer, S., Shen, S.-Z., and Henderson, C. H.: Precarious ephemeral refugia during the earliest Triassic, Geology, 45, 607–610, 2017.
Gómez Correa, M. A., Frank, A., Wiedenbeck, M., Gliwa, J., Korn, D., Prinoth, H., Kustatscher, E., and Foster, W. J.: Insights into thermal stress during the Permian-Triassic mass extinction using ostracods from the Southern Alps (Italy), EGU General Assembly 2025, Vienna, Austria, 27 Apr–2 May 2025, EGU25-13354, https://doi.org/10.5194/egusphere-egu25-13354, 2025.
Groves, J. R., Rettori, R., Payne, J. L., Boyce, M. D., and Altiner, D.: End-Permian mass extinction of lagenide foraminifers in the southern Alps (northern Italy), Journal of Paleontology, 81, 415–434, 2007.
Hautmann, M., Bagherpour, B., Brosee, M., Frisk, Å., Hofmann, R., Baud, A., Nützel, A., Goudemand, N., and Bucher, H.: Competition in slow motion: the unusual case of benthic marine communities in the wake of the end-Permian mass extinction, Palaeontology, 58, 871–901, 2015.
He, W., Twitchett, R. J., Zhang, Y., Shi, G. R., Feng, Q.-L., Yu, J.-X., Wu, S.-B., and Peng, X.-F.: Controls on body size during the Late Permian mass extinction event, Geobiology, 8, 391–402, 2010.
He, W., Zhang, K., Yang, T., and Wu, S.: Morphological evolution of Claraia species from the Late Permian (Changhsingian) to the Early Triassic (Induan) and the response to the Permian–Triassic stressed environment, in: STRATI 2013, edited by: Rocha, R., Pais, J., Kullberg, J., and Finney, S., Springer Geology, 1159–1163, https://doi.org/10.1007/978-3-319-04364-7_197, 2014.
Hofmann, R., Hautmann, M., and Bucher, H.: Recovery dynamics of benthic marine communities from the Lower Triassic Werfen Formation, northern Italy, Lethaia, 48, 474–496, 2015.
Holser, W. T., Schönlaub, H. P., Boeckelmann, K., and Magaritz, M.: The Permian-Triassic of the Gartnerköfel-1 core (Carnic Alps, Austria): Synthesis and conclusions, Abh. Geol. B.-A., 45, 213–232, 1991.
Horacek, M., Brandner, R., and Abart, R.: Carbon isotope record of the P/T boundary and the Lower Triassic in the Southern Alps: Evidence for rapid changes in storage of organic carbon, Palaeogeogr. Palaeocl., 252, 347–354, 2007.
Huang, Y., Chen, Z.-Q., Roopnarine, P., Benton, M. J., Zhao, L., Feng, X., and Li, Z.: The stability and collapse of marine ecosystems during the Permian-Triassic mass extinction, Current Biology, 33, 1002–1011, 2023a.
Huang, Y., Tong, J., Tian, L., Song, H., Chu, D., Miao, X., and Song, T.: Temporal shell-size variations of bivalves in South China from the Late Permian to the Early Middle Triassic, Palaeogeogr. Palaeocl., 609, 111307, https://doi.org/10.1016/j.palaeo.2022.111307, 2023b.
Isson, T. T., Planavsky, N. J., Coogan, L. A., Stewart, E. M., Ague, J. J., Bolton, E. W., Zhang, S., McKenzie, N. R., and Kump, L. R.: Evolution of the Global Carbon Cycle and Climate Regulation on Earth, Global Biogeochemical Cycles, 34, e2018GB006061, https://doi.org/10.1029/2018GB006061, 2020.
Jarrett, M.: Lilliput Effect Dynamics across the Cretaceous-Paleogene Mass Extinction: Approaches, Prevalence, and Mechanisms, PhD thesis, University of South Florida, https://www.proquest.com/openview/ca73dfd305bb907ecb4992e89406ac9c/1?pq-origsite=gscholar&cbl=18750&casa_token=UDGPOXZBHnUAAAAA:CaDD6O3uxwMAZw1at-eNJTaKMPsXaKzCw5ox6lzbSESnj_Jw7TR-pz-RR8CBc_VLZG04KsdO4YNb (last access: 4 February 2026), 2016.
Jurikova, H., Gutjahr, M., Wallmann, K., Flögel, S., Liebetrau, V., Posenato, R., Angiolini, L., Garbelli, C., Brand, U., Wiedenbeck, M., and Eisenhauer, A.: Permian–Triassic mass extinction pulses driven by major marine carbon cycle perturbations, Nature Geoscience, 13, 745–750, 2020.
Karapunar, B., Wang, X., Frank, A. B., Gürsoy, M., Buchwald, S. Z., Gómez Correa, M. A., Liu, Z., Xu, X., Meng, L., Demir, D., Koşun, E., and Foster, W.: Environmental and ecological changes across the Permian–Triassic transition in Türkiye: integratingvirtual outcrop models and new fieldwork data, EarthArXiv [preprint], https://doi.org/10.31223/X5KQ9B, 2025a.
Karapunar, B., Wolniewicz, A. S., Romano, C., Osvárt, P., Rochín-Bañaga, H., Kustatscher, E., Buchwald, S. Z., Galasso, F., Davis, D., López-Arbarello, A., Prinoth, H., Bernardi, M., and Foster, W. J.: New insights into the extinction and recovery of marine vertebrates across the Permian-Triassic mass extinction event in the Dolomites, Southern Alps, Italy, BioRxiv [preprint], https://doi.org/10.1101/2025.08.23.671916, 2025b.
Kearsey, T., Twitchett, R. J., Price, G. D., and Grimes, S. T.: Isotope excursions and palaeotemperature estimates from the Permian/Triassic boundary in the Southern Alps (Italy), PPalaeogeogr. Palaeocl., 279, 29–40, 2009.
Kiessling, W., Schobben, M., Ghaderi, A., Hairapetian, V., Leda, L., and Korn, D.: Pre-mass extinction decline of latest Permian ammonoids, Geology, 46, 283–286, 2018.
Kocsis, A. T., Reddin, C. J., Aloy, J., and Kiessling, W.: The r package divDyn for quantifying diversity dynamics using fossil sampling data, Methods in Ecology and Evolution, 10, 765–746, 2018.
Kovács, S., Sudar, M., Grădinaru, E., Gawlick, H.-J., Karamata, S., Haas, J., Péró, C., Gaetani, M., Mell, J., Polák, M., Aljinović, D., Ogorelec, B., Kolar-Jurkovšek, T., Jurkovšek, B., and Buser, S.: Triassic Evolution of the Tectonostratigraphic Units of the Circum-Pannonian Region, Jb. Geol. B.-A., 151, 199–280, 2011.
Kruft Welton, R. A., Hoppit, G., Schmidt, D. N., Witts, J. D., and Moon, B. C.: Reviews and syntheses: The clam before the storm – a meta-analysis showing the effect of combined climate change stressors on bivalves, Biogeosciences, 21, 223–239, https://doi.org/10.5194/bg-21-223-2024, 2024.
Kump, L. R., Bralower T. J., and Ridgwell, A.: Ocean acidification in deep time, Oceanography, 22, 94–107, 2009.
Kustatscher, E., Novack, H., Bernardi, M., Krainer, K., Petti, F. M., Prinoth, H., Roghi, G., Preto, N., Tomasoni, R., Rigo, M., and Spötl, C.: The Permian-Triassic boundary and the end-Permian mass extinction in the southern and eastern Alps, Geo. Alp, 16, 73–76, 2019.
Lockwood, R.: Body size, extinction events, and the early Cenozoic record of veneroid bivalves: a new role for recoveries?, Paleobiology, 31, 578–590, 2005.
McRoberts, C. A.: Triassic bivalves and the initial marine Mesozoic revolution: a role for predators?, Geology, 29, 359–362, 2001.
Metcalfe, B., Twitchett, R. J., and Price-Lloyd, N.: Changes in size and growth rate of `Lilliput' animals in the earliest Triassic, Palaeogeogr. Palaeocl., 308, 171–180, 2011.
Mette, W. and Roozbahani, P.: Late Permian (Changsingian) ostracods of the Bellerophon Formation at Seis (Siusi) (Dolomites, Italy), Journal of Micropalaeontology, 31, 73–87, https://doi.org/10.1144/0262-821X11-010, 2012.
Monarrez, P. B., Heim, N. A., and Payne, J. L.: Mass extinctions alter extinction and origination dynamics with respect to body size, Proc. R. Soc. B, 288, 20211681, https://doi.org/10.1098/rspb.2021.1681, 2020.
Morten, S. D. and Twitchett, R. J.: Fluctuations in the body size of marine invertebrates through the Pliensbachian–Toarcian extinction event, Palaeogeogr. Palaeocl., 284, 29–38, 2009.
Muttoni, G., Gaetani, M., Kent, D. V., Sciunnach, D., Angiolini, L., Berra, F., Garzanti, E., Mattei, M., and Zanchi, A.: Opening of the Neo-Tethys Ocean and the Pangea B to Pangea A transformation during the Permian, GeoArabia, 14, 17–48, 2009.
Nakazawa, K.: Permian bivalves from West Spitsbergen, Svalbard islands, Norway, Paleontological Research, 3, 1–17, 1999.
Nakazawa, K. and Newell, N. D.: Permian bivalves of Japan, Memoirs of the Faculty of Science, Kyoto University, Series of Geology and Mineralogy, 35, 1–108, 1968.
Nakazawa, T., Ishida, N., Kato, M., and Yannamura, N.: Larger body size with higher predation rate, Ecology of Freshwater Fish, 16, 362–372, 2007.
Nätscher, P., Dera, G., Reddin, C. J., Rita, P., and De Baets, K.: Morphological response accompanying size reduction of belemnites during an Early Jurassic hyperthermal event modulated by life history, Scientific Reports, 11, 14480, https://doi.org/10.1038/s41598-021-93850-0, 2021.
Nätscher, P., Gliwa, J., De Baets, K., Ghaderi, A., and Korn, D.: Exceptions to the temperature–size rule: no Lilliput Effect in end-Permian ostracods (Crustacea) from Aras Valley (northwest Iran), Paleontology, 66, e12667, https://doi.org/10.1111/pala.12667, 2023.
Nätscher, P., De Baets, K., and Kiessling, W.: Unique fingerprint of marine ectotherm body size change during hyperthermal crises, biorXiv, https://doi.org/10.1101/2025.07.25.666730, 2025.
Neri, C. and Posenato, R.: New biostratigraphical data on uppermost Werfen Formation of western Dolomites (Trento, Italy), Geol. Paläont. Mitt. Innsbruck, 14, 83–107, 1985.
Newell, N. D. and Boyd, D. W.: Parallel evolution in early Trigoniacean bivalves, Bulletin of the American Museum of Natural History, 154, 57–158, 1975.
Nützel, A., Mannani, M., Senowbari-Daryan, B., and Yazdi, M.: Gastropods from the Late Triassic Nayband Formation (Iran), their relationships to other Tethyan faunas and remarks on the Triassic gastropod body size problem, N. Jb. Geol. Paläont. (Abh.), 256, 213–228, 2010.
Opazo, L. F. and Twitchett R. J.: Bivalve body-size distribution through the Late Triassic mass extinction event, Paleobiology, 48, 420–445, 2022.
Payne, J. L., Bush, A. M., Heim, N. A., Knope, M. L., and McCauley, D. J.: Ecological selectivity of the emerging mass extinction in the oceans, Science, 353, 1284–1288, 2016.
Payne, N. L. and Smith, J. A.: An alternative explanation for global trends in thermal tolerance, Ecology Letters, 20, 70–77, 2017.
Penn, J. L., Deutsch C., Payne J. J., and Sperling E. D.: Temperature-dependent hypoxia explains biogeography and severity of end-Permian marine mass extinction, Science, 362, 1130, https://doi.org/10.1126/science.aat1327, 2018.
Pérez-Valera, F., Pérez-López, A., Götz, A. E., Ros-Franch, S., Márquez-Aliaga, A., Baeza-Carratalá, J. F., and Pérez-Valera, J. A.: First record of Anisian deposits in the Betic External Zone of southern Spain and its paleogeographical implications, Sedimentary Geology, 449, 106374, https://doi.org/10.1016/j.sedgeo.2023.106374, 2023.
Piazza, V., Duarte, l. V., Renaudie, J., and Aberhan, M.: Reductions in body size of benthic macroinvertebrates as a precursor of the early Toarcian (Early Jurassic) extinction event in the Lusitanian Basin, Portugal, Paleobiology, 45, 296–316, 2019.
Piazza, V., Ullmann, C. V., and Aberhan, M.: Temperature-related body size change of marine benthic macroinvertebrates across the Early Toarcian Anoxic Event, Scientific Reports, 10, 4675, https://doi.org/10.1038/s41598-020-61393-5, 2020.
Pörtner, H.-O., Scholes, R. J., Arneth, A., Barnes, D. K. A., Burrows, M. T., Diamond, S. E., Duarte, C. M., Kiessling, W., Leadley, P., Managi, S., and McElwee, P.: Overcoming the coupled climate and biodiversity crises and their societal impacts, Science, 380, eabl4881, https://doi.org/10.1126/science.abl4881, 2023.
Posenato, R.: Tirolites (Ammonoidea) from the Dolomites, Bakony and Dalmatia: taxonomy and biostratigraphy, Eclogae Geol. Helv., 85, 893–929, 1992.
Posenato, R.: Global correlations of mid Early Triassic events: the Induan/Olenekian boundary in the Dolomites (Italy), Earth Sci. Rev., 91, 93–105, 2008.
Posenato, R.: Survival patterns of macrobenthic marine assemblages during the end-Permian mass extinction in the western Tethys (Dolomites, Italy), Palaeogeogr. Palaeocl., 280, 150–167, 2009.
Posenato, R.: The end-Permian mass extinction (EPME) and the Early Triassic biotic recovery in the western Dolomites (Italy): state of the art, Bollettino della Società Paleontologica Italiana, 58, 11–34, 2019.
Posenato, R., Pelikan, P., and Hips, K.: Bivalves and brachiopods near the Permian-Triassic boundary from Bükk Mountains (Balvany-North section, northern Hungary), Revista Italiana di Paleontologia e Stratigrafia, 111, 215–232, 2005.
Prinoth, H. and Posenato, R.: Bivalves from the Changhsingian (upper Permian) Bellerophon Formation of the Dolomites (Italy): ancestors of Lower Triassic post-extinction benthic communities, Papers in Palaeontology, 9, e1486, https://doi.org/10.1002/spp2.1486, 2023.
Qiu, Z., Song, H., Hu, C., Wignall, P. B., and Song, H.: Carbonate thermoluminescence and its implication for marine productivity change during the Permian–Triassic transition, Palaeogeogr. Palaeocl., 526, 72–79, 2019.
Rauzi, S., Foster, W. J., Takahashi, S., Hori, R. S., Beaty, B. J., Tarhan, L. G., and Isson, T. T.: Lithium isotopic evidence for enhanced reverse weathering during the Early Triassic warm period, P. Natl. Acad. Sci. USA, 121, e2318860121, https://doi.org/10.1073/pnas.2318860121, 2024.
R Core Team: R: A Language and Environment for Statistical Computing, R Foundation for Statistical Computing [data set], Vienna, Austria, https://www.R-project.org/ (last access: 4 February 2026), 2019.
Rego, B. L., Wang, S. C., Altiner, D., and Payne, J. L.: Within- and among-genus components of size evolution during mass extinction, recovery, and background intervals: a case study of Late Permian through Late Triassic foraminifera, Paleobiology, 38, 627–643, 2012.
Rita, P., Nätscher P., Duarte, L. V., Weis, R., and De Baets, K.: Mechanisms and drivers of belemnite body-size dynamics across the Pliensbachian–Toarcian crisis, Royal Society Open Science, 6, 190494, https://doi.org/10.1098/rsos.190494, 2019.
Romano, C., Goudemand, N., Vennemann, T. W., Ware, D., Schneebeli-Hermann, E., Hochuli, P. A., Brühwiler, T., Brinkmann, W., and Bucher, H.: Climatic and biotic upheavals following the end-Permian mass extinction, Nat. Geosci., 6, 57–60, 2012.
Sandoval-Castillo, J., Gates, K., Brauer, C. J., Smith, S., Bernatchez, L., and Beheregaray, B.: Adaptation of plasticity to projected maximum temperatures and across climatically defined bioregions, P. Natl. Acad. Sci. USA, 117, 17112–17121, 2020.
Scheyer, T. M., Romano, C., Jenks, J., and Bucher, H.: Early Triassic marine biotic recovery: the predators' perspective, PLoS One, 9, e88987, https://doi.org/10.1371/journal.pone.0088987, 2014.
Schobben, M., Joachimski, M. M., Korn, D., Leda, L., and Korte, C.: Palaeotethys seawater temperature rise and an intensified hydrological cycle following the end-Permian mass extinction, Gondwana Res., 26, 675–683, 2014.
Schubert, J. K. and Bottjer, D. J.: Aftermath of the Permian-Triassic mass extinction event: Paleoecology of lower Triassic carbonates in the western USA, Palaeogeogr. Palaeocl., 116, 1–39, 1995.
Shen, J., Schoepfer, S. D., Feng, Q., Zhou, L., Yu, J., Song, H., Wie, H., and Algeo, T. J.: Marine productivity changes during the end-Permian crisis and Early Triassic recovery, Earth-Science Reviews, 149, 136–162, 2015.
Siegert, S., Kraus, S. H., Mette, W., Struck, U., and Korte, C.: Organic carbon isotope values from the Late Permian Seis/Siusi succession (Dolomites, Italy): Implications for palaeoenvironmental changes, Foss. Rec., 14, 207–217, https://doi.org/10.1002/mmng.201100008, 2011.
Song, H., Tong, J., and Chen, Z.-Q.: Evolutionary dynamics of the Permian–Triassic foraminifer size: evidence for Lilliput effect in the end-Permian mass extinction and its aftermath, Palaeogeogr. Palaeocl., 308, 98–110, 2011.
Spiejer, R., Schneibner, C., Stassen, P., and Morsi, A. M. M.: Response of marine ecosystems to deep-time global warming: a synthesis of biotic patterns across the Paleocene-Eocene thermal maximum (PETM), Austrian Journal of Earth Sciences, 105, 6–16, 2012.
Stanley, S. M.: An explanation for Cope's rule, Evolution, International Journal of Organic Evolution, 27, 1–26, 1973.
Stefani, M., Brack, P., Gianolla, P., Keim, L., Mauer, F., Neri, C., Preto, N., Riva, A., Roghi, G., and Russo, F.: Triassic carbonate platforms of the Dolomites: Carbonate production, relative sea-level fluctuations and the shaping of the depositional architecture, in: Field Trip Guide Book, 1–44, 2004.
Sun, Y., Joachimski, M. M., Wignall, P. B., Yan, C., Chen, Y., Jiang. H., Wang, L., and Lai, X.: Lethally Hot Temperatures During the Early Triassic Greenhouse, Science, 338, 366–370, 2012.
Tong, J., Zhang, S., Zuo, J., and Xiong, X.: Events during Early Triassic recovery from the end-Permian extinction, Global and Planetary Change, 55, 66–80, 2007.
Twitchett, R. J.: Incompleteness of the Permian-Triassic fossil record: a consequence of productivity decline?, Geol. J., 36, 341–353, 2001.
Twitchett, R. J.: The Lilliput effect in the aftermath of the end-Permian extinction event, Palaeogeogr. Palaeocl., 252, 132–144, 2007.
Twitchett, R. J. and Wignall, P. B.: Trace fossils and the aftermath of the Permo-Triassic mass extinction: evidence from northern Italy, Palaeogeogr. Palaeocl., 24, 137–151, 1996.
Urbanek, A.: Biotic crises in the history of Upper Silurian graptoloids: a palaeobiological model, Hist. Biol., 7, 29–50, 1993.
Wu, Y., Cui, Y., Chu, D., Song, H., Tong, J., Dal Corso., J., and Ridgwell A.: Volcanic CO2 degassing postdates thermogenic carbon emission during the end-Permian mass extinction, Science Advances, 9, eabq4082, https://doi.org/10.1126/sciadv.abq4082, 2023.
Yasuhara, M., Wei, C.-L., Kucera, M., Costello, M. J., Tittensor, D. P., Kiessling, W., Bonebrake, T. C., Tabor, C. R., Feng, R., Baselga, A., Kretschmer, K., Kusumoto, B., and Kubota, Y.: Past and future decline of tropical pelagic biodiversity, P. Natl. Acad. Sci. USA, 117, 12891–12896, 2020.
Zhang, Y., Shi, G. R., He, W.-H., Wu, H.-T., Lei, K.-X., Du, C.-C., Yang, T.-L., Yue, M.-L., and Xiao, Y.-F.: Significant pre-mass extinction animal body-size changes: Evidences from the Permian–Triassic boundary brachiopod faunas of South China, Palaeogeogr. Palaeocl., 448, 85–95, 2016.
Zhang, Y., Wen, H., Zhu, C., Fan, H., and Cloquet, C.: Cadmium isotopic evidence for the evolution of marine primary productivity and the biological extinction event during the Permian-Triassic crisis from the Meishan section, South China, Chemical Geology, 481, 110–118, 2018.




