the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
From Alnus to Pinus: temperate peatland ecosystem transformation triggered by human-driven landscape change
Tomasz Związek
Marcin Klisz
Sandra Słowińska
Dominik Róg
Milena Obremska
Dominika Łuców
Jarosław Pietruczuk
Joachim Popek
Katarzyna Piotrowicz
Kamil Pilch
Krzysztof Szewczyk
Agnieszka Halaś
Michał Słowiński
Peatlands are invaluable archives of palaeoenvironmental and climate dynamics, play a central role in the global carbon cycle and hydrological processes, preserve biological diversity, and act as climatic microrefugia. Over millennia, these ecosystems have been heavily modified by human land use, including drainage, overgrazing, and peat extraction, leading to large-scale degradation in many regions. Understanding the long-term dynamics of peatlands is crucial for determining their conservation and restoration needs, as well as for predicting their evolution, including responses to climate change, community changes, and carbon sequestration potential. In this study, we adopted an interdisciplinary approach to investigate the relationships between climate, vegetation, tree growth, hydrology, and human activities in a peatland ecosystem in one of the poorly explored regions of Central Europe, the Solska Forest in southeastern Poland. To reconstruct local ecosystem and landscape dynamics and assess possible climatic and anthropogenic impacts, we integrated various proxy data from natural and human archives: long-term meteorological data (1792–2020), tree ring data (1729–2022) from living peatland pines, palaeoecological data from peat sediment (pollen, plant macrofossils, testate amoebae, and charcoal), and archival written and cartographic sources. Our environmental reconstruction, spanning over 2300 years (ca. 330 BCE–2022 CE), identified three distinct periods in the peatland's history: ca. 330 BCE–1400 CE, ca. 1400–1830 CE, and ca. 1830–2022 CE. These phases are characterized by varying archival coverage, including a sedimentary hiatus between 1400 and 1830 CE, likely associated with a fire disturbance documented at the study site in the first half of the 19th century. However, the stratigraphic gap was largely bridged by integrating tree ring, climate, and historical data. This synthesis revealed considerable hydrological instability of the peatland and documented its complete transition from black alder bog forest to Scots pine bog forest. To our knowledge, this provides the first palaeoecological evidence of such a conversion in temperate Europe. This ecosystem shift was likely triggered by several interlinked factors, primarily anthropogenic land use change, including settlement development, deforestation, and the introduction of timber-oriented forest management. These drivers led to the landscape-scale expansion of pine forests and subsequent environmental acidification that facilitated Sphagnum encroachment. Furthermore, the roles of fire and drainage in this transition were clearly demonstrated. Our results, including the tree ring record of successful Scots pine establishment and historical data on settlement development throughout the 1700s, suggest that this environmental transformation occurred before the end of the period ca. 1400–1830 CE, between the 16th and 18th centuries. In addition, our findings indicate that both the transition period (> 400 years) and the current ecosystem type (ca. 200 years) represent a much shorter part of the peatland's history (> 600 years) than the preceding, substantially different ecosystem state (> 1700 years). We also address the methodological challenges of multi-proxy studies of landscape dynamics, such as reconciling inconsistent quantitative and qualitative data or managing periods of low archival resolution. Nevertheless, linking natural and human archives enabled us to gain a deeper understanding of a complex environmental system, with added value from combining different approaches. Moreover, by confirming the multifaceted interactions between different biotic and abiotic factors affecting both landscape and peatland ecosystems, we emphasized the continuing need for further research on peatland ecology, including past and current changes. Our evidence of a complete ecosystem shift from Alnus to Pinus and Sphagnum dominance under the synergistic influence of different factors, particularly the undeniable human impact, highlights the importance of a temporal perspective and long-term environmental data in conservation and land management, as such records are vital for assessing reference conditions and defining protection and restoration goals.
- Article
(9233 KB) - Full-text XML
-
Supplement
(2153 KB) - BibTeX
- EndNote
Peatland ecosystems are widely recognized as habitats crucial for a broad variety of species, an important part of hydrological systems, palaeoenvironmental archives, potential climatic microrefugia, and long-term regulators of the global carbon cycle (MacDonald et al., 2006; Rydin and Jeglum, 2013; Leifeld and Menichetti, 2018; Słowińska et al., 2022). Despite their limited distribution (approx. 3 % of the world's land surface), peatlands are the largest terrestrial carbon reservoir (Joosten et al., 2016). They are highly sensitive to climate change, which can substantially affect their carbon budget and, as a result, transform them into carbon sources (Belyea and Malmer, 2004; Jassey et al., 2018). Like many other ecosystems worldwide, peatlands have been significantly modified by human land use for millennia, often involving activities with highly negative impacts such as overgrazing, peat extraction, or drainage (Päivänen and Hånell, 2012; Joosten, 2016; Manton et al., 2025). Due to its high population density, long cultural history, and climatic suitability for agriculture, Europe is currently the continent with the greatest proportional loss of peatlands, with 44 % being degraded, i.e., no longer peat accumulating (Joosten, 2016). In many European countries, intensive peatland drainage for both agriculture and forestry took place in the second half of the 20th century (Päivänen and Hånell, 2012; Joosten, 2016). Yet, the extraordinary value of peatland ecosystems has been acknowledged in recent decades, resulting in broad conservation and restoration efforts across the continent (Andersen et al., 2017; Jurasinski et al., 2020), clearly highlighting the importance of rewetting (Jurasinski et al., 2024; Manton et al., 2025). However, peatlands in the continental fen and bog region, which covers a large part of Eastern Europe (including eastern Poland, southern Belarus, northern Ukraine, and a substantial section of Central European Russia), are highly degraded (52 %) and still insufficiently protected (15 %) (Tanneberger et al., 2021).
In Europe, peatland forests, particularly bog woodlands, represent habitats of regional importance, legally protected under the European Union Habitats Directive (Annex I, habitat type code: 91D0) (Pawlaczyk, 2010; Anonymous, 2013; Grzybowski and Glińska-Lewczuk, 2020). In the continental biogeographical region of the European Union, nearly half of this habitat type is located in Poland (Anonymous, 2013–2018), making the country one of the conservation hotspots. However, environmental changes in peatland forests, such as decreasing humidity, peat decay, increasing fertility, and decreasing acidity, which result in significant functional and structural changes, have been recorded even in the best preserved woodlands (Czerepko, 2008; Yermokhin et al., 2021). Data on the long-term dynamics of peatland forests is thus urgently needed to provide important baseline information for the conservation and management of these highly valuable habitats (cf. Lindbladh et al., 2013; Tanneberger et al., 2021; Grzybowski and Glińska-Lewczuk, 2020).
Forested or not, peatlands are driven by several hydrological feedbacks that regulate their response to varying environmental factors, including seasonal fluctuations in water table depth and various disturbances, which can cause profound changes in peatland functioning and structure, such as vegetation and carbon budget (Waddington et al., 2015). This makes peatlands highly complex environments, where lag and feedback effects, together with interactive effects of different drivers, are common (Linderholm et al., 2002; Smiljanić et al., 2014; Janecka et al., 2025). Multi-proxy studies have proven valuable in broadening the understanding of the intricate peatland ecology and dynamics, including fire history (Niklasson et al., 2002; Šamonil et al., 2018), moisture dynamics (Edvardsson et al., 2019; Taminskas et al., 2019), anthropogenic impact (Lamentowicz et al., 2009; Edvardsson et al., 2018), vegetation and peatland development (Eckstein et al., 2009; Edvardsson et al., 2014; Šamonil et al., 2018; Stančikaitė et al., 2019), tree demography (Linderholm and Leine, 2004; Edvardsson et al., 2015a), and climate variability (Edvardsson et al., 2012a, 2018). In Europe, the number of these studies substantially increased during the 21st century and resulted in several datasets covering much of northern Fennoscandia, Great Britain, and the area around the southern Baltic Sea (southern Sweden, northern Germany, Lithuania, northern Poland). Yet, broad geographic areas are still underutilized in this respect, for example large sections of continental Europe, including Belgium, central Germany, Belarus, Ukraine, central and central-southern Poland (Edvardsson et al., 2022). Hence, a substantial share of European peatland ecosystems (Joosten, 2016) persists not thoroughly explored and understood.
Despite different time resolution between palaeoecological and dendrochronological records (Edvardsson et al., 2022), tree ring data from both living (Niklasson et al., 2002; Edvardsson et al., 2019) and subfossil (Eckstein et al., 2009; Edvardsson et al., 2012a, b) trees were often applied in multi-proxy studies of peatland ecosystems. Tree growth in peatlands is affected by local water table fluctuations (Boggie, 1972; Smiljanić et al., 2014; Edvardsson et al., 2019), which are closely linked to climatic variables such as air temperature, precipitation, or snow cover (Linderholm et al., 2002; Dauškane et al., 2011; Dinella et al., 2021). However, this complex and multifaceted relationship remains not fully recognized, mainly due to the already mentioned hydrological feedback (Waddington et al., 2015) and lag effects in peatland ecosystems (Linderholm et al., 2002; Edvardsson et al., 2015b, 2019; Dinella et al., 2019). Generally, high water table level may inhibit tree growth (Dang and Lieffers, 1989; Linderholm, 1999; Smiljanić et al., 2014; Edvardsson and Hansson, 2015) since waterlogged, anaerobic environment is strongly restricting root development or even leads to the dying of fine roots (Boggie, 1972; Laiho and Finér, 1996). Because of that, peatland trees: (1) often respond negatively (i.e., with reduced growth) to spring/summer precipitation (Linderholm, 1999; Linderholm et al., 2002; Dauškane et al., 2011; Edvardsson et al., 2015b; Blanchet et al., 2017) – just opposite to populations of the same species growing on mineral soils (Linderholm et al., 2002; Janecka et al., 2025), and (2) show a positive growth response to dry conditions (Potapov et al., 2019), for example as a result of peatland drainage (Linderholm, 1999; Potapov et al., 2019). On the other hand, prolonged periods of low water table level may cause water stress in peatland trees (Dang et al. 1991; Pepin et al., 2002). Hence, a contrasting, positive growth response to precipitation in the growing season can be also observed (Linderholm et al., 2002; Vitas and Erlickytė, 2007; Cedro and Lamentowicz, 2011; Ignatiev and Yermokhin, 2022). Such diverse data justify the need for further multi-proxy studies, preferably adopting an interdisciplinary approach that extends beyond palaeoecology and dendrochronology, and employs a high number of proxies (Edvardsson et al., 2014, 2018, 2019; Stančikaitė et al., 2019; Bąk et al., 2024). To date, such studies remain rather scarce and geographically unbalanced (Edvardsson et al., 2022), especially in the case of studies using tree ring data from living peatland trees (Edvardsson et al., 2019; Taminskas et al., 2019).
Human impact on terrestrial ecosystems worldwide has spanned millennia (Leuschner and Ellenberg, 2017b; Ellis et al., 2021). Even before the industrial era human societies were modifying land cover, fire regimes, vegetation communities, and global carbon budget (Kaplan et al., 2009; McMichael and Bush, 2019; Izdebski et al., 2022; Sayedi et al., 2024). Increasing human population density and industrial development were followed by further substantial changes in disturbance regimes, land use and land cover, including deforestation, rise of urban and cropland areas, and ecosystem transformation in effect of drainage (Ellis and Ramankutty, 2008; Kaplan et al., 2009; Joosten, 2016; Williams et al., 2020). Due to its duration and global extent, disentangling human impact from the other environmental factors shaping long-term landscape dynamics may be often challenging if not impossible, which calls for an integrative approach acknowledging the interconnection of environment and societies (Naveh, 1995; Bürgi and Russell, 2001; Dearing et al., 2015). However, human and natural archives differ significantly in availability, time span covered, objectivity, precision, spatial and temporal resolution, and the presence of quantitative information (Forman and Russell, 1983; Ruffner and Abrams, 1998), making long-term cross-disciplinary studies involving environmental sciences and the humanities challenging and relatively infrequent (Verheyen et al., 1999; Dearing et al., 2008; Szabó, 2010; Lamentowicz et al., 2020; Bąk et al., 2024). Hence, broadening the knowledge on anthropogenic influence on different habitats and regions seems valuable for a full picture of the past, current, and future trajectories of ecosystem processes (Dearing et al., 2015).
A long-term perspective is particularly vital for peatland ecosystems, as understanding their past and present dynamics is essential for predicting future development, including responses to climate change, vegetation shifts, carbon sequestration potential, and restoration and conservation needs (Lindbladh et al., 2013; Marcisz et al., 2022). To address the aforementioned long-term human–environment interactions (e.g., Joosten, 2016; Bąk et al., 2024), we applied an interdisciplinary approach, combining palaeoecological, dendrochronological, meteorological, and historical data to explore the linkages between tree growth, hydrology, climate, and possible human impact in a peatland located in one of the less studied regions of Central Europe: southeastern Poland (Edvardsson et al., 2022). Notably, a unique feature of Poland is that, following the geopolitical changes of the late 18th century, different parts of its territory were incorporated into the distinct economic and administrative systems of the neighbouring Enlightenment-era monarchies: Prussia, Russia, and Austria (Davies, 2005; Lukowski and Zawadzki, 2006). This significantly influenced local land management and, consequently, the landscape of the Polish lands, including forests and peatlands (Broda, 2000; Jaszczak, 2008a, b, c; Bąk et al., 2024; Przybylski et al., 2025).
Given the complexities of peatland ecology and functioning (e.g., Waddington et al., 2015; Edvardsson et al., 2019; Marcisz et al., 2022) and their global and regional vulnerability (e.g., Joosten, 2016; Jassey et al., 2018; Manton et al., 2025), we hypothesized that the peatland ecosystem under study has experienced substantial structural and functional changes, likely including disturbances, throughout its development history, driven not only by natural abiotic factors but also by pronounced human impact. Therefore, in this study we aimed to (1) reconstruct long-term ecosystem dynamics by integrating diverse proxy records from both natural and human archives, (2) assess peatland ecosystem stability, and (3) evaluate the underlying drivers of ecosystem transformation, including anthropogenic influence.
2.1 Study area
2.1.1 Geography, vegetation and climate
Wielkie Bagno (Eng. Great Swamp) peatland is located near town Biłgoraj in the Solska Forest (50°31′N, 22°50′ E) in southeastern Poland, which is a large forest area covering over 1400 km2 in the Biłgoraj Plain, stretching from the Vistula River in the west up to the border of Ukraine in the east (50°48′ N, 21°56′ E–50°13′ N, 23°26′ E). Along with the neighbouring Roztocze region it is a regionally important biodiversity hotspot due to its extraordinary ecosystem diversity, including forests, peatlands, meadows, steppe communities, and arable land. Solska Forest is a continuous woodland composed mainly of Scots pine (Pinus sylvestris L.) forests on mineral, mineral-organic, and organic (i.e., peat) soils, varying from dry to moist, humid, and bog forest communities, including peatlands. Deciduous tree species such as European beech (Fagus sylvatica L.), oak (Quercus spp.), and black alder (Alnus glutinosa (L.) Gaertn.) occur in smaller patches, in majority in black alder bog forests (Chmielewski and Sowińska, 2008, 2011; Maciejewski and Szwagrzyk, 2011) (Fig. 1). Wielkie Bagno peatland is a large (approx. 250 ha) basin filled with peat and mud sediments, surrounded by fluvial sands of floodplain terraces and eolian sands, including eolian sands in dunes, overlying Pleistocene lacustrine silts, fluvial sands and gravels (including those with peat and mud layers), and Tertiary clays, sandy clays, and mudstones with sandstone interbeds (Popielski, 1992). The main part of the basin is generally flat (elevation of approx. 208–218 m a.s.l.), surrounded by dunes and with a dune extending along a west-east axis across the southern section (Fig. 1c). Wielkie Bagno peatland and its immediate surroundings are covered by a network of ditches (Fig. 1 and Fig. S1 in the Supplement).
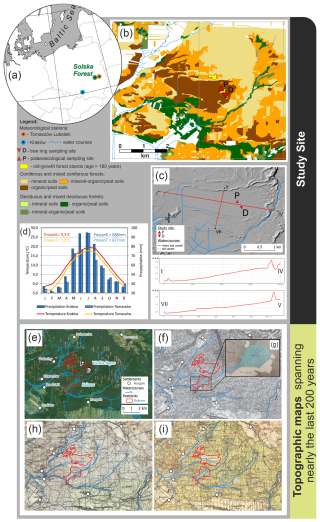
Figure 1Our study site and its surroundings in different time periods. (a) Location of the Solska Forest and meteorological stations (Kraków, Tomaszów Lubelski) that derived climate data used in this study; (b–c) study area and sampling sites presented on: (b) the forest type map (Forest Data Bank, Pol. Bank Danych o Lasach, https://www.bdl.lasy.gov.pl/portal/udostepnianie-en, last access: 19 April 2024) and (c) the digital terrain model (GDAL/OGR contributors, GDAL – Geospatial Data Abstraction Library, Open Source Geospatial Foundation, https://gdal.org, last access: 6 January 2025); (d) average air temperature and total precipitation in Kraków and Tomaszów Lubelski in 1951–2017; (e) ortophotomap from June 2023 (map data © 2023 Google); (f) topographical map of the Congress Kingdom, published in 1843; (g) archival map (APL, AOZ, IMK, sign. 3, sheet 421, archival source 15, AS15, full list of archival sources given prior to the reference list), published in the second half of the 19th century; (h) map of west Russia (Ger. Karte des westchlichen Russlands), published in approx. 1919; (i) map by the Military Geographical Institute (WIG), published in the 1930s. Hydrology data source: https://www.geoportal.gov.pl (last access: 19 April 2024). Historical maps acquired from the public domain.
The study area is located in the humid continental climate zone (Dfb) with warm summers and cold winters according to the Köppen-Geiger climate classification (1951–2000, Kottek et al., 2006). In the period 1951–2017, the mean annual air temperature in the study area was 7.2 °C, with monthly mean air temperatures in January and July of −3.9 and 17.9 °C, respectively (Tomaszów Lubelski meteorological station, Institute of Meteorology and Water Management-National Research Institute, IMGW-PIB, 50°27′ N, 23°24′ E). The average annual precipitation total for the same period was 637.0 mm (Fig. 1d). Precipitation predominated in the warm half of the year (407.4 mm) compared to the cold half of the year (229.6 mm), which is typical for a continental climate. However, the climate of peatlands and Scots pine bog forests may be cooler than in the open areas where the usual weather stations are located (Olszewski, 1986; Słowińska et al., 2022).
Our sampling site was selected based on the following specific criteria: (1) the presence of old-growth tree populations (> 180 years), providing a long tree ring chronology, and (2) location within a peatland area, allowing peat sampling in close proximity (< 500 m) to the sampled trees. Tree ring (50°31′23.88′′ N, 22°51′56.52′′ E) and palaeoecological (50°31′31.08′′ N, 22°51′46.799′′ E) sampling within the Wielkie Bagno peatland (Fig. 1) took place in two neighbouring pure pine stands on peat soil, which are 182 and 190 years old and represent a Scots pine bog forest (Vaccinio uliginosi-Pinetum; Leuschner and Ellenberg, 2017b) according to the forest inventory data (Forest Data Bank, Pol. Bank Danych o Lasach, https://www.bdl.lasy.gov.pl/portal/en, last access: 19 April 2024). However, the pine populations of our study site should be described as multi-aged since several older trees were also present as confirmed by their crown and bark features typical for ancient conifers (Andersson and Niklasson, 2004). Ground layer was composed of Vaccinium uliginosum, Ledum palustre, Eriophorum vaginatum, Molinia caerulea, and Vaccinium myrtillus. Bottom layer was dominated by Sphagnum spp. mosses (Fig. S1).
2.1.2 Regional historical background
Solska Forest is a remnant of the vast Sandomierz Forest, which was largely uninhabited during the Middle Ages (Buraczyński, 2008). Settlement development first intensified at the end of the 16th century with the emergence of an important trade route in the region (Szczygieł, 1985).
Between the 16th and 18th centuries, the landscape of the Polish-Lithuanian Commonwealth was characterized by vast estates owned by influential noble families, who derived their power from substantial land revenues. One such family was the Zamoyski family (Rajca, 1972). Solska Forest became part of the Zamoyski Family Estate (Pol. Ordynacja Zamoyskich) as early as the turn of the 16th and 17th centuries. The new owners intensified settlement development in the area by building sawmills and establishing seasonal forest settlements for the production of potash, ash, and wood tar, particularly towards the end of the 17th century. After a substantial decrease in available forest resources, local inhabitants, known as hutters (Pol. budziarze), had to adapt their economy and land use by turning to field and meadow farming, cattle and sheep breeding, traditional forest beekeeping, and only limited forest exploitation, mainly tar production (Róg, 2021).
The late 18th century brought significant political and territorial changes to this part of Europe. The southern lands of the Polish-Lithuanian Commonwealth, including the Zamoyski Family Estate and our study site, were incorporated into the Habsburg Monarchy in 1772 and consequently became subject to Austrian legislation and several economic reforms (Piller, 1782; Davies, 2005; Jones, 2016; Carvalho, 2018). In forest management, these changes involved the introduction of the Enlightenment-era model, based on the German silviculture system (Hölzl, 2010).
The early 19th century was marked by further geopolitical changes affecting our study area. The Polish lands, originally incorporated by Austria in 1795, by the end of the first decade of the 19th century became part of the Duchy of Warsaw, created by Napoleon in 1807. However, after Napoleon's defeat and the resolutions of the Congress of Vienna in 1815, the Duchy was abolished and its lands, including the Zamoyski Family Estate and our study site, came under the rule of the Russian tsars. This change in government introduced new legal and administrative regulations, including a management system for income-generating properties such as forests (Grodziski, 1971; Jewuła et al., 2015).
The 20th century brought further historical challenges. After World War I, some forest areas had to be sold to cover the costs of rebuilding the Zamoyski Family Estate after the war. In the 1920s, the increasing demand for timber led to considerable logging. Rational forest management was introduced in the mid-1930s (Kozaczka, 2002). During World War II, the entire estate was managed by the Nazi occupying forces (Klukowski, 1945–1947). The end of the Zamoyski Family Estate was brought by the change in the socio-political system in Poland in 1944, which took private estates, including both agricultural lands and forest areas, into public ownership and management. Since then the land forming the estate was not the property of the Zamoyski family anymore (Kozaczka, 2003; Jędrejek, 2012).
Currently, the study area is part of the Tereszpol Municipality. Since 1944, the forests surrounding our study site remain state-owned and are managed by the Polish State Forest Administration. They are part of the Zwierzyniec Forest District (Forest Data Bank, https://www.bdl.lasy.gov.pl/portal/mapy, last access: 19 April 2024) and are used for forest management.
2.2 Climate data
To assess long-term climate fluctuations in the region and the climate–tree growth relationships at the study site over the longest period covered by available instrumental data, average monthly, seasonal, and annual values of air temperature (1792–2020) and atmospheric precipitation (1811–2020) from the meteorological station of the Department of Climatology at Jagiellonian University in Kraków were used. The meteorological station is located in the city centre, in the Botanical Garden of the Jagiellonian University (50°04′ N, 19°58′ E, 220 m a.s.l.). Sensors are installed at a height of 12 m above ground. Meteorological data from Kraków are one of the longest instrumental weather series in Europe and have been widely used in numerous scientific studies as they well represent the climatic conditions of Central European lowlands within a radius of 300 km (Hess, 1974; Kożuchowski et al., 1994; Trepińska et al., 1997; Trepińska, 2000), including our study site, located approximately 280 km to the east. To verify the representativeness of the Kraków data for our study area, we compared air temperature and precipitation data from Kraków and Tomaszów Lubelski (IMGW-PIB), the nearest weather station to our study site (Fig. 1a), for the overlapping period of 1951–2017 (67 years). We assessed the significance of differences between the two stations in monthly temperature (T) values (mean, minimum, and maximum) and precipitation (P) totals across various periods (monthly, seasonal, warm and cold half-years, and annual) using the Mann–Whitney U test. The statistical analyses were conducted using the nlme package (Pinheiro et al., 2025) of the R software (R, Version 3.1-168).
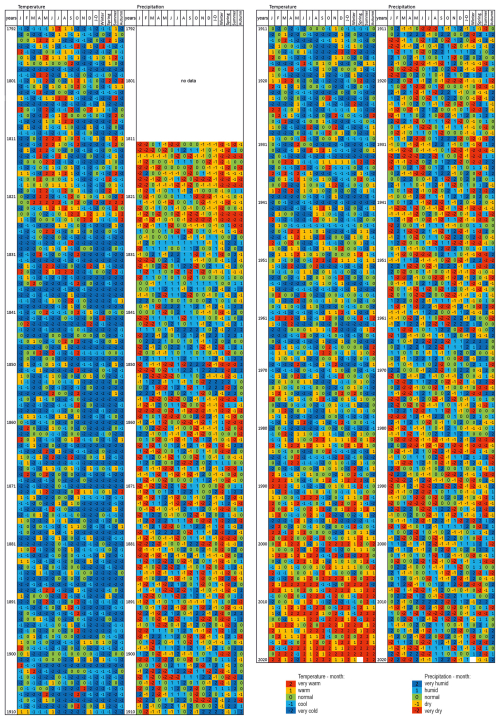
A percentile (quantile) classification of thermal and pluvial conditions was made for months, seasons, and years in Kraków and Tomaszów Lubelski. The classification allowed for the assessment of mean air temperature values and precipitation totals for the specified period in comparison to the reference period (Miętus et al., 2002; Czernecki and Miętus, 2011). In our case, we assumed a common period for both stations, i.e., 1951–2017. The empirical percentiles were determined in 20 % increments, ranging from 20 % to 80 %. Subsequently, the complete range of temperature and precipitation variability over a specified period and at a given station was divided into five percentile intervals. The following categories for temperature were assigned: < 20 % very cold (−2); 20.01 %–40 % cold (−1); 40.01 %–60 % normal (0); 60.01 %–80 % warm; > 80 % very warm. In regard to precipitation, they were as follows: < 20 % very dry (−2); 20.01 %–40 % dry (−1); 40.01 %–60 % normal (0); 60.01 %–80 % wet; > 80 % very wet (Figs. 3 and S2). Such classifications are used, among others, by the IPCC (2007) to assess climate change in specific time periods.
2.3 Dendrochronology
2.3.1 Fieldwork, sample preparation, and chronology building
To reduce individual growth variability, tree ring samples were collected from 21 Scots pine trees in June 2022, selected according to their biosocial (dominant or co-dominant trees), health (vital individuals without visible damage), and stand structure (avoidance of trees at the edge of gaps and stand margins) status. From each sample tree two increment cores were taken from two perpendicular directions using 5.15 mm diameter Pressler borers at a height of 1.3 m (Cook and Kairiukstis, 1990). After discarding samples too eroded for analysis, wood samples from 19 trees were mounted on wooden supports, sanded with a series of progressively finer sandpapers (grit size up to 1000) to improve the visibility of the tree ring boundaries and scanned at 2400 dpi (Epson Expression XL12000). Identification of the boundaries between annual rings was done with a binocular (80 × magnification LEICA S8APO) to enable detection of false rings (due to intra-annual density fluctuations in the early wood), wedging rings, and missing rings. Cross dating was performed using standard dendrochronological approaches (Stokes and Smiley, 1968; Yamaguchi, 1991) and verified with “CDendro” software (Larsson and Larsson, 2018) after annual ring widths were measured to an accuracy of 0.01 mm using “CooRecorder” software (Larsson and Larsson, 2018; Maxwell and Larsson, 2021). Tree recruitment dates (i.e., tree age) were determined based on the pith dates at sampling height (Heyerdahl et al., 2014). In case of increment cores, where the pith was missing, distance to pith was estimated based on growth and curvature of the earliest observed rings in the sample during the ring width measuring procedure (Larsson and Larsson, 2018). Determination of pith date was possible for 13 sample trees which did not include inner rot precluding a reliable assessment of the distance to pith in reference to the ring curvature. Tree ring width series were detrended to remove any biological (age) trend and other low-frequency fluctuations caused by non-climatic factors (Cook and Peters, 1981; Speer, 2010). Individual tree ring series were detrended with a 30-year cubic smoothing spline with a frequency cut-off of 50 % (Bunn, 2008). To remove the first-order autocorrelation in the dimensionless ring width index (RWI) series, we applied autoregressive modelling. Finally, a bi-weighted robust mean was applied to the pre-whitened series of individual trees to develop a mean stand chronology (Cook and Kairiukstis, 1990). The quality of the generated chronologies was checked with the so-called Ger. Gleichläufigkeit (GLK, i.e., the coherence coefficient) (Eckstein and Bauch, 1969; Buras and Wilmking, 2015) and the mean correlation between individual tree ring series (mean rbt, an indicator of the strength of the common signal) (Wigley et al., 1984; Cook and Kairiukstis, 1990). To test the suitability of the constructed chronologies for climate–growth analyses, we applied subsample signal strength (SSS; Buras, 2017), first-order autocorrelation (AR1, an indicator of the effect of the previous year's conditions on the current year's growth), and signal-to-noise ratio (SNR, the proportion of explainable variation in the chronology due to climate divided by the unexplained variation) (Wilczyński and Kulej, 2013). A threshold value of more than 0.85 was used as an entry criterion for the SSS (see Fig. S4). These steps were completed with the dplR package (Bunn et al., 2020) of the R statistical software (R Core Team, 2021).
2.3.2 Climate–tree growth relationship
To assess how climate affects inter-annual growth variability of peatland trees at the study site, we used validated climate data from the Kraków meteorological station (see Sect. 2.2) and calculated Pearson correlation coefficients between the site indexed ring width chronology and monthly climate variables: mean air temperature, precipitation sum, and the standardized precipitation evapotranspiration index (SPEI), aggregated over three and six months. To calculate the SPEI indices, we estimated the water balance as the difference between monthly precipitation and potential evapotranspiration. Positive value of the index (SPEI > 0) reflects the positive water balance, higher precipitation than potential evapotranspiration (Vicente-Serrano et al., 2010) (cf. Fig. S3). To assess the lag effect of the negative water balance we aggregated the monthly SPEI over three and six months (SPEI3 and SPEI6, respectively). To calculate climate–growth correlation we used the monthly_response() function implemented in the R package dendroTools, considering all previous and current seasons from the previous January to the current December (Jevšenak and Levanič, 2018; Jevšenak, 2020). As the relationship between tree growth and external factors is non-linear (Wilmking et al., 2020), we tested the temporal variability of the relationship between climate and growth by systematically subsetting sub-periods of RWI values and climate variables from the total analysed period, using a 30-year running window with a one year offset. For each subset period, we calculated climate variable–growth correlations (Jevšenak and Levanič, 2018; Jevšenak, 2020).
2.4 Palaeoecology of the peat archive
2.4.1 Core collection, lithology, chronology, and numerical analysis
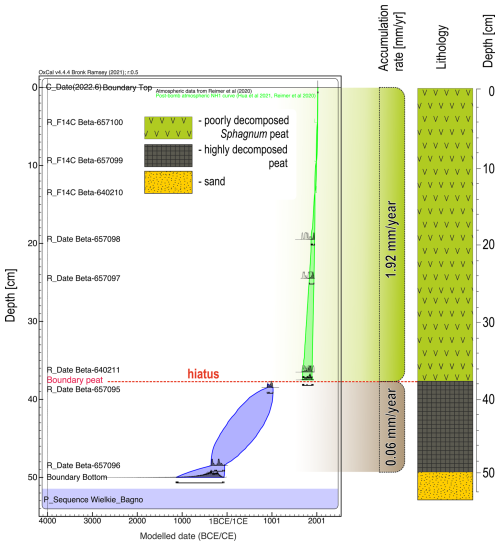
In June of 2022, a peat core with a diameter of 5 cm and a length of 50 cm was collected with an Instorf corer, reaching the bottom of the peat deposit at the sampling point (Fig. 7). The extraction core was placed in a PVC tube and then transported to the cold room of the Institute of Geography and Spatial Organization of the Polish Academy of Sciences, where it was stored at 4 °C until subsampling. The organic sediment (peat) was cut into 1 cm thick slices, which were then analysed at a resolution of 1 cm. Of these samples, 50 were analysed for pollen, macrofossils, and charcoal, and 25 for testate amoebae. The lithology of the analysed peat core was as follows: (1) highly decomposed peat at a depth of 50 to 37 cm, and (2) poorly decomposed Sphagnum peat from the depth of 37 cm to the surface. The transition horizon between these two lithological segments was marked by increased amorphous matter and a distinct layer of macroscopic charcoal. Eight radiocarbon dates were obtained from the peat core, using Sphagnum stems and pollen extract (Table 1). For chronological control of the transition between the two lithological segments, we intentionally selected two radiocarbon samples: one from the underlying highly decomposed peat and another from the overlying Sphagnum peat. This bracketing strategy was intended to constrain the timing of the end of the former state and the onset of the new Sphagnum-dominated phase. Conversely, we did not sample charcoal from the transition horizon for radiocarbon dating, as this would primarily provide the age of the burned wood (potentially including inbuilt age and reworking), and would not reliably constrain the timing of the fire event or the ecosystem transition. The pollen extract was prepared according to the procedure specified in the Oxford Long-Term Ecology Laboratory's “Pollen preparation procedure for radiocarbon dating” protocol, based on Brown et al. (1989). Pollen was extracted from 2 cm3 of sediment. The chronology of the profile was established from these eight radiocarbon dates. The age–depth model was constructed using OxCal (OxCal v4.4.4, 2023) (Fig. 7). The IntCal20 (Reimer et al., 2020) and Bomb21NH1 (Hua et al., 2021) atmospheric curves were used to calibrate the dates. To present variation in biological assemblages across the peat sediment layers, non-metric multidimensional scaling (NMDS) ordination on the Bray-Curtis dissimilarity (Ricotta and Podani, 2017) was applied on the dataset using the vegan package (Oksanen et al., 2025) in R software (R Core Team, 2021). The NMDS was based on pollen data (only taxa that exceeded 1 % of total pollen sum in at least one sample were selected), plant macrofossil data, and micro- and macrocharcoal influx. To identify assemblage groupings, we applied k-means clustering to the NMDS ordination scores (Hartigan and Wong, 1979). To determine the optimal number of clusters, we used the elbow method, which involves calculating the total within-cluster sum of squares (WSS) for a range of cluster numbers (k = 1 to 10). Based on this analysis, we selected k = 5 as the most optimal clustering solution. The NMDS results were plotted in R using ggplot2 package (Wickham, 2016) as the ordination plot to show variation among samples and as the stratigraphic plot of NMDS1 axis scores plotted against depth to show temporal changes.
Table 1The list of radiocarbon dates from the Wielkie Bagno peatland (Solska Forest) with calibrations. The pollen extract was prepared according to the procedure specified in the Oxford Long-Term Ecology Laboratory's “Pollen preparation procedure for radiocarbon dating” protocol, based on Brown et al. (1989).
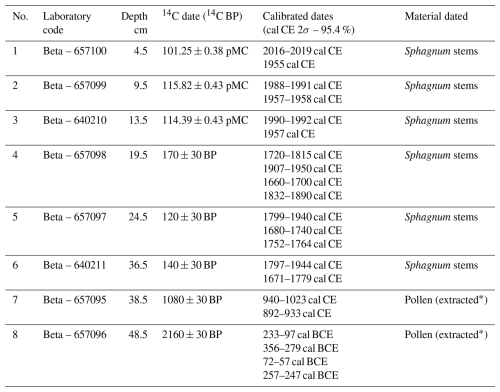
∗ From 2 cm3 of sediment.
2.4.2 Pollen and microcharcoal analysis
Samples for pollen and microcharcoal analysis were collected from the peat core at 1 cm intervals (a total of 50 samples) and prepared using standard laboratory procedures (Berglund and Ralska-Jasiewiczowa, 1986). To determine microcharcoal concentration and calculate annual influx, Lycopodium markers were used (Stockmarr, 1971). Pollen and spore identification was carried out using photographic reference collections and keys (Fægri et al., 1989; Moore et al., 1991; Beug, 2004). For each sample, at least 500 tree pollen grains were counted. The percentage share of plant taxa was calculated based on the sum of arboreal pollen (AP) and non-arboreal pollen (NAP), i.e., the AP+NAP sum. Pollen grains and spores of local aquatic and telmatic plants were excluded from this sum. Zonation was confirmed by CONISS cluster analysis (Grimm, 1987). During analysis, non-pollen palynomorphs and microscopic charcoal particles (size: > 10 µm) were also counted from the same slides as the pollen. The curve of human indicators total (HIT) included the following taxa: Plantago media, Plantago major, Plantago lanceolata-type, Rumex acetosella, Rumex acetosa, Ambrosia-type, Matricaria-type, Artemisia, Chenopodiaceae, Urtica, Polygonum aviculare-type, Scleranthus, Spergularia-type, Centaurea cyanus, Fagopyrum, Cannabis sativa, Secale cereale, Triticum-type, Cerealia undiff., and Zea mays.
2.4.3 Plant macrofossils analysis
Material for the plant macrofossils analysis was collected at 1 cm intervals from the peat core (0–50 cm). All macrofossil samples were washed on sieves with a mesh diameter of 0.2 and 0.125 mm (Birks, 2002). Five microscope slides were prepared from each sample. Each specimen was inspected under an Opta-tech MB 300 series optical microscope at 200 × and 400 × magnifications in five fields of view. Plant macrofossils were identified using several macrofossil taxonomic keys (Lubliner-Mianowska, 1951, 1957; Dombrovskaya et al., 1959; Szafran, 1963; Grosse-Brauckmann, 1972, 1974; Grosse-Brauckmann and Streitz, 1992; Tobolski, 2000; Birks, 2007).
2.4.4 Testate amoebae analysis
Testate amoebae analysis was used to reconstruct hydrological variability. Material for the analysis was collected at 2 cm intervals from the peat core. The 2 cm3 of the peat were mixed in a 500 mL beaker, and next shaken in distilled water, and washed through a sieve with a mesh of 300 µm (Booth et al., 2010; Payne and Mitchell, 2009). The sediment was investigated using light microscope Nicon eclipse 50i at a magnification of 200 × and 400 ×. Tests were counted and identified to a minimal total of 100 individuals per sample using the available identification guides (Mazei and Tsyganov, 2006; Todorov and Bankov, 2019; Siemensma, 2019). The exception was the lower part of the core (from 37 to 48 cm), where the abundance of amoebas was extremely low (single test on one microscopic slide). Therefore, these samples were excluded from the analysis and from the percentage diagram. To calculate zonation of testate amoebae a CONISS method (Grimm, 1987) was applied based on a stratigraphically constrained cluster analysis. The reconstruction of hydrological variability (depth to water table, DWT) of the peatland was carried out based on the European training set compiled by Amesbury et al. (2016) with C2 software (Juggins, 2003). The Shannon diversity and Dominance D = 1-Simpson index of the testate amoeba communities were calculated in the PAST program (Hammer et al., 2001). Community of testate amoebae was divided into four categories based on the amoeba test (i.e., shell) construction (idiosomic, organic-coated idiosomic, agglutinated, and organic) according to Marcisz et al. (2021) and Mitchell et al. (2008). Mixotrophic testate amoeba species (Hyalosphenia papilio, Amphitrema wrightianum, Heleopera sphagni, Placocista spinosa, and Archerella flavum) were also summed up.
2.4.5 Macrocharcoal analysis
The record of macrocharcoal distribution was obtained from 50 samples (each 2 cm3) taken at 1 cm vertical intervals in peat profile. The collected samples were first bleached (Halsall et al., 2018; Hawthorne et al., 2018) and sieved through a 500 and 150 mm mesh. Charcoal particles with a size of > 100 µm were counted using a stereomicroscope at 200 × and 400 × magnifications. To account for variations in sedimentation in the sequence (Davis and Deevey, 1964), the macrocharcoal data were transformed into charcoal accumulation rate (i.e., charcoal influx, CHAR, particles cm−2 yr−1) by multiplying the concentrations of charcoal (CHAC, particles cm−3) with the sediment accumulation rates (cm yr−1). In addition, based on their sizes, macrocharcoal particles were divided into two groups: 150–500 µm and > 500 µm. Subdividing macrocharcoal size classes can provide information on the potential distance of fires from the studied site (e.g., Clark, 1988; Vannière et al., 2008; Conedera et al., 2009). While macroscopic charcoal particles are generally interpreted as a proxy for local fires, occurring within a few hundred metres (or as close as 1 m) of the sampling point (e.g., Ohlson and Tryterud, 2000; Higuera et al., 2005; Tinner et al., 2006; Conedera et al., 2009), they can also reflect a more distant, regional fire signal. This is due to the complex patterns of charcoal deposition and transport, which are influenced by fuel type, fire intensity, burn area, and meteorological conditions (e.g., Tinner et al., 2006; Peters and Higuera, 2007; Oris et al., 2014; Vachula and Rehn, 2023).
2.5 Historical sources – Cartography and written evidences
To assess human impact, archives were searched for any archival sources such as documents, historical maps, and printed archival sources, with information on land use, demography (including settlement development), and economy of our study site and its surroundings. In search of the archival data, we focused on information from economic inventories that would depict the economic processes, the dynamics of environmental change, demography, and the transformation of settlement structures in the Solska Forest. We also searched the rich cartographic archives for plans and maps of the Wielkie Bagno peatland which could visualize the above mentioned processes. The searches were conducted at the Central Archives of Historical Records in Warsaw (AGAD) and the State Archives in Lublin (APL). In the latter one, particular emphasis was placed on the Archives of the Zamoyski Family Estate (Pol. Archiwum Ordynacji Zamoyskich, AOZ). In addition, we also searched and critically reviewed the existing historical studies on the Solska Forest.
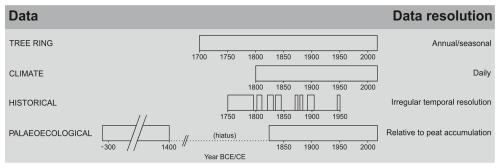
3.1 Temporal resolution of multi-proxy records provided by different archives
The peat archive allowed reconstruction of the peatland ecosystem dynamics during the periods of ca. 330 BCE–1400 CE and ca. 1830–2022 CE. The clear changes in the peat core based on the analysis of several palaeoecological proxies documented the two main phases of development and transformation of the Wielkie Bagno peatland: (1) WB-1, 49–37 cm (time period: ca. 330 BCE–1400 CE) and (2) WB-2, 37–0 cm (time period: ca. 1830–2022 CE). Three subphases were distinguished in the second phase: (2a) WB-2a, 37–16 cm (time period: ca. 1830–1947 CE), (2b) WB-2b, 16–7 cm (time period: ca. 1947–2004 CE), and (2c) WB-2c, 7–0 cm (time period: ca. 2004–2022 CE). The tree ring archive covered the period of 1729–2022 CE, extending the palaeoecological record back a century further in one of the above parts and broadening the picture of the period between the two main phases (WB-1 and WB-2). The climate records covered the period of 1792–2020 CE for temperature and 1812–2020 CE for precipitation. The human archives covered periods that overlapped with the natural archives. However, they provided more scattered data (Fig. 2). Most of the historical data covered the period from the mid-18th to the early 20th century and complemented the natural archives (Fig. 2, Tables S3 and S4).
3.2 Climate conditions
3.2.1 Comparison of climatic data from Kraków and Tomaszów Lubelski
A statistically significant thermal contrast was observed between Tomaszów Lubelski and Kraków. From 1951 to 2017, the mean annual air temperature in Tomaszów Lubelski was approximately 2 °C lower than that of Kraków (Fig. 1d). However, the correlation between the air temperature values (monthly, annual, and seasonal) at the meteorological stations in Kraków and Tomaszów Lubelski was found to be highly significant (correlation coefficients > 0.8–0.9). In contrast, the annual precipitation total, as well as the precipitation totals for the winter, spring, and autumn months, did not exhibit statistically significant differences. The correlation between precipitation data and the meteorological stations in Tomaszów Lubelski and Kraków was notably weaker than that observed for temperature data, particularly in the spring and summer months. However, in many instances, the values were statistically significant at the 0.05 level (Table S1).
3.2.2 Classification of thermal and pluvial conditions in Kraków and Tomaszów Lubelski
The classification of thermal and pluvial conditions carried out for each month, season, and year in the analysed datasets revealed values within the norm and deviations from it, both positive and negative. For air temperature, the increasing frequency of warm and very warm periods since the end of the 1980s and of cool and very cold periods in the years 1826–1920 can be clearly seen by the increased frequency of occurrence. Wet months and seasons occurred more frequently in the years: 1829–1849, 1896–1910, 1962–1966, 1996–1997, and 2010. Dry and very dry summer months have occurred more frequently since the 1980s. If we compare the classification results for the data from Kraków and Tomaszów Lubelski, we can see that there are no very clear differences between the values, both in terms of air temperature and precipitation (Figs. 3 and S2).
3.3 Tree ring data
3.3.1 Site chronology
Scots pine ring-width chronology covered the period 1729–2022 (GLK = 0.685; Rbar = 0.491; AR1 = 0.775; EPS = 0.948; SNR = 18.334; SSS = 0.959) (Fig. 4). The annual ring widths ranged from 0.06 to 6.96 mm, with an average of 0.81 mm. Peatland pine chronology revealed the following pointer years that agreed with data from other study sites in the region (NE Poland, Lithuania, Latvia; Cedro and Lamentowicz, 2008; Dauškane et al., 2011; Edvardsson et al., 2015b, 2019; Ewa Zin, Kamil Pilch, and Marcin Klisz, unpublished data): (1) positive: 1851, 1864, 1919, 1924, 1926, 1953, 1968, 1977, 1988, and (2) negative: 1928, 1930–1931, 1937, 1952, 1956, 1963, 1965. Sampled pine population recruited in two different time periods. Tree establishment data (at sampling height of 1.3 m) revealed continuous pine regeneration in the 18th century (during 1720s–1770s) and a cohort originating from the first half of the 19th century (1823–1827). One of the sample trees had a fire scar in the middle earlywood of 1830 indicating a fire, which occurred during the season of cambial activity (Baisan and Swetnam, 1990). This fire was recorded by other pines as a post-fire growth reaction, often combined with fire-induced disturbances in tree ring morphology (Niklasson and Granström, 2000; Zin et al., 2015).
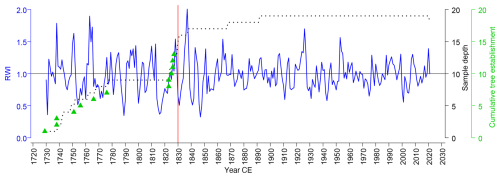
Figure 4Scots pine (Pinus sylvestris L.) chronology (blue line) and tree recruitment dates (green triangles) of living peatland trees in Wielkie Bagno, Solska Forest. RWI – ring width index. Sample depth is denoted by a dotted line and fire date recorded in the tree ring material by red vertical line.
3.3.2 Climate sensitivity of peatland pines
Climate–growth analyses revealed the dominant positive effect of the aggregated drought indices (both SPEI3 and SPEI6) of the second half of the current year (June–December), demonstrating the growth-promoting lagged effect of the wet conditions. The influence of temperature on growth of peatland trees was more diverse, documenting the significant effect of the current year conditions. Higher early spring temperatures (February–March) induced tree growth while warmer conditions in the latter period (May–December) had the opposite, growth inhibiting effect. Precipitation showed the weakest influence on tree growth, consistent with the influence of SPEI. The pluvial conditions of the current season had a dominant, growth-promoting effect. Precipitation in the previous year November and the current October showed positive correlation with tree growth, while precipitation in July of the previous year evidenced an opposite, negative correlation (Fig. 5).
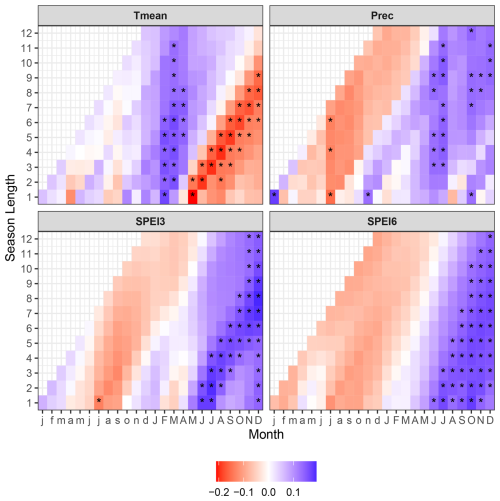
Figure 5Correlation between indexed tree ring width (RWI) of peatland pines and mean monthly air temperature (Tmean), monthly precipitation sum (Prec), and standardized precipitation evapotranspiration indices aggregated over three (SPEI3) and six (SPEI6) months, calculated for the period from January of the previous year (horizontal axis, lowercase letters) to December of the current year (horizontal axis, uppercase letters). Asterisks (∗) indicate the correlation significance level (p < 0.05).
3.3.3 Temporal variation of the climate–growth relationship
The growth-accelerating effect of the positive water balance (SPEI3, SPEI6) in spring–summer of the current season observed until the 1980s of the 20th century was inverted to a growth-limiting, however, statistically insignificant effect in the recent decades. This was paired with a clear growth enhancing effect of the positive water balance of the autumn months (October–November). Interestingly, a positive influence of wet early summer conditions was also noted in the earliest period, i.e., in the first half of the 19th century (Fig. S5). A markedly similar trend of temporal changes in climate sensitivity was observed for the effect of precipitation (Fig. 6). The temperature influence on tree growth of peatland pines revealed a more complex picture throughout the analysed period (1812–2022). The negative impact of high temperatures in spring–summer continued until the 1920s, where it changed towards the stronger effect of summer temperatures (August). In the recent decades, since the second half of the 20th century, negative correlation of growth with temperature was observed. Noteworthy, a comparable effect of temperature in autumn (October–November) was recorded in the second half of the 19th century (Fig. 6).
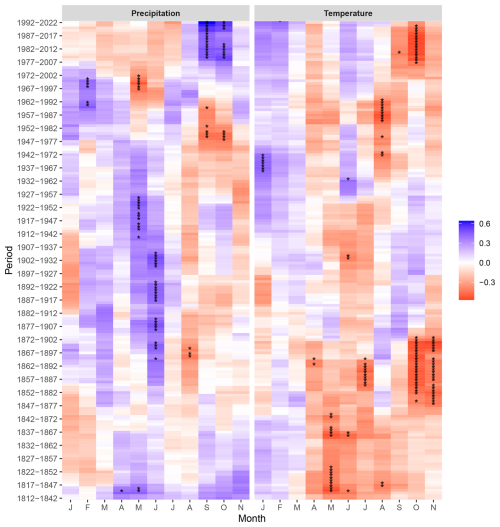
Figure 6Correlation in the 30-year moving window during the period 1812–2022 between indexed tree ring width (RWI) of peatland pines and monthly precipitation sum, and mean monthly air temperature, calculated for the current year (January–November, J–N). Asterisks (∗) indicate the significant correlations (p < 0.05).
3.4 Palaeoecology
3.4.1 Age–depth model and sediment composition
In the peat core, the section between 50 and 37 cm was already very different from the other sections during drilling due to the degree of decomposition, the structure of the sediment, and the macroscopic difference in the peat type. The highly decomposed peat at a depth of 50 to 37 cm was characterized by a slow accumulation rate (about 170 yr cm−1), while the second part of the profile showed a markedly increased peat growth rate of about 5 yr cm−1 (Fig. 7). Sediment accumulation was not continuous in this part of the Wielkie Bagno peatland. Between ca. 1830 and 1396 CE, a hiatus was observed in the transition between the two lithological segments, in which increased amorphous matter and a pronounced horizon rich in macroscopic charcoal were present. The hiatus interpretation is primarily based on radiocarbon bracketing (the missing time interval) and the abrupt shift in peat type (Fig. 7).
3.4.2 Pollen data (regional vegetation) and microcharcoal distribution
Based on changes in the percentage of taxa, three local pollen assemblage zones (LPAZ) were separated (Fig. 8), with two sub-phases in LPAZ-1 and two sub-phases in LPAZ-2:
-
LPAZ-1: 50–37 cm, Carpinus
Level with a high proportion of Carpinus pollen grains (13.9 %–19 %) in which two sub-phases were distinguished:
- 1a.
50–44 cm, Quercus: percentages of Carpinus reached a maximum of 19 %. Quercus content was between 6.2 % and 8.1 %. Ulmus curve was stable and reached values between 1.3 % and 1.7 %;
- 1b.
44–37 cm, Fagus: Carpinus (7.9 %–12.6 %) and Quercus (3.3 %–5.5 %) percentages decreased. Fagus content was from 3 % to 9 %. The curve of Ulmus declined below 1 %.
The upper limit of the level LPAZ-1 was marked by a decrease in the Carpinus curve.
- 1a.
-
LPAZ-2: 37–13 cm, Pinus–NAP
Proportion of Pinus pollen grains gradually increased from 42 % at the beginning of the phase to 63.6 % at the top of the level. NAP percentages were high, between 10.6 % and 23.6 %. Continuous curves of anthropogenic plant indicators (Rumex, Plantago lanceolata-type, Cannabis sativa, Secale cereale, Cerealia undiff.) were present. Two subphases were distinguished:
- 2a.
37–22 cm, Carpinus–Fagus: the proportion of Carpinus and Fagus pollen grains was 2.9 %–7.2 % for hornbeam and 2.5 %–6.1 % for beech. High percentage of Calluna pollen grains was recorded (up to 7.4 %). Proportion of anthropogenic indicators (HIT) reached 9.8 %;
- 2b.
22–13 cm, NAP: share of NAP was between 17.6 % and 23.7 % with predominance of anthropogenic indicators (up to 13.8 %). The proportion of hornbeam and beech was less than 2 %. Calluna did not exceed 2.7 %.
The upper limit of the level was determined by the decrease of the NAP.
- 2a.
-
LPAZ-3: 13–0 cm, Pinus
Content of Pinus pollen grains was predominant and reached a maximum of 83.1 %. NAP decreased to less than 10 %. HIT content gradually declined from 11 % at the beginning of the LPAZ-3 to 1.3 % in the top sample (Fig. 8).
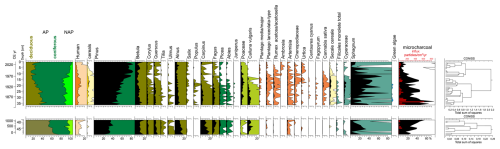
Figure 8Simplified percentage pollen diagram from the Wielkie Bagno peatland, showing selected taxa, microcharcoal concentration, and influx. Pollen and microcharcoal percentages are shown in black, 10 times exaggeration is marked by colours, and charcoal influx is depicted as red bars.
The distribution of microcharcoal particles, both concentration and influx, showed considerable variation throughout the profile, with the highest values recorded at depths of approximately 45, 40, 35–30, 25–20, and 17–12 cm. The maximum concentration (over 70 %) and the maximum influx (over 600 particles cm−3 yr−1) were found at a depth of 33 cm. A decrease in the amount of microcharcoal was observed from a depth of approx. 12 cm (Fig. 8).
3.4.3 Plant macrofossil data (local vegetation)
As a result of the plant macrofossil analysis (Fig. 9), five local macrofossil assemblage zones (LMAZ) were determined:
-
LMAZ-1, 50–37 cm: this phase was characterized by a high proportion of roots (from 35 % to 76 %) and a large number of uncategorized parts. Throughout the entire section, Sphagnum (including Secc. Cuspidata and Secc. Palustria) occurred at a level of 1 %–5 %. In the roof part of this phase, there was a sharp increase in the share of Eriophorum, from 1 % to 13 %. Remains of Alnus and Pinus bark were found throughout the entire section. Their share was not large, only Pinus at a depth of 37–38 cm had a share of 18 %. However, this phase was characterized by the largest quantity of Alnus macrofossil remains.
-
LMAZ-2, 37–18 cm: this phase began with a significant increase in the share of Sphagnum Secc. Cuspidata (increase from 12 % to over 70 %). Compared to the previous phase, there was a clear decrease in the share of roots and uncategorized parts. Eriophorum, which occurred along the entire length of this phase, was characterized by a significantly greater share in the initial and final phases. The share of Eriophorum was clearly smaller at a depth of 24–31 cm and then fell below 10 %.
-
LMAZ-3, 18–7 cm: the boundary of this phase at a depth of 18 cm was determined by a significant decrease in the share of Eriophorum, whose share in this phase dropped to 0 %–2 % at subsequent depths. Sphagnum Secc. Cuspidata was still at a high level. Fragments of Pinus bark were found at all depths, with the maximum occurring at a depth of 14–15 cm (27 %). At a depth of 11–12 cm, Polytrichum commune appeared. However, its share in this phase was still small (1 %–4 %).
-
LMAZ-4, 7–2 cm: this phase was characterized by a clear and sudden increase in the share of Polytrichum commune and the appearance of Aulacomnium palustre at the expense of Sphagnum Secc. Cuspidata, whose share dropped even below 10 %. Eriophorum did not occur in this phase. In addition to Pinus bark, there were also numerous needles and a pine bud scale.
-
LMAZ-5, 0–2 cm: again, Sphagnum Secc. Cuspidata had a share of over 70 %. The presence of Polytrichum commune and Aulacomnium palustre decreased to 0 %.
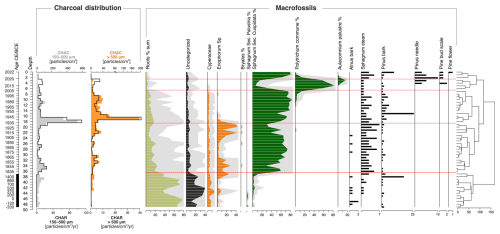
Figure 9Macrofossil diagram and charcoal distribution: influx (CHAR, particles cm−2 yr−1) and concentration (CHAC, particles cm−3) for macrocharcoal particles of two size classes, 150–500 µm (grey) and > 500 µm (orange), from the Wielkie Bagno peatland. Red dashed lines indicate the boundaries between five local macrofossil assemblage zones (LMAZ).
3.4.4 Macrocharcoal data (long-term fire evidence)
The period from approximately 330 BCE to 1400 CE was characterized by low and gradually increasing microcharcoal influx (CHAR) and concentration (CHAC). Macrocharcoal influx values for particles larger than 500 µm ranged from 0.01 to 0.07 particles cm−2 yr−1, while fragments in the 150–500 µm range varied between 0.1 and 0.5 particles cm−2 yr−1. A sudden shift occurred immediately after the hiatus at a depth of 37 cm (ca. 1830 CE), with a notable increase in fire activity, as macrocharcoal influx rose to 1.1–40 particles cm−2 yr−1. In the 100–500 µm fraction, influx values ranged from 0.1 to 108.5 particles cm−2 yr−1. The peak concentration and influx of macrocharcoal occurred at depths of 17 and 16 cm, corresponding to ca. 1941–1946 CE (Fig. 9).
3.4.5 Variation in biological assemblages across the peat sediment layers
The non-metric multidimensional scaling (NMDS) analysis based on pollen, plant macrofossil, and micro- and macrocharcoal influx data revealed clear compositional variation among biological assemblages across the peat sediment layers (Fig. 10). The k-means clustering approach identified five distinct clusters (Fig. 10a). Samples from cluster 4 and 1 stood out from the rest, representing, respectively, the oldest sediment layers below the hiatus, before ca. 1830 CE (37–50 cm), and the most recent surface layers after ca. 2015 CE (0–5 cm) (Fig. 10b). The remaining clusters were located closer to each other in the NMDS space, indicating higher similarity. However, cluster 2 stood out slightly from the others. Cluster 3 and 5 were the most similar, corresponding to the middle part of the record (ca. 1830–1960 CE), with cluster 3 including samples just after the hiatus (ca. 1830–1915 CE), and cluster 5 mainly representing samples after ca. 1915 CE. Samples in cluster 5 and 4 showed the highest internal similarity, with mean within-cluster Euclidean distances of 0.096 and 0.102, respectively. In contrast, the most modern samples from 0–12 cm depth (after ca. 1960 CE) were characterized by the highest internal compositional diversity of 0.202 (cluster 1) and 0.198 (cluster 2) (Fig. 10).
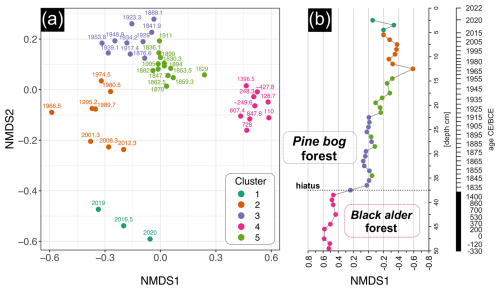
Figure 10The non-metric multidimensional scaling (NMDS) and k-means clustering of biological assemblages across peat sediment layers from the Wielkie Bagno peatland (stress = 0.14). The analysis was based on pollen data (> 1 %), plant macrofossil data, and micro- and macrocharcoal influx. (a) The NMDS ordination plot showing compositional variation among samples. Points are labeled by calibrated year (CE or BCE) and colored according to cluster group (k = 5). (b) Stratigraphic plot of NMDS1 axis scores by depth, illustrating temporal changes in biological assemblages' composition.
3.4.6 Hydrology reconstruction – testate amoebae data
Altogether, 43 species of testate amoebae were recorded in the peat core. The changes in the community of testate amoebae indicated four zones (Fig. 11):
-
TA-1, 48–36 cm: the lowest parts of the peat core were characterized by an extremely low concentration of testate amoebae. Single tests of Cryptodifflugia oviformis, Amphitrema wrightianum, Archerella flavum, Centropyxis aculeata-type, and Phryganella acropodia were recorded.
-
TA-2, 36–16 cm: this zone was dominated by organic testate amoebae (around 54 %–89 %) and characterized by a strong dominance of one species – Cryptodifflugia oviformis (around 35 %–88 % of the total number of testate amoeba tests). It was accompanied by Galeripora discoides (around 2 %–12 %). The remaining community of testate amoebae was mainly constituted by Amphitrema wrightianum, Archerella flavum, Centropyxis aculeata-type, Difflugia sp./Netzelia lithopila, Difflugia pulex-type, and Phryganella acropodia. The proportion of mixotrophic testate amoebae ranged from 1 % to 15 % and showed a decreasing trend. The Shannon diversity index of the testate amoeba communities ranged between 0.5 and 2.5. Depth to the water table (DWT) was unstable and oscillated between around 17.2 and 32.1 cm.
-
TA-3, 16–7 cm: in this zone the greatest change in the testate amoeba communities was observed. A rapid decrease of organic testate amoebae (around 35 %–68 %) and a rapid increase of idiosomic testate amoebae to around 54 % were recorded. Cryptodifflugia oviformis first decreased to around 23 %, and next increased to around 66 %. Its decline in the first half of the phase contrasted with an increase of Galeripora discoides to around 29 %. The contribution of Alabasta militaris significantly increased to around 30 %. The rest of the testate amoeba community was mainly constituted by Difflugia leidyi, Euglypha ciliata-type, and Euglypha compressa. A pronounced decline in mixotrophic testate amoebae was recorded, with their abundance becoming nearly absent. The Shannon diversity index of the communities ranged between around 1.2 and 2.2. A trend of a slight increase in the DWT was observed in this zone. DWT oscillated between around 17 and 32.1 cm.
-
TA-4, 7–0 cm: in the top layer of the peat core idiosomic testate amoebae were found to be dominant (around 86 %), including species such as Alabasta militaris and Nebela tincta, accounting for around 33 % and 27 % of the total community, respectively. Cryptodifflugia oviformis disappeared in this zone, whereas the contribution of Galeripora discoides increased to around 24 %. Towards the end of the zone, an increase in mixotrophic testate amoebae was observed to 4 %. The Shannon diversity index was high, varying between 2.3 and 2.5, whereas the DWT increased slightly (oscillation between around 13.9 and 20.1 cm).

Figure 11Testate amoebae percentage diagram for the Wielkie Bagno peatland with 10 times exaggeration (presented as black lines) and with quantitative reconstructions of depths to water table (DWT), diversity (Shannon index), sum of mixotrophic species and divided into test constructions of testate amoebae.
3.5 Human archives
Query in archives evidenced 133 cartography and written sources relevant for our study area. The majority of historical data was related to land use/economy (Table S2). The most detailed quantitative data were found for the land use and settlement intensity in the second half of the 1700s in the direct surroundings of our study site, documenting an increase by over half (51.9 %) in the rent paid for fields and meadows in the Tereszpol area between 1757 and 1775 (Table S3), as well as an increase in the number of hutters in both, the Zwierzyniec Key of the Zamoyski Family Estate (including Wielkie Bagno and Tereszpol) and in the vicinity of the Tereszpol village in the period 1759–1792 (Table S4). We recorded 20 historical maps depicting our study site and/or its immediate surroundings (Fig. 1f–i).
In this interdisciplinary study, data from natural (climate, peat sediment, tree rings from living peatland pines) and human archives (Fig. 2) allowed us to document a complete shift in a peatland ecosystem from Alnus to Pinus and Sphagnum dominance, which occurred on a time scale of over two millennia of our reconstruction. This transition from the alder bog forest (i.e., alder carr, alder swamp forest) to the Scots pine bog forest resulted in substantial changes in ecosystem's diversity and functioning (Figs. 7 and 10–11). To our knowledge, this is the first palaeoecological evidence of such a conversion in temperate Europe (cf. Barbier and Visset, 1997). Despite the challenges in unequivocal interpretations of certain aspects and some limitations of our data, we were able to demonstrate that the human impact on this transformation was undeniable.
4.1 Dynamics of the peatland ecosystem revealed by multiple archives
The archives we studied covered the last > 2300 years with different temporal resolution and overlaps (Fig. 2). Based on the longest record from the peat archive and the associated palaeoecological proxies, coupled with data from the other archives, we distinguished three main periods of our environmental reconstruction: (1) the oldest period, covering approx. 1700 years (ca. 330 BCE–1400 CE), (2) the transition period of approx. 430 years (ca. 1400–1830 CE), and (3) the last period of approx. 200 years (ca. 1830–2022 CE).
4.1.1 Alder (Alnus) bog forest (ca. 330 BCE–1400 CE)
The oldest period of our reconstruction was only covered by the peat archive (Fig. 2). A 25 cm long section of highly decomposed peat documents the beginning of the functioning of the Wielkie Bagno ecosystem in this part of the peatland basin. The very low peat accumulation rate of about 170 yr cm−1 on average (0.006 cm yr−1, Fig. 7) is one to two orders of magnitude lower than peat accumulation rates of approx. 0.05–0.4 cm yr−1, reported in the region (Lamentowicz et al., 2007; Mažeika et al., 2009; Dobrowolski et al., 2010; Stivrins et al., 2025). This suggests that conditions for organic matter accumulation were unfavourable and that continuous decomposition was taking place, as also evidenced by the extremely low concentration of testate amoebae we recorded (Fig. 11). There are several indications that our study site was covered by Alnus glutinosa bog woodland (i.e., alder carr, black alder swamp forest, black alder bog forest) during this period (Fig. 2). Alder bog forest is characterized by wet soils and frequent water level fluctuations, related to a high groundwater table and seasonal fluctuations of stagnant water (Faliński, 1986; Solińska-Górnicka, 1987; Leuschner and Ellenberg, 2017a). The quantity of Alnus macrofossil remains was highest in this phase. Further, the aforementioned low peat accumulation rate, high occurrence of roots and uncategorized parts, together with a low share of Sphagnum and a substantial share of Cyperaceae (including Eriophorum) macrofossils documented unstable moisture conditions (Shaver et al., 1986; Rydin and Jeglum, 2013; cf. Pędziszewska and Latałowa, 2016) (Fig. 9). The palynological data reflected a substantial share of deciduous forests in the area. In addition to Alnus, Carpinus, Quercus, Fagus, and Betula also had a relatively high share (10 %–20 %). Conifers in the surrounding landscape were represented by Abies, Picea, and Pinus, with the latter accounting for the largest proportion (share of approx. 25 %–40 %). This is consistent with historical accounts, revealing a diverse tree species composition and a high share of deciduous tree taxa in the Solska Forest before the 16th century (Kubrak, 2010). Interestingly, the relatively high proportion of Calluna pollen as well as high microcharcoal concentration (Fig. 8) indicate noticeable fire activity in the landscape around our study site during this phase of its history (Rösch, 2000; Pędziszewska and Latałowa, 2016). However, the very low macrocharcoal concentration (Fig. 9) suggests limited local fire occurrence, which was probably related to the low flammability of the ecosystem at this time. Considering the large number of sand dunes surrounding the Wielkie Bagno peatland (Popielski, 1992; Fig. 1c), these were probably the sites most strongly associated with fire at this time. Although the region was still largely uninhabited during this period (Szczygieł, 1985; Buraczyński, 2008; Kubrak, 2010), the anthropogenic pollen indicators (Artemisia, Plantago lanceolata-type, cereals) in the profile proved the human presence in the landscape. Urtica pollen showed a slight increase at 44 cm (ca. 200–370 BCE), suggesting a rise in the peatland trophy (Behre, 1981; Zarzycki et al., 2002) (Fig. 8). This coincided with a small decrease in the share of roots and an increase of uncategorized parts, Eriophorum and Cyperaceae, indicating a possible drop in the peatland wetness and providing further evidence of the hydrological instability of the ecosystem.
4.1.2 Transition period (ca. 1400–1830 CE)
Due to the discontinuous accumulation and the resulting hiatus, there is no peat sediment documenting this period (Figs. 2 and 7). However, the other archives have partially filled this gap in the stratigraphy, including annually resolved tree ring data for the last one hundred years of this phase, as well as mostly qualitative and often intermittent historical records encompassing mainly the period from the mid-18th to the mid-19th century. Information on earlier periods is limited (Fig. 2).
Yet, historical accounts demonstrate the crucial role of human activities in shaping the Solska Forest during the 16th–18th centuries. Settlement development in the area increased from the second half of the 16th century due to the expansion of the manorial-serf economy and the gradual shift from pastoralism and beekeeping to agriculture. This was closely linked to the emergence of a highly receptive market in Western Europe for grain, timber, and forest products such as ash, potash, and tar, leading to significant deforestation in the area, initially (and especially) on the more fertile soils. Large-scale deforestation approx. 15 km south of our study site, caused by potash and charcoal production, occurred in the late 1600s. One of the oldest settlements neighbouring the Wielkie Bagno peatland, the village of Tereszpol, located approx. 5.8 km northwest of our study site, was founded at the end of the 17th century. Its surroundings were intensively used for the production of ash, charcoal, wood tar, and wood shingles from the beginning of the 18th century, resulting in substantial deforestation (Kubrak, 2010) and increased colonization of the area (archival source 1, AS1, full list of archival sources given prior to the reference list; Tables S3 and S4). The most destructive forms of forest utilization in the Solska Forest during the 17th–18th centuries were ash burning and potash production, which led to deforestation and were facilitated by rapid settlement development in cleared areas (for example, the village of Aleksandrów, settled in 1791 approx. 8 km south of our study site) and the establishment of new towns and cities, including those close to our study site such as Biłgoraj (in 1578) and Józefów (in 1725) (Kubrak, 2010).
Historical data show also that, along with deforestation linked to settlement activity, the development of the forest industry, and forest use by peasants and townspeople, the tree species composition in the Solska Forest changed. The mixed forests, which consisted of tree species such as fir, spruce, beech, oak, hornbeam, pine, birch, lime, and yew, were gradually transformed as a result of selective use of certain preferred taxa such as oak or yew, of damage, especially to tree regeneration, caused by the forest grazing of domestic animals, and of ash burning (exceptionally affecting beech, but also oak and hornbeam). In consequence, the proportion of deciduous tree species decreased and the degree of forest openness increased, which in turn favoured the advance of light-demanding taxa such as Pinus, Betula, and Corylus. The establishment of pine was particularly facilitated by fires, as it colonized burned areas faster than the other tree species. By the end of the 18th century, Scots pine was already predominant in the Solska Forest, while fir, beech, spruce, and hornbeam were only found locally (Kubrak, 2010). Forest management maps of the Wielkie Bagno peatland confirmed the dominance of pine with admixture of spruce and beech in the late 18th century (AS2; Janeczek et al., 2015). The map and its description from 1779 documented our study site and its immediate surroundings as a very wet and marshy area interspersed with dense coniferous forests, pastures, and meadows (Janeczek et al., 2015). Land survey data from the mid-1780s for the Tereszpol area, collected for the Austrian land cadastre (Ger. Josephinisches Lagebuch) (AS3) following the first partition of Poland in 1772 and the resulting new governmental and legal order (Piller, 1782; Davies, 2005; Jones, 2016; Carvalho, 2018), generally classified the surroundings of our study site as a plain with sandy soils. The pastures and forest clearings in the area were given a low utilization value, and several hundred of hectares were categorized as waterlogged and economically unreasonable. Forests were classified as pine forest, with aspen in admixture, suitable for timber production over several decades. Based on tree ring samples taken from five different sites in the area, the approximate age of the stand was determined to be 31–55 years (AS3). This indicates that the forests in the vicinity of the Wielkie Bagno peatland established around 1730–1750, which corresponds well with our tree ring data (Fig. 4).
Because of the hiatus in our peat archive (Fig. 2), we cannot corroborate this picture with pollen, macrofossil, and sedimentary charcoal data, which are missing for this period of the ecosystem's history. However, our palaeoecological record for the neighbouring periods (Figs. 8–9), together with the tree ring archive covering the last hundred years of this phase (Fig. 4), supports the findings from historical accounts on factors affecting our study site and its surroundings until the late 18th century (Kubrak, 2010; AS1–AS3; Tables S3 and S4), highlighting the value of both archival sources and interdisciplinary, multi-proxy studies.
The number of proxies provided by various archives increases in the early 19th century (Fig. 2). This phase of our reconstruction indicates a cohort regeneration of Scots pine in the 1820s, as documented by the tree ring data (Fig. 4), likely associated with several possible factors that substantially altered local environmental conditions, such as peatland drying, including drainage (Linderholm and Leine, 2004; Freléchoux et al., 2000, 2003, 2004; Edvardsson et al., 2012a, b, 2014, 2015a), clear cutting (Freléchoux et al., 2003, 2004), or fire (Filicetti and Nielsen, 2020). Based on tree ring data on early Scots pine growth under similar habitat conditions in the Białowieża Forest, northeastern Poland, which showed seven years as the time needed to reach breast height (i.e., 1.3 m) (Ewa Zin, Kamil Pilch, and Marcin Klisz, unpublished data), and on the relatively high growth rate of our sample trees at a young age (Fig. S6), we propose that seven years may be a reliable value for determining actual tree recruitment dates. Hence, the pine cohort at our study site most likely originated from 1816–1820, during a period of unfavourable tree growth conditions, as evidenced by a growth depression recorded in older pines, revealed by both the site chronology (Fig. 4) and the individual tree growth series (Fig. S6). This growth depression followed a positive pointer year, 1813 (recorded by all sample trees), and a notable signature year, 1812, the latter characterized by both positive and negative growth reactions (Figs. 4 and S6) recorded by three (33.33 %) and six (66.67 %) sample trees, respectively, suggesting fire disturbance rather than climate as the possible main driver of tree growth (Zin et al., 2015).
We acknowledge that our dendrochronological fire record was limited by our sampling design. The procedure used was not designed for a tree ring fire history study (Arno and Sneck, 1977; McBride, 1983; Van Horne and Fulé, 2006), so it could not provide a complete inventory of fire years, especially considering the complex process of fire scar formation in trees (McBride, 1983; Farris et al., 2010; Piha et al., 2013). Nevertheless, fires in the Solska Forest during this period of our reconstruction are mentioned in the historical data (AS6–AS7; Kubrak, 2010). Moreover, fire disturbances have been documented as frequent and probably crucial for the development and dynamics of peatland ecosystems in temperate latitudes (Marcisz et al., 2015; Sutheimer et al., 2021). They have also been shown to both limit and enhance Scots pine growth, not only in mineral soils (Blanck et al., 2013; Zin et al., 2015), but also in peatlands (González de Andrés et al., 2022). Therefore, we interpret the marked growth anomaly recorded in 1812 as most consistent with a fire disturbance affecting our study site. The absence of fire scars in our wood samples (Fig. 4) does not preclude a fire event: low-intensity surface fires, confined to the ground (or peat) layer, or fires that did not sufficiently damage tree stems, may leave little or no scarring (Piha et al., 2013). Importantly, the occurrence of fire in situ within a peatland would imply exceptionally dry conditions, as peat substrates and wetland vegetation typically limit ignition and fire spread unless the water table is strongly lowered (Sillasoo et al., 2007; Väliranta et al., 2007). In addition, it is also possible that the growth response observed in our sample trees in 1812 was a reaction to a disturbance in the previous year, 1811, as the effects of fire on radial tree growth can manifest with a one-year delay (González de Andrés et al., 2022). Remarkably, both the historical and climate data for 1811–1812 support our conclusion that a fire disturbance was very likely at our study site in 1811 or 1812. The activity protocols for the Zamoyski Family Estate frequently mention the severe drought in the summer of 1811, leading to crop failures and numerous forest fires, and report crop failures in 1812. Records from the municipality of Tereszpol village, located near our study site, contain complaints about drought and crop failures at the turn of July and August 1811 (AS6–AS11). This is consistent with the climate data for the region documenting very dry and very warm summers during 1811–1812 (Fig. 3).
Since peatland fires are well documented as effective peat consumers regardless of combustion type (smouldering or flaming) (Lavoie et al., 2005; Benscoter and Vitt, 2008; Turetsky et al., 2015), the fire in 1811 or 1812 is also a very likely cause of the hiatus in our peat archive (cf. Marcisz et al., 2015). Another fire that could be responsible is the one in 1830, which marks the end of this period in our reconstruction and which all our sample trees survived (Fig. 4). This fire was recorded by almost all of them (93.33 %) as a narrow or wide ring (cf. Fig. S6), or as a fire scar by one of our sample trees (Fig. 4). As for 1812, we interpret this different growth response in 1830 as a fire effect, relying on existing data showing both positive and negative post-fire growth responses in Scots pine (Blanck et al., 2013; Zin et al., 2015; González de Andrés et al., 2022). Interestingly, a negative growth response was found in both 1830 and the following year, 1831 (Figs. 4 and S6), with both years classified as negative pointer years (data not shown), confirming the one-year lag in fire impact (González de Andrés et al., 2022). Further arguments in favour of fire as the cause of the hiatus in our peat sediment chronology are a pronounced horizon rich in macroscopic charcoal observed in the peat core at the transition between the two lithological segments (see Sect. 2.4.1), and climate data from Kraków documenting the first decades of the 19th century (1812–1828) as a distinctly dry and warm period (Figs. 3 and S3), which could have resulted in increased fire occurrence (González de Andrés et al., 2022).
Nevertheless, drought alone cannot be completely ruled out as a possible reason for the hiatus in our sedimentary record (Van Geel et al., 1980), since a drop in the water table leads to peat oxidation and mineralization, resulting in peat decomposition and loss (Päivänen and Hånell, 2012; Joosten, 2016). However, we consider this explanation unlikely due to the high interannual hydroclimatic variability during the generally dry early 19th century (Fig. S3), as evidenced by our climate data (Figs. 3 and S3) and historical accounts (AS4–AS14). Wet conditions were documented in 1813 and 1815 (Figs. 3 and S3), and locally also in 1814 (AS14). Furthermore, floods in the Solska Forest, including areas several to a dozen kilometres from our study site and rivers such as the Łada, Branwia, Bukowa, San, and Tanew, were recorded in 1813 (August), 1814, and 1815 (AS11–AS12; AS14).
Climate data (Figs. 3 and S3) also support the interpretation of the tree growth–climate relationship in the early decades of the 19th century. We demonstrated a positive correlation between the radial increment of peatland pines and hydrological parameters: precipitation, SPEI3, and SPEI6 in spring and summer (Figs. 6 and S5), consistent with data from southwest Siberia, the northeastern Italian Alps, Sweden, Lithuania, Belarus, and northern Poland (Linderholm et al., 2002; Vitas and Erlickytė, 2007; Cedro and Lamentowicz, 2011; Edvardsson and Hansson, 2015; Dinella et al., 2021; González de Andrés et al., 2022; Ignatiev and Yermokhin, 2022). This effect may be related to water stress experienced by peatland trees during dry periods when the water table is low, which can reduce physiological processes such as stomatal conductance or photosynthesis (Dang et al., 1991; Pepin et al., 2002), as well as plant growth, including root development (Pearson et al., 2013). Water availability during the growing season is typically a limiting factor for Scots pine radial growth at mineral soil sites, as widely reported across Eurasia, including Poland (Linderholm et al., 2002; Opała, 2015; Misi et al., 2019; Harvey et al., 2020; Waszak et al., 2021; González de Andrés et al., 2022; Janecka et al., 2025). In our study, the very high growth in 1813 observed in all sample trees (Figs. 4 and S6) may reflect a positive growth response to high summer precipitation (Fig. 3; AS11–AS12), a reaction to the fire disturbance in 1811 or 1812 (as mentioned above), or a combination of both. However, the lack of other environmental proxies (testate amoebae, sedimentary charcoal, or fire scar data) limits our ability to definitively disentangle these effects.
4.1.3 Sphagnum & Pinus expansion – transformation into Scots pine bog forest (ca. 1830–2022 CE)
The next phase in the development of the Wielkie Bagno peatland was characterized by the gradual increase in the dominance of Sphagnum and Pinus in the peat profile. Noteworthy, already the end of the earlier phase in the peat archive was marked by a slight increase of Sphagnum and the highest proportion of Eriophorum macrofossils (approx. 20 %), suggesting the beginning of a gradual development towards more acidic, ombrotrophic conditions (Fig. 9). For the last period of our environmental reconstruction, we were initially able to distinguish three sub-periods based on the macrofossil data, which were later confirmed by the other peat sediment proxies and are reflected in the climate, tree ring, and historical data.
Ca. 1830–1947 CE
The period 1830–1947 CE was characterized by a gradual decline of deciduous trees in the landscape such as Quercus, Carpinus, Corylus, Fagus, Betula, and Alnus, and a continuous increase in the Pinus occurrence. Share of the non-arboreal pollen (NAP) was high, including Poaceae, cereals, and several other anthropogenic indicators such as Plantago lanceolata-type, Rumex, and Cannabis sativa indicating the increasing human impact on the study site and its surroundings (Behre, 1981; Poska et al., 2004) (Fig. 8). The changing share of Eriophorum macrofossils, with high values in the beginning and in the end of this period, proved unstable moisture conditions (Shaver et al., 1986; Rydin and Jeglum, 2013; Marcisz et al., 2015; Słowiński et al., 2016) (Fig. 9), which is also supported by our reconstruction of depth to the water table (DWT). This period is marked by the presence of Galeripora discoides, which may indicate hydrological instability of the Wielkie Bagno peatland during that time (Lamentowicz et al., 2009; Sullivan and Booth, 2011; Łuców et al., 2021, 2022). However, the testate amoeba community was dominated by Cryptodifflugia oviformis, a species typically associated with dry habitats (Mazei and Tsyganov, 2006; Łuców et al., 2020), suggesting prevailing dry conditions despite signs of hydrological instability. The overall trend of declining water levels observed on Wielkie Bagno (Fig. 11) aligns with the broader understanding that most European peatlands were drier than in the past during this period (Swindles et al, 2019).
Numerous European studies, also from northern Poland, have shown that peatland hydrology is driven by both climate and human impacts, often interacting (e.g., Lamentowicz et al., 2009; van der Knaap et al., 2011; Gałka et al., 2014; Łuców et al., 2021). We cannot exclude a possible climate forcing upon fluctuations of the water levels we found (Fig. 11), since several exceptionally cold and dry years fell in the periods characterized by the strongest water level declines in our record (notably 1856, 1858, and 1869 in the 1850s/60s; 1894 in the 1890s; and 1942, 1944, and 1948 in the 1940s), as shown by the climate data (Fig. 3). Nevertheless, we consider the connection between the lowering of the water table in the Wielkie Bagno peatland and the drainage activities to be very probable. The historical data we examined revealed that drainage measures were carried out in our study site and its immediate vicinity from the late 19th to the first half of the 20th century (Fig. 1f–i). As a very wet and swampy area (Janeczek et al., 2015), the Wielkie Bagno peatland and its surroundings posed significant management challenges. Estate maps from the second half of the 19th century showed that attempts to utilize the wetlands and floodplains around our study site were successfully implemented. The Rakowe Bagno peatland, located directly south of the Wielkie Bagno peatland, was drained between around 1850 and 1939 to create land for cattle grazing (AS15; Fig. 1g). Further drainage activities were undertaken after the end of the partition period (i.e., after World War I), when the new Polish authorities continued the earlier drainage plans. Two maps from the first half of the 20th century show a considerable increase in the drainage system, especially in the number of ditches located directly in the Wielkie Bagno and the Rakowe Bagno peatlands (Fig. 1h–i). Remarkably, the ditch located in the central part of the Wielkie Bagno peatland, along its west-east axis, remains visible in the terrain topography, as evidenced by the digital terrain model (Fig. 1c).
The hydrological instability of our study site as proven by the DWT fluctuations we found (Fig. 11), coupled with the climatic conditions during this period (Figs. 3 and S3), were important for shaping the temporal variability in climate sensitivity of peatland trees during this phase (Figs. 6 and S5). The climate data documented the beginning of this period (1829–1855) as humid (Figs. 3 and S3). The entire period (1830–1947) proved to be generally cold, with a slight warming in the last phase, in the 1930s–1940s (Fig. 3), which is consistent with existing climate reconstructions (Büntgen et al., 2021b; Björklund et al., 2023). Under the overall wet and cold conditions in the first part of this period, until the late 19th century, our tree ring archive showed a growth-limiting effect of high temperatures during the growing season (spring–summer) (Fig. 6), which is consistent with data from drained peatlands in northern Poland (Cedro and Lamentowicz, 2008, 2011; Cedro and Sotek, 2016), suggesting that drainage measures in our study area had the predominant effect on pine growth at this time. In the second half of the 19th century, this effect was combined with the negative correlation between mean air temperature in autumn of the current year (October–November) and tree growth (Fig. 6), which agrees with data from southwest Sweden, where a comparable relationship was found for September–October. However, as this study (Janecka et al., 2025) did not discuss the eventual impacts of drainage (especially in the area surrounding the study site), it is not readily possible to judge whether the recorded pattern is a direct analogue for our results. By contrast, the relationship between tree growth and climate that we found in the last phase of this period, from the late 19th to the first half of the 20th century, demonstrating the positive correlation with precipitation and SPEI in early summer (May–July) (Figs. 6 and S5), can be associated with aforementioned drainage activities in our study area (Fig. 1f–i), lowering of the DWT (Fig. 11), and the generally dry conditions (Figs. 3 and S3). Interestingly, it aligns with our results for the first decades of the 19th century (1812–1828) (Figs. 6 and S5) and is consistent with numerous studies across Eurasia (e.g., Edvardsson and Hansson, 2015; Dinella et al., 2021; González de Andrés et al., 2022; see earlier discussion, Sect. 4.1.2).
The gradual decline in microcharcoal particles and Calluna pollen (Fig. 8) suggests a progressive decrease in landscape-level fire activity (e.g., Clark, 1988; Vannière et al., 2008; Conedera et al., 2009) during this phase of peatland history, which agrees well with data demonstrating a continuous reduction in the forest fire frequency over the last centuries as a consequence of land use change and fire suppression both regionally (Rolstad et al., 2017; Ryzhkova et al., 2020; Zin et al., 2022) and globally (Nowacki and Abrams, 2008; Wallenius, 2011). Nevertheless, the sediment charcoal data (both microcharcoal and macrocharcoal) confirm that fire disturbance still occurred in our study area and its surroundings during this period, especially in the 1830s–1860s, in the late 19th century (1880s–1890s), and around 1940–1945, as shown by the peaks in charcoal distribution we recorded (Figs. 8–9). The highest distribution of the macrocharcoal particles (both in terms of influx and concentration), documented around 1940–1945 (Fig. 9), was probably related to various wartime events, including resistance movements or other military operations that took place in the vicinity of the Wielkie Bagno peatland during World War II (Klukowski, 1945–1947). As already mentioned, our tree ring data confirmed fire disturbance at our study site in the first half of the 19th century (Fig. 4; Sect. 4.1.2).
Besides records of drainage during this period of our reconstruction (Fig. 1f–i), the human archives also provided valuable data on the forest structure and logging campaigns. The forest inventory of 1924 (AS16) shows that the stands around the peatland were dominated by pine populations of various age classes from 70 to 160 years, which is consistent with forestry documents from the late 18th century that demonstrate a high proportion of pine forests in the area even earlier (AS2; Sect. 4.1.2). As the leaving of seed trees was used in forest management in the early 20th century (AS16), some individuals may have been older. Thus, the 1924 forest inventory data reflect effective pine recruitment in the 1760s–1850s, probably even earlier. This agrees with our tree ring data (Fig. 4) and confirms the conditions for successful tree establishment in our study site both in this and the earlier period of its history. However, there were also periods of increased tree felling during this phase. In 1844, the decision of the then landowner to reduce the number of professional forest managers and transfer responsibility for the forests to the peasants resulted in considerable deforestation and devastation (Rajca, 1972). The abolition of serfdom in the 1860s was followed by increasing, often unregulated or illegal logging by peasants who saw tree felling as a way of realizing their new land rights (Mazurek, 1957). In the early 20th century, the forests surrounding our study site became part of the Tereszpol Forest District and were divided into concessions and forest sub-districts (AS20). After World War I, some of the estate's forests were sold to support its rebuilding. The increasing demand for timber in the 1920s triggered further intensive logging that levelled off after the introduction of rational forest management in the 1930s (Kozaczka, 2002).
Ca. 1947–2004 CE
The second half of the 20th century brought further changes in the development of the Wielkie Bagno peatland. A high proportion of Sphagnum was accompanied by the disappearance of Eriophurum macroremains and a further increase in Pinus pollen (Figs. 8–9), documenting the gradual acidification of the site (Clymo, 1984; Koks et al., 2025), which was possibly a consequence of the increasing share of pine-dominated forests in the region, including our study area (e.g., Lamentowicz et al., 2007; Słowiński et al., 2019; Bąk et al., 2024; Marcisz et al., 2025). This process is also reflected in the strong decrease in the proportion of Populus, Picea, and Abies pollen during this period, as well as in a further decline in deciduous tree taxa (such as Quercus, Carpinus, Corylus, Fagus, and Alnus). The presence of anthropogenic pollen indicators proves the human impact on the landscape surrounding our study site. Interestingly, this phase of peatland history is characterized by a very sharp reduction in Calluna pollen (Fig. 8). This could be related to the general decline in fire occurrence during this period, as shown by the decrease in micro- and macrocharcoal distribution, with only sporadic fire activity (Figs. 8–9), apparently indicating further land use changes and fire suppression measures in the region (cf. Ryzhkova et al., 2020; Zin et al., 2022) that would likely eliminate frequent surface fires, which in turn would lead to decreasing forest openness (Niklasson et al., 2010; Zin et al., 2015), being definitely unfavourable for the light-demanding heather Calluna vulgaris (Ellenberg et al., 2001; Zarzycki et al., 2002; cf. Skre et al., 1998). Around 1980 (depth: 11 cm), a decrease in Pinus pollen was observed with a simultaneous increase in non-arboreal pollen (such as Poa) (Fig. 8), indicating an increase in the landscape openness (Sugita et al., 1999). This could be the result of clear-cutting in the vicinity of our study site. Indeed, about 450–500 m southwest, there is a group of pine-dominated stands currently 37–46 years old (total area: 10.25 ha) and several other patches of younger pine stands (aged 30–50 years) surrounding our peat sampling site (Forest Data Bank, https://www.bdl.lasy.gov.pl/portal/mapy, last access: 19 April 2024), which are a legacy of logging operations in the 1970s–1990s.
The testate amoebae data showed a rise in water level, as demonstrated by the DWT reconstruction. Additionally, the strong increase in the proportion of Galeripora discoides (an indicator of wet conditions), combined with a decrease in Cryptodifflugia oviformis (an indicator of dry conditions) (Fig. 11), provides further evidence of water level fluctuations during this phase of our reconstruction (Mazei and Tsyganov, 2006; Lamentowicz et al., 2009; Sullivan and Booth, 2011; Łuców et al., 2020, 2021). Furthermore, the decline and near disappearance of mixotrophic taxa in the second half of the 20th century (Fig. 11) can be linked to drying conditions and environmental disturbances (Marcisz et al., 2016; Zhang et al., 2020). These changes in peatland hydrology may be related to both hydroclimatic conditions and nationwide land use transformations during this period, including shifts in population density due to migration associated with new state boundaries, fluctuations in arable land, and large-scale afforestation campaigns (Broda, 2000; Gorzelak, 2004). Climate data show that this period was relatively dry and has warmed since the 1980s (Figs. 3 and S2), consistent with regional and global trends (Degirmendžić et al., 2004; Büntgen et al., 2021a; Björklund et al., 2023). Together with the aforementioned DWT dynamics (Fig. 11), this aligns with the tree growth response we have documented. During this period, we observed a clear negative correlation between pine growth and summer (August) temperatures, along with a positive correlation with precipitation in late winter–early spring (February). Our results are comparable with data from northern Poland, where a negative impact of high temperatures in summer (June, August) (Cedro and Sotek, 2016) and a positive (although not always significant) relationship between tree growth and precipitation in February and March (Cedro and Lamentowicz, 2008, 2011) were found in some Scots pine populations growing in peatlands. Likewise, a growth-promoting effect of precipitation in February or March has been documented for Pinus sylvestris in peat soils in southern Sweden (Linderholm et al., 2002; Edvardsson and Hansson, 2015). These results may be particularly related to snow cover dynamics. The advanced biological spring and the general decrease in the frequency and duration of snow cover in recent decades reduce snowmelt water availability and can lead to early spring drought (Degirmendžić et al., 2004; Kreyling, 2010), which would explain the positive response of tree growth to precipitation. Conversely, the growth-accelerating effect of warm winters, indicating an earlier onset of secondary meristem activity or photosynthetic activity in winter, can lead to higher storage of non-structural carbohydrates, facilitating tree growth in spring irrespective of precipitation (Michelot et al., 2012; Almagro et al., 2025). Notably, we also recorded a negative effect of wet conditions (precipitation and SPEI3) in spring, summer, and autumn (Figs. 6 and S5). Besides the complex relationship between tree growth and hydroclimatic conditions, this may reflect a possible recovery of the Wielkie Bagno peatland after previous disturbances, such as drainage activities in the earlier period (Fig. 1f–i). Our data are consistent with results from Sweden, Lithuania, and western Siberia, where a negative correlation between Pinus sylvestris radial growth and spring (March) and summer (August) precipitation has been documented in undisturbed peatlands (Linderholm, 1999; Edvardsson et al., 2015b; Blanchet et al., 2017; Janecka et al., 2025).
Ca. 2004–2022 CE
The most recent period in the history of the Wielkie Bagno peatland (ca. 2004–2022 CE) was characterized by a decreasing share of non-arboreal pollen (NAP) and a decline in anthropogenic indicators, including cereals and taxa such as Plantago and Rumex (Fig. 8). This may reflect a reduction in agriculture in the region, leading to land abandonment and a consequent increase in forest cover (Gorzelak, 2004). In general, the regional vegetation and landscape features documented in the previous phase, such as the increasing proportion of Pinus and limited fire occurrence (Figs. 8–9), persisted. The observed DWT fluctuations and the high proportion of Galeripora discoides (Fig. 11) also demonstrate the hydrological instability of our study site (Lamentowicz et al., 2009; Sullivan and Booth, 2011; Łuców et al., 2021) as in the preceding period. During this phase, macrofossils of Polytrichum commune and Aulacomnium palustre appeared suddenly at the expense of Sphagnum Secc. Cuspidata (Fig. 9), indicating a disturbance in peatland hydrology and nutrient content (Toet et al., 2006; Purre and Ilomets, 2018) around 2005–2020 CE. An increase in mixotrophic testate amoebae in the upper part of the record (Fig. 11) may indicate a return to more stable and wetter conditions (Marcisz et al., 2020b; Łuców et al., 2022). However, the dominance of idiosomic testate amoebae (Fig. 11), including dry indicators such as Alabasta militaris and Nebela tincta (Koenig et al., 2018), suggests drying of the Wielkie Bagno peatland during this period. This is consistent with data from other studies in northern Poland (Gałka et al., 2022) and Europe (Swindles et al., 2019), which document peatland drying in recent decades as a result of climate change, particularly warming (Swindles et al., 2019).
As elsewhere (Ustrnul et al., 2021; Marosz et al., 2023), the climate data in our study area also show a substantial increase in temperature since the beginning of the 21st century (Figs. 3 and S2). We associate this with the negative correlation between pine growth and temperature, and the growth-accelerating effect of a positive water balance (precipitation, SPEI3) in autumn (September–November) of the current season, which we have documented (Fig. 5). Remarkably, the positive correlation of Scots pine growth with precipitation and the negative correlation with temperature during the growing season (spring–summer) have been reported from both mineral and peat soil sites, albeit with highly variable and not always statistically significant results (Linderholm et al., 2002; Cedro and Lamentowicz, 2011; Edvardsson and Hansson, 2015; Edvardsson et al., 2015b; Blanchet et al., 2017; Janecka et al., 2025). A possible explanation for our results (Fig. 5) is that the trees extend their growing season beyond summer due to climate change (Radosław Puchałka and Marcin Klisz, unpublished data) and thus also respond to hydroclimatic conditions in the autumn months. Photoperiod and air temperature have been proven to interactively affect the growth cessation of both conifers and deciduous trees. Although data are much scarcer than for growth onset, it has been demonstrated that global warming can either accelerate or delay the growth cessation of temperate trees, depending on the species or ecotype (Hänninen and Tanino, 2011). For three conifers, including Scots pine, it has been demonstrated that under a temperate climate, xylogenesis in latewood (both cell enlargement and cell wall thickening), lasted until September–November in the early 21st century (Cuny and Rathgeber, 2016). There is also evidence that growth cessation of Pinus sylvestris in peat soils can occur as late as September, even though data on the wood phenology of peatland pines are extremely limited and cover very short periods (no longer than two years) (Smiljanić and Wilmking, 2018; Francon et al., 2024). However, the last two decades of peatland history also showed a growth-limiting (albeit not significant) influence of wet conditions (positive water balance, high precipitation) in spring–summer (Fig. 5), which was already observed in the previous phase. As such a relationship has also been found in undisturbed peatlands (Linderholm, 1999; Edvardsson et al., 2015b; Blanchet et al., 2017; Janecka et al., 2025), we see this as a possible indication of ecosystem recovery, supported by the recorded rise in mixotrophic testate amoebae (Fig. 11) (Marcisz et al., 2020b; Łuców et al., 2022). Interestingly, this finding does not align with the aforementioned evidence of drying documented during this period (Figs. 8–9 and 11), which highlights the complexity of both peatland hydrology and the peatland tree growth response, as discussed in numerous studies (e.g., Linderholm et al., 2002; Edvardsson and Hansson, 2015; Dinella et al., 2019). Furthermore, it suggests that tree ring width alone may not be a sufficient proxy for assessing the climate–growth relationship of peatland trees (Dinella et al., 2021; Janecka et al., 2025).
4.2 The turning point – ecosystem change: its reasons and effects
The documented shift from an Alnus- to a Pinus- and Sphagnum-dominated ecosystem (Fig. 10), which occurred during the transition period of our reconstruction (ca. 1400–1830 CE), likely had complex and interacting origins. Therefore, it may be difficult, if not impossible, to identify a single main cause, especially since this period is not fully recorded in the archives we have analysed (Fig. 2). However, as reflected in the ecosystem and landscape dynamics discussed previously (Sect. 4.1), we propose that this habitat transformation began in the 16th to 18th centuries, and it is impossible to deny the role of humans in this ecosystem change, with several factors involved that we will review in more detail. In addition to human influence at the local and landscape levels, other interrelated environmental phenomena and processes must also be considered.
4.2.1 Human impact on the landscape: settlement pressure, deforestation, and timber-oriented forest management
Our study demonstrates the crucial role of human impact in shaping the long-term dynamics of the Wielkie Bagno peatland and its surroundings, as has been shown for the entire terrestrial biosphere (Ellis et al., 2021). Although the Solska Forest was not densely populated until relatively late – the end of the 16th century (Szczygieł, 1985; Buraczyński, 2008) – the area was not entirely free from human pressure, as confirmed by the presence of anthropogenic pollen indicators in the oldest part of our reconstruction (Fig. 8). Nevertheless, the relatively low level of human activity recorded at that time was clearly not conducive to significant landscape changes, including deforestation (Samojlik et al., 2013a), similar to other locations in northeastern Poland, where continuous forest cover was never fully disturbed (Zimny et al., 2017; Jaroszewicz et al., 2019), or where forest decline occurred much later (16th–17th centuries) compared to sites in the north-western and north-central parts of the region (7th–9th centuries) (Marcisz et al., 2020a; Czerwiński et al., 2022).
Our study site and its surroundings were possibly used by inhabitants of neighbouring villages and forest settlements from the mid-17th century (AS17). Local settlement development followed the expansion of the traditional forest industry in the area (Kubrak, 2010; Róg, 2021). Clusters of forest settlements were located along the nearby Ratwica and Rakówka streams, among others. By the beginning of the 18th century, some tar workers lived in the nearby village of Tereszpol and the surrounding forests (AS18). The inhabitants of Tereszpol and Hedwiżyn established enclaves of forest meadows near our study site (AS19). Our results on dynamic settlement development in the second half of the 18th century (Tables S3–S4) correspond well with regional data. In Europe, a steady increase in human population has been documented since 1700 CE, together with an increase in anthropogenic ecosystem changes (Ellis et al., 2021), including substantial deforestation of large parts of the continent (Kaplan et al., 2009). For northern Poland, several studies have confirmed the growing human impact on forest and peatland ecosystems, with comparable timing of its onset: 1720s CE (Marcisz et al., 2020a), ca. 1750 CE (Słowiński et al., 2019), 1790s/1800 CE (Czerwiński et al., 2021), 1830s (Bąk et al., 2024). At sites near the Wielkie Bagno peatland, an increase in human activity was recorded in the uppermost sections of the peat profiles, as shown by pollen of anthropogenic indicators such as Artemisia, Asteraceae, Plantago lanceolata, Rumex, Urtica, Chenopodiaceae, and cereals. These profile sections have all been assigned to the Subatlantic chronozone, although different Common Era dates have been attributed to them due to the varying temporal resolution of radiocarbon dating and the resulting time spans covered by the peat archives (Bałaga, 1998; Korzeń et al., 2015; Margielewski et al., 2015, 2022). The most comparable time frame was reported from the Kobyle Jezioro peatland (approx. 45 km southeast), where the beginning of the youngest period in its history was dated to 60 ± 70 BP (i.e., OxCal calibrated 1814 CE, OxCal v4.4.4, 2023) (Korzeń et al., 2015). Similarly, in the Obary and Imielty Ług peatlands, located west of our study site (approx. 18 and 47 km, respectively), the highest proportion of anthropogenic plants was documented in the most recent phase of vegetation development. However, the absence of radiocarbon dating precludes precise dating of this period (Mamakowa, 1962).
In all the above palaeoecological datasets, an increase in human impact indicators is associated with a decrease in the proportion of deciduous tree taxa and an increase in the share of Pinus pollen, often coupled with a rise in non-arboreal pollen of terrestrial plants indicating landscape openness (e.g., Korzeń et al., 2015; Margielewski et al., 2015, 2022; Słowiński et al., 2019; Bąk et al., 2024). This is fully consistent with our palaeoecological record (Fig. 8) and the previously discussed landscape-scale environmental changes that affected our study area during the transition period of our reconstruction (Sect. 4.1.2). Deciduous trees such as hornbeam (Carpinus), birch (Betula), beech (Fagus), and oak (Quercus) were used extensively for firewood, charcoal, ash, potash (especially white potash), wood tar, construction timber (including shipbuilding material), shoemaking and tanning, and everyday objects (Kubrak, 2010; Samojlik et al., 2013b; Cywa, 2018), resulting in the decline of deciduous forests in the landscape. Generally, increasing human presence often leads to greater landscape openness caused by deforestation related to timber harvesting (Broda, 2000; Kubrak, 2010; Czerwiński et al., 2022), agricultural development (Kaplan et al., 2009; Ellis et al., 2021), or anthropogenic fire (Marcisz et al., 2015; Dietze et al., 2019; Bonk et al., 2022), which favours the spread of light-demanding pioneer tree species (Svenning, 2002; Dyer, 2010; Kubrak, 2010) such as Scots pine, especially on poor and dry soils, where it can outcompete other tree taxa due to its broad edaphic amplitude (Przybylski, 1993) or its high tolerance to frequent fire disturbances of both low and high intensity (Kubrak, 2010; Niklasson et al., 2010; Keeley, 2012; Zin et al., 2015).
However, the increase in Pinus presence in the landscape may also result directly from human activities. Pinus sylvestris has been widely introduced into the forests of Central Europe over the past 200 years (Broda, 1993; Schabel, 2001; Timbal et al., 2005; McGrath et al., 2015), leading to its large-scale dominance in the region, including Poland (Timbal et al., 2005; Leuschner and Ellenberg, 2017c; Zajączkowski et al., 2023). This was closely linked to the concept of sustainability (Ger. Nachhaltigkeit) and sustainable forest use, initiated by von Carlowitz (1713) and further developed in German-speaking Central Europe in the late 18th and 19th centuries. Its basis was rationalization, measurement, and calculation, with the main focus on the production of wood biomass and its economic value. Consequently, rules for quantitative forest management were established, including the division of forests into management units, the formulation of rotation ages, and the creation of forest yield tables allowing effortless estimation of wood mass. Besides sustained yield, minimum diversity was one of the key principles, as it enabled easy assessment, control, and prediction of timber biomass. Therefore, even-aged monocultures were strongly favoured (Lowood, 1990). The German school of forest management spread to neighbouring areas, including the Zamojski Family Estate, whose owners valued highly qualified managers from abroad and gradually introduced the principles of quantitative forest management into their estate from the 1770s onwards (Rajca, 1972).
Land use changes, such as deforestation, can lead to hydrological shifts that promote the formation of new peatland types (Warner et al., 1989). Studies north of our study area (northern Poland) have shown that the introduction of pine monocultures into forest management has played a significant role in the dynamics of peatland ecosystems (e.g., Słowiński et al., 2019; Łuców et al., 2021: Bąk et al., 2024, 2025). A high proportion of Pinus plantations in the catchment area can lead to peatland acidification, which in turn can stimulate the expansion of Sphagnum (Lamentowicz et al., 2007; Bąk et al., 2024, 2025; Marcisz et al., 2025; Tipton et al., 2026). Given the dunes surrounding the Wielkie Bagno peatland (Fig. 1c) and documented evidence from Poland that runoff from pine-covered sandy areas causes peat acidification, facilitating peat moss establishment (Marek, 1965), we consider this scenario highly likely at our site. In addition, colonization by Sphagnum can exacerbate acidification (Gorham et al., 1987) through cation exchange between the cell walls of the moss plants and the peatland water (Clymo, 1964, 1987; Wheeler, 1992). Conifer monocultures are generally known to cause environmental acidification by altering soil properties (Biały, 1999) and modifying the chemistry of both surface waters and groundwater, including a reduction in pH (Allen and Chapman, 2001; Neal et al., 2010; Drinan et al., 2013). The second process is influenced by several factors (Larssen and Holme, 2006), such as the acidic litterfall from coniferous trees (Alexander and Cresser, 1995; Burgess-Conforti et al., 2019), as well as the fact that conifer forests uptake more ions, such as compounds of atmospheric pollutants (nowadays mainly nitrogen, N) and marine ions (e.g., sodium, Na), than non-forested areas (Reynolds et al., 1994, 1997; Harriman et al., 2003). The cation exchange of sodium ions to hydrogen and aluminium ions in the soil can further amplify the acidification of surface waters (Larssen and Holme, 2006).
4.2.2 Role of fire across spatial scales
Besides human impact, another factor promoting the spread of pine in the landscape could be fire (as discussed in Sect. 4.1.2). Pinus sylvestris is one of the most fire-adapted European tree taxa, as confirmed by data at broad temporal (centennial to millennial) and spatial (local and regional) scales across its geographic range in Eurasia (Niklasson and Drakenberg, 2001; McRae et al., 2006; Olsson et al., 2010; Blanck et al., 2013; Novenko et al., 2018), including temperate Europe (Niklasson et al., 2010; Novák et al., 2012; Adámek et al., 2015, 2016; Spînu et al., 2020). Besides increasing landscape or forest openness, which favours the establishment of the light-demanding Scots pine (Lindbladh et al., 2003), fire has also been proven to facilitate pine regeneration on mineral soils by improving seedbed conditions (Kuuluvainen and Rouvinen, 2000; Hille and den Ouden, 2004; cf. Kubrak, 2010) or removing other, more fire-sensitive tree species that could be potential competitors (Niklasson et al., 2010; Spînu et al., 2020). Interestingly, fire activity can affect peatland hydrology and functioning in several ways (e.g., Väliranta et al., 2007; Benscoter et al., 2015; Lukenbach et al., 2016; Filicetti and Nielsen, 2020; Davies et al., 2023). At the landscape scale, fire can reduce tree cover, which affects both local evapotranspiration and runoff regimes (as reduction in vegetation decreases evapotranspiration and increases runoff, e.g., Piao et al., 2007; Sterling et al., 2013; Hrachowitz et al., 2021), causing an increase in peatland water levels (Sillasoo et al., 2007; Väliranta et al., 2007; Marcisz et al., 2015). However, fire activity can also be associated with periods of peatland drying (Väliranta et al., 2007; Marcisz et al., 2015).
In our study, fire record from the peat archive showed that the highest fire activity, documented at depths of ca. 33, 26, and 16 cm (Figs. 8–9), coincided with low water table levels (Fig. 11). During dry periods, more peatland fuels are available for burning, the likelihood of fire spread from neighbouring sites on mineral soils increases, and the probability of substantial acrotelm consumption rises. As a result, hollows may be locally formed (Benscoter et al., 2005, 2015; Sillasoo et al., 2007; Väliranta et al., 2007), which in turn have a positive effect on peatland surface wetness, demonstrating that the relationship between fire disturbance and peatland moisture is complex and difficult to interpret unambiguously (Sillasoo et al., 2007; Väliranta et al., 2007). Moreover, the link between fire and subsequent vegetation succession, including tree regeneration, is also intricate, as it involves – in addition to peatland hydrology – environmental variables such as burn severity, pre-fire species composition and cover, availability of microsites for recruitment, or inter- and intra-species competition (Ohlson et al., 2001; Brūmelis et al., 2009; Lukenbach et al., 2016; Davies et al., 2023). In Europe, fires have been shown to trigger successional changes in peatland vegetation and influence the regeneration of Scots pine (Durno and McVean, 1959; Bellamy, 1962; Ågren et al., 1983; Brūmelis et al., 2009). In addition, the importance of interactions between Pinus sylvestris and Sphagnum mosses has been demonstrated (Ohlson et al., 2001). Sphagnum has been shown to make up important recruitment sites for pine (Ohlson et al., 2001; Brūmelis et al., 2009), with an average germination success of up to 75 % (Gunnarsson and Rydin, 1998; Ohlson, 1999). However, severe mortality of Scots pine seedlings (especially with a stem diameter of less than 10 mm) resulting from being bogged down by Sphagnum (Gunnarsson and Rydin, 1998; Ohlson, 1999; Ohlson et al., 2001) due to its higher growth rate is very common, which proves that reduced growth of Sphagnum is a prerequisite for successful pine regeneration in peatlands. At the same time, the establishment and growth of Pinus sylvestris has been demonstrated to be able to terminate the Sphagnum dominance by impeding its growth and peat accumulation rate (once pine trees have reached a stem diameter of about 20 mm) (Ohlson et al., 2001). As fire has been proven to reduce water availability for Sphagnum mosses (Lukenbach et al., 2016), this disturbance could be a factor facilitating pine recruitment. Yet, post-fire water availability for mosses, tightly linked to the hydrophysical properties of peat (water repellency and moisture retention), is shaped by both, the pre-disturbance vegetation species composition and structure (microtopography), and the burn severity, leading to variable post-fire recovery pathways of the ground layer community in peatlands (Benscoter and Vitt, 2008; Lukenbach et al., 2016).
4.2.3 Drainage as the secondary factor
Drainage was undoubtedly another factor of anthropogenic origin that was of great importance for the dynamics of our study area (see earlier discussion, Sect. 4.1.3). Drainage of peatlands has an enormous impact on their structure and functioning, including hydrology, vegetation, decomposition, thermal conditions, and nutrient budget along with carbon balance. Furthermore, it also has an immense effect on the catchment hydrology (Holden et al., 2004; Ramchunder et al., 2009; Päivänen and Hånell, 2012; Kruczkowska et al., 2021). Following the lowering of the water table as a result of drainage, a number of processes are initiated in the peat that alter its physical and chemical properties under the interacting influence of environmental and substrate factors (Holden et al., 2004). In general, the water level drawdown enables oxic decomposition in a thicker surface peat layer (Päivänen and Hånell, 2012). Increased oxygen levels promote the mineralisation of nutrients such as carbon-bound sulphur and nitrogen and organically bound phosphorus, which can lead to their loss and reduce peat fertility (Holden et al., 2004). However, the amount of nutrients such as nitrogen and phosphorus in the surface peat layers may also increase as a result of drainage, mainly related to increased bulk density (Holden et al., 2004; Päivänen and Hånell, 2012). Another ecological consequence of lowering the water table is strong vegetation dynamics, including secondary vegetation succession of the ground layer from peatland taxa towards forest vegetation (Laine et al., 1995; Päivänen and Hånell, 2012) and the development of the tree layer through increased tree encroachment and tree growth (Laine et al., 1995; Linderholm and Leine, 2004; Freléchoux et al., 2000; Päivänen and Hånell, 2012), which was the main rationale behind large-scale peatland drainage for forestry worldwide (Päivänen and Hånell, 2012; Joosten, 2016). Interestingly, forest drainage operations can both increase and decrease environmental acidification (Kazda, 1995; Minkkinen et al., 2008; Päivänen and Hånell, 2012; Nisbet and Evans, 2014; Kļaviņa et al., 2025).
However, we assume that the human-driven landscape change and the resulting environmental consequences occurred prior to the intensive drainage measures in our study area, which began in the late 19th century (as discussed in Sect. 4.1.3). The absence of ditches on the map of the Wielkie Bagno peatland and its surroundings from the 1840s (Fig. 1f) is not necessarily due to the lack of drainage activities at that time, but may simply result from the cartographic principles and accuracy of the map. On another map from a slightly later period, the second half of the 19th century, a ditch network in the Rakowe Bagno peatland immediately south of our study site is shown in great detail (Fig. 1g), which in turn is not depicted on a map from a later period, the 1910s (Fig. 1h), confirming that the degree of map accuracy can sometimes be decisive for the landscape features recorded. Nevertheless, in the daily activity protocols of the Zamoyski Family Estate from 1810 to 1814, there is no evidence of drainage operations in the vicinity of our study site. Furthermore, severe floods were reported from this period (AS4–AS13), which would probably have been less possible with the drainage systems in place to facilitate landscape-level water runoff. Additionally, we documented successful pine recruitment in our study site much earlier, in the 1700s (and as early as the 1720s–1730s) (Fig. 4), which is not common in a black alder bog forest Carici elongatae-Alnetum (Solińska-Górnicka, 1987; Leuschner and Ellenberg, 2017a), suggesting that an ecosystem transition must have been initiated earlier.
Notably, the general pattern of the peatland pine climate–growth relationship we documented is typical for mineral soils: a positive influence of moist conditions (SPEI, precipitation) and a negative impact of high temperatures during the warm season months, combined with a growth-promoting effect of warmer conditions in early spring (February–March) (Fig. 5) (e.g., Linderholm, 1999; Edvardsson et al., 2015b; Misi et al., 2019; Waszak et al., 2021; Bąk et al., 2024). This could indicate that the trees established during a dry period, for example, following peatland drainage (Linderholm and Leine, 2004; Freléchoux et al., 2000, 2003, 2004; Edvardsson et al., 2012a, b, 2014, 2015a). However, we consider this unlikely. As discussed earlier, large-scale drainage measures in our study area probably occurred later than the establishment of the oldest pines in the population we studied (Figs. 1e–h and 3). Of course, the lack of historical evidence does not prove the absence of an environmental factor (here: drainage). Nevertheless, data demonstrating continuous recruitment of Pinus sylvestris in pristine peatlands without drainage activities during the last 200–400+ years (Ågren et al., 1983; Ågren and Zackrisson, 1990; Brūmelis et al., 2009; González de Andrés et al., 2022) show that this tree species is able to establish successfully in peatland ecosystems even without their draining. A possible explanation for our findings could be that our sampling site was located at the edge of the Wielkie Bagno peatland (Fig. 1), where the peat depth was not particularly great, i.e., 0.5 m. This is consistent with the results of a recent study in southern Sweden, which documented certain similarities (although not always significant and not for all hydroclimate variables analysed) in the tree growth–climate relationship in Scots pines at the peatland edge and on the neighbouring mineral soil site, which differed from the relationship in trees in the peatland's centre (Janecka et al., 2025). Considering that studies comparing the climate sensitivity of trees in peatlands and on adjacent mineral soils usually use one sampling site per substrate (Linderholm et al., 2002; Cedro and Lamentowicz, 2011; Edvardsson et al., 2015b; Blanchet et al., 2017), without distinguishing between the edge and the centre of the peatland, it is difficult to find further empirical evidence to support our reasoning.
4.2.4 Possible impact of climate and autogenic peatland development
Climate cannot be excluded as a factor contributing to the documented ecosystem shift. The transition period, from around 1400 to 1830 CE, overlaps with the Little Ice Age (1450–1850 CE, Björklund et al., 2023), which was characterized by low warm season temperatures in the region (Büntgen et al., 2011; Björklund et al., 2023). A colder climate would favour Scots pine, a tree species with a very wide thermal amplitude and high tolerance to low temperatures and frost (Przybylski, 1993; Leuschner and Ellenberg, 2017b), currently predicted to reduce its future range in Europe due to climate change (Dyderski et al., 2018, 2025). However, as shown by our tree ring record (Fig. 4), the relatively short period of dry and warm conditions in the early 19th century (Figs. 3 and S3) had no negative impact on the population dynamics of Pinus sylvestris in the Wielkie Bagno peatland. Given evidence that climate warming can exacerbate acidification (Murdoch et al., 2000; Evans et al., 2008; Whitehead et al., 2009), we consider a positive influence of cool thermal conditions rather unlikely.
Moreover, a high level of hydroclimatic variability throughout the entire period of our reconstruction, spanning over 2300 years (ca. 330 BCE–2022 CE), is clearly evidenced by both our data and regional records. These document not only exceptional dry and wet years such as 1811–1812, 1813, and 1815 (Figs. 3 and S3; AS4–AS14; see also Sect. 4.1.2), but also several pluvials and droughts in Europe, including megadroughts around 1000–1200, 1400–1480, and 1770–1840 CE (Cook et al., 2015, 2022; Büntgen et al., 2011, 2021a). Notably, the latter two overlap with the period during which the ecosystem transition occurred (ca. 1400–1830 CE) (Fig. 10). Additionally, existing data do not indicate that this period was drier than the modern era of anthropogenic climate change (Cook et al., 2015, 2022; Büntgen et al., 2011, 2021a). If climate had been the primary driver of the documented peatland transformation, we would expect this to have happened substantially earlier during the Late Holocene, namely around 4000–2000 years ago, as elsewhere in the region (Lamentowicz et al., 2015; Väliranta et al., 2017; Stivrins et al., 2025). Consequently, we suggest that factors beyond climate must have played a significant role in the ecosystem shift we documented.
In addition to external forcing, autogenic processes play a crucial role in peatland development (Belyea and Baird, 2006; Waddington et al., 2015), with various successional pathways possible (e.g., Marek, 1965; Väliranta et al., 2017; Blaus et al., 2020). However, the transition from a base-rich, minerotrophic fen to an acidic, ombrotrophic bog (cf. Wheeler and Proctor, 2000) is a common and widely reported trajectory. Despite the high decomposition rate and generally slow peat accumulation during the fen stage, with values of 0.05–0.4 cm yr−1 reported from Poland, Lithuania, Latvia, and Ukraine (Lamentowicz et al., 2007; Mažeika et al., 2009; Dobrowolski et al., 2010; Stivrins et al., 2025), the gradual thickening of the peat layer reduces the influence of mineral-rich groundwater, thereby decreasing available nutrients and surface pH. This lowers the decomposition rate, further accelerates peat accumulation, and eventually eliminates the impact of groundwater, resulting in full ombrotrophy (Lamentowicz et al., 2007; Väliranta et al., 2017). Notably, alder carr (i.e., wooded fen) is identified as a possible successional stage within the fen–bog transition (Marek, 1965). In temperate–boreal peatlands across the Baltic region and Finland, low fen peat accumulation rates and transitions in sedimentation regimes and peatland types have been broadly documented, often highlighting the complex interplay of autogenic and allogenic processes, including climatic variability, as key drivers (Lamentowicz et al., 2007; Hiiemaa et al., 2014; Väliranta et al., 2017; Stivrins et al., 2025). Therefore, neither climate nor autogenic succession can be omitted from the factors involved in the observed ecosystem change.
Although the vegetation shift we documented was likely the outcome of interacting natural and anthropogenic forces, we emphasize that the landscape-scale human impact and its cascading environmental effects, which occurred during the relatively short transition period of our reconstruction (cf. Fig. 2; Sect. 4.1), were the decisive drivers that triggered the fundamental ecological transformation. We hypothesize that land use changes, together with the landscape-scale increase in Pinus occurrence and the resulting acidification of catchment waters, followed by Sphagnum encroachment (and further acidification), possibly combined with concurrent drainage activities, contributed to the ecosystem changes observed at our study site (cf. Bąk et al., 2024; Wochal et al., 2025). Notably, the direct transition from alderwoods to Sphagnum-dominated acidic peatlands (i.e., bogs) or Scots pine bog forests (Vaccinio uliginosi-Pinetum) has been documented in Europe (McVean, 1956; Marek, 1965; Solińska-Górnicka, 1987). Alongside other possible determinants, as discussed above, this is likely strongly related to the spread of Sphagnum and Eriophorum, especially considering that the proximity of Sphagnum can cause very high mortality of alder seedlings (McVean, 1956).
4.3 Temporal perspective in conservation
The ecological importance of peatlands and peatland forests is widely recognized in Europe, making them one of the most significant targets in nature conservation, ecosystem restoration, and climate change mitigation efforts (Rydin and Jeglum, 2013; Joosten, 2016; Grzybowski and Glińska-Lewczuk, 2020; Jurasinski et al., 2020; Tanneberger et al., 2021). In addition, the importance of the temporal perspective in their conservation and restoration has already been emphasized by demonstrating how difficult it can be to define ecosystem reference conditions that should be targeted in these activities (Marcisz et al., 2022). Although there is palaeoecological evidence for millennia-long ecosystem stability in temperate Europe (Novák et al., 2012), including peatlands (Marcisz et al., 2020a), our results, similar to studies from northern Poland or Estonia (Lamentowicz et al., 2020; Czerwiński et al., 2021; Łuców et al., 2022; Wochal et al., 2025), clearly show that ecosystem transition (Fig. 10) is not uncommon in the long-term peatland dynamics. When they are caused by humans, novel anthroecosystems are created which are now being protected or restored (Marcisz et al., 2022). This is in line with the increasing amount of data challenging the static approach in vegetation ecology (Chiarucci et al., 2010) by documenting significant changes in ecosystems over longer time periods (Willis and Birks, 2006; Nowacki and Abrams, 2008, 2015; Spînu et al., 2020), as well as with the theory of alternative stable states/alternative equilibria (Scheffer et al., 1993; Beisner et al., 2003; Scheffer and van Nes, 2007) and the general understanding that disturbance and ecosystem change are inevitable features of nature and the environment and are therefore key mechanisms of vegetation dynamics (Pickett et al., 2009; Kuuluvainen, 2016; Graham et al., 2021). In fact, alternative stable states have been already recognized and are being implemented in peatland restoration (e.g., Temmink et al., 2021).
One of the key issues in nature conservation (and ecosystem management) is defining the conservation targets by answering the basic question about what is to be conserved: a species, community, ecosystem, landscape, processes (Groves et al., 2002) and how, with active or passive approach being the most frequent choices (e.g., Hartup et al., 2022). Often it couples with further points to consider such as degree of naturalness or level of variability (Willis and Birks, 2006), which may prove a given ecosystem to represent a certain successional stage, not necessarily pristine (Marcisz et al., 2022; Wochal et al., 2025). Our findings demonstrate how landscape-scale environmental changes can impact local ecosystems in a timescale longer than human memory (Fig. 2) and are another proof for the importance of long-term data in science-based resource management, conservation, and restoration (Heyerdahl and Card, 2000; Willis and Birks, 2006; Froyd and Willis, 2008; Marcisz et al., 2022), especially in such complex ecosystems as peatlands (Päivänen and Hånell, 2012; Waddington et al., 2015; Joosten, 2016). As ecosystem change in peatlands can occur in both directions, either through a switch to degradation or regeneration, and through a tipping point either from a non-degraded to a degraded state or vice versa (Milner et al., 2021), the need for further interdisciplinary multi-proxy studies that include both reconstructions of past dynamics and monitoring of current changes remains very high.
4.4 Challenges and opportunities in palaeoecological research: an interdisciplinary approach
This study has encountered numerous challenges, including incomplete or discontinuous archives and the integration of quantitative and qualitative data (Fig. 2). Despite these difficulties, it has created new opportunities for palaeoecological research by adopting a broad perspective that extends beyond classical palaeoecology and by incorporating disciplines such as climatology, dendroecology, dendroclimatology, history, and cartography. Rather than focusing solely on expanding existing datasets, its primary goal has been a critical reassessment of analytical approaches and methodologies, seeking deeper understanding and added value from combining different perspectives – an essential step for refining our understanding of past landscape dynamics (Ellis et al., 2021; Słowiński et al., 2021, 2024; Związek et al., 2024). This approach has facilitated a rigorous re-evaluation of prior assumptions, at times revealing inconsistencies in previous interpretations and highlighting the need for methodological refinements. Additionally, integrating quantitative and qualitative data of different resolutions (Fig. 2) has provided a broader perspective on the possibilities and scope of result interpretation, both in terms of analytical accuracy and the evaluation of reconstructed uncertainties, such as those concerning changes in water levels or air temperature across different archives and sources. Integrating data with varying temporal and spatial resolutions is becoming a key challenge in contemporary palaeoecology (Seddon et al., 2014). Researchers emphasize the need to develop methods that allow the combination of palaeoenvironmental records differing in precision and origin (e.g., lake and peat sediments, dendrochronological records, historical sources, or cartographic materials) (Słowiński et al., 2019; Edvardsson et al., 2022; Związek et al., 2024). In this context, our study serves as an example of such synthesis, enabling the comparison of data across various temporal and spatial scales and facilitating the identification of long-term trends and a more nuanced understanding of environmental dynamics. Furthermore, the diversity of temporal scales – ranging from daily climatic data (e.g., Słowińska et al., 2022), through annual or seasonal dendrochronological records (e.g., Zin et al., 2022; Klisz et al., 2023), to non-uniform datasets shaped by peat decomposition and accumulation (e.g., Łuców et al., 2020), and the temporally limited historical records (e.g., Róg, 2021) – has allowed for an interdisciplinary reconstruction of landscape change. As highlighted by Seddon et al. (2014), advancing methodologies for assessing synchrony and relationships between different sources of palaeoecological data remains a crucial research priority. In our case, the integration of multi-scale analyses has revealed patterns and processes that would likely have remained hidden if individual archives had been examined in isolation. In summary, such studies (e.g., Szabó, 2010; Lamentowicz et al., 2020; Bąk et al., 2024) present inherent challenges due to the complexity of the analyses and the necessity of collaboration among experts from various scientific disciplines. However, it is precisely this interdisciplinary approach that enables the expansion of interpretative frameworks and the formulation of novel research questions. Recent literature underscores the importance of reducing uncertainty in palaeoenvironmental reconstructions through robust chronological frameworks and a critical evaluation of methodological biases (Seddon et al., 2014). Our work contributes to these ongoing efforts of analyzing and interpreting complex environmental systems. Moreover, it offers a fresh perspective on palaeoecological questions, allowing critical engagement with established interpretations rather than simply reproducing them, which is essential for advancing scientific discourse.
Our study documents the transition of a peatland ecosystem from black alder bog forest to Scots pine bog forest (Fig. 10). This shift was likely driven by several interconnected factors, primarily land use changes, specifically the landscape-scale increase in the proportion of pine forests and the resulting environmental acidification that triggered Sphagnum encroachment. Our results, including the tree ring record of successful Scots pine establishment (Fig. 4) and historical data on settlement development throughout the 1700s (Tables S3 and S4), suggest that this environmental transformation occurred before the end of the second reconstruction period (ca. 1400–1830 CE; Fig. 2), between the 16th and 18th centuries. Notably, both the ecosystem shift and the current ecosystem type represent a much shorter period in its history (> 300–400 years) than the preceding, substantially different ecosystem state (> 1700 years). Furthermore, our reconstruction using various natural and human archives covering the last > 2300 years revealed considerable hydrological instability of the peatland. It also highlighted certain advantages and challenges of multi-proxy studies of landscape history and ecosystem dynamics, such as the different temporal resolution and coverage of the archives studied (including the problem of incomplete data) and the different information provided by quantitative and qualitative data, especially considering that human archives may be biased towards exceptional, catastrophic environmental events (e.g., McClain et al., 2021). In line with existing studies, we confirmed the importance of long-term environmental records for conservation ecology and land management (e.g., Willis and Birks, 2006; Froyd and Willis, 2008; Marcisz et al., 2022) and emphasized the continuing need for further research on peatland ecology, including past and current changes.
-
AS1: Inspection of the Zwierzyniec Estate (Rewizja klucza zwierzynieckiego), 1702–1762, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), ref. no. 2.1.13/1538, f. 10.
-
AS2: Collection of Maps of the Entailed Forests in Galicia, Created in March 1793 (Zbiór mapp lasów ordynackich w Galicyi in martio 1793 uczynionych), 1786–1793, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), Forest and Estate Maps Collection (IMK), ref. no. 7.4.4.1/1149.
-
AS3: Survey Protocol of the Village of Tereszpol (Protokół pomiarowy wsi Tereszpol), 1787, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), ref. no. 2.1.3/824.
-
AS4: Protocol of the Activities of the Economic Administration of the Zamoyski Entail Estate, vol. II (Protokół czynności Rządu Ekonomicznego Państwa Ordynacji Zamoyskiej, t. II), 1810, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), ref. no. 3.1/1755.
-
AS5: Protocol of the Activities of the Economic Administration of the Zamoyski Entail Estate, vol. I (Protokół czynności Rządu Ekonomicznego Państwa Ordynacji Zamoyskiej, t. I), 1810, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), ref. no. 3.1/1756.
-
AS6: Protocol of the Economic Activities for the Year 1811 (Protokół czynności ekonomicznych roku 1811), 1811, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), ref. no. 3.1/1757.
-
AS7: Protocol of More Important Matters Beginning on 1 January 1811 (Protokół ważniejszych interesów od dnia 1 stycznia 1811 roku poczynający się), 1811, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), ref. no. 3.1/1758.
-
AS8: Economic Protocol for the Year 1812 (Protokół ekonomiczny na rok 1812), 1812, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), ref. no. 3.1/1759.
-
AS9: Economic Protocol Beginning in January 1812: Grzegorz Korab Dolański, Administrator of the Estates of the Entail... (Protokół ekonomiczny od stycznia 1812 roku zaczynający się Grzegorz Korab Dolański administrator dóbr Ordynacji...), 1812–1813, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), ref. no. 3.1/1760.
-
AS10: Journal of the Activities of the General Administration of the Estates of His Lordship the Ordynat (Dziennik czynności Rządu Generalnego Dóbr JW Ordynata), 1811–1812, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), ref. no. 3.1/1761.
-
AS11: Journal of the Activities of the General Administration (Dziennik czynności administracji generalnej), 1812–1813, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), ref. no. 3.1/1762.
-
AS12: Journal of the Activities of the General Administration (Dziennik czynności administracji generalnej), 1813, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), ref. no. 3.1/1763.
-
AS13: Journal of the Activities of the General Administration... (Dziennik czynności Administracji Generalnej...), 1814, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), ref. no. 3.1/1764.
-
AS14: Register of Hearths, Population, Draught Teams, Sown Area, and Hay Yield in 1814, Tarnogród County, gmina of Księżpol (Wykaz dymów, ludności, zaprzężaju, wysiewów i zbioru siana w roku 1814, powiat tarnogrodzki, gmina Księżpol), 1814, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), ref. no. 3.9.7/3699, f. 25.
-
AS15: [Plan of the Zamoyski Entail Estates] ([Plan dóbr Ordynacji Zamojskiej]), 1851–1900, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), Forest and Estate Maps Collection (IMK), ref. no. 7.1/3.
-
AS16: Forest Management Plan of the Forests of the Zamoyski Entail Estate – Tereszpol Forest District (Plan gospodarczy lasów dóbr Ordynacji Zamojskiej – leśnictwo Tereszpol), 1924, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), Forest and Estate Maps Collection (IMK), ref. no. 7.4.4.4/2865.
-
AS17: Records of Individual Zamoyski Estates (Akta poszczególnych dóbr Zamoyskich), 1588–1673, The Central Archives of Historical Records in Warsaw (AGAD), Zamoyski Archive (AZ), ref. no. 2595.
-
AS18: Inspection of the Zwierzyniec Estate (Rewizja klucza zwierzynieckiego), 1702–1762, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), ref. no. 2.1.13/1538, f. 39r.
-
AS19: Regulation Map of the Tereszpol Estate with Adjacent Lands (Mapa regulacyjna dóbr Tereszpola z przyległościami...), 1827, State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), Forest and Estate Maps Collection (IMK), ref. no. 7.4.1/282.
-
AS20: Plan of the Tereszpol Forest District of the Zamoyski Entail Estate (Plan leśnictwa Tereszpol Ordynacji Zamojskiej), State Archive in Lublin (APL), Archive of the Zamoyski Family Estate (AOZ), Forest and Estate Maps Collection (IMK), ref. no. 7.4.4.3/1311.
The open dataset supporting the findings of this study is available from Zenodo at: https://doi.org/10.5281/zenodo.19330702 (Zin et al., 2026).
The supplement related to this article is available online at https://doi.org/10.5194/bg-23-3091-2026-supplement.
EZ, TZ, MK, and MS designed the study. EZ, TZ, MK, DR, and MS selected the study site. EZ, TZ, MK, DR, JPo, KPil, and MS carried out fieldwork. SS and KPio conducted the analysis of climatic data, EZ, MK, and KPil conducted the analysis of tree ring data, MO conducted the pollen and microcharcoal analysis, MS and JPi conducted the plant macrofossil analysis, MS and KS conducted the macrocharcoal analysis, AH conducted the non-metric multidimensional scaling (NMDS) and k-means clustering, DŁ conducted the testate amoebae analysis and depth to water table (DWT) reconstruction, TZ, DR, and JPo conducted the analysis of historical data. EZ, TZ, MK, SS, DR, MO, AH, DŁ, KPio, KPil, and MS designed and prepared figures and tables. EZ wrote the initial draft (with input from co-authors in the description of methods and results). EZ, TZ, MK, MS, SS, DR, JPo, DŁ, KPio, and KPil reviewed and edited the manuscript. All authors contributed to the discussion of the manuscript and approved the submitted version.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
We thank the Polish State Forest Administration, Zwierzyniec Forest District, for allowing us to collect tree ring samples. We would like to express our sincere gratitude to the Municipal Cultural Centre in Biłgoraj (Pol. Gminny Ośrodek Kultury w Biłgoraju) for their kind support and assistance during our fieldwork in the Solska Forest. We acknowledge fruitful discussions with Artur Obidziński on vegetation succession and with Sylwia Wierzcholska on bryophyte ecology. We thank Łukasz Kuberski for technical support in the preliminary site selection using Forest Data Dank (Pol. Bank Danych o Lasach, https://www.bdl.lasy.gov.pl/portal/en). We are grateful to Urszula Wojciechowska for help with fieldwork; and to Malwina Pilch and Paweł Nowak for their assistance with logistics during fieldwork.
This research has been supported by the Polish Ministry of Science and Higher Education within Forest Research Institute’s (IBL) statutory activities (grant no. 900613 to EZ, MK, KPil), the Forest Research Institute (IBL) (Own Research Fund, grants nos. 261509 to EZ, KPil and 260233 to EZ, MK, KPil), and the Polish National Science Center (NCN) (grants nos. 2023/49/N/ST10/04035 to KS, 2022/47/D/HS3/02947 to EZ, MK, KPil, TZ, MS, 2022/45/B/ST10/03423 to AH, MS, SS, 2021/43/O/HS3/01373 to DR, 2020/39/D/HS3/01298 to JPo, and 2018/31/B/ST10/02498 to MS, KS).
This paper was edited by Petr Kuneš and reviewed by Normunds Stivrins and Katarzyna Marcisz.
Adámek, M., Bobek, P., Hadincová, V., Wild, J., and Kopecký, M.: Forest fires within a temperate landscape: A decadal and millennial perspective from a sandstone region in Central Europe, Forest Ecol. Manage., 336, 81–90, https://doi.org/10.1016/j.foreco.2014.10.014, 2015.
Adámek, M., Hadincová, V., and Wild, J.: Long-term effect of wildfires on temperate Pinus sylvestris forests: Vegetation dynamics and ecosystem resilience, Forest Ecol. Manage., 380, 285–295, 2016.
Ågren, J. and Zackrisson, O.: Age and Size Structure of Pinus Sylvestris Populations on Mires in Central and Northern Sweden, J. Ecol., 78, 1049–1062, https://doi.org/10.2307/2260951, 1990.
Ågren, J., Isaksson, L., and Zackrisson, O.: Natural age and size of Pinus sylvestris and Picea abies on a mire in the inland part of northern Sweden, Holarctic Ecol., 6, 228–237, 1983.
Alexander, C. E. and Cresser, M. S.: An assessment of the possible impact of expansion of native woodland cover on the chemistry of Scottish freshwaters, Forest Ecol. Manage., 73, 1–27, https://doi.org/10.1016/0378-1127(94)03476-D, 1995.
Allen, A. and Chapman, D.: Impacts of afforestation on groundwater resources and quality, Hydrogeol. J., 9, 390–400, https://doi.org/10.1007/s100400100148, 2001.
Almagro, D., Martin-Benito, D., Rossi, S., Conde, M., Fernández-de-Uña, L., and Gea-Izquierdo, G.: Long-Term Cambial Phenology Reveals Diverging Growth Responses of Two Tree Species in a Mixed Forest Under Climate Change, Glob. Change Biol., 31, e70503, https://doi.org/10.1111/gcb.70503, 2025.
Amesbury, M. J., Swindles, G. T., Bobrov, A., Charman, D. J., Holden, J., Lamentowicz, M., Mallon, G., Mazei, Y., Mitchell, E. A. D., Payne, R. J., Roland, T. P., Turner, T. E., and Warner, B. G.: Development of a new pan-European testate amoeba transfer function for reconstructing peatland palaeohydrology, Quaternary Sci. Rev., 152, 132–151, https://doi.org/10.1016/j.quascirev.2016.09.024, 2016.
Andersen, R., Farrell, C., Graf, M., Muller, F., Calvar, E., Frankard, P., Caporn, S., and Anderson, P.: An overview of the progress and challenges of peatland restoration in Western Europe, Restor. Ecol., 25, 271–282, https://doi.org/10.1111/rec.12415, 2017.
Andersson, M. and Niklasson, M.: Rekordgammal tall på Hornslandet i Hälsingland, Svensk Botanisk Tidskrift, 98, 333–338, 2004.
Anonymous: Interpretation Manual of European Union Habitats, version EUR 28, https://eunis.eea.europa.eu/references/2435 (last access: 19 April 2024), 2013.
Anonymous: Conservation status 2013–2018: experts web viewer, Article 17 web tool, Habitat assessments at EU biogeographical level, https://nature-art17.eionet.europa.eu/article17/habitat/summary/?period=5&group=Forests&subject=91D0®ion=CON (last access: 19 April 2024), 2013–2018.
Arno, S. F. and Sneck, K. M.: A method for determining fire history in coniferous forests of the mountain West USA, USDA Forest Service General Technical Report, Intermountain Forest and Range Experiment Station, 28 pp., 1977.
Baisan, C. H. and Swetnam, T. W.: Fire History on a Desert Mountain-Range - Rincon Mountain Wilderness, Arizona, USA, Can. J. Forest Res., 20, 1559–1569, https://doi.org/10.1139/X90-208, 1990.
Bąk, M., Lamentowicz, M., Kołaczek, P., Wochal, D., Matulewski, P., Kopeć, D., Wietecha, M., Jaster, D., and Marcisz, K.: Assessing the impact of forest management and climate on a peatland under Scots pine monoculture using a multidisciplinary approach, Biogeosciences, 21, 5143–5172, https://doi.org/10.5194/bg-21-5143-2024, 2024.
Bąk, M., Lamentowicz, M., Kołaczek, P., Wochal, D., Jakubowicz, M., Andrews, L., and Marcisz, K.: Twentieth-century ecological disasters in central European monoculture pine plantations led to critical transitions in peatlands, Biogeosciences, 22, 3843–3866, https://doi.org/10.5194/bg-22-3843-2025, 2025.
Bałaga, K.: Post-glacial vegetational changes in the Middle Roztocze (E Poland), Acta Palaeobotanica, 38, 175–192, 1998.
Barbier, D. and Visset, L.: Logné, a peat bog of European ecological interest in the Massif Armorican, Western France: Bog development, vegetation and land-use history, Veg. Hist. Archaeobot., 6, 69–77, https://doi.org/10.1007/BF01261955, 1997.
Behre, K.-E.: The interpretation of anthropogenic indicators in pollen diagrams, Pollen et Spores, 23, 225–245, 1981.
Beisner, B., Haydon, D., and Cuddington, K.: Alternative stable states in ecology, Front. Ecol. Environ., 1, 376–382, https://doi.org/10.1890/1540-9295(2003)001[0376:assie]2.0.co;2, 2003.
Bellamy, D. J.: Some observations on the peat bogs of the wilderness of Pisz, Przegląd Geograficzny, 34, 691–716, 1962.
Belyea, L. R. and Baird, A. J.: Beyond “the limits to peat bog growth”: cross-scale feedback in peatland development, Ecol. Monogr., 76, 299–322, https://doi.org/10.1890/0012-9615(2006)076[0299:BTLTPB]2.0.CO;2, 2006.
Belyea, L. R. and Malmer, N.: Carbon sequestration in peatland: patterns and mechanisms of response to climate change, Glob. Change Biol., 10, 1043–1052, https://doi.org/10.1111/j.1529-8817.2003.00783.x, 2004.
Benscoter, B. W. and Vitt, D. H.: Spatial Patterns and Temporal Trajectories of the Bog Ground Layer Along a Post-Fire Chronosequence, Ecosystems, 11, 1054–1064, https://doi.org/10.1007/s10021-008-9178-4, 2008.
Benscoter, B. W., Kelman Wieder, R., and Vitt, D. H.: Linking microtopography with post-fire succession in bogs, J. Veg. Sci., 16, 453–460, https://doi.org/10.1111/j.1654-1103.2005.tb02385.x, 2005.
Benscoter, B. W., Greenacre, D., and Turetsky, M. R.: Wildfire as a key determinant of peatland microtopography, Can. J. Forest Res., 45, 1132–1136, https://doi.org/10.1139/cjfr-2015-0028, 2015.
Berglund, B. E. and Ralska-Jasiewiczowa, M.: Pollen analysis and pollen diagrams, Handbook of Holocene Palaeoecology and Palaeohydrology, 455, 484–486, 1986.
Beug, H.-J.: Leitfaden der Pollenbestimmung für Mitteleuropa und angrenzende Gebiete, Verlag Friedrich Pfeil, Munich, ISBN 3-89937-043-0, 2004.
Biały, K.: Dowolność wyróżniania typów siedliskowych lasu i projektowania składów docelowych drzewostanów w obrębie gleb bielicoziemnych, Sylwan, 143, 65–72, 1999.
Birks, H. H.: Plant macrofossils, in: Tracking Environmental Change Using Lake Sediments, Volume 3: Terrestrial, Algal, and Siliceous Indicators, edited by: Smol, J. P., Birks, H. J. B., and Last, W. M., Kluwer Academic Publishers, Dordrecht, the Netherlands, 49–74, https://doi.org/10.1007/0-306-47668-1, ISBN 978-1-4020-0681-4, 2002.
Birks, H. J. B.: Estimating the amount of compositional change in late-Quaternary pollen-stratigraphical data, Veg. Hist. Archaeobot., 16, 197–202, https://doi.org/10.1007/s00334-006-0079-1, 2007.
Björklund, J., Seftigen, K., Stoffel, M., Fonti, M. V., Kottlow, S., Frank, D. C., Esper, J., Fonti, P., Goosse, H., Grudd, H., Gunnarson, B. E., Nievergelt, D., Pellizzari, E., Carrer, M., and von Arx, G.: Fennoscandian tree-ring anatomy shows a warmer modern than medieval climate, Nature, 620, 97–103, https://doi.org/10.1038/s41586-023-06176-4, 2023.
Blanchet, G., Guillet, S., Calliari, B., Corona, C., Edvardsson, J., Stoffel, M., and Bragazza, L.: Impacts of regional climatic fluctuations on radial growth of Siberian and Scots pine at Mukhrino mire (central-western Siberia), Sci. Total Environ., 574, 1209–1216, https://doi.org/10.1016/j.scitotenv.2016.06.225, 2017.
Blanck, Y., Rolstad, J., and Storaunet, K. O.: Low- to moderate-severity historical fires promoted high tree growth in a boreal Scots pine forest of Norway, Scand. J. Forest Res., 28, 126–135, https://doi.org/10.1080/02827581.2012.706635, 2013.
Blaus, A., Reitalu, T., Amon, L., Vassiljev, J., Alliksaar, T., and Veski, S.: From bog to fen: palaeoecological reconstruction of the development of a calcareous spring fen on Saaremaa, Estonia, Veg. Hist. Archaeobot., 29, 373–391, https://doi.org/10.1007/s00334-019-00748-z, 2020.
Boggie, R.: Effect of Water-Table Height on Root Development of Pinus Contorta on Deep Peat in Scotland, Oikos, 23, 304–312, https://doi.org/10.2307/3543168, 1972.
Bonk, A., Słowiński, M., Żarczyński, M., Oliński, P., Kupryjanowicz, M., Fiłoc, M., and Tylmann, W.: Tracking fire activity and post-fire limnological responses using the varved sedimentary sequence of Lake Jaczno, Poland, Holocene, 32, 515–528, https://doi.org/10.1177/09596836221080755, 2022.
Booth, R. K., Lamentowicz, M., and Charman, D. J.: Preparation and analysis of testate amoebae in peatland paleoenvironmental studies, Mires Peat, 7 (2010/11), 1–7, 2010.
Broda, J.: Sosna w czasach historycznych, in: Biologia sosny zwyczajnej, edited by: Białobok, S., Boratyński, A., and Bugała, W., Sorus, Poznań – Kórnik, 17–31, ISBN 83-85599-21-5, 1993.
Broda, J.: Historia leśnictwa w Polsce, Wydawnictwo Akademii Rolniczej im. Augusta Cieszkowskiego w Poznaniu, Poznań, ISBN 83-7160-216-2, 2000.
Brown, T. A., Nelson, D. E., Mathewes, R. W., Vogel, J. S., and Southon, J. R.: Radiocarbon Dating of Pollen by Accelerator Mass Spectrometry, Quaternary Res., 32, 205–212, https://doi.org/10.1016/0033-5894(89)90076-8, 1989.
Brūmelis, G., Strazds, M., and Eglava, Ž.: Stand structure and spatial pattern of regeneration of Pinus sylvestris in a natural treed mire in Latvia, Silva Fennica, 43, 767–781, https://doi.org/10.14214/sf.172, 2009.
Bunn, A., Korpela, M., Biondi, F., Campelo, F., Mérian, P., Qeadan, F., Zang, C., Buras, A., Cecile, J., and Mudelsee, M.: Package “dplR”: Dendrochronology Program Library in R (1.7.2), dplR: Dendrochronology Program Library in R (v1.7.2), Comprehensive R Archive Network (CRAN), 2020.
Bunn, A. G.: A dendrochronology program library in R (dplR), Dendrochronologia, 26, 115–124, https://doi.org/10.1016/j.dendro.2008.01.002, 2008.
Büntgen, U., Tegel, W., Nicolussi, K., McCormick, M., Frank, D., Trouet, V., Kaplan, J. O., Herzig, F., Heussner, K.-U., Wanner, H., Luterbacher, J., and Esper, J.: 2500 Years of European Climate Variability and Human Susceptibility, Science, 331, 578–582, https://doi.org/10.1126/science.1197175, 2011.
Büntgen, U., Urban, O., Krusic, P. J., Rybníček, M., Kolář, T., Kyncl, T., Ač, A., Koňasová, E., Čáslavský, J., Esper, J., Wagner, S., Saurer, M., Tegel, W., Dobrovolný, P., Cherubini, P., Reinig, F., and Trnka, M.: Recent European drought extremes beyond Common Era background variability, Nat. Geosci., 14, 190–196, https://doi.org/10.1038/s41561-021-00698-0, 2021a.
Büntgen, U., Allen, K., Anchukaitis, K. J., Arseneault, D., Boucher, É., Bräuning, A., Chatterjee, S., Cherubini, P., Churakova, O. V., Corona, C., Gennaretti, F., Grießinger, J., Guillet, S., Guiot, J., Gunnarson, B., Helama, S., Hochreuther, P., Hughes, M. K., Huybers, P., Kirdyanov, A. V., Krusic, P. J., Ludescher, J., Meier, W. J. H., Myglan, V. S., Nicolussi, K., Oppenheimer, C., Reinig, F., Salzer, M. W., Seftigen, K., Stine, A. R., Stoffel, M., St. George, S., Tejedor, E., Trevino, A., Trouet, V., Wang, J., Wilson, R., Yang, B., Xu, G., and Esper, J.: The influence of decision-making in tree ring-based climate reconstructions, Nat. Commun., 12, 3411, https://doi.org/10.1038/s41467-021-23627-6, 2021b.
Buraczyński, J.: Roztocze – Dzieje Osadnictwa, Drukarnia i Wydawnictwo Akademickie Wyższej Szkoły Społeczno-Przyrodniczej im. Wincentego Pola, Lublin, ISBN 978-83-6059-420-9, 2008.
Buras, A.: A comment on the expressed population signal, Dendrochronologia, 44, 130–132, https://doi.org/10.1016/j.dendro.2017.03.005, 2017.
Buras, A. and Wilmking, M.: Correcting the calculation of Gleichläufigkeit, Dendrochronologia, 34, 29–30, https://doi.org/10.1016/j.dendro.2015.03.003, 2015.
Burgess-Conforti, J. R., Moore, P. A., Owens, P. R., Miller, D. M., Ashworth, A. J., Hays, P. D., Evans-White, M. A., and Anderson, K. R.: Are soils beneath coniferous tree stands more acidic than soils beneath deciduous tree stands?, Environ. Sci. Pollut. R., 26, 14920–14929, https://doi.org/10.1007/s11356-019-04883-y, 2019.
Bürgi, M. and Russell, E. W. B.: Integrative methods to study landscape changes, Land Use Pol., 18, 9–16, https://doi.org/10.1016/S0264-8377(00)00041-7, 2001.
Carvalho, T.: Joseph II et la physiocratie. Enquête sur un malentendu historique, in: Annuaire de la Société autrichienne pour l'étude du dix-huitième siècle (Jahrbuch der Österreichischen Gesellschaft zur Erforschung des 18. Jahrhunderts), 89–107, 2018.
Cedro, A. and Lamentowicz, M.: The last hundred years' dendroecology of Scots pine (Pinus sylvestris L.) on a Baltic bog in Northern Poland: Human impact and hydrological changes, Balt. For., 14, 26–33, 2008.
Cedro, A. and Lamentowicz, M.: Contrasting responses to environmental changes by pine (Pinus sylvestris L.) growing on peat and mineral soil: An example from a Polish Baltic bog, Dendrochronologia, 29, 211–217, https://doi.org/10.1016/j.dendro.2010.12.004, 2011.
Cedro, A. and Sotek, Z.: Natural and Anthropogenic Transformations of A Baltic Raised Bog (Bagno Kusowo, North West Poland) in the Light of Dendrochronological Analysis of Pinus sylvestris L, Forests, 7, 202, https://doi.org/10.3390/f7090202, 2016.
Chiarucci, A., Araújo, M. B., Decocq, G., Beierkuhnlein, C., and Fernández-Palacios, J. M.: The concept of potential natural vegetation: an epitaph?, J. Veg. Sci., 21, 1172–1178, https://doi.org/10.1111/j.1654-1103.2010.01218.x, 2010.
Chmielewski, T. J. and Sowińska, B.: Social expectations concerning landscape quality objectives for the Roztocze – Solska Forest region, Teka Komisji Ochrony I Kształtowania Środowiska Przyrodniczego – OL PAN, 5, 41–49, 2008.
Chmielewski, T. J. and Sowińska, B.: Landscape ecological structure of the Roztocze and Solska Forest regions: a comparative study of models from 1988 and 2011, Teka Komisji Ochrony I Kształtowania Środowiska Przyrodniczego – OL PAN, 8, 13–23, 2011.
Clark, J. S.: Particle motion and the theory of charcoal analysis: Source area, transport, deposition, and sampling, Quaternary Res., 30, 67–80, https://doi.org/10.1016/0033-5894(88)90088-9, 1988.
Clymo, R. S.: The Origin of Acidity in Sphagnum Bogs, Bryologist, 67, 427–431, https://doi.org/10.2307/3240768, 1964.
Clymo, R. S.: Sphagnum-dominated peat bog: a naturally acid ecosystem, Philos. T. Roy. Soc. Lond. B, 305, 487–499, https://doi.org/10.1098/rstb.1984.0072, 1984.
Clymo, R. S.: Interactions of Sphagnum with Water and Air, in: Effects of Atmospheric Pollutants on Forests, Wetlands and Agricultural Ecosystems, edited by: Hutchinson, T. C. and Meema, K. M., NATO ASI Series, vol. 16, Springer, Berlin, Heidelberg, 513–529, https://doi.org/10.1007/978-3-642-70874-9_37, 1987.
Conedera, M., Tinner, W., Neff, C., Meurer, M., Dickens, A. F., and Krebs, P.: Reconstructing past fire regimes: methods, applications, and relevance to fire management and conservation, Quaternary Sci. Rev., 28, 555–576, https://doi.org/10.1016/j.quascirev.2008.11.005, 2009.
Cook, B. I., Smerdon, J. E., Cook, E. R., Williams, A. P., Anchukaitis, K. J., Mankin, J. S., Allen, K., Andreu-Hayles, L., Ault, T. R., Belmecheri, S., Coats, S., Coulthard, B., Fosu, B., Grierson, P., Griffin, D., Herrera, D. A., Ionita, M., Lehner, F., Leland, C., Marvel, K., Morales, M. S., Mishra, V., Ngoma, J., Nguyen, H. T. T., O'Donnell, A., Palmer, J., Rao, M. P., Rodriguez-Caton, M., Seager, R., Stahle, D. W., Stevenson, S., Thapa, U. K., Varuolo-Clarke, A. M., and Wise, E. K.: Megadroughts in the Common Era and the Anthropocene, Nat. Rev. Earth Environ., 3, 741–757, https://doi.org/10.1038/s43017-022-00329-1, 2022.
Cook, E. R. and Kairiukstis, L. A. (Eds.): Methods of Dendrochronology, Kluwer, Academic Publishers, Dordrecht, ISBN 0-7923-0586-8, 1990.
Cook, E. R. and Peters, K.: The smoothing spline: a new approach to standardizing forest interior tree-ring width series for dendroclimatic studies, Tree-Ring Bulletin, 41, 45–53, 1981.
Cook, E. R., Seager, R., Kushnir, Y., Briffa, K. R., Büntgen, U., Frank, D., Krusic, P. J., Tegel, W., van der Schrier, G., Andreu-Hayles, L., Baillie, M., Baittinger, C., Bleicher, N., Bonde, N., Brown, D., Carrer, M., Cooper, R., Čufar, K., Dittmar, C., Esper, J., Griggs, C., Gunnarson, B., Günther, B., Gutierrez, E., Haneca, K., Helama, S., Herzig, F., Heussner, K.-U., Hofmann, J., Janda, P., Kontic, R., Köse, N., Kyncl, T., Levanič, T., Linderholm, H., Manning, S., Melvin, T. M., Miles, D., Neuwirth, B., Nicolussi, K., Nola, P., Panayotov, M., Popa, I., Rothe, A., Seftigen, K., Seim, A., Svarva, H., Svoboda, M., Thun, T., Timonen, M., Touchan, R., Trotsiuk, V., Trouet, V., Walder, F., Ważny, T., Wilson, R., and Zang, C.: Old World megadroughts and pluvials during the Common Era, Sci. Adv., 1, e1500561, https://doi.org/10.1126/sciadv.1500561, 2015.
Cuny, H. E. and Rathgeber, C. B. K.: Xylogenesis: Coniferous Trees of Temperate Forests Are Listening to the Climate Tale during the Growing Season But Only Remember the Last Words!, Plant Physiol., 171, 306–317, https://doi.org/10.1104/pp.16.00037, 2016.
Cywa, K.: Trees and shrubs used in medieval Poland for making everyday objects, Veg. Hist. Archaeobot., 27, 111–136, https://doi.org/10.1007/s00334-017-0644-9, 2018.
Czerepko, J.: A long-term study of successional dynamics in the forest wetlands, Forest Ecol. Manage., 255, 630–642, https://doi.org/10.1016/j.foreco.2007.09.039, 2008.
Czernecki, B. and Miętus, M.: Porównanie stosowanych klasyfikacji termicznych na przykładzie wybranych regionów Polski, Przegląd Geofizyczny, 56, 201–233, 2011.
Czerwiński, S., Guzowski, P., Lamentowicz, M., Gałka, M., Karpińska-Kołaczek, M., Poniat, R., Łokas, E., Diaconu, A.-C., Schwarzer, J., Miecznik, M., and Kołaczek, P.: Environmental implications of past socioeconomic events in Greater Poland during the last 1200 years. Synthesis of paleoecological and historical data, Quaternary Sci. Rev., 259, 106902, https://doi.org/10.1016/j.quascirev.2021.106902, 2021.
Czerwiński, S., Marcisz, K., Wacnik, A., and Lamentowicz, M.: Synthesis of palaeoecological data from the Polish Lowlands suggests heterogeneous patterns of old-growth forest loss after the Migration Period, Sci. Rep., 12, 8559, https://doi.org/10.1038/s41598-022-12241-1, 2022.
Dang, Q. L. and Lieffers, V. J.: Climate and annual ring growth of black spruce in some Alberta peatlands, Can. J. Botany, 67, 1885–1889, https://doi.org/10.1139/b89-239, 1989.
Dang, Q. L., Lieffers, V. J., Rothwell, R. L., and Macdonald, S. E.: Diurnal variation and interrelations of ecophysiological parameters in three peatland woody species under different weather and soil moisture conditions, Oecologia, 88, 317–324, https://doi.org/10.1007/BF00317573, 1991.
Dauškane, I., Brūmelis, G., and Elferts, D.: Effect of climate on extreme radial growth of Scots pine growing on bogs in Latvia, Estonian Journal of Ecology, 60, 236–248, https://doi.org/10.3176/eco.2011.3.06, 2011.
Davies, G. M., Gray, A., Power, S. C., and Domènech, R.: Resilience of temperate peatland vegetation communities to wildfire depends upon burn severity and pre-fire species composition, Ecol. Evol., 13, e9912, https://doi.org/10.1002/ece3.9912, 2023.
Davies, N.: God's Playground A History of Poland: Volume II: 1795 to the Present, OUP Oxford, ISBN 978-0-19-925340-1, 2005.
Davis, M. B. and Deevey, E. S.: Pollen Accumulation Rates: Estimates from Late-Glacial Sediment of Rogers Lake, Science, 145, 1293–1295, https://doi.org/10.1126/science.145.3638.1293, 1964.
Dearing, J., Acma, B., Bub, S., Chambers, F., Chen, X., Cooper, J., Crook, D., Dong, X., Dotterweich, M., Edwards, M., Foster, T., Gaillard, M.-J., Galop, D., Gell, P., Gil, A., Jeffers, E., Jones, R., Anupama, K., Langdon, P., Marchant, R., Mazier, F., McLean, C., Nunes, L., Sukumar, R., Suryaprakash, I., Umer, M., Yang, X., Wang, R., and Zhang, K.: Social-ecological systems in the Anthropocene: The need for integrating social and biophysical records at regional scales, Anthropocene Review, 2, 220–246, https://doi.org/10.1177/2053019615579128, 2015.
Dearing, J. A., Jones, R. T., Shen, J., Yang, X., Boyle, J. F., Foster, G. C., Crook, D. S., and Elvin, M. J. D.: Using multiple archives to understand past and present climate–human–environment interactions: the lake Erhai catchment, Yunnan Province, China, J. Paleolimnol., 40, 3–31, https://doi.org/10.1007/s10933-007-9182-2, 2008.
Degirmendžić, J., Kożuchowski, K., and Żmudzka, E.: Changes of air temperature and precipitation in Poland in the period 1951–2000 and their relationship to atmospheric circulation, Int. J. Climatol., 24, 291–310, https://doi.org/10.1002/joc.1010, 2004.
Dietze, E., Brykała, D., Schreuder, L. T., Jażdżewski, K., Blarquez, O., Brauer, A., Dietze, M., Obremska, M., Ott, F., Pieńczewska, A., Schouten, S., Hopmans, E. C., and Słowiński, M.: Human-induced fire regime shifts during 19th century industrialization: A robust fire regime reconstruction using northern Polish lake sediments, Plos One, 14, e0222011, https://doi.org/10.1371/journal.pone.0222011, 2019.
Dinella, A., Giammarchi, F., Tonon, G., and De Micco, V.: Are living peatland trees a reliable natural archive for climate reconstruction?, IAWA J., 40, 366–379, https://doi.org/10.1163/22941932-40190228, 2019.
Dinella, A., Giammarchi, F., Prendin, A. L., Carrer, M., and Tonon, G.: Xylem traits of peatland Scots pines reveal a complex climatic signal: A study in the Eastern Italian Alps, Dendrochronologia, 67, 125824, https://doi.org/10.1016/j.dendro.2021.125824, 2021.
Dobrowolski, R., Ziółek, M., Bałaga, K., Melke, J., and Bogucki, A.: Radiocarbon age and geochemistry of the infillings of small closed depressions from Western Polesie (Poland SE, Ukraine NW), Geochronometria, 36, 39–46, https://doi.org/10.2478/v10003-010-0010-8, 2010.
Dombrovskaya, A., Koreneva, M., and Tyuremnov, S.: Atlas rastitel'nykh ostatkov, vstrechaemykh v torfe, Gosenergoizdat, Moscow, 1959.
Drinan, T. J., Graham, C. T., O'Halloran, J., and Harrison, S. S. C.: The impact of catchment conifer plantation forestry on the hydrochemistry of peatland lakes, Sci. Total Environ., 443, 608–620, https://doi.org/10.1016/j.scitotenv.2012.10.112, 2013.
Durno, S. E. and McVean, D. N.: Forest History of the Beinn Eighe Nature Reserve, New Phytol., 58, 228–236, https://doi.org/10.1111/j.1469-8137.1959.tb05353.x, 1959.
Dyderski, M. K., Paź, S., Frelich, L. E., and Jagodziński, A. M.: How much does climate change threaten European forest tree species distributions?, Glob. Change Biol., 24, 1150–1163, https://doi.org/10.1111/gcb.13925, 2018.
Dyderski, M. K., Paź-Dyderska, S., Jagodziński, A. M., and Puchałka, R.: Shifts in native tree species distributions in Europe under climate change, J. Environ. Manage., 373, 123504, https://doi.org/10.1016/j.jenvman.2024.123504, 2025.
Dyer, J. M.: Land-use legacies in a central Appalachian forest: differential response of trees and herbs to historic agricultural practices, Appl. Veg. Sci., 13, 195–206, https://doi.org/10.1111/j.1654-109X.2009.01061.x, 2010.
Eckstein, D. and Bauch, J.: Beitrag zur Rationalisierung eines dendrochronologischen Verfahrens und zur Analyse seiner Aussagesicherheit, Forstwiss. Centralbl., 88, 230–250, https://doi.org/10.1007/BF02741777, 1969.
Eckstein, J., Leuschner, H. H., Bauerochse, A., and Sass-Klaassen, U.: Subfossil bog-pine horizons document climate and ecosystem changes during the Mid-Holocene, Dendrochronologia, 27, 129–146, https://doi.org/10.1016/j.dendro.2009.06.007, 2009.
Edvardsson, J. and Hansson, A.: Multiannual hydrological responses in Scots pine radial growth within raised bogs in southern Sweden, Silva Fenn, 49, 1–14, https://doi.org/10.14214/sf.1354, 2015.
Edvardsson, J., Linderson, H., Rundgren, M., and Hammarlund, D.: Holocene peatland development and hydrological variability inferred from bog-pine dendrochronology and peat stratigraphy – a case study from southern Sweden, J. Quaternary Sci., 27, 553–563, https://doi.org/10.1002/jqs.2543, 2012a.
Edvardsson, J., Leuschner, H. H., Linderson, H., Linderholm, H. W., and Hammarlund, D.: South Swedish bog pines as indicators of Mid-Holocene climate variability, Dendrochronologia, 30, 93–103, https://doi.org/10.1016/j.dendro.2011.02.003, 2012b.
Edvardsson, J., Poska, A., Van der Putten, N., Rundgren, M., Linderson, H., and Hammarlund, D.: Late-Holocene expansion of a south Swedish peatland and its impact on marginal ecosystems: Evidence from dendrochronology, peat stratigraphy and palaeobotanical data, Holocene, 24, 466–476, https://doi.org/10.1177/0959683613520255, 2014.
Edvardsson, J., Šimanauskienė, R., Taminskas, J., Baužienė, I., and Stoffel, M.: Increased tree establishment in Lithuanian peat bogs – Insights from field and remotely sensed approaches, Sci. Total Environ., 505, 113–120, https://doi.org/10.1016/j.scitotenv.2014.09.078, 2015a.
Edvardsson, J., Rimkus, E., Corona, C., Šimanauskienė, R., Kažys, J., and Stoffel, M.: Exploring the impact of regional climate and local hydrology on Pinus sylvestris L. growth variability – A comparison between pine populations growing on peat soils and mineral soils in Lithuania, Plant Soil, 392, 345–356, https://doi.org/10.1007/s11104-015-2466-9, 2015b.
Edvardsson, J., Stančikaitė, M., Miras, Y., Corona, C., Gryguc, G., Gedminienė, L., Mažeika, J., and Stoffel, M.: Late-Holocene vegetation dynamics in response to a changing climate and anthropogenic influences – Insights from stratigraphic records and subfossil trees from southeast Lithuania, Quaternary Sci. Rev., 185, 91–101, https://doi.org/10.1016/j.quascirev.2018.02.006, 2018.
Edvardsson, J., Baužienė, I., Lamentowicz, M., Šimanauskienė, R., Tamkevičiūtė, M., Taminskas, J., Linkevičienė, R., Skuratovič, Ž., Corona, C., and Stoffel, M.: A multi-proxy reconstruction of moisture dynamics in a peatland ecosystem: A case study from Čepkeliai, Lithuania, Ecol. Indic., 106, 105484, https://doi.org/10.1016/j.ecolind.2019.105484, 2019.
Edvardsson, J., Helama, S., Rundgren, M., and Nielsen, A. B.: The Integrated Use of Dendrochronological Data and Paleoecological Records From Northwest European Peatlands and Lakes for Understanding Long-Term Ecological and Climatic Changes – A Review, Frontiers in Ecol. Evol., 10, https://doi.org/10.3389/fevo.2022.781882, 2022.
Ellenberg, H., Weber, H. E., Düll, R., Wirth, V., and Werner, W.: Zeigerwerte von Pflanzen in Mitteleuropa. 3., durchgesehene Aufl., Verlag Erich Goltze GmbH & Co, Göttingen, ISBN 978-3-88452-518-0, 2001.
Ellis, E. C. and Ramankutty, N.: Putting people in the map: anthropogenic biomes of the world, Front. Ecol. Environ., 6, 439–447, https://doi.org/10.1890/070062, 2008.
Ellis, E. C., Gauthier, N., Klein Goldewijk, K., Bliege Bird, R., Boivin, N., Díaz, S., Fuller, D. Q., Gill, J. L., Kaplan, J. O., Kingston, N., Locke, H., McMichael, C. N. H., Ranco, D., Rick, T. C., Shaw, M. R., Stephens, L., Svenning, J.-C., and Watson, J. E. M.: People have shaped most of terrestrial nature for at least 12 000 years, P. Natl. Acad. Sci. USA, 118, e2023483118, https://doi.org/10.1073/pnas.2023483118, 2021.
Evans, C. D., Reynolds, B., Hinton, C., Hughes, S., Norris, D., Grant, S., and Williams, B.: Effects of decreasing acid deposition and climate change on acid extremes in an upland stream, Hydrol. Earth Syst. Sci., 12, 337–351, https://doi.org/10.5194/hess-12-337-2008, 2008.
Fægri, K., Kaland, P. E., and Krzywinski, K.: Textbook of pollen analysis (based on original work by K. Fægri and J. Iversen), 4th edn., revised, John Wiley & Sons Ltd., Chichester, England/New York, ISBN 0 471 92178 5, 1989.
Faliński, J. B.: Vegetation Dynamics in Temperate Lowland Primeval Forest. Ecological Studies in Białowieża Forest, Dr W. Junk Publishers, Dordrecht, ISBN 90-6193-534-2, 1986.
Farris, C. A., Baisan, C. H., Falk, D. A., Yool, S. R., and Swetnam, T. W.: Spatial and temporal corroboration of a fire-scar-based fire history in a frequently burned ponderosa pine forest, Ecol. Appl., 20, 1598–1614, https://doi.org/10.1890/09-1535.1, 2010.
Filicetti, A. T. and Nielsen, S. E.: Tree regeneration on industrial linear disturbances in treed peatlands is hastened by wildfire and delayed by loss of microtopography, Can. J. Forest Res., 50, 936–945, https://doi.org/10.1139/cjfr-2019-0451, 2020.
Forman, R. T. T. and Russell, E. W. B.: Evaluation of Historical Data in Ecology, Bulletin of the Ecological Society of America, 64, 5–7, 1983.
Francon, L., Edvardsson, J., Corona, C., and Stoffel, M.: The timing of wood formation in peatland trees as obtained with different approaches, Dendrochronologia, 85, 126210, https://doi.org/10.1016/j.dendro.2024.126210, 2024.
Freléchoux, F., Buttler, A., Schweingruber, F. H., and Gobat, J.-M.: Stand structure, invasion, and growth dynamics of bog pine (Pinus uncinata var. rotundata) in relation to peat cutting and drainage in the Jura Mountains, Switzerland, Can. J. Forest Res., 30, 1114–1126, https://doi.org/10.1139/x00-039, 2000.
Freléchoux, F., Buttler, A., Gillet, F., Gobat, J.-M., and Schweingruber, F. H.: Succession from bog pine (Pinus uncinata var. rotundata) to Norway spruce (Picea abies) stands in relation to anthropic factors in Les Saignolis bog, Jura Mountains, Switzerland, Ann. For. Sci., 60, 347–356, 2003.
Freléchoux, F., Buttler, A., Schweingruber, F. H., and Gobat, J.-M.: Spatio-temporal pattern of bog pine (Pinus uncinata var. rotundata) at the interface with the Norway spruce (Picea abies) belt on the edge of a raised bog in the Jura Mountains, Switzerland, Ann. For. Sci., 61, 309–318, 2004.
Froyd, C. A. and Willis, K. J.: Emerging issues in biodiversity & conservation management: The need for a palaeoecological perspective, Quaternary Sci. Rev., 27, 1723–1732, https://doi.org/10.1016/j.quascirev.2008.06.006, 2008.
Gałka, M., Tobolski, K., Górska, A., Milecka, K., Fiałkiewicz-Kozieł, B., and Lamentowicz, M.: Disentangling the drivers for the development of a Baltic bog during the Little Ice Age in northern Poland, Quatern. Int., 328–329, 323–337, https://doi.org/10.1016/j.quaint.2013.02.026, 2014.
Gałka, M., Knorr, K.-H., Tobolski, K., Gallego-Sala, A., Kołaczek, P., Lamentowicz, M., Kajukało-Drygalska, K., and Marcisz, K.: How far from a pristine state are the peatlands in the Białowieża Primeval Forest (CE Europe) – Palaeoecological insights on peatland and forest development from multi-proxy studies, Ecol. Indic., 143, 109421, https://doi.org/10.1016/j.ecolind.2022.109421, 2022.
González de Andrés, E., Shestakova, T. A., Scholten, R. C., Delcourt, C. J. F., Gorina, N. V., and Camarero, J. J.: Changes in tree growth synchrony and resilience in Siberian Pinus sylvestris forests are modulated by fire dynamics and ecohydrological conditions, Agr. Forest Meteorol., 312, 108712, https://doi.org/10.1016/j.agrformet.2021.108712, 2022.
Gorham, E., Janssens, J. A., Wheeler, G. A., and Glaser, P. H.: The Natural and Anthropogenic Acidification of Peatlands, in: Effects of Atmospheric Pollutants on Forests, Wetlands and Agricultural Ecosystems, edited by: Hutchinson, T. C. and Meema, K. M., NATO ASI Series, vol 16, Springer, Berlin, Heidelberg, 493–512, https://doi.org/10.1007/978-3-642-70874-9_36, 1987.
Gorzelak, A.: Zalesianie i zadrzewianie kraju, in: Z dziejów Lasów Państwowych i leśnictwa polskiego, Tom 3(1) Lata powojenne i współczesność, edited by: Bernadzki, E., 97–121, ISBN 83-88478-40-0, 2004.
Graham, E. B., Averill, C., Bond-Lamberty, B., Knelman, J. E., Krause, S., Peralta, A. L., Shade, A., Smith, A. P., Cheng, S. J., Fanin, N., Freund, C., Garcia, P. E., Gibbons, S. M., Van Goethem, M. W., Guebila, M. B., Kemppinen, J., Nowicki, R. J., Pausas, J. G., Reed, S. P., Rocca, J., Sengupta, A., Sihi, D., Simonin, M., Słowiński, M., Spawn, S. A., Sutherland, I., Tonkin, J. D., Wisnoski, N. I., Zipper, S. C., , C. C., Staal, A., Arora, B., Oldfield, C., Dwivedi, D., Larson, E., Santillan, E., Aaron Hogan, J., Atkins, J., Zheng, J., Lembrechts, J., Patel, K., Copes-Gerbitz, K., Winker, K., Mudge, L., Wong, M., Nuñez, M., Luoto, M., and Barnes, R.: Toward a Generalizable Framework of Disturbance Ecology Through Crowdsourced Science, Front. Ecol. Evol., 9, https://doi.org/10.3389/fevo.2021.588940, 2021.
Grimm, E. C.: CONISS: a FORTRAN 77 program for stratigraphically constrained cluster analysis by the method of incremental sum of squares, Comput. Geosci., 13, 13–35, https://doi.org/10.1016/0098-3004(87)90022-7, 1987.
Grodziski, S.: Historia ustroju społeczno-politycznego Galicji 1772–1848, Zakład Narodowy im. Ossolińskich. Wydawnictwo Polskiej Akademii Nauk, Wrocław, 1971.
Grosse-Brauckmann, G.: Über pflanzliche Makrofossilien mitteleuropäischer Torfe, TELMA – Berichte der Deutschen Gesellschaft für Moor-und Torfkunde, 2, 19–55, 1972.
Grosse-Brauckmann, G.: Über pflanzliche Makrofossilien mitteleuropäischer Torfe, TELMA – Berichte der Deutschen Gesellschaft für Moor-und Torfkunde, 4, 51–117, 1974.
Grosse-Brauckmann, G. and Streitz, B.: Pflanzliche Makrofossilien mitteleuropäischer Torfe. III Früchte, samen und einige gewebe (fotos von fossilen pflanzenresten), TELMA – Berichte der Deutschen Gesellschaft für Moor-und Torfkunde, 22, 53–102, 1992.
Groves, C. R., Jensen, D. B., Valutis, L. L., Redford, K. H., Shaffer, M. L., Scott, J. M., Baumgartner, J. V., Higgins, J. V., Beck, M. W., and Anderson, M. G.: Planning for Biodiversity Conservation: Putting Conservation Science into Practice: A seven-step framework for developing regional plans to conserve biological diversity, based upon principles of conservation biology and ecology, is being used extensively by the nature conservancy to identify priority areas for conservation, Bioscience, 52, 499–512, https://doi.org/10.1641/0006-3568(2002)052[0499:Pfbcpc]2.0.Co;2, 2002.
Grzybowski, M. and Glińska-Lewczuk, K.: The principal threats to the peatlands habitats, in the continental bioregion of Central Europe – A case study of peatland conservation in Poland, J. Nat. Conserv., 53, 125778, https://doi.org/10.1016/j.jnc.2019.125778, 2020.
Gunnarsson, U. and Rydin, H.: Demography and recruitment of Scots pine on raised bogs in eastern Sweden and relationships to microhabitat differentiation, Wetlands, 18, 133–141, https://doi.org/10.1007/BF03161450, 1998.
Halsall, K., Ellingsen, V., Asplund, J., Bradshaw, R., and Ohlson, M.: Fossil charcoal quantification using manual and image analysis approaches, Holocene, 28, 1345–1353, https://doi.org/10.1177/0959683618771488, 2018.
Hammer, Ø., Harper, D. A. T., and Ryan, P. D.: Past: Paleontological Statistics Software Package for Education and Data Analysis, Palaeontol. Electron., 4, 1–9, 2001.
Hänninen, H. and Tanino, K.: Tree seasonality in a warming climate, Trends Plant Sci., 16, 412–416, https://doi.org/10.1016/j.tplants.2011.05.001, 2011.
Harriman, R., Watt, A. W., Christie, A. E. G., Moore, D. W., McCartney, A. G., and Taylor, E. M.: Quantifying the effects of forestry practices on the recovery of upland streams and lochs from acidification, Sci. Total Environ., 310, 101–111, https://doi.org/10.1016/S0048-9697(02)00626-5, 2003.
Hartigan, J. A. and Wong, M. A.: A K-Means Clustering Algorithm, J. Roy. Stat. Soc. C-App., 28, 100–108, 1979.
Hartup, J., Ockendon, N., and Pettorelli, N.: Active versus passive restoration: Forests in the southern Carpathian Mountains as a case study, J. Environ. Manage., 322, 116003, https://doi.org/10.1016/j.jenvman.2022.116003, 2022.
Harvey, J. E., Smiljanić, M., Scharnweber, T., Buras, A., Cedro, A., Cruz-García, R., Drobyshev, I., Janecka, K., Jansons, Ā., Kaczka, R., Klisz, M., Läänelaid, A., Matisons, R., Muffler, L., Sohar, K., Spyt, B., Stolz, J., van der Maaten, E., van der Maaten-Theunissen, M., Vitas, A., Weigel, R., Kreyling, J., and Wilmking, M.: Tree growth influenced by warming winter climate and summer moisture availability in northern temperate forests, Glob. Change Biol., 26, 2505–2518, https://doi.org/10.1111/gcb.14966, 2020.
Hawthorne, D., Courtney Mustaphi, C. J., Aleman, J. C., Blarquez, O., Colombaroli, D., Daniau, A.-L., Marlon, J. R., Power, M., Vannière, B., Han, Y., Hantson, S., Kehrwald, N., Magi, B., Yue, X., Carcaillet, C., Marchant, R., Ogunkoya, A., Githumbi, E. N., and Muriuki, R. M.: Global Modern Charcoal Dataset (GMCD): A tool for exploring proxy-fire linkages and spatial patterns of biomass burning, Quatern. Int., 488, 3–17, https://doi.org/10.1016/j.quaint.2017.03.046, 2018.
Hess, M.: Klimat Krakowa, Folia Geographica, Series Geographica-Physica, 8, 45–102, 1974.
Heyerdahl, E. K. and Card, V.: Implications of paleorecords for ecosystem management, Trends Ecol. Evol., 15, 49–50, https://doi.org/10.1016/S0169-5347(99)01779-6, 2000.
Heyerdahl, E. K., Loehman, R. A., and Falk, D. A.: Mixed-severity fire in lodgepole pine dominated forests: are historical regimes sustainable on Oregon's Pumice Plateau, USA?, Can. J. Forest Res., 44, 593–603, https://doi.org/10.1139/cjfr-2013-0413, 2014.
Higuera, P. E., Sprugel, D. G., and Brubaker, L. B.: Reconstructing fire regimes with charcoal from small-hollow sediments: a calibration with tree-ring records of fire, Holocene, 15, 238–251, https://doi.org/10.1191/0959683605hl789rp, 2005.
Hiiemaa, H., Mustasaar, M., Kohv, M., Hang, T., Jõeleht, A., Lasberg, K., and Kalm, V.: Geological settings of the protected Selisoo mire (northeastern Estonia) threatened by oil shale mining, Est. J. Earth Sci., 63, 97–107, https://doi.org/10.3176/earth.2014.09, 2014.
Hille, M. and den Ouden, J.: Improved recruitment and early growth of Scots pine (Pinus sylvestris L.) seedlings after fire and soil scarification, Eur. J. Forest Res., 123, 213–218, https://doi.org/10.1007/s10342-004-0036-4, 2004.
Holden, J., Chapman, P. J., and Labadz, J. C.: Artificial drainage of peatlands: hydrological and hydrochemical process and wetland restoration, Progress in Physical Geography: Earth and Environment, 28, 95–123, https://doi.org/10.1191/0309133304pp403ra, 2004.
Hölzl, R.: Historicizing Sustainability: German Scientific Forestry in the Eighteenth and Nineteenth Centuries, Sci. Cult., 19, 431–460, https://doi.org/10.1080/09505431.2010.519866, 2010.
Hrachowitz, M., Stockinger, M., Coenders-Gerrits, M., van der Ent, R., Bogena, H., Lücke, A., and Stumpp, C.: Reduction of vegetation-accessible water storage capacity after deforestation affects catchment travel time distributions and increases young water fractions in a headwater catchment, Hydrol. Earth Syst. Sci., 25, 4887–4915, https://doi.org/10.5194/hess-25-4887-2021, 2021.
Hua, Q., Turnbull, J. C., Santos, G. M., Rakowski, A. Z., Ancapichún, S., De Pol-Holz, R., Hammer, S., Lehman, S. J., Levin, I., Miller, J. B., Palmer, J. G., and Turney, C. S. M.: Atmospheric radiocarbon for the period 1950–2019, Radiocarbon, 64, 723–745, https://doi.org/10.1017/rdc.2021.95, 2021.
Ignatiev, Y. and Yermokhin, M.: Impact of climatic factors on the tree ring dynamics of Scots pine (Pinus sylvestris L.) in bog forests of northern Belarus, Botanika (Issledovaniya): sbornik nauchnykh trudov, 51, 77–84, 2022.
IPCC: Climate Change 2007: Synthesis Report. Contribution of Working Groups I, II and III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, edited by: Core Writing Team, Pachauri, R. K., and Reisinger, A., IPCC, Geneva, Switzerland, 104 pp., ISBN 92-9169-122-4, 2007.
Izdebski, A., Guzowski, P., Poniat, R., Masci, L., Palli, J., Vignola, C., Bauch, M., Cocozza, C., Fernandes, R., Ljungqvist, F. C., Newfield, T., Seim, A., Abel-Schaad, D., Alba-Sánchez, F., Björkman, L., Brauer, A., Brown, A., Czerwiński, S., Ejarque, A., Fiłoc, M., Florenzano, A., Fredh, E. D., Fyfe, R., Jasiunas, N., Kołaczek, P., Kouli, K., Kozáková, R., Kupryjanowicz, M., Lagerås, P., Lamentowicz, M., Lindbladh, M., López-Sáez, J. A., Luelmo-Lautenschlaeger, R., Marcisz, K., Mazier, F., Mensing, S., Mercuri, A. M., Milecka, K., Miras, Y., Noryśkiewicz, A. M., Novenko, E., Obremska, M., Panajiotidis, S., Papadopoulou, M. L., Pędziszewska, A., Pérez-Díaz, S., Piovesan, G., Pluskowski, A., Pokorny, P., Poska, A., Reitalu, T., Rösch, M., Sadori, L., Sá Ferreira, C., Sebag, D., Słowiński, M., Stančikaitė, M., Stivrins, N., Tunno, I., Veski, S., Wacnik, A., and Masi, A.: Palaeoecological data indicates land-use changes across Europe linked to spatial heterogeneity in mortality during the Black Death pandemic, Nat. Ecol. Evol., 6, 297–306, https://doi.org/10.1038/s41559-021-01652-4, 2022.
Janecka, K., Treydte, K., Piccinelli, S., Francon, L., Argelich Ninot, M., Edvardsson, J., Corona, C., Lehsten, V., and Stoffel, M.: Peatland trees record strong and temporally stable hydroclimate information in tree-ring δ13C and δ18O, Clim. Past, 21, 1679–1697, https://doi.org/10.5194/cp-21-1679-2025, 2025.
Janeczek, A., Dybaś, B., and Walczy, Ł.: Galicja na józefińskiej mapie topograficznej: 1779–1783. T. 8, cz. B, Faksymilia arkuszy 160–162, 180–182, 202–205, 226–229, 251–255, 283–286, Instytut Archeologii i Etnologii PAN, Warszawa, ISBN 978-83-65548-01-6, 2015.
Jaroszewicz, B., Cholewińska, O., Gutowski, J. M., Samojlik, T., Zimny, M., and Latałowa, M.: Białowieża Forest – A Relic of the High Naturalness of European Forests, Forests, 10, 849, https://doi.org/10.3390/f10100849, 2019.
Jassey, V. E. J., Reczuga, M. K., Zielińska, M., Słowińska, S., Robroek, B. J. M., Mariotte, P., Seppey, C. V. W., Lara, E., Barabach, J., Słowiński, M., Bragazza, L., Chojnicki, B. H., Lamentowicz, M., Mitchell, E. A. D., and Buttler, A.: Tipping point in plant–fungal interactions under severe drought causes abrupt rise in peatland ecosystem respiration, Glob. Change Biol., 24, 972–986, https://doi.org/10.1111/gcb.13928, 2018.
Jaszczak, R.: Urządzanie lasu w Polsce do 1939 roku. Część I – początki urządzania lasu na ziemiach polskich, Sylwan, 152, 13–21, https://doi.org/10.26202/sylwan.2006126, 2008a.
Jaszczak, R.: Urządzanie lasu w Polsce do 1939 roku. Część II – urządzanie lasu w Królestwie Polskim, Sylwan, 152, 3–13, https://doi.org/10.26202/sylwan.2006179, 2008b.
Jaszczak, R.: Urządzanie lasu w Polsce do 1939 roku. Część III – urządzanie lasu na ziemiach polskich w zaborze austriackim i pruskim, Sylwan, 152, 3–10, https://doi.org/10.26202/sylwan.2006180, 2008c.
Jędrejek, G.: Regulacje prawne dotyczące Ordynacji Zamojskiej, Roczniki Nauk Prawnych, 22, 7–19, 2012.
Jevšenak, J.: New features in the dendroTools R package: Bootstrapped and partial correlation coefficients for monthly and daily climate data, Dendrochronologia, 63, 125753, https://doi.org/10.1016/j.dendro.2020.125753, 2020.
Jevšenak, J. and Levanič, T.: dendroTools: R package for studying linear and nonlinear responses between tree-rings and daily environmental data, Dendrochronologia, 48, 32–39, https://doi.org/10.1016/j.dendro.2018.01.005, 2018.
Jewuła, Ł., Kargol, T., and Ślusarek, K.: Dwór, wieś i plebania w przestrzeni społecznej zachodniej Małopolski w latach 1772–1815, Towarzystwo Wydawnicze Historia Iagellonica, Kraków, ISBN 978-83-65080-09-7, 2015.
Jones, P. M.: Agricultural Enlightenment. Knowledge, Technology, and Nature, 1750–1840, Oxford University Press, Oxford, ISBN 978-0-19-871607-5, 2016.
Joosten, H.: Peatlands across the globe, in: Peatland Restoration and Ecosystem Services: Science, Policy and Practice, edited by: Bonn, A., Allott, T., Evans, M., Joosten, H., and Stoneman, R., Ecological Reviews, Cambridge University Press, Cambridge, 19–43, https://doi.org/10.1017/CBO9781139177788.003, 2016.
Joosten, H., Sirin, A., Couwenberg, J., Laine, J., and Smith, P.: The role of peatlands in climate regulation, in: Peatland Restoration and Ecosystem Services: Science, Policy and Practice, edited by: Bonn, A., Allott, T., Evans, M., Joosten, H., and Stoneman, R., Cambridge University Press, 63–76, https://doi.org/10.1017/CBO9781139177788.005, 2016.
Juggins, S.: C2 User guide. Software for ecological and palaeoecological data analysis and visualisation, University of Newcastle, Newcastle upon Tyne, UK, 69 pp., 2003.
Jurasinski, G., Ahmad, S., Anadon-Rosell, A., Berendt, J., Beyer, F., Bill, R., Blume-Werry, G., Couwenberg, J., Günther, A., Joosten, H., Koebsch, F., Koehn, D., Koldrack, N., Kreyling, J., Leinweber, P., Lennartz, B., Liu, H., Michaelis, D., Mrotzek, A., and Wrage-Mönnig, N.: From Understanding to Sustainable Use of Peatlands: The WETSCAPES Approach, Soil Systems, 4, https://doi.org/10.3390/soilsystems4010014, 2020.
Jurasinski, G., Barthelmes, A., Byrne, K. A., Chojnicki, B. H., Christiansen, J. R., Decleer, K., Fritz, C., Günther, A. B., Huth, V., Joosten, H., Juszczak, R., Juutinen, S., Kasimir, Å., Klemedtsson, L., Koebsch, F., Kotowski, W., Kull, A., Lamentowicz, M., Lindgren, A., Lindsay, R., Linkevičienė, R., Lohila, A., Mander, Ü., Manton, M., Minkkinen, K., Peters, J., Renou-Wilson, F., Sendžikaitė, J., Šimanauskienė, R., Taminskas, J., Tanneberger, F., Tegetmeyer, C., van Diggelen, R., Vasander, H., Wilson, D., Zableckis, N., Zak, D. H., and Couwenberg, J.: Active afforestation of drained peatlands is not a viable option under the EU Nature Restoration Law, Ambio, 53, 970–983, https://doi.org/10.1007/s13280-024-02016-5, 2024.
Kaplan, J. O., Krumhardt, K. M., and Zimmermann, N.: The prehistoric and preindustrial deforestation of Europe, Quaternary Sci. Rev., 28, 3016–3034, https://doi.org/10.1016/j.quascirev.2009.09.028, 2009.
Kazda, M.: Changes in alder fens following a decrease in the ground water table: results of a geographical information system application, J. Appl. Ecol., 32, 100–110, https://doi.org/10.2307/2404419, 1995.
Keeley, J. E.: Ecology and evolution of pine life histories, Ann. Forest Sci., 69, 445–453, https://doi.org/10.1007/s13595-012-0201-8, 2012.
Kļaviņa, Z., Kļaviņš, I., Lībiete, Z., and Šteinberga, I.: Acidification level variability in hemiboreal production forest drained peatland catchments and after different intensity regeneration fellings using critical loads modelling approach, J. Environ. Manage., 374, 124118, https://doi.org/10.1016/j.jenvman.2025.124118, 2025.
Klisz, M., Puchałka, R., Jakubowski, M., Koprowski, M., Netsvetov, M., Prokopuk, Y., and Jevšenak, J.: Local site conditions reduce interspecific differences in climate sensitivity between native and non-native pines, Agr. Forest Meteorol., 341, 109694, https://doi.org/10.1016/j.agrformet.2023.109694, 2023.
Klukowski, Z.: Wydawnictwo materiałów do dziejów Zamojszczyzny w latach wojny 1939–1944. T. 1–4, Drukarnia Powiatowa Rady Narodowej, Zamość, 1945–1947.
Koenig, I., Mulot, M., and Mitchell, E. A. D.: Taxonomic and functional traits responses of Sphagnum peatland testate amoebae to experimentally manipulated water table, Ecol. Indic., 85, 342–351, https://doi.org/10.1016/j.ecolind.2017.10.017, 2018.
Koks, A. H. W., Käärmelahti, S. A., Temmink, R. J. M., Smolders, A. J. P., van de Riet, B. P., Lamers, L. P. M., Peters, R. C. J. H., Fritz, C., and van Dijk, G.: Acidifying surface water and water level management promote Sphagnum health for peatland restoration and paludiculture, Ecol. Eng., 216, 107579, https://doi.org/10.1016/j.ecoleng.2025.107579, 2025.
Korzeń, K., Margielewski, W., and Nalepka, D.: Neoholocene palaeoenvironmental changes in the Southern Roztocze region (SE Poland): The Kobyle Jezioro raised bog case study, Quatern. Int., 386, 191–202, https://doi.org/10.1016/j.quaint.2015.06.001, 2015.
Kottek, M., Grieser, J., Beck, C., Rudolf, B., and Rubel, F.: World Map of the Köppen-Geiger climate classification updated, Meteorol. Z., 15, 259–263, https://doi.org/10.1127/0941-2948/2006/0130, 2006.
Kozaczka, M.: Gospodarka leśna Ordynacji Zamojskiej (1918–1939), Kwartalnik Historyczny, 109, 61–75, 2002.
Kozaczka, M.: Ordynacja Zamojska: 1919–1945, Wydawnictwo “Norbertinum”, Lublin, ISBN 83-7222-147-2, 2003.
Kożuchowski, K., Trepińska, J., and Wibig, J.: The air temperature in Cracow from 1826 to 1990: Persistence, fluctuations and the urban effect, Int. J. Climatol., 14, 1035–1049, https://doi.org/10.1002/joc.3370140908, 1994.
Kreyling, J.: Winter climate change: a critical factor for temperate vegetation performance, Ecology, 91, 1939–1948, https://doi.org/10.1890/09-1160.1, 2010.
Kruczkowska, B., Jonczak, J., Słowińska, S., Bartczak, A., Kramkowski, M., Uzarowicz, Ł., Tyszkowski, S., and Słowiński, M.: Stages of soil development in the coastal zone of a disappearing lake – a case study from central Poland, J. Soil. Sediment., 21, 1420–1436, https://doi.org/10.1007/s11368-021-02880-8, 2021.
Kubrak, Z.: Wpływ osadnictwa i przemysłu leśnego na proces wylesienia i przemiany drzewostanu w Puszczy Solskiej (XVI–XVIII w.), in: Wierny swemu dziedzictwu. Księga jubileuszowa dedykowana profesorowi Józefowi Półćwiartkowi, edited by: Nabywaniec, S., Lorens, B., and Zabraniak, S., Wydawnictwo Uniwersytetu Rzeszowskiego, Rzeszów, 175–189, ISBN 978-83-7338-589-4, 2010.
Kuuluvainen, T.: Conceptual models of forest dynamics in environmental education and management: keep it as simple as possible, but no simpler, Forest Ecosystems, 3, 1–9, https://doi.org/10.1186/s40663-016-0075-6, 2016.
Kuuluvainen, T. and Rouvinen, S.: Post-fire understorey regeneration in boreal Pinus sylvestris forest sites with different fire histories, J. Veg. Sci., 11, 801–812, https://doi.org/10.2307/3236550, 2000.
Laiho, R. and Finér, L.: Changes in root biomass after water-level drawdown on pine mires in southern Finland, Scand. J. Forest Res., 11, 251–260, https://doi.org/10.1080/02827589609382934, 1996.
Laine, J., Vasander, H., and Sallantaus, T.: Ecological effects of peatland drainage for forestry, Environ. Rev., 3, 286–303, https://doi.org/10.1139/a95-015, 1995.
Lamentowicz, M., Tobolski, K., and Mitchell, E.: Palaeoecological evidence for anthropogenic acidification of a kettle-hole peatland in northern Poland, Holocene, 17, 1185–1196, https://doi.org/10.1177/0959683607085123, 2007.
Lamentowicz, M., Milecka, K., Gałka, M., Cedro, A., Pawlyta, J., Piotrowska, N., Lamentowicz, Ł., and Van der Knaap, W. O.: Climate and human induced hydrological change since AD 800 in an ombrotrophic mire in Pomerania (N Poland) tracked by testate amoebae, macro-fossils, pollen and tree rings of pine, Boreas, 38, 214–229, https://doi.org/10.1111/j.1502-3885.2008.00047.x, 2009.
Lamentowicz, M., Gałka, M., Lamentowicz, Ł., Obremska, M., Kühl, N., Lücke, A., and Jassey, V. E. J.: Reconstructing climate change and ombrotrophic bog development during the last 4000years in northern Poland using biotic proxies, stable isotopes and trait-based approach, Palaeogeogr. Palaeocl., 418, 261–277, https://doi.org/10.1016/j.palaeo.2014.11.015, 2015.
Lamentowicz, M., Marcisz, K., Guzowski, P., Gałka, M., Diaconu, A.-C., and Kołaczek, P.: How Joannites' economy eradicated primeval forest and created anthroecosystems in medieval Central Europe, Sci. Rep., 10, 18775, https://doi.org/10.1038/s41598-020-75692-4, 2020.
Larssen, T. and Holme, J.: Afforestation, seasalt episodes and acidification – A paired catchment study in western Norway, Environ. Pollut., 139, 440–450, https://doi.org/10.1016/j.envpol.2005.06.012, 2006.
Larsson, L. A. and Larsson, P. O.: CDendro and CooRecorder (v. 9.3.1), Cybis Elektronik and Data AB, Saltsjöbaden, Sweden, 2018.
Lavoie, M., Paré, D., Fenton, N., Groot, A., and Taylor, K.: Paludification and management of forested peatlands in Canada: a literature review, Environ. Rev., 13, 21–50, https://doi.org/10.1139/a05-006, 2005.
Leifeld, J. and Menichetti, L.: The underappreciated potential of peatlands in global climate change mitigation strategies, Nat. Commun., 9, 1071, https://doi.org/10.1038/s41467-018-03406-6, 2018.
Leuschner, C. and Ellenberg, H.: Ecology of Central European Forests: Vegetation Ecology of Central Europe, Volume I, Springer International Publishing, Cham, https://doi.org/10.1007/978-3-319-43042-3, 2017a.
Leuschner, C. and Ellenberg, H.: The Central European Vegetation as the Result of Millennia of Human Activity, in: Ecology of Central European Forests: Vegetation Ecology of Central Europe, Volume I, Springer International Publishing, Cham, 31–116, https://doi.org/10.1007/978-3-319-43042-3_3, 2017b.
Leuschner, C. and Ellenberg, H.: Forest Plantations and Clearings, in: Ecology of Central European Forests: Vegetation Ecology of Central Europe, Volume I, edited by: Leuschner, C., and Ellenberg, H., Springer International Publishing, Cham, 607–632, https://doi.org/10.1007/978-3-319-43042-3_8, 2017c.
Lindbladh, M., Niklasson, M., and Nilsson, S. G.: Long-time record of fire and open canopy in a high biodiversity forest in southeast Sweden, Biol. Conserv., 114, 231–243, https://doi.org/10.1016/S0006-3207(03)00043-0, 2003.
Lindbladh, M., Fraver, S., Edvardsson, J., and Felton, A.: Past forest composition, structures and processes – How paleoecology can contribute to forest conservation, Biol. Conserv., 168, 116–127, https://doi.org/10.1016/j.biocon.2013.09.021, 2013.
Linderholm, H. W.: Climatic and Anthropogenic Influences on Radial Growth of Scots Pine at Hanvedsmossen, a Raised Peat Bog, in South Central Sweden, Geogr. Ann. A, 81, 75–86, https://doi.org/10.1111/j.0435-3676.1999.00050.x, 1999.
Linderholm, H. W. and Leine, M.: An assessment of twentieth century tree-cover changes on a southern Swedish peatland combining dendrochronoloy and aerial photograph analysis, Wetlands, 24, 357–363, https://doi.org/10.1672/0277-5212(2004)024[0357:AAOTCT]2.0.CO;2, 2004.
Linderholm, H. W., Moberg, A., and Grudd, H.: Peatland pines as climate indicators? A regional comparison of the climatic influence on Scots pine growth in Sweden, Can. J. Forest Res., 32, 1400–1410, https://doi.org/10.1139/x02-071, 2002.
Lowood, H. E.: The Calculating Forester: Quantification, Cameral Science, and the Emergence of Scientific Forestry Management in Germany, in: The Quantifying spirit in the 18th century, edited by: Frängsmyr, T., Heilbron, J. L., and Rider, R. E., University of California Press, Berkeley – Los Angeles – Oxford, 315–342, ISBN 0-520-07022-4, 1990.
Lubliner-Mianowska, K.: Mchy liściaste: klucz do oznaczania pospolitych gatunków niżowych ziem polskich, Państwowe Zakłady Wydawnictw Szkolnych, Warszawa, 1951.
Lubliner-Mianowska, K.: Torfowce: opisy i klucze do oznaczania gatunków krajowych, Państwowe Wydawnictwo Naukowe, Warszawa, 1957.
Łuców, D., Küttim, M., Słowiński, M., Kołaczek, P., Karpińska-Kołaczek, M., Küttim, L., Salme, M., and Lamentowicz, M.: Searching for an ecological baseline: Long-term ecology of a post-extraction restored bog in Northern Estonia, Quatern. Int., 607, 65–78, https://doi.org/10.1016/j.quaint.2021.08.017, 2022.
Łuców, D., Lamentowicz, M., Kołaczek, P., Łokas, E., Marcisz, K., Obremska, M., Theuerkauf, M., Tyszkowski, S., and Słowiński, M.: Pine Forest Management and Disturbance in Northern Poland: Combining High-Resolution 100-Year-Old Paleoecological and Remote Sensing Data, Front. Ecol. Evol., 9, https://doi.org/10.3389/fevo.2021.747976, 2021.
Łuców, D., Lamentowicz, M., Obremska, M., Arkhipova, M., Kittel, P., Łokas, E., Mazurkevich, A., Mróz, T., Tjallingii, R., and Słowiński, M.: Disturbance and resilience of a Sphagnum peatland in western Russia (Western Dvina Lakeland) during the last 300 years: A multiproxy, high-resolution study, Holocene, 30, 1552–1566, https://doi.org/10.1177/0959683620941064, 2020.
Lukenbach, M. C., Devito, K. J., Kettridge, N., Petrone, R. M., and Waddington, J. M.: Burn severity alters peatland moss water availability: implications for post-fire recovery, Ecohydrology, 9, 341–353, https://doi.org/10.1002/eco.1639, 2016.
Lukowski, J. and Zawadzki, H.: A Concise History of Poland, 2, Cambridge Concise Histories, Cambridge University Press, Cambridge, https://doi.org/10.1017/CBO9780511813856, 2006.
MacDonald, G. M., Beilman, D. W., Kremenetski, K. V., Sheng, Y., Smith, L. C., and Velichko, A. A.: Rapid Early Development of Circumarctic Peatlands and Atmospheric CH4 and CO2 Variations, Science, 314, 285–288, https://doi.org/10.1126/science.1131722, 2006.
Maciejewski, Z. and Szwagrzyk, J.: Long-term changes in stand composition of natural forest associations on the Roztocze Highlands (Eastern Poland), Pol. J. Ecol., 59, 535–549, 2011.
Mamakowa, K.: Roślinność Kotliny Sandomierskiej w późnym glacjale i Holocenie, Acta Palaeobotanica, 3, 1–57, 1962.
Manton, M., Grigaliūnas, V., Jarašius, L., Sendžiėkaitė, J., Tamulaitytė, G., and Grodzka-Łukaszewska, M.: Drained and forgotten peat extraction sites: economic and carbon impacts of peat and water loss in spontaneously forested Lithuanian peatlands, Scientific Review Engineering and Environmental Sciences (SREES), 34, 376–397, https://doi.org/10.22630/srees.10809, 2025.
Marcisz, K., Tinner, W., Colombaroli, D., Kołaczek, P., Słowiński, M., Fiałkiewicz-Kozieł, B., Łokas, E., and Lamentowicz, M.: Long-term hydrological dynamics and fire history over the last 2000 years in CE Europe reconstructed from a high-resolution peat archive, Quaternary Sci. Rev., 112, 138–152, https://doi.org/10.1016/j.quascirev.2015.01.019, 2015.
Marcisz, K., Colombaroli, D., Jassey, V. E. J., Tinner, W., Kołaczek, P., Gałka, M., Karpińska-Kołaczek, M., Słowiński, M., and Lamentowicz, M.: A novel testate amoebae trait-based approach to infer environmental disturbance in Sphagnum peatlands, Sci. Rep., 6, 33907, https://doi.org/10.1038/srep33907, 2016.
Marcisz, K., Kołaczek, P., Gałka, M., Diaconu, A.-C., and Lamentowicz, M.: Exceptional hydrological stability of a Sphagnum-dominated peatland over the late Holocene, Quaternary Sci. Rev., 231, 106180, https://doi.org/10.1016/j.quascirev.2020.106180, 2020a.
Marcisz, K., Jassey, V. E. J., Kosakyan, A., Krashevska, V., Lahr, D. J. G., Lara, E., Lamentowicz, Ł., Lamentowicz, M., Macumber, A., Mazei, Y., Mitchell, E. A. D., Nasser, N. A., Patterson, R. T., Roe, H. M., Singer, D., Tsyganov, A. N., and Fournier, B.: Testate Amoeba Functional Traits and Their Use in Paleoecology, Front. Ecol. Evol., 8, https://doi.org/10.3389/fevo.2020.575966, 2020b.
Marcisz, K., Buczek, K., Gałka, M., Margielewski, W., Mulot, M., and Kołaczek, P.: Past testate amoeba communities in landslide mountain fens (Polish Carpathians): The relationship between shell types and sediment, Holocene, 31, 954–965, https://doi.org/10.1177/0959683621994647, 2021.
Marcisz, K., Czerwiński, S., Lamentowicz, M., Łuców, D., and Słowiński, M.: How paleoecology can support peatland restoration, Past Global Changes Magazine, 30, 12–13, https://doi.org/10.22498/pages.30.1.12, 2022.
Marcisz, K., Bąk, M., Lamentowicz, M., Kołaczek, P., Theurer, T., Matulewski, P., and Mauquoy, D.: Substantial changes in land and forest management led to critical transitions in peatland functioning over the last 700 years, Sci. Rep., 15, 18211, https://doi.org/10.1038/s41598-025-02580-0, 2025.
Marek, S.: Biologia i stratygrafia torfowisk olszynowych w Polsce, Zeszyty Problemowe Postępów Nauk Rolniczych, 57, 1–264, 1965.
Margielewski, W., Krąpiec, M., Jankowski, L., Urban, J., and Zernitskaya, V.: Impact of aeolian processes on peat accumulation: Late Glacial–Holocene history of the Hamernia peat bog (Roztocze region, south-eastern Poland), Quatern. Int., 386, 212–225, https://doi.org/10.1016/j.quaint.2015.07.016, 2015.
Margielewski, W., Krąpiec, M., Buczek, K., Korzeń, K., Szychowska-Krapiec, E., Pociecha, A., Pilch, J., Obidowicz, A., Sala, D., and Klimek, A.: Bog pine and deciduous trees chronologies related to peat sequences stratigraphy of the Podemszczyzna peatland (Sandomierz Basin, southeastern Poland), Radiocarbon, 64, 1–19, https://doi.org/10.1017/RDC.2022.38, 2022.
Marosz, M., Miętus, M., and Biernacik, D.: Features of Multiannual Air Temperature Variability in Poland (1951–2021), Atmosphere, 14, 282, https://doi.org/10.3390/atmos14020282, 2023.
Maxwell, R. S. and Larsson, L.-A.: Measuring tree-ring widths using the CooRecorder software application, Dendrochronologia, 67, 125841, https://doi.org/10.1016/j.dendro.2021.125841, 2021.
Mazei, Y. and Tsyganov, A.: Freshwater Testate Amoebae, KMK Scientific press, Moscow, 302 pp., ISBN 978-5-873317-336-2, 2006.
Mažeika, J., Guobytė, R., Kibirkštis, G., Petrošius, R., Skuratovič, Ž., and Taminskas, J.: The use of carbon-14 and tritium for peat and water dynamics characterization: case of Čepkeliai peatland, southeastern Lithuania, Geochronometria, 34, 41–48, https://doi.org/10.2478/v10003-009-0007-3, 2009.
Mazurek, Z.: Walka chłopów Ordynacji Zamojskiej o prawa serwitutowe w końcu XIX wieku, Annales Universitatis Mariae Curie-Skłodowska. Sectio F, Nauki Filozoficzne i Humanistyczne, 12, 197–220, 1957.
McBride, J. R.: Analysis of tree rings and fire scars to establish fire history, Tree-Ring Bulletin, 43, 51–67, 1983.
McClain, W. E., Ruffner, C. M., Ebinger, J. E., and Spyreas, G.: Patterns of Anthropogenic Fire within the Midwestern Tallgrass Prairie 1673–1905: Evidence from Written Accounts, Nat. Area J., 41, 283–300, https://doi.org/10.3375/20-5, 2021.
McGrath, M. J., Luyssaert, S., Meyfroidt, P., Kaplan, J. O., Bürgi, M., Chen, Y., Erb, K., Gimmi, U., McInerney, D., Naudts, K., Otto, J., Pasztor, F., Ryder, J., Schelhaas, M.-J., and Valade, A.: Reconstructing European forest management from 1600 to 2010, Biogeosciences, 12, 4291–4316, https://doi.org/10.5194/bg-12-4291-2015, 2015.
McMichael, C. N. H. and Bush, M. B.: Spatiotemporal patterns of pre-Columbian people in Amazonia, Quaternary Res., 92, 53–69, https://doi.org/10.1017/qua.2018.152, 2019.
McRae, D. J., Conard, S. G., Ivanova, G. A., Sukhinin, A. I., Baker, S. P., Samsonov, Y. N., Blake, T. W., Ivanov, V. A., Ivanov, A. V., Churkina, T. V., Hao, W. M., Koutzenogij, K. P., and Kovaleva, N.: Variability of fire behavior, fire effects, and emissions in Scotch pine forests of Central Siberia, Mitig. Adapt. Strat. Gl., 11, 45–74, https://doi.org/10.1007/s11027-006-1008-4, 2006.
McVean, D. N.: Ecology of Alnus Glutinosa (L.) Gaertn: V. Notes on Some British Alder Populations, J. Ecol., 44, 321–330, https://doi.org/10.2307/2256824, 1956.
Michelot, A., Simard, S., Rathgeber, C., Dufrêne, E., and Damesin, C.: Comparing the intra-annual wood formation of three European species (Fagus sylvatica, Quercus petraea and Pinus sylvestris) as related to leaf phenology and non-structural carbohydrate dynamics, Tree Physiol., 32, 1033–1045, https://doi.org/10.1093/treephys/tps052, 2012.
Miętus, M., Owczarek, M., and Filipiak, J.: Warunki termiczne na obszarze Wybrzeża i Pomorza w świetle wybranych klasyfikacji, Materiały Badawcze IMGW, Seria: Meteorologia, 36, Instytut Meteorologii i Gospodarki Wodnej, Warszawa, 2002.
Milner, A. M., Baird, A. J., Green, S. M., Swindles, G. T., Young, D. M., Sanderson, N. K., Timmins, M. S. I., and Gałka, M.: A regime shift from erosion to carbon accumulation in a temperate northern peatland, J. Ecol., 109, 125–138, https://doi.org/10.1111/1365-2745.13453, 2021.
Minkkinen, K., Byrne, K. A., and Trettin, C. C.: Climate Impacts of Peatland Forestry, in: Peatlands and climate change, edited by: Strack, M., International Peat Society, Jyväskylä, 98–122, ISBN 978-952-99401-1-0, 2008.
Misi, D., Puchałka, R., Pearson, C., Robertson, I., and Koprowski, M.: Differences in the Climate-Growth Relationship of Scots Pine: A Case Study from Poland and Hungary, Forests, 10, 243, https://doi.org/10.3390/f10030243, 2019.
Mitchell, E. A. D., Payne, R. J., and Lamentowicz, M.: Potential implications of differential preservation of testate amoeba shells for paleoenvironmental reconstruction in peatlands, J. Paleolimnol., 40, 603–618, https://doi.org/10.1007/s10933-007-9185-z, 2008.
Moore, P. D., Webb, J. A., and Collinson, M. E.: Pollen Analysis, Blackwell Scientific Publications, Oxford, ISBN, 2nd edn. 0-632-02176-4, 1991.
Murdoch, P. S., Baron, J. S., and Miller, T. L.: Potential effects of climate change on surface-water quality in North America, JAWRA J. Am. Water Resour. As., 36, 347–366, https://doi.org/10.1111/j.1752-1688.2000.tb04273.x, 2000.
Naveh, Z.: Interactions of landscapes and cultures, Landscape Urban Plan., 32, 43–54, https://doi.org/10.1016/0169-2046(94)00183-4, 1995.
Neal, C., Robinson, M., Reynolds, B., Neal, M., Rowland, P., Grant, S., Norris, D., Williams, B., Sleep, D., and Lawlor, A.: Hydrology and water quality of the headwaters of the River Severn: Stream acidity recovery and interactions with plantation forestry under an improving pollution climate, Sci. Total Environ., 408, 5035–5051, https://doi.org/10.1016/j.scitotenv.2010.07.047, 2010.
Niklasson, M. and Drakenberg, B.: A 600-year tree-ring fire history from Norra Kvills National Park, southern Sweden: implications for conservation strategies in the hemiboreal zone, Biol. Conserv., 101, 63–71, https://doi.org/10.1016/S0006-3207(01)00050-7, 2001.
Niklasson, M. and Granström, A.: Numbers and sizes of fires: Long-term spatially explicit fire history in a Swedish boreal landscape, Ecology, 81, 1484–1499, https://doi.org/10.2307/177301, 2000.
Niklasson, M., Lindbladh, M., and Bjorkman, L.: A long-term record of Quercus decline, logging and fires in a southern Swedish Fagus-Picea forest, J. Veg. Sci., 13, 765–774, https://doi.org/10.1658/1100-9233(2002)013[0765:Alroqd]2.0.Co;2, 2002.
Niklasson, M., Zin, E., Zielonka, T., Feijen, M., Korczyk, A. F., Churski, M., Samojlik, T., Jędrzejewska, B., Gutowski, J. M., and Brzeziecki, B.: A 350-year tree-ring fire record from Białowieża Primeval Forest, Poland: implications for Central European lowland fire history, J. Ecol., 98, 1319–1329, https://doi.org/10.1111/j.1365-2745.2010.01710.x, 2010.
Nisbet, T. and Evans, C. D.: Forestry and surface water acidification, Research note (Great Britain. Forestry Commission), FCRN016, Forestry Commission, [U.K.], ISBN 978-0-85538-900-0, 2014.
Novák, J., Sádlo, J., and Svobodová-Svitavská, H.: Unusual vegetation stability in a lowland pine forest area (Doksy region, Czech Republic), Holocene, 22, 947–955, https://doi.org/10.1177/0959683611434219, 2012.
Novenko, E. Y., Tsyganov, A. N., Payne, R. J., Mazei, N. G., Volkova, E. M., Chernyshov, V. A., Kupriyanov, D. A., and Mazei, Y. A.: Vegetation dynamics and fire history at the southern boundary of the forest vegetation zone in European Russia during the middle and late Holocene, Holocene, 28, 308–322, https://doi.org/10.1177/0959683617721331, 2018.
Nowacki, G. J. and Abrams, M. D.: The demise of fire and “Mesophication” of forests in the eastern United States, Bioscience, 58, 123–138, https://doi.org/10.1641/b580207, 2008.
Nowacki, G. J. and Abrams, M. D.: Is climate an important driver of post-European vegetation change in the Eastern United States?, Glob. Chang. Biol., 21, 314–334, https://doi.org/10.1111/gcb.12663, 2015.
Ohlson, M.: Differentiation in adaptive traits between neighbouring bog and mineral soil populations of Scots pine Pinus sylvestris, Ecography, 22, 178–182, https://doi.org/10.1111/j.1600-0587.1999.tb00466.x, 1999.
Ohlson, M. and Tryterud, E.: Interpretation of the charcoal record in forest soils: forest fires and their production and deposition of macroscopic charcoal, Holocene, 10, 519–525, https://doi.org/10.1191/095968300667442551, 2000.
Ohlson, M., Økland, R. H., Nordbakken, J.-F., and Dahlberg, B.: Fatal interactions between Scots pine and Sphagnum mosses in bog ecosystems, Oikos, 94, 425–432, https://doi.org/10.1034/j.1600-0706.2001.940305.x, 2001.
Oksanen, J., Simpson, G. L., Blanchet, F. G., Kindt, R., Legendre, P., Minchin, P. R., O'hara, R., Solymos, P., Stevens, M. H. H., Szoecs, E., Wagner, H., Barbour, M., Bedward, M., Bolker, B., Borcard, D., Borman, T., Carvalho, G., Chirico, M., De Caceres, M., Durand, S., Evangelista, H., FitzJohn, R., Friendly, M., Furneaux, B., Hannigan, G., Hill, M., Lahti, L., McGlinn, D., Ouellette, M., Ribeiro Cunha, E., Smith, T., Stier, A., Ter Braak, C., and Weedon, J.: Vegan: community ecology package, R package version 2.7-0, CRAN [code], https://doi.org/10.32614/CRAN.package.vegan, 2025.
Olsson, F., Gaillard, M.-J., Lemdahl, G., Greisman, A., Lanos, P., Marguerie, D., Marcoux, N., Skoglund, P., and Wäglind, J.: A continuous record of fire covering the last 10 500 calendar years from southern Sweden – The role of climate and human activities, Palaeogeogr. Palaeocl., 291, 128–141, https://doi.org/10.1016/j.palaeo.2009.07.013, 2010.
Olszewski, J. L.: Rola ekosystemów leśnych w modyfikacji klimatu lokalnego Puszczy Białowieskiej, Prace Habilitacyjne – Polska Akademia Nauk. Zakład Badania Ssaków, Zakład Narodowy im. Ossolińskich – Wydaw. Polskiej Akademii Nauk, Wrocław, ISBN 83-04-02437-3, 1986.
Opała, M.: The 443-Year Tree-Ring Chronology for the Scots Pine from Upper Silesia (Poland) as A Dating Tool and Climate Proxy, Geochronometria, 42, 41–52, https://doi.org/10.1515/geochr-2015-0005, 2015.
Oris, F., Ali, A. A., Asselin, H., Paradis, L., Bergeron, Y., and Finsinger, W.: Charcoal dispersion and deposition in boreal lakes from 3 years of monitoring: Differences between local and regional fires, Geophys. Res. Lett., 41, 6743–6752, https://doi.org/10.1002/2014GL060984, 2014.
Päivänen, J. and Hånell, B.: Peatland Ecology and Forestry – a Sound Approach, University of Helsinki, Department of Forest Sciences, Helsinki, ISBN 978-952-10-4531-8, 2012.
Pawlaczyk, P.: Bory i lasy bagienne, in: Monitoring siedlisk przyrodniczych. Przewodnik metodyczny. Część I, edited by: Mróz, W., GIOŚ, Warszawa, 216–235, ISBN 978-83-61227-52-6, 2010.
Payne, R. J. and Mitchell, E. A. D.: How many is enough? Determining optimal count totals for ecological and palaeoecological studies of testate amoebae, J. Paleolimnol., 42, 483–495, https://doi.org/10.1007/s10933-008-9299-y, 2009.
Pearson, M., Saarinen, M., Nummelin, L., Heiskanen, J., Roitto, M., Sarjala, T., and Laine, J.: Tolerance of peat-grown Scots pine seedlings to waterlogging and drought: Morphological, physiological, and metabolic responses to stress, Forest Ecol. Manage., 307, 43–53, https://doi.org/10.1016/j.foreco.2013.07.007, 2013.
Pędziszewska, A. and Latałowa, M.: Stand-scale reconstruction of late Holocene forest succession on the Gdańsk Upland (N. Poland) based on integrated palynological and macrofossil data from paired sites, Veg. Hist. Archaeobot., 25, 239–254, https://doi.org/10.1007/s00334-015-0546-7, 2016.
Pepin, S., Plamondon, A., and Britel, A.: Water relations of black spruce trees on a peatland during wet and dry years, Wetlands, 22, 225–233, https://doi.org/10.1672/0277-5212(2002)022[0225:WROBST]2.0.CO;2, 2002.
Peters, M. E. and Higuera, P. E.: Quantifying the source area of macroscopic charcoal with a particle dispersal model, Quaternary Res., 67, 304–310, https://doi.org/10.1016/j.yqres.2006.10.004, 2007.
Piao, S., Friedlingstein, P., Ciais, P., de Noblet-Ducoudré, N., Labat, D., and Zaehle, S.: Changes in climate and land use have a larger direct impact than rising CO2 on global river runoff trends, P. Natl. Acad. Sci. USA, 104, 15242–15247, https://doi.org/10.1073/pnas.0707213104, 2007.
Pickett, S. T. A., Cadenasso, M. L., and Meiners, S. J.: Ever since Clements: From Succession to Vegetation Dynamics and Understanding to Intervention, Appl. Veg. Sci., 12, 9–21, 2009.
Piha, A., Kuuluvainen, T., Lindberg, H., and Vanha-Majamaa, I.: Can scar-based fire history reconstructions be biased? An experimental study in boreal Scots pine, Can. J. Forest Res., 43, 669–675, https://doi.org/10.1139/cjfr-2012-0471, 2013.
Piller, J. J.: Continuatio edictorum et mandatorum universalium in Regnis Galicae et Lodomeriae a die 1 mensis Januar anno 1776 Emanatorum = Kontynuacya wyrokow y rozkazow powszechnych w Galicyi y Lodomeryi krolestwach od dnia 1. miesiąca stycznia roku 1776 wypadłych, in: Kontynuacya wyrokow y rozkazow powszechnych w Galicyi y Lodomeryi krolestwach od dnia 1. miesiąca stycznia roku 1776 wypadłych., edited by: Piller, J. J., Typis Josephae Piller C.R. Gubern. Typogr., Leopoli – Lwów – Lemberg, 151–173, 1782.
Pinheiro, J., Bates, D., and R Core Team: nlme: Linear and nonlinear mixed effects models, R Package Version 3.1–168, CRAN [code], https://doi.org/10.32614/CRAN.package.nlme, 2025.
Popielski, W.: Detailed Geological Map of Poland 1 : 50 000. Sheet 893 – Tereszpol (M-34-58-B), Państwowy Instytut Geologiczny, 1992.
Poska, A., Saarse, L., and Veski, S.: Reflections of pre- and early-agrarian human impact in the pollen diagrams of Estonia, Palaeogeogr. Palaeocl., 209, 37–50, https://doi.org/10.1016/j.palaeo.2003.12.024, 2004.
Potapov, A., Toomik, S., Yermokhin, M., Edvardsson, J., Lilleleht, A., Kiviste, A., Kaart, T., Metslaid, S., Järvet, A., and Hordo, M.: Synchronous Growth Releases in Peatland Pine Chronologies as an Indicator for Regional Climate Dynamics – A Multi-Site Study Including Estonia, Belarus and Sweden, Forests, 10, 1097, https://doi.org/10.3390/f10121097, 2019.
Przybylski, P., Związek, T., Kowalczyk, J., and Słowiński, M.: Research perspectives on historical legacy of the Scots pine (Pinus sylvestris L.): Genes as the silent actor in the transformation of the Central European forests in the last 200 years, Elem. Sci. Anth., 13, 00041, https://doi.org/10.1525/elementa.2024.00041, 2025.
Przybylski, T.: Autekologia i synekologia, in: Biologia sosny zwyczajnej, edited by: Białobok, S., Boratyński, A., and Bugała, W., Sorus, Poznań – Kórnik, 255–281, ISBN 83-85599-21-5, 1993.
Purre, A.-H. and Ilomets, M.: Relationships between bryophyte production and substrate properties in restored milled peatlands, Restor. Ecol., 26, 858–864, https://doi.org/10.1111/rec.12656, 2018.
Rajca, C.: Gospodarka leśna w Ordynacji Zamojskiej w pierwszej połowie XIX wieku. Uwagi o urządzeniu i organizacji, Roczniki Humanistyczne, 20, 207–217, 1972.
Ramchunder, S. J., Brown, L. E., and Holden, J.: Environmental effects of drainage, drain-blocking and prescribed vegetation burning in UK upland peatlands, Progress in Physical Geography: Earth and Environment, 33, 49–79, https://doi.org/10.1177/0309133309105245, 2009.
R Core Team: R: A language and environment for statistical computing, R Foundation for Statistical Computing, Vienna, Austria, https://www.R-project.org/ (last access: 3 April 2025), 2021.
Reimer, P. J., Austin, W. E. N., Bard, E., Bayliss, A., Blackwell, P. G., Bronk Ramsey, C., Butzin, M., Cheng, H., Edwards, R. L., Friedrich, M., Grootes, P. M., Guilderson, T. P., Hajdas, I., Heaton, T. J., Hogg, A. G., Hughen, K. A., Kromer, B., Manning, S. W., Muscheler, R., Palmer, J. G., Pearson, C., van der Plicht, J., Reimer, R. W., Richards, D. A., Scott, E. M., Southon, J. R., Turney, C. S. M., Wacker, L., Adolphi, F., Büntgen, U., Capano, M., Fahrni, S. M., Fogtmann-Schulz, A., Friedrich, R., Köhler, P., Kudsk, S., Miyake, F., Olsen, J., Reinig, F., Sakamoto, M., Sookdeo, A., and Talamo, S.: The IntCal20 Northern Hemisphere Radiocarbon Age Calibration Curve (0–55 cal kBP), Radiocarbon, 62, 725–757, https://doi.org/10.1017/RDC.2020.41, 2020.
Reynolds, B., Ormerod, S. J., and Gee, A. S.: Spatial patterns concentrations in upland Wales in relation to catchment forest cover and forest age, Environ. Pollut., 84, 27–33, https://doi.org/10.1016/0269-7491(94)90067-1, 1994.
Reynolds, B., Fowler, D., Smith, R. I., and Hall, J. R.: Atmospheric inputs and catchment solute fluxes for major ions in five Welsh upland catchments, J. Hydrol., 194, 305–329, https://doi.org/10.1016/S0022-1694(96)03226-X, 1997.
Ricotta, C. and Podani, J.: On some properties of the Bray-Curtis dissimilarity and their ecological meaning, Ecol. Complex., 31, 201–205, https://doi.org/10.1016/j.ecocom.2017.07.003, 2017.
Róg, D.: Zagadnienia identyfikacji osadnictwa budziarskiego w drugiej połowie XVIII i na początku XIX w. na przykładzie Puszczy Solskiej, Kwartalnik Historii Kultury Materialnej, 69, 193–218, https://doi.org/10.23858/KHKM69.2021.2.004, 2021.
Rolstad, J., Blanck, Y.-l., and Storaunet, K. O.: Fire history in a western Fennoscandian boreal forest as influenced by human land use and climate, Ecol. Monogr., 87, 219–245, https://doi.org/10.1002/ecm.1244, 2017.
Rösch, M.: Long-term human impact as registered in an upland pollen profile from the southern Black Forest, south-western Germany, Veg. Hist. Archaeobot., 9, 205–218, https://doi.org/10.1007/Bf01294635, 2000.
Ruffner, C. M. and Abrams, M. D.: Lightning Strikes and Resultant Fires from Archival (1912–1917) and Current (1960–1997) Information in Pennsylvania, J. Torrey Bot. Soc., 125, 249–252, https://doi.org/10.2307/2997223, 1998.
Rydin, H. and Jeglum, J. K.: The Biology of Peatlands, 2nd end., Oxford University Press, Oxford, https://doi.org/10.1093/acprof:osobl/9780199602995.001.0001, 2013.
Ryzhkova, N., Pinto, G., Kryshen, A., Bergeron, Y., Ols, C., and Drobyshev, I.: Multi-century reconstruction suggests complex interactions of climate and human controls of forest fire activity in a Karelian boreal landscape, North-West Russia, Forest Ecol. Manage., 459, 117770, https://doi.org/10.1016/j.foreco.2019.117770, 2020.
Samojlik, T., Rotherham, I. D., and Jędrzejewska, B.: Quantifying Historic Human Impacts on Forest Environments: A Case Study in Białowieża Forest, Poland, Environ. Hist., 18, 576–602, https://doi.org/10.1093/envhis/emt039, 2013a.
Samojlik, T., Jędrzejewska, B., Michniewicz, M., Krasnodębski, D., Dulinicz, M., Olczak, H., Karczewski, A., and Rotherham, I. D.: Tree species used for low-intensity production of charcoal and wood-tar in the 18th-century Białowieża Primeval Forest, Poland, Phytocoenologia, 43, 1–12, https://doi.org/10.1127/0340-269x/2013/0043-0511, 2013b.
Šamonil, P., Moravcová, A., Pokorný, P., Žáčková, P., Kašpar, J., Vašíčková, I., Daněk, P., Novák, J., Hájková, P., Adam, D., and Leuschner, H. H.: The disturbance regime of an Early Holocene swamp forest in the Czech Republic, as revealed by dendroecological, pollen and macrofossil data, Palaeogeogr. Palaeocl., 507, 81–96, https://doi.org/10.1016/j.palaeo.2018.07.001, 2018.
Sayedi, S. S., Abbott, B. W., Vannière, B., Leys, B., Colombaroli, D., Romera, G. G., Słowiński, M., Aleman, J. C., Blarquez, O., Feurdean, A., Brown, K., Aakala, T., Alenius, T., Allen, K., Andric, M., Bergeron, Y., Biagioni, S., Bradshaw, R., Bremond, L., Brisset, E., Brooks, J., Brugger, S. O., Brussel, T., Cadd, H., Cagliero, E., Carcaillet, C., Carter, V., Catry, F. X., Champreux, A., Chaste, E., Chavardès, R. D., Chipman, M., Conedera, M., Connor, S., Constantine, M., Courtney Mustaphi, C., Dabengwa, A. N., Daniels, W., De Boer, E., Dietze, E., Estrany, J., Fernandes, P., Finsinger, W., Flantua, S. G. A., Fox-Hughes, P., Gaboriau, D. M., M.Gayo, E., Girardin, M. P., Glenn, J., Glückler, R., González-Arango, C., Groves, M., Hamilton, D. S., Hamilton, R. J., Hantson, S., Hapsari, K. A., Hardiman, M., Hawthorne, D., Hoffman, K., Inoue, J., Karp, A. T., Krebs, P., Kulkarni, C., Kuosmanen, N., Lacourse, T., Ledru, M.-P., Lestienne, M., Long, C., López-Sáez, J. A., Loughlin, N., Niklasson, M., Madrigal, J., Maezumi, S. Y., Marcisz, K., Mariani, M., McWethy, D., Meyer, G., Molinari, C., Montoya, E., Mooney, S., Morales-Molino, C., Morris, J., Moss, P., Oliveras, I., Pereira, J. M., Pezzatti, G. B., Pickarski, N., Pini, R., Rehn, E., Remy, C. C., Revelles, J., Rius, D., Robin, V., Ruan, Y., Rudaya, N., Russell-Smith, J., Seppä, H., Shumilovskikh, L., T.Sommers, W., Tavşanoğlu, Ç., Umbanhowar, C., Urquiaga, E., Urrego, D., Vachula, R. S., Wallenius, T., You, C., and Daniau, A.-L.: Assessing changes in global fire regimes, Fire Ecol., 20, 18, https://doi.org/10.1186/s42408-023-00237-9, 2024.
Schabel, H. G.: Deer and Dauerwald in Germany: Any Progress?, Wildl. Soc. Bull., 29, 888–898, http://www.jstor.org/stable/3784416, 2001.
Scheffer, M. and van Nes, E. H.: Shallow lakes theory revisited: various alternative regimes driven by climate, nutrients, depth and lake size, Hydrobiologia, 584, 455–466, https://doi.org/10.1007/s10750-007-0616-7, 2007.
Scheffer, M., Hosper, S. H., Meijer, M. L., Moss, B., and Jeppesen, E.: Alternative equilibria in shallow lakes, Trends Ecol. Evol., 8, 275–279, https://doi.org/10.1016/0169-5347(93)90254-M, 1993.
Seddon, A. W. R., Mackay, A. W., Baker, A. G., Birks, H. J. B., Breman, E., Buck, C. E., Ellis, E. C., Froyd, C. A., Gill, J. L., Gillson, L., Johnson, E. A., Jones, V. J., Juggins, S., Macias-Fauria, M., Mills, K., Morris, J. L., Nogués-Bravo, D., Punyasena, S. W., Roland, T. P., Tanentzap, A. J., Willis, K. J., Aberhan, M., van Asperen, E. N., Austin, W. E. N., Battarbee, R. W., Bhagwat, S., Belanger, C. L., Bennett, K. D., Birks, H. H., Bronk Ramsey, C., Brooks, S. J., de Bruyn, M., Butler, P. G., Chambers, F. M., Clarke, S. J., Davies, A. L., Dearing, J. A., Ezard, T. H. G., Feurdean, A., Flower, R. J., Gell, P., Hausmann, S., Hogan, E. J., Hopkins, M. J., Jeffers, E. S., Korhola, A. A., Marchant, R., Kiefer, T., Lamentowicz, M., Larocque-Tobler, I., López-Merino, L., Liow, L. H., McGowan, S., Miller, J. H., Montoya, E., Morton, O., Nogué, S., Onoufriou, C., Boush, L. P., Rodriguez-Sanchez, F., Rose, N. L., Sayer, C. D., Shaw, H. E., Payne, R., Simpson, G., Sohar, K., Whitehouse, N. J., Williams, J. W., and Witkowski, A.: Looking forward through the past: identification of 50 priority research questions in palaeoecology, J. Ecol., 102, 256–267, https://doi.org/10.1111/1365-2745.12195, 2014.
Shaver, G. R., Chapin, F., and Gartner, B. L.: Factors Limiting Seasonal Growth and Peak Biomass Accumulation in Eriophorum Vaginatum in Alaskan Tussock Tundra, J. Ecol., 74, 257–278, https://doi.org/10.2307/2260362, 1986.
Siemensma, F. J.: Microworld, world of amoeboid organisms, Kortenhoef, the Netherlands, https://arcella.nl (last access: 10 March 2023), 2019.
Sillasoo, U., Mauquoy, D., Blundell, A., Charman, D., Blaauw, M., Daniell, J. R. G., Toms, P., Newberry, J., Chambers, F. M., and Karofeld, E.: Peat multi-proxy data from Männikjärve bog as indicators of late Holocene climate changes in Estonia, Boreas, 36, 20–37, https://doi.org/10.1111/j.1502-3885.2007.tb01177.x, 2007.
Skre, O., Wielgolaski, F. E., and Moe, B.: Biomass and chemical composition of common forest plants in response to fire in western Norway, J. Veg. Sci., 9, 501–510, https://doi.org/10.2307/3237265, 1998.
Słowińska, S., Słowiński, M., Marcisz, K., and Lamentowicz, M.: Long-term microclimate study of a peatland in Central Europe to understand microrefugia, Int. J. Biometeorol., 66, 817–832, https://doi.org/10.1007/s00484-022-02240-2, 2022.
Słowiński, M., Marcisz, K., Płóciennik, M., Obremska, M., Pawłowski, D., Okupny, D., Słowińska, S., Borówka, R., Kittel, P., Forysiak, J., Michczyńska, D. J., and Lamentowicz, M.: Drought as a stress driver of ecological changes in peatland – A palaeoecological study of peatland development between 3500 BCE and 200 BCE in central Poland, Palaeogeogr. Palaeocl., 461, 272–291, https://doi.org/10.1016/j.palaeo.2016.08.038, 2016.
Słowiński, M., Lamentowicz, M., Łuców, D., Barabach, J., Brykała, D., Tyszkowski, S., Pieńczewska, A., Śnieszko, Z., Dietze, E., Jażdżewski, K., Obremska, M., Ott, F., Brauer, A., and Marcisz, K.: Paleoecological and historical data as an important tool in ecosystem management, J. Environ. Manage., 236, 755–768, https://doi.org/10.1016/j.jenvman.2019.02.002, 2019.
Słowiński, M., Brauer, A., Guzowski, P., Związek, T., Obremska, M., Theuerkauf, M., Dietze, E., Schwab, M., Tjallingii, R., Czaja, R., Ott, F., and Błaszkiewicz, M.: The role of Medieval road operation on cultural landscape transformation, Sci. Rep., 11, 20876, https://doi.org/10.1038/s41598-021-00090-3, 2021.
Słowiński, M., Związek, T., Swoboda, P., Niedzielski, M. A., Słowińska, S., Konopski, M., Jonczak, J., Kruczkowska, B., Chojnacka, A., Róg, D., Szewczyk, K., and Brykała, D.: Human impacts on environment in the preindustrial forest landscapes in Poland – An overview, Elem. Sci. Anth., 12, https://doi.org/10.1525/elementa.2023.00065, 2024.
Smiljanić, M. and Wilmking, M.: Drivers of stem radial variation and its pattern in peatland Scots pines: A pilot study, Dendrochronologia, 47, 30–37, https://doi.org/10.1016/j.dendro.2017.12.001, 2018.
Smiljanić, M., Seo, J.-W., Läänelaid, A., van der Maaten-Theunissen, M., Stajić, B., and Wilmking, M.: Peatland pines as a proxy for water table fluctuations: Disentangling tree growth, hydrology and possible human influence, Sci. Total Environ., 500–501, 52–63, https://doi.org/10.1016/j.scitotenv.2014.08.056, 2014.
Solińska-Górnicka, B.: Alder (Alnus glutinosa) carr in Poland, Tuexenia, 7, 329–346, 1987.
Speer, J. H.: Fundamentals of tree-ring research, The University of Arizona Press, Tucson, ISBN 978-0-8165-2684-0, 2010.
Spînu, A. P., Niklasson, M., and Zin, E.: Mesophication in temperate Europe: A dendrochronological reconstruction of tree succession and fires in a mixed deciduous stand in Białowieża Forest, Ecol. Evol., 10, 1029–1041, https://doi.org/10.1002/ece3.5966, 2020.
Stančikaitė, M., Gedminienė, L., Edvardsson, J., Stoffel, M., Corona, C., Gryguc, G., Uogintas, D., Zinkutė, R., Skuratovič, Ž., and Taraškevičius, R.: Holocene vegetation and hydroclimatic dynamics in SE Lithuania – Implications from a multi-proxy study of the Čepkeliai bog, Quatern. Int., 501, 219–239, https://doi.org/10.1016/j.quaint.2017.08.039, 2019.
Sterling, S. M., Ducharne, A., and Polcher, J.: The impact of global land-cover change on the terrestrial water cycle, Nat. Clim. Chang., 3, 385–390, https://doi.org/10.1038/nclimate1690, 2013.
Stivrins, N., Kalnina, L., Cerina, A., Reire, E., Kreslina, S., Ozola, I., Soms, J., and Veski, S.: Climate change impact on peatland dynamics during the Holocene in Latvia, Northeastern Europe, Catena, 254, 108965, https://doi.org/10.1016/j.catena.2025.108965, 2025.
Stockmarr, J.: Tables with spores used in absolute pollen analysis, Pollen et Spores, 13, 615–621, 1971.
Stokes, M. A. and Smiley, T. L.: An Introduction to Tree-Ring Dating, University of Chicago Press, Chicago, 1968.
Sugita, S., Gaillard, M.-J., and Broström, A.: Landscape openness and pollen records: a simulation approach, Holocene, 9, 409–421, https://doi.org/10.1191/095968399666429937, 1999.
Sullivan, M. E. and Booth, R. K.: The Potential Influence of Short-term Environmental Variability on the Composition of Testate Amoeba Communities in Sphagnum Peatlands, Microb. Ecol., 62, 80–93, https://doi.org/10.1007/s00248-011-9875-y, 2011.
Sutheimer, C. M., Meunier, J., Hotchkiss, S. C., Rebitzke, E., and Radeloff, V. C.: Historical fire regimes of North American hemiboreal peatlands, Forest Ecol. Manage., 498, 119561, https://doi.org/10.1016/j.foreco.2021.119561, 2021.
Svenning, J. C.: A review of natural vegetation openness in north-western Europe, Biol. Conserv., 104, 133–148, https://doi.org/10.1016/S0006-3207(01)00162-8, 2002.
Swindles, G. T., Morris, P. J., Mullan, D. J., Payne, R. J., Roland, T. P., Amesbury, M. J., Lamentowicz, M., Turner, T. E., Gallego-Sala, A., Sim, T., Barr, I. D., Blaauw, M., Blundell, A., Chambers, F. M., Charman, D. J., Feurdean, A., Galloway, J. M., Gałka, M., Green, S. M., Kajukało, K., Karofeld, E., Korhola, A., Lamentowicz, Ł., Langdon, P., Marcisz, K., Mauquoy, D., Mazei, Y. A., McKeown, M. M., Mitchell, E. A. D., Novenko, E., Plunkett, G., Roe, H. M., Schoning, K., Sillasoo, Ü., Tsyganov, A. N., van der Linden, M., Väliranta, M., and Warner, B.: Widespread drying of European peatlands in recent centuries, Nat. Geosci., 12, 922–928, https://doi.org/10.1038/s41561-019-0462-z, 2019.
Szabó, P.: Driving forces of stability and change in woodland structure: A case-study from the Czech lowlands, Forest Ecol. Manage., 259, 650–656, https://doi.org/10.1016/j.foreco.2009.11.026, 2010.
Szafran, B.: Bryophyta I. Musci – Mchy, Flora Słodkowodna Polski, 16, Państwowe Wydawnictwo Naukowe, Warszawa, 1963.
Szczygieł, R.: W Dawnej Rzeczypospolitej (1578–1795), in: Dzieje Biłgoraja, edited by: Markiewicz, J., Szczygieł, R., and Śladkowski, W., Wydawnictwo Lubelskie, Lublin, 11–72, ISBN 83-222-0307-1, 1985.
Taminskas, J., Edvardsson, J., Linkevičienė, R., Stoffel, M., Corona, C., and Tamkevičiūtė, M.: Combining multiple proxies to investigate water table fluctuations in wetlands: A case study from the Rėkyva wetland complex, Lithuania, Palaeogeogr. Palaeocl., 514, 453–463, https://doi.org/10.1016/j.palaeo.2018.11.004, 2019.
Tanneberger, F., Moen, A., Barthelmes, A., Lewis, E., Miles, L., Sirin, A., Tegetmeyer, C., and Joosten, H.: Mires in Europe – Regional Diversity, Condition and Protection, Diversity, 13, 381, https://doi.org/10.3390/d13080381, 2021.
Temmink, R. J. M., Cruijsen, P. M. J. M., Smolders, A. J. P., Bouma, T. J., Fivash, G. S., Lengkeek, W., Didderen, K., Lamers, L. P. M., and van der Heide, T.: Overcoming establishment thresholds for peat mosses in human-made bog pools, Ecol. Appl., 31, e02359, https://doi.org/10.1002/eap.2359, 2021.
Timbal, J., Bonneau, M., Landmann, G., Trouvilliez, J., and Bouhot-Delduc, L.: European non-boreal conifer forests, in: Ecosystems of the World. Coniferous Forests, edited by: Andersson, F., Ecosystems of the World, Coniferous Forests, Elsevier, Amsterdam – San Diego – Oxford – London, 131–162, ISBN 978-0-444-81627-6, 2005.
Tinner, W., Hofstetter, S., Zeugin, F., Conedera, M., Wohlgemuth, T., Zimmermann, L., and Zweifel, R.: Long-distance transport of macroscopic charcoal by an intensive crown fire in the Swiss Alps – implications for fire history reconstruction, Holocene, 16, 287–292, https://doi.org/10.1191/0959683606hl925rr, 2006.
Tipton, J., Marcisz, K., Łuców, D., Obremska, M., and Lamentowicz, M.: Testate amoebae as indicators of critical transitions in historical forest management: A case study of Scots pine monoculture, Holocene, 09596836261422219, https://doi.org/10.1177/09596836261422219, 2026.
Tobolski, K.: Przewodnik do oznaczania torfów i osadów jeziornych, Wydawnictwo Naukowe PWN, Warszawa, ISBN 83-01-13215-9, 2000.
Todorov, M. and Bankov, N.: An Atlas of Sphagnum-Dwelling Testate Amoebae in Bulgaria, Advanced Books, 1, e38685, https://doi.org/10.3897/ab.e38685, 2019.
Toet, S., Cornelissen, J. H. C., Aerts, R., van Logtestijn, R. S. P., de Beus, M., and Stoevelaar, R.: Moss Responses to Elevated CO2 and Variation in Hydrology in a Temperate Lowland Peatland, Plant Ecol., 182, 27–40, https://doi.org/10.1007/s11258-005-9029-8, 2006.
Trepińska, J.: Anomalie, cykle, trendy termiczne w klimatologii na przykładzie fluktuacji termicznych w Europie Środkowej w XIX i XX wieku, Acta Universitatis Nicolai Copernici, Geografia, 31, 307–326, 2000.
Trepińska, J., Ustrnul, Z., and Kowanetz, L.: Variability of the Air Temperature in Central Europe in the Years 1792–1995, Geographia Polonica, 70, 43–52, 1997.
Turetsky, M. R., Benscoter, B., Page, S., Rein, G., van der Werf, G. R., and Watts, A.: Global vulnerability of peatlands to fire and carbon loss, Nat. Geosci., 8, 11–14, https://doi.org/10.1038/ngeo2325, 2015.
Ustrnul, Z., Wypych, A., and Czekierda, D.: Air Temperature Change, in: Climate Change in Poland: Past, Present, Future, edited by: Falarz, M., Springer International Publishing, Cham, 275–330, https://doi.org/10.1007/978-3-030-70328-8_11, 2021.
Vachula, R. S. and Rehn, E.: Modeled dispersal patterns for wood and grass charcoal are different: Implications for paleofire reconstruction, Holocene, 33, 159–166, https://doi.org/10.1177/09596836221131708, 2023.
Väliranta, M., Korhola, A., Seppä, H., Tuittila, E.-S., Sarmaja-Korjonen, K., Laine, J., and Alm, J.: High-resolution reconstruction of wetness dynamics in a southern boreal raised bog, Finland, during the late Holocene: a quantitative approach, Holocene, 17, 1093–1107, https://doi.org/10.1177/0959683607082550, 2007.
Väliranta, M., Salojärvi, N., Vuorsalo, A., Juutinen, S., Korhola, A., Luoto, M., and Tuittila, E.-S.: Holocene fen–bog transitions, current status in Finland and future perspectives, Holocene, 27, 752–764, https://doi.org/10.1177/0959683616670471, 2017.
van der Knaap, W. O., Lamentowicz, M., van Leeuwen, J. F. N., Hangartner, S., Leuenberger, M., Mauquoy, D., Goslar, T., Mitchell, E. A. D., Lamentowicz, Ł., and Kamenik, C.: A multi-proxy, high-resolution record of peatland development and its drivers during the last millennium from the subalpine Swiss Alps, Quaternary Sci. Rev., 30, 3467–3480, https://doi.org/10.1016/j.quascirev.2011.06.017, 2011.
Van Geel, B., Bohncke, S. J. P., and Dee, H.: A palaeoecological study of an upper late glacial and holocene sequence from “de borchert”, the Netherlands, Rev.Palaeobot. Palyno., 31, 367–448, https://doi.org/10.1016/0034-6667(80)90035-4, 1980.
Van Horne, M. L. and Fulé, P. Z.: Comparing methods of reconstructing fire history using fire scars in a southwestern United States ponderosa pine forest, Can. J. Forest Res., 36, 855–867, https://doi.org/10.1139/X05-289, 2006.
Vannière, B., Colombaroli, D., Chapron, E., Leroux, A., Tinner, W., and Magny, M.: Climate versus human-driven fire regimes in Mediterranean landscapes: the Holocene record of Lago dell'Accesa (Tuscany, Italy), Quaternary Sci. Rev., 27, 1181–1196, https://doi.org/10.1016/j.quascirev.2008.02.011, 2008.
Verheyen, K., Bossuyt, B., Hermy, M., and Tack, G.: The land use history (1278–1990) of a mixed hardwood forest in western Belgium and its relationship with chemical soil characteristics, J. Biogeogr., 26, 1115–1128, https://doi.org/10.1046/j.1365-2699.1999.00340.x, 1999.
Vicente-Serrano, S. M., Beguería, S., and López-Moreno, J. I.: A Multiscalar Drought Index Sensitive to Global Warming: The Standardized Precipitation Evapotranspiration Index, J. Climate, 23, 1696–1718, https://doi.org/10.1175/2009JCLI2909.1, 2010.
Vitas, A. and Erlickytė, R.: Influence of droughts to the radial growth of Scots Pine (Pinus sylvestris L.) at different site conditions, Balt. For., 13, 10–16, 2007.
von Carlowitz, H. C.: Sylvicultura Oeconomica, Oder Haußwirtschaftliche Nachricht und Naturgemäßige Anweisung Zur Wilden Baum-Zucht, J. Fr. Braun, Leipzig, 1713.
Waddington, J. M., Morris, P. J., Kettridge, N., Granath, G., Thompson, D. K., and Moore, P. A.: Hydrological feedbacks in northern peatlands, Ecohydrology, 8, 113–127, https://doi.org/10.1002/eco.1493, 2015.
Wallenius, T.: Major Decline in Fires in Coniferous Forests – Reconstructing the Phenomenon and Seeking for the Cause, Silva Fenn, 45, 139–155, https://doi.org/10.14214/sf.36, 2011.
Warner, B. G., Kubiw, H. J., and Hanf, K. I.: An anthropogenic cause for quaking mire formation in southwestern Ontario, Nature, 340, 380–384, https://doi.org/10.1038/340380a0, 1989.
Waszak, N., Robertson, I., Puchałka, R., Przybylak, R., Pospieszyńska, A., and Koprowski, M.: Investigating the Climate-Growth Response of Scots Pine (Pinus sylvestris L.) in Northern Poland, Atmosphere, 12, 1690, https://doi.org/10.3390/atmos12121690, 2021.
Wheeler, A. J.: Vegetational succession, acidification and allogenic events as recorded in Flandrian peat deposits from an isolated Fenland embayment, New Phytol., 122, 745–756, https://doi.org/10.1111/j.1469-8137.1992.tb00103.x, 1992.
Wheeler, B. D. and Proctor, M. C. F.: Ecological gradients, subdivisions and terminology of north-west European mires, J. Ecol., 88, 187–203, https://doi.org/10.1046/j.1365-2745.2000.00455.x, 2000.
Whitehead, P. G., Wilby, R. L., Battarbee, R. W., Kernan, M., and Wade, A. J.: A review of the potential impacts of climate change on surface water quality, Hydrol. Sci. J., 54, 101–123, https://doi.org/10.1623/hysj.54.1.101, 2009.
Wickham, H.: ggplot2: Elegant Graphics for Data Analysis, 2nd edn., Springer-Verlag, New York, https://doi.org/10.1007/978-3-319-24277-4, 2016.
Wigley, T. M. L., Briffa, K. R., and Jones, P. D.: On the Average Value of Correlated Time Series, with Applications in Dendroclimatology and Hydrometeorology, J. Appl. Meteorol. Clim., 23, 201–213, https://doi.org/10.1175/1520-0450(1984)023<0201:OTAVOC>2.0.CO;2, 1984.
Wilczyński, S. B. and Kulej, M.: The influence of climate on the radial increment of larches of different provenances on the basis of the experiment in the Carpathian Mountains in Southern Poland, Eur. J. Forest Res., 132, 919–929, https://doi.org/10.1007/s10342-013-0731-0, 2013.
Williams, B. A., Venter, O., Allan, J. R., Atkinson, S. C., Rehbein, J. A., Ward, M., Di Marco, M., Grantham, H. S., Ervin, J., and Goetz, S. J.: Change in terrestrial human footprint drives continued loss of intact ecosystems, One Earth, 3, 371–382, https://doi.org/10.1016/j.oneear.2020.08.009, 2020.
Willis, K. J. and Birks, H. J. B.: What Is Natural? The Need for a Long-Term Perspective in Biodiversity Conservation, Science, 314, 1261–1265, https://doi.org/10.1126/science.1122667, 2006.
Wilmking, M., van der Maaten-Theunissen, M., van der Maaten, E., Scharnweber, T., Buras, A., Biermann, C., Gurskaya, M., Hallinger, M., Lange, J., Shetti, R., Smiljanic, M., and Trouillier, M.: Global assessment of relationships between climate and tree growth, Glob. Change Biol., 26, 3212–3220, https://doi.org/10.1111/gcb.15057, 2020.
Wochal, D., Marcisz, K., Barabach, J., Bąk, M., and Lamentowicz, M.: The Fen that vanished: The untold story of drainage and peat extraction in Bagno Chlebowo peatland with implications for nature conservation, Global Ecology and Conservation, 61, e03647, https://doi.org/10.1016/j.gecco.2025.e03647, 2025.
Yamaguchi, D. K.: A Simple Method for Cross-Dating Increment Cores from Living Trees, Can. J. Forest Res., 21, 414–416, https://doi.org/10.1139/X91-053, 1991.
Yermokhin, M., Barsukova, T., Uhlianets, S., Lukin, V., Knysh, N., Dudkina, L., Mychko, V., and Bernackijj, D.: Dynamics and condition of bog and boggy pine forests in Belovezhskaya Pushcha, Botanika (Issledovaniya): sbornik nauchnykh trudov, 50, 171–194, 2021.
Zajączkowski, G., Jabłoński, M., Jabłoński, T., Szmidla, H., Kowalska, A., Małachowska, J., Piwnicki, J., and Kaliszewski, A.: Raport o stanie lasów w Polsce 2022, Centrum Informacyjne Lasów Państwowych, Warszawa, https://www.bdl.lasy.gov.pl/portal/Media/Default/Publikacje/raport_o_stanie_lasow_2022.pdf (last access: 24 April 2026), 2023.
Zarzycki, K., Trzcińska-Tacik, H., Różański, W., Szeląg, Z., Wołek, J., and Korzeniak, U.: Ecological indicator values of vascular plants of Poland, Biodiversity of Poland, 2, W. Szafer Institute of Botany, Polish Academy of Sciences, Kraków, ISBN 83-85444-95-5, 2002.
Zhang, H., Amesbury, M. J., Piilo, S. R., Garneau, M., Gallego-Sala, A., and Väliranta, M. M.: Recent Changes in Peatland Testate Amoeba Functional Traits and Hydrology Within a Replicated Site Network in Northwestern Québec, Canada, Front. Ecol. Evol., 8, https://doi.org/10.3389/fevo.2020.00228, 2020.
Zimny, M., Latałowa, M., and Pędziszewska, A.: The Late-Holocene history of forests in the Strict Reserve of Białowieża National Park, in: The Forests of the Strict Reserve of Białowieża National Park, edited by: Keczyński, A., Białowieski Park Narodowy, Białowieża, 29–59, ISBN 978-83-64513-15-2, 2017.
Zin, E., Drobyshev, I., Bernacki, D., and Niklasson, M.: Dendrochronological reconstruction reveals a mixed-intensity fire regime in Pinus sylvestris-dominated stands of Białowieża Forest, Belarus and Poland, J. Veg. Sci., 26, 934–945, https://doi.org/10.1111/jvs.12290, 2015.
Zin, E., Kuberski, Ł., Drobyshev, I., and Niklasson, M.: First Spatial Reconstruction of Past Fires in Temperate Europe Suggests Large Variability of Fire Sizes and an Important Role of Human-Related Ignitions, Front. Ecol. Evol., 10, https://doi.org/10.3389/fevo.2022.768464, 2022.
Zin, E., Związek, T., Klisz, M., Słowińska, S., Róg, D., Obremska, M., Łuców, D., Pietruczuk, J., Popek, J., Piotrowicz, K., Pilch, K., Szewczyk, K., Halaś, A., and Słowiński, M.: Dataset for “From Alnus to Pinus: temperate peatland ecosystem transformation triggered by human-driven landscape change”, Zenodo [data set], https://doi.org/10.5281/zenodo.19330702, 2026.
Związek, T., Łuców, D., Popek, J., Klisz, M., Obremska, M., Sobechowicz, Ł., Solon, J., Słowiński, M., Przybylski, P., Tyburski, Ł., Zin, E., Jastrzębowski, S., Płaczkowska, E., Pilch, K., Szewczyk, K., Konczal, A. A., Rutkowski, P., Główka, D., and Swoboda, P.: Addressing multiple perspectives in studying environmental changes in forest landscapes during the modernization period (18th–19th centuries), Anthropocene Review, 11, 302–328, https://doi.org/10.1177/20530196231205485, 2024.