the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Seafloor chemosynthetic habitats and AOM-influenced sediment microbiome at a cold-water coral site off the Vesterålen coast, northern Norway
Claudio Argentino
Luca Fallati
Sebastian Petters
Hans C. Bernstein
Ines Barrenechea Angeles
Jorge Corrales-Guerrero
Alessandra Savini
Benedicte Ferré
Giuliana Panieri
Cold seeps associated with cold-water corals have been reported worldwide. Yet, there are still knowledge gaps regarding ecological relationships due to contrasting observations. As a part of a larger multidisciplinary project on cold seeps off the Vesterålen coast (northern Norway), we analysed sediment (carbon-nitrogen systematics, foraminifera) and pore fluid (sulfate, dissolved inorganic carbon, methane) geochemistry and seafloor habitats (orthomosaics and habitat maps). Microbial mats are the dominant seep-related community, forming white patches of a few ten cm in diameter, mostly distributed along the edges of methane-derived authigenic carbonates and on sedimented spots. Mats are associated with sulfate-methane transition zones as shallow as 5 cm and high production of 13C-depleted dissolved inorganic carbon, an evidence of ongoing anaerobic oxidation of methane, also recorded in foraminiferal tests. We also report the discovery of a macroscopic white biofilm, observed while slicing a pushcore onboard. Organic matter analyses indicated that the sediment interval hosting this biofilm is associated with a sharp drop in δ13C values, as negative as −43.4 ‰. Results from 16S rRNA gene analyses on the uppermost 10 cm in the same core showed a significant shift in microbial community. Protebacteria-dominated communities near the seafloor transition to a Halobacterota-dominated composition in correspondence of the biofilm interval, with amplicon sequence variants pointing to ANME-1b anaerobic methanotrophs. The presence of a methane-charged sediment substrate leading to carbonate crust formation, the heterogeneous topography and food supply by high-energy currents appears to be a prerequisite for cold-water corals development and their spatial distribution in this area. Our results provide biogeochemical and seafloor habitat context to cold-water corals, contributing to the understanding of potential influence of methane seepage on corals. Furthermore, the produced seafloor orthomosaics and habitat maps will facilitate the monitoring of habitat changes in response to future climate-driven environmental disturbances.
- Article
(8422 KB) - Full-text XML
-
Supplement
(601 KB) - BibTeX
- EndNote
Cold seeps are seafloor areas characterized by the emission of hydrocarbons, mostly methane, into the water column. These environments are found along continental margins worldwide (Phrampus et al., 2020) and typically support chemosynthetic communities that rely on chemical energy from reduced compounds (H2S, CH4) contained in the fluids (Joye, 2020). Their association with reef-building corals inhabiting deep (>40 m) and cold waters, known as cold-water corals (CWCs), has been reported worldwide, i.e., from offshore New Zealand (Liebetrau et al., 2010), Gulf of Cadiz (Pirlet et al., 2012; Rincón-Tomás et al., 2019), Gulf of Mexico (Schroeder, 2002), South China Sea (Xu et al., 2019) and offshore Norway (Ferré et al., 2024; Hovland, 1990; Hovland and Thomsen, 1997). However, the biogeochemical relationships between cold seeps and CWCs remain unclear.
CWCs have been found at all continental margins, at water depths down to 3000 m and temperatures below 5 °C (Freiwald, 2011). They include a variety of scleractinian corals which do not rely on photosynthesis to produce energy, but feed on plankton and other organic particles transported by currents (Hovland, 2008; Roberts et al., 2009). CWCs create a three-dimensional framework spanning tens of meters to kilometres in lateral extent and up to 40 m in thickness (Freiwald, 2011; Sanna et al., 2023). It provides protective habitats for fish and other invertebrates, as well as a hard substrate for the settlement of other organisms, i.e., foraminifera, hydrozoans, octocorals, serpulids, molluscs, bryozoans, brachiopods, and sponges (Freiwald, 2011). CWCs also represent nursery grounds and reproduction environments for commercial fish species (Husebø et al., 2002). Therefore, CWCs play a key role in deep-sea biodiversity and ecosystem functioning. Among them, Desmophyllum pertusum (known as Lophelia pertusa) is the most widespread and dispersed representative, very common in the North Atlantic (Roberts et al., 2009) and along the Norwegian coasts (Fosså et al., 2002; Sanna et al., 2023; Sundahl et al., 2020). It is known that besides adequate environmental conditions (temperature, food supplied by water current), CWCs require a hard substrate for colonization and growth of a large colony (Roberts et al., 2009). The substrate must be large and stable enough to resist strong currents. From that perspective, cold seeps represent optimal areas for their settling and growth as they are often associated with extensive carbonate pavements and/or mounds. During methane migration through the sediment, microbial communities consisting of methanotrophic archaea (ANME) and sulfate-reducing bacteria (SRB) (Boetius et al., 2000; Boetius and Wenzhöfer, 2013) carry out the anaerobic oxidation of methane (AOM) which is a globally widespread biofilter of this greenhouse gas (Egger et al., 2018; Lapham et al., 2024). AOM occurs in a relatively narrow zone within the sedimentary column, located at variable depths beneath the seafloor ranging from a few cm to several hundred meters, mainly depending on the magnitude of methane fluxes (Egger et al., 2018). Below the seafloor, sulfate is the quantitatively dominant electron acceptor available for microbes to oxidize methane, and sulfate-driven AOM releases bisulfide (HS−) and dissolved inorganic carbon in the form of bicarbonate (HCO) following Reaction (R1):
AOM-related sulfide fluxes (mainly in the form of H2S under in situ conditions, Reaction R2) toward the seafloor sustain chemosynthetic habitats, including chemosymbiotic mussels, clams, tubeworms, and microbial mats (Argentino et al., 2022; Fischer et al., 2012; Sahling et al., 2002; Sen et al., 2018b). The protracted release of bicarbonate via AOM into pore water leads to an increase in alkalinity, which eventually triggers the precipitation of authigenic carbonates (Blumenberg et al., 2015; Crémière et al., 2016; Karaca et al., 2010). Methane-derived authigenic carbonates (MDAC) range from microscopic concretions to kilometres-wide and several-meters-high deposits becoming exposed at the seafloor by winnowing and erosion (Judd et al., 2020; Judd and Hovland, 2007; Tseng et al., 2023). MDAC outcrops are ideal places for the settling and growth of CWCs which benefit from optimal exposure to bottom currents rich in nutrients (Rincón-Tomás et al., 2019). More recent studies suggested that CWCs can gain benefits not only from the presence of hard substrate (Deng et al., 2019; Rincón-Tomás et al., 2019; Xu et al., 2019) but also from chemosynthetically-derived food (Osman et al., 2023; Sert et al., 2025). Other studies found that the major food source in deep-sea corals is photosynthetically-derived food, even near cold seeps (Becker et al., 2009). Ocean acidification (OA) poses a potential threat to all coral reefs by reducing their growth rate (Kline et al., 2019) and inducing dissolution of their aragonitic skeletons (Eyre et al., 2018; Wisshak et al., 2012). Methane seeps can contribute to ocean acidification when methane (CH4) is oxidized to carbon dioxide (CO2) in the water column (Biastoch et al., 2011; Garcia-Tigreros et al., 2021; Hauri et al., 2024) following Reaction (R3):
Conducting further investigations on cold seep biogeochemistry at CWC sites is needed, since the reduction of CWC ecosystems brings societal and economic implications, in particular in the pharmaceutical, medical and food industry (Beaumont et al., 2007; Foley et al., 2010; Hilmi et al., 2013).
To respond to that call, a CWC-seep system offshore Vesterålen, Northern Norway, became the subject of the multidisciplinary project EMAN7 – Environmental impact of Methane seepage and sub-seabed characterization at LoVe-Node 7 – aiming to investigate the interplay between CWCs and methane emissions. Within EMAN7 project, we conducted an expedition onboard RV Kronprins Haakon in summer 2022 (CAGE22-3, Ferré et al., 2022) during which we also performed Remotely-Operated Vehicle (ROV) seafloor imagery acquisitions and targeted sediment samplings near Desmophyllum pertusum coral mounds. In this study, we combined the information obtained from seafloor orthomosaics and habitat maps with the geochemistry (pore fluids, organic matter, foraminiferal proxies) of shallow sediment cores. This multidisciplinary and multiproxy approach enabled us to understand how methane spatial and temporal dynamics regulate the type and distribution of benthic habitats and subsurface carbon cycling at this site. Moreover, we used 16S rRNA gene sequencing to explore downcore shifts in microbial diversity and representative taxonomy through a push core showing the presence of a macroscopic white biofilm at 10 cm depth. This study provides a comprehensive overview of the biogeochemical context and seafloor environment where the Vesterålen CWCs have developed, thus aiding in the assessment of the potential influence of methane seepage on corals. Additionally, the generated seafloor orthomosaics and habitat maps will support the monitoring of habitat changes in response to future climate-induced environmental perturbations.
The targeted cold seeps investigated here are located on the Norwegian continental shelf at ∼ 68.9° N and ∼ 14.3° E (Fig. 1A), 15.5 km from the Vesterålen coast (Fig. 1B). This area is geomorphologically associated with the Hola Trough, an elongated ∼ 12 km-wide depression carved by ice during Pleistocene glaciations (Ottesen et al., 2005). Water depths at the seep site range between 200 and 230 m, and the seafloor is wiped by strong bottom currents up to 1 m s−1 (Ferré et al., 2024). CTD measurements conducted in 2022 during the cruise CAGE22-3 indicated water temperatures of 6.7 °C near the seafloor increasing to 8 °C at the sea surface, with salinity levels of 34–35 psu (Ferré et al., 2024). The seepage systems have been known since 2008 (Chand et al., 2008) and have been the focus of studies on hydrocarbon composition (Sauer et al., 2015) and seepage paleo-reconstructions (Crémière et al., 2016). The origin of the gas showed a predominantly thermogenic component, including methane with δ13C values between −60 ‰ and −52 ‰ and δD between −225 ‰ and −191 ‰, associated with higher alkanes (C2+) (Sauer et al., 2015). This gas migrates from late Jurassic to early Cretaceous source rocks and has been seeping since at least ∼ 11 ka (Crémière et al., 2016), producing extensive MDAC pavements. The Hola Trough is of great biological interest due to the presence of more than 300 Desmophyllum pertusum reefs distributed over an area of almost 50 km2 with individual reefs having heights up to 17 m and widths >300 m in the longest direction (NNW-SSE; Bøe et al., 2016). In the investigated seepage area, CWCs form two main mounds (appearing on the left side of the microbathymetry in Fig. 2C) but minor coral build-ups are also present over a larger area hosting gas seeps (Ferré et al., 2022, 2024). The ROV survey performed during CAGE22-3 (Ferré et al., 2022) enabled high-resolution seafloor mosaicking, habitat mapping, and video-guided coring at specific microhabitats.
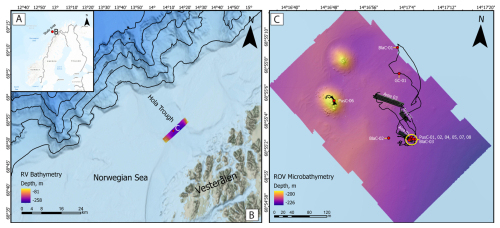
Figure 1(A) Geographical context of the study area located offshore Northern Norway. (B) Bathymetric map of the Hola Through and Norwegian shelf off the Vesterålen islands. Ship-based multibeam bathymetry is shown with Viridis color palette accessible to people with color vision deficiencies. (C) High-resolution ROV microbathymetry (Ferré et al., 2022, 2024) showing the sediment coring locations (red dots) and the three mosaic areas of Fig. 2. PusC-06 was excluded from analysis due to disturbance caused by reworking during sampling. Two CWC mounds marked by higher topography (yellow color) appear on the top-left corner. The ROV track is also displayed.
3.1 Seafloor ROV video acquisition and Structure from Motion processing
Seafloor imagery was collected in 2022 during the CAGE22-3 cruise (Ferré et al., 2022). During the second ROV dive of the cruise, three sub-areas (Area 01, Area 02, Area 03) were surveyed using high-resolution video acquisition for photogrammetric mosaicking (Fig. 1C). They were selected based on intense seep activity previously identified by screening the water column for gas flares (high backscatter) using R/V-mounted multibeam echosounder (MBES) data (Ferré et al., 2024). ROV Ægir 6000, a 150 Hp work-class vehicle equipped with various samplers and sensors and operated by the Norwegian Marine Robotics Laboratory (NORMAR) at the University of Bergen (UiB), was deployed with an EM 2024 Kongsberg MBES mounted on it and a dedicated photogrammetry sledge. The MBES was used to create a high-resolution microbathymetric map of the surveyed region (Fig. 1). Mounted on the ROV, this MBES captures detailed topographic data at sub-meter to decimetre scales, offering essential context for interpreting seafloor features in the imagery and improving the spatial precision of photogrammetric outputs. The photogrammetry sledge included a downward-facing Imenco® Spinner II Shark HD zoom camera mounted perpendicular to the seafloor. Illumination was provided by two high-output strobes (>2500 W) and a pair of deep-sea power lasers spaced 14 cm apart, ensuring consistent lighting and enabling scale calibration. ROV video acquisition, with the downward-facing camera, was conducted along parallel transects spaced 2 m apart, with the ROV maintaining a steady speed of 0.15 m s−1 and an altitude of 2.5 m. This configuration resulted in a field of view slightly greater than 2 m and ensured sufficient image overlap for photogrammetric reconstruction. These acquisition parameters were validated in previous field campaigns (Fallati et al., 2023; Panieri et al., 2024). In total, four ∼ 35 m transects were collected in Area 01, five ∼ 30 m transects in Area 02, and six ∼ 60 m transects in Area 03. ROV positioning for both MBES and photogrammetric surveys was achieved using the HIPAP 501 USBL (Ultra Short Baseline) system, ensuring high-precision localisation. These positional data were later integrated into the geospatial processing workflow to scale and georeference the resulting models accurately. ROV videos were processed by automatically extracting one frame per second using VLC Media Player's Scene Video Filter, producing high-definition (1920 × 1080 px) PNG images sorted by survey area. These images were imported into Agisoft Metashape Professional 2.0 (Agisoft 2018) and processed following a standard Structure from Motion (SfM) workflow (Fallati et al., 2020; Lim et al., 2020; Montes-Herrera et al., 2024; Price et al., 2019). After camera alignment, dense point clouds were generated to produce ultra-high-resolution digital elevation models (DEMs) and orthomosaics. The spacing between the laser pointers served as a ground-truth scale bar for each model. To georeference the models, USBL-derived coordinates and timestamps were linked to selected video frames, primarily those at the ends and centres of each transect. These data points were further cross-validated against geomorphic features visible in both the ROV-derived microbathymetry and the SfM-generated DEMs. This multisource integration enabled the accurate spatial placement of the models within the WGS 84/UTM Zone 33N coordinate system. Substrate semi-automatic classification was performed using Object-Based Image Analysis (OBIA) in the eCognition Developer (Trimble) environment. This approach segments the orthomosaics into meaningful image objects based on spectral, textural, and spatial characteristics (Benz et al., 2004; Blaschke, 2010; Hay and Castilla, 2008; Hossain and Chen, 2019), allowing for the supervised classification of distinct substrate classes, such as fine-grained sediments mixed with gravels and pebbles, microbial mats, boulders, and benthic organisms. The interpretation of the observed hardgrounds as MDAC pavements was based on previous extensive studies at this site which reported low δ13C values for these materials ranging from −38.0 ‰ to −22.2 ‰ and the incorporation of biomarkers typical of AOM-performing microbial consortia (Crémière et al., 2016; Sauer et al., 2017).
3.2 Sediment coring
Sediment samplings near the mounds and adjacent sediment were hindered by the presence of a stiff layer of coral debris and/or hardgrounds partially exposed at the seafloor. Moving away from the mounds, we managed to obtain cores for biogeochemical investigations and decided to focus on these areas for samplings and orthomosaicking. Six push cores, three blade cores, and one gravity core were collected during the expedition CAGE22-3 (Ferré et al., 2022) (Fig. 1C). The blade corer consisted of a 32 cm-long frame with a rectangular base of 25 × 10 cm. It was pushed into the seabed using the ROV manipulator arm and a locking system was activated to avoid sample loss. This technique is especially suitable for coarse-grained sediments. We collected three blade cores CAGE22-3-KH-01-BlaC-01, CAGE22-3-KH-01-BlaC-02, and CAGE22-3-KH-01-BlaC-03, hereafter named BlaC-01, BlaC-02 and BlaC-03, respectively. BlaC-01 was collected as a reference core for geochemical interpretations from a seafloor spot barren of chemosynthetic fauna (Ferré et al., 2022). BlaC-02 and BlaC-03 were collected from microbial mats. We also conducted push coring using a 60 cm-long cylindrical tube (8 cm inner diameter) made of fiberglass. The push corer was pushed into the sediment by the ROV during samplings and enabled us to obtain longer cores. We collected six push cores: CAGE22-3-KH-01-PusC-01, CAGE22-3-KH-01-PusC-02, CAGE22-3-KH-01-PusC-04, CAGE22-3-KH-01-PusC-05, CAGE22-3-KH-01-PusC-07 and CAGE22-3-KH-01-PusC-08, hereafter named PusC-01, PusC-02, PusC-04, PusC-05, PusC-07 and PusC-08, respectively and all collected from microbial mats. We also report the data from gravity corer CAGE22-3-KH-01-GC-01 (GC-01), which was collected using a 6 m-long iron barrel hosting a PVC liner with an inner diameter of 10 cm. On deck, the cores were split into 1 m-long sections (or shorter depending on the recovery), capped, labelled, and stored at 4 °C. Pore fluid analyses were conducted on all the cores, while PusC-08 and BlaC-01 were additionally selected for sediment geochemistry and foraminifera analyses. Preliminary microbiological analyses were only performed on PusC-08 after discovering a biofilm at 10 cm, suggesting strong biogeochemical and microbiological gradients.
3.3 Pore fluid samplings and analyses (CH4, SO, DIC, δ13CDIC)
Pore water extractions were conducted in a cold room (4 °C) immediately after coring operations. We sampled every 2 cm using rhizon soil moisture samplers (5 cm-long filter; 0.15 µm mesh) and 10 mL sterile plastic syringes. We split the pore water samples into two aliquots: one aliquot for dissolved inorganic carbon (DIC) analyses was transferred into 2 mL glass scintillation vials to which we added 10 µL of HgCl2 saturated solution. DIC samples were then stored in the dark at 4 °C; the other aliquot for sulfate analysis was transferred into 5 mL Eppendorf tubes and stored at −20 °C. DIC concentrations and isotopic composition (δ13C) were measured on a Thermo Fisher Scientific MAT 253 IRMS coupled to a Gasbench II at the Stable Isotope Laboratory (SIL) of the Department of Geosciences, UiT. Isotopic values were normalized to Vienna Pee Dee Belemnite standard (VPDB) using three in-house calcite reference materials covering a δ13C range from −48.95 ‰ to 1.96 ‰. DIC concentration was calculated by comparing the IRMS peak areas for the samples with calibration curves made from two NaHCO3(aq) stock solutions. Repeatability precision (1 s) of δ13C and DIC concentration based on three duplicate samples was better than or equal to 0.23 ‰ and 1.2 mM, respectively. The sulfate concentration was measured via ion chromatography at TosLab in Tromsø (NO). Accuracy and precision were estimated based on repeated measurements of certified materials. The measured values agreed within the uncertainty of the certified value and are commonly associated with a precision of 15 % (relative standard error RSE). Bulk sediment samples were collected from the bottom of push cores and from depths ranging from 5 to 15 cm in blade cores using a cut-off syringe. Around 5 mL of sediment were transferred to 20 mL serum vials containing two glass beads and 5 mL of 1 M NaOH. The vials were immediately closed with a rubber septum and aluminum crimp seal and stored upside-down at 4 °C. Methane concentration was measured at the SIL laboratory using a Thermo Fisher Scientific GC Trace 1310 gas chromatograph equipped with a Rt-Alumina BOND/Na2SO4 PLOT column and calibrated using 100 and 1000 ppm Calgaz and Linde calibration gases. Air was measured for quality control throughout the session. Injected volumes ranged from 10 to 1000 µL and measurement temperatures were set to 50–150 °C.
3.4 Benthic foraminifera isolation and isotope analyses (δ13C, δ18O)
We conducted down-core foraminiferal investigations in PusC-08 (microbial mat microhabitat) and in reference core BlaC-01. The isotopic composition of foraminifera was used to trace methane-derived carbon incorporation and/or MDAC precipitation as secondary overgrowth. This geochemical proxy is widely used in paleo-seep reconstructions (Argentino et al., 2021, 2024; Boretto et al., 2026; Schneider et al., 2018; Yao et al., 2020). We combined foraminifera geochemistry with pore water data to determine whether isotopic anomalies in foraminifera are associated with modern or ancient Sulfate-Methane Transition Zones (SMTZ). Push core PusC-08 and blade core BlaC-01 were sliced onboard every cm. For each sample, we weighed around 50 g of dry sediment and wet-sieved it through 63 and 125 µm sieves. We dried the residue at 50 °C and hand-picked benthic foraminifera with identification at genus or species level. Picking was conducted from the fraction >125 µm using a stereomicroscope. From each aliquot we selected 5 to 15 individuals for isotope analysis. The dominant species in most of the samples were Cibicidoides lobatulus and Cassidulina spp. Some of the samples were barren of these target foraminifera, thus resulting in a total of 41 and 13 samples for PusC-08 and BlaC-01, respectively. Isotope analyses were conducted at the SIL laboratory using a Thermo Fisher Scientific MAT 253 IRMS with a Gasbench II. Precision on two replicate reference materials (n=6) was better than 0.1 ‰ for both carbon and oxygen. All isotope results were normalized to Vienna Pee Dee Belemnite standard (VPDB) using three in-house reference materials ranging in δ13C and δ18O from −48.95 ‰ to 1.96 ‰ and from −18.59 ‰ to −2.15 ‰, respectively.
3.5 Sediment chemical analyses (TOC, TNd, δ13Corg, δ15Nd)
We conducted geochemical investigations of the organic matter in core PusC-08 and BlaC-01 to identify the presence of methanotrophic biomass in the sediment. In combination with pore water data, it is possible to discriminate between modern or ancient AOM-impacted intervals. Negative δ13Corg values lower than the ranges for marine and terrestrial end-members can be used as a proxy for AOM-associated microbial biomass in the sediment (Argentino et al., 2023, 2024; Ghosh et al., 2025; Szymczycha et al., 2025). We analysed a total of 45 sediment samples from PusC-08 and 15 samples from BlaC-01 collected every cm down the cores. Prior to the analyses, the samples were treated with 6 N HCl to remove the carbonate component following the protocol reported in Argentino et al. (2023). Analyses were conducted on a Thermo Fisher Scientific MAT 253 Isotope Ratio Mass Spectrometer (IRMS) coupled to a Flash HT Plus Elemental Analyzer hosted at the SIL laboratory. The δ13C and δ15N values were obtained by normalization to international standards, Vienna Pee Dee Belemnite, VPDB (δ13C) and Air-N2 (δ15N), using three in-house reference materials ranging in δ13C and δ15N from −40.44 ‰ to −9.14 ‰ and from −4.91 ‰ to 20.73 ‰, respectively. The total nitrogen in the decarbonated material (TNd) is hereafter assumed to represent organic nitrogen based on the close-to-zero-intercept of TNd versus total organic carbon (TOC) for TOC = 0 (e.g. Knies et al., 2007). This assumption allowed us to consider TNd values as mixtures of different organic sources, and the bulk nitrogen isotopic composition (δ15Nd) can be used for organic source interpretations (Argentino et al., 2023; Hu et al., 2020). Repeatability precision (1 s) of four duplicate samples measured in the same analytical session was better than or equal to 1.7 ‰, 0.12 %, 0.66 ‰, and 0.011 % for δ13Corg, TOC, δ15Nd, TNd, respectively. The atomic ratio was calculated using the atomic mass weighted ratio of TOC and TNd as = (TOC 12.011) (TN 14.007).
3.6 DNA extraction and sequencing
Sediment samples for DNA analyses were collected from PusC-08 immediately after core recovery using sterile spoons and stored frozen at −80 °C until processing. Samples were collected every cm in the interval 0–10 cm, to characterize any vertical gradient from the seafloor down to the biofilm-rich interval at 10 cm. For DNA extraction, 0.2–0.5 g of sediments was extracted using the DNeasy Power Soil Kit (Qiagen, Carlsbad, CA) and stored at −80 °C. DNA quality was measured by a NanoDrop Spectrophotometer and a Qubit Fluorometer (Invitrogen). The V4 region of the 16S SSU rRNA gene was amplified following the Earth Microbiome Project (EMP) (Gilbert et al., 2014) 16S Standard191Illumina library preparation protocol, using the forward-barcoded 515f (5′-3′192GTGYCAGCMGCCGCGGTAA) and 806r (5′-3′ GGACTACNVGGGTWTCTAAT). The quality of PCR products was assessed using 1 % agarose gel electrophoresis and GelRed staining, and three replicates per sample were chosen for sequencing. Illumina MiSeq sequencing was performed at the Environmental Sample Preparation and Sequencing Facility at Argonne National Laboratory (Lemont, IL, USA). The amplicon analyses were performed with the QIIME2 environment and using QIIME2 plugins as previously described in Aalto et al. (2022). Briefly, Illumina forward and reverse reads and the corresponding barcode files were imported and demultiplexed using the Earth Microbiome Project paired-end flag. All reads were quality filtered, de-replicated, and chimera-checked using all default parameters in DADA2 v2021.2.0 (Callahan et al., 2016). The reads were merged and amplicon sequence variants (ASVs) were determined using DADA2 v2021.2.0. The DADA2 statistic on sequence reads is provided in Data set S1. A 16S from the SILVA v138.1 database was trained using RESCRIPt (Quast et al., 2013; Robeson et al., 2021). The ASVs were classified with the self-trained classifier database (Yilmaz et al., 2014). Sequences are being processed for archiving with the European Nucleotide Archive and are available under accession number PRJEB96327.
Statistical analyses
All ASVs not assigned to the expected kingdom (Archaea and Bacteria) were removed. Downstream analysis was completed in R (R Core Team, 2021), using the “microeco” v0.11.0 (Liu et al., 2021) and “vegan” v2.5-7 packages (Oksanen et al., 2001). In particular, the R packages were used to calculate relative organismic abundances, observed alpha diversity, beta diversity using unweighted UniFrac, PERMANOVA, and Pearson correlation.
4.1 Orthomosaics and benthic coverage maps
The three orthomosaics (Fig. 2.A1, A2, A3), generated at millimetric resolution, cover a total area of 1307 m2 and generally show a hard seafloor composed of compact patches of MDAC, colonized by numerous benthic organisms, such as sponges, sea anemones, and sea fans. Outside the MDAC areas, the seafloor is characterized by fine sediments mixed with gravels and pebbles, which dominate the mapped area and include coral rubble.

Figure 2(A) Close-up of the microbathymetry area with high-resolution orthomosaics of the three surveyed subareas (Area 1, Area 2, Area 3). (B) Benthic coverage maps produced through OBIA showing the seafloor composition and distribution of methane-derived authigenic carbonate crusts (MDAC), fine sediments with gravels and pebbles, benthic organisms, and microbial mat patches. The analysis of the orthomosaics (A1, A2, A3) and the benthic coverage maps (B1, B2, B3) reveal subcircular microbial mats located near MDAC outcrops (C1, C2, C3). For a better view of small-scale features, we recommend accessing the dataset at https://doi.org/10.18710/1FCVAE (Argentino et al., 2026).
The high resolution of the orthomosaics enabled the detection and mapping of small, subcircular microbial mat patches, mainly found along the edges of the carbonate crusts (Fig. 2.C1, C2, C3). These patches vary in surface area from 0.002 to 0.17 m2, with an average of 0.02 m2 and an average perimeter of 0.58 m.
OBIA-derived benthic coverage maps identify the main seafloor classes in the high-resolution orthomosaics. Area 1 (Fig. 2.A1–B1), covering 312 m2, primarily consists of fine sediments with gravels, occupying about 267 m2. Microbial mats are located near the carbonate crusts (4.6 m2) in the south-central part of the area, with a total surface area of 0.26 m2. In the area, two benthic nodes (landers) are present, and well visible in the orhomosaic (Fig. 2.A1) as part of the project LoVeOcean, Lofoten-Vesterålen Ocean Observatory (https://loveocean.no/, last access: 24 August 2022).
Area 2 is smaller (Fig. 2.A2–B2), at 258 m2, and features a central, slightly elevated subcircular MDAC patch (42.2 m2), surrounded by fine sediments with gravels and pebbles (211 m2). This area also displays a more complex three-dimensional structure and hosts numerous benthic organisms and small microbial mat patches, which together cover 0.22 m2.
Area 3 comprises the largest portion of the mapped seafloor (Fig. 2.A3–B3), spanning 738 m2. Most of this area is dominated by fine sediments with gravels (437.7 m2), along with extensive, discontinuous MDAC outcrops (282 m2) concentrated in the northwestern section. Small, subcircular bacterial mats are also visible along the rims of the carbonate crusts, with a total surface coverage of 1.65 m2.
4.2 Pore water geochemistry and methane concentration
All pore water profiles from the 10 investigated cores are reported in the Supplement (Fig. S1) and Fig. 3. Two cores were selected based on completeness of the data (no gaps in either sulfate or DIC profiles) to discuss the subsurface biogeochemistry of microbial mats (Fig. 3). Sulfate concentration in surface sediments (0–1 cm below the seafloor) varies from a normal seawater value ∼ 29 mM (Millero, 2005) to as low as 24.3 mM (PusC-01; presence of a microbial mat at the seafloor). Sulfate values <1 mM are observed in two cores, PusC-02 (Fig. S1) and PusC-04 (Fig. 3), at 24 and 10 cm, respectively. Concentrations of dissolved inorganic carbon in surface sediments (0–1 cm) ranges from 2.2 mM (BlaC-01) to 5.1 mM (PusC-01), with overall increasing downcore trends (Fig. S1). DIC isotopic composition (δ13C) in surface sediment ranges from −22.5 ‰ (PusC-01) to −1.1 ‰ (BlaC-01) (Fig. S1). Isotopic values decrease with depth, with the most negative value δ13C = −29.8 ‰ measured at 6 cm in PusC-04 and at 18 cm in PusC-08, which also corresponds to an inversion in isotopic trends (becoming higher down the core). Methane concentration in samples collected from the bottom of the push cores and at 5–15 cm in blade cores are reported in the Supplement (Table S1). Methane concentrations show high values in pushcores, ranging 1.2 mM (PusC-02) to 3.4 mM (PusC-04). Blade cores contain trace amounts of methane, with measured values of 0.003 mM (BlaC-01; 10 cm depth), 0.010 mM (BlaC-02; 5 cm depth) and 1.1 mM (BlaC-03; 15 cm depth).
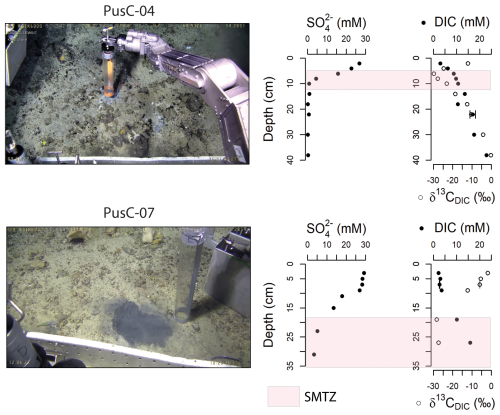
Figure 3Seafloor images and pore water geochemical profiles of selected sediment cores collected from the CWC area. Locations in relation to cold water coral mounds is shown on the microbathymetry of Fig. 1C and on the mosaics and habitat maps of Fig. 2. The SMTZ is marked with pink color in concomitance of a drop in sulfate concentrations and δ13CDIC, and an increase in DIC concentrations. Pore water profiles for all the investigated cores are reported in Fig. S1. Error bars refer to standard uncertainty and correspond to one standard deviation.
4.3 Foraminifera geochemistry
The carbon isotopic composition δ13C of Lobatula lobatula, an epifaunal species, and Cassidulina spp., an infaunal genus, shows a marked difference between reference core BlaC-01 and PusC-08. In BlaC-01, δ13C values remain relatively steady throughout the core with L. lobatula ranging from 0.8 ‰ to 1.9 ‰, and Cassidulina spp. ranging from −1.5 ‰ to −1.0 ‰. In push core PusC-08, the δ13C values for both taxa show a wider range and significant depletion. For L. lobatula, values ranged from −18.5 ‰ to 1.7 ‰ and for Cassidulina spp. from −15.0 ‰ to 1.0 ‰. A drop in isotopic values is observed at around 18 cm for both taxa, although more evident in Cassidulina spp. In the blade core, L. lobatula and Cassidulina spp. have δ18O values within the intervals 2.4 ‰–3.5 ‰ and 4.1 ‰–4.3 ‰. In PusC-08, the range of δ18O values are 2.0 ‰–4.1 ‰ and 1.8 ‰–4.6 ‰. BlaC-01 displays rather flat downcore isotope profiles (Fig. 4A), while PusC-08 shows a sharp increase in δ18O and decrease in δ13C starting at 20 cm below the seafloor (Fig. 4B).
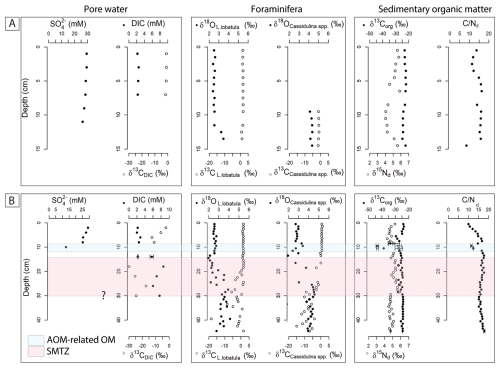
Figure 4Pore water, benthic foraminifera and sediment geochemical profiles of reference core BlaC-01 (A) and PusC-08 (B). The SMTZ is marked with pink color in PusC-08, whereas it has not been intercepted in BlaC-01. Question mark at the base of the SMTZ indicates uncertainty on the interpretation of its thickness due to missing pore water data below 30 cm. We marked in blue color the interval containing AOM-related organic matter in PusC-08, as determined by carbon-nitrogen interpretations (see full text). Error bars refer to standard uncertainty and correspond to one standard deviation.
4.4 Carbon-nitrogen geochemistry
The contents of total organic carbon in BlaC-01 range from 0.07 % to 0.58 % and are associated with δ13Corg values from −24.8 ‰ to −22.1 ‰. TNd contents range from 0.01 % to 0.06 %, with isotopic values δ15Nd between 4.1 ‰ and 5.8 ‰. The resulting values range from 8.5 to 16.3. PusC-08 has TOC contents ranging from 0.09 % to 1.78 %, associated with δ13Corg values from −43.4 ‰ to −23.6 ‰. TNd and δ15Nd varies between 0.01 % and 0.18 %, and between 4.3 ‰ and 5.9 ‰. values range from 10.0 to 17.9. A sharp decrease in δ13Corg and values is observed between 5–11 cm which matches positive peaks in TOC, TNd and δ15Nd (Fig. 4B). Similar patterns, though less pronounced, are observed at approximately 17, 25, and 40 cm. During onboard sediment slicing, we observed a macroscopic biofilm at a depth of 10 cm having white color and exhibiting a soft, slimy texture (Fig. S2).
4.5 Sediment microbiome
The phylum-level taxonomic composition of 16S rRNA gene–informed ASVs showed a steady, depth-structured shift across the 10 cm sediment column in PusC-08 (Fig. 5A). Triplicates from each depth were largely consistent, with the exception of single outlier replicates at 8 and 9 cm in phylum-level abundances. Across depths, relative abundances changed progressively: Proteobacteria exceeded 50 % at 1 cm but declined with depth, such that by 8 cm the community was largely composed of other phyla. In contrast, Desulfobacterota and Campylobacterota increased from the shallow to deeper layers. Around 7 cm, the profile shifted again, with Halobacterota increasing sharply below this horizon. Looking at the same microbial community on a family level (Fig. 5B), the aforementioned depth-structured shift was less apparent and steady. However, notable shifts could be documented. Thioglobaceae decreased over the first 3 cm of depth whereas Sulfurovaeae increased moving down to 7 cm. Below 7 cm of sediment depth, a clear cut in the community structure could be observed after which organisms of the ANME families could be detected.
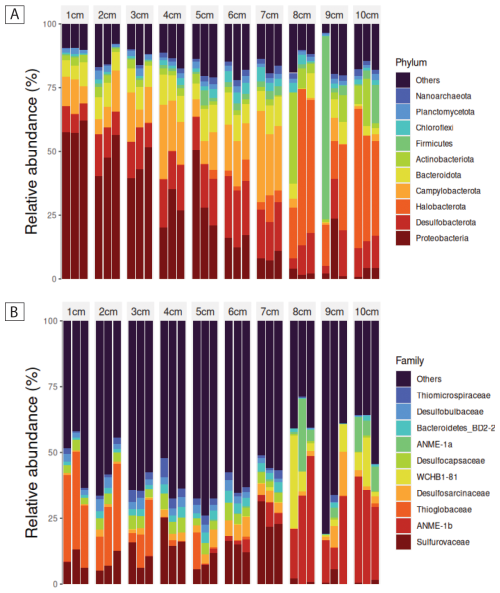
Figure 5(A) Relative abundance of 16S rRNA gene-informed ASVs on phylum level in the first 10 cm of PusC-08. (B) Relative abundance of 16S rRNA gene-informed ASVs on family level in the first 10 cm of PusC-08.
Alpha- and beta-diversity patterns indicate a depth-structured reorganization of the microbiome across the push-core. Alpha diversity (observed ASVs) rose steadily from 1 to 6 cm, consistent with increasing richness/niche diversification in the upper layers, then declined below 6 cm, coincident with the depth at which the taxonomic profile began to turn over. Beta diversity resolved three depth-associated clusters: a cohesive group comprising all samples from 1–6 cm; a clearly separated group comprising 8–10 cm; and 7 cm samples positioned between them, consistent with a transitional assemblage. Together, these patterns in PusC-08 point to a stratified microbiome with a boundary around 6–7 cm, above which communities are richer and more similar to one another, and below which communities are less diverse and compositionally distinct.
5.1 Cold seep biogeochemistry and microhabitats
Chemosynthetic habitats found at cold seeps rely on hydrogen sulfide fluxes from the SMTZ which are maintained by AOM-sustained sulfate reduction (Foucher et al., 2007; Levin, 2005; Levin et al., 2016). There is a causative link between magnitude of methane fluxes, depth of the SMTZ, sulfide fluxes and type of chemosynthetic communities at the seafloor (Argentino et al., 2022; Fischer et al., 2012; Levin, 2005; Levin et al., 2016; Sahling et al., 2002). Hydrogen sulfide in aquatic environments is generally viewed as a toxicant able to interrupt cellular respiration due to its lipid solubility (Bagarinao, 1992; Riesch et al., 2015; Tobler et al., 2016). Therefore, at cold seeps, only organisms that have adapted to varying levels of sulfide exposure are present and their distribution follows subsurface geochemical gradients. Microbial mats are found in correspondence of the most intense seepage areas with shallower SMTZs (∼ 5–10 cm below the seafloor), whereas higher organisms (clams, mussels, tubeworms) in symbiosis with sulfur-oxidizing bacteria colonize lower-seepage intensity areas associated with deeper SMTZs (∼ 10–50 cm below the seafloor) (e.g. Fischer et al., 2012; Sen et al., 2018b; Lee et al., 2019; Argentino et al., 2022; Barrenechea et al., 2025). We identified the SMTZs in our cores where a drop in sulfate content matched a decrease in δ13CDIC and an increase in DIC concentration, evidence of ongoing AOM which consumes sulfate and releases 13C-depleted bicarbonate. In the study area, the SMTZ ranges from ∼ 5 cm (PusC-01, PusC-4) to ∼ 25 cm (PusC-02, PusC-07, PusC-08) (Figs. S1, 3). No SMTZs were intercepted in the other cores. Besides BlaC-01 (and probably gravity core GC-01), all the other cores were collected from microbial mats. However, while the SMTZ depths in PusC-01 and PusC-4 are typical of those expected from microbial mat habitats, the other cores from mats showed deeper SMTZs. To interpret the latter case, we considered the morphology and extent of the microhabitats and local biogeochemical and environmental conditions. The high-resolution seafloor imagery and habitat maps show that the sampled mats form small (∼ 200 cm2) and highly heterogeneous (shape, color) features with reduced mat thickness giving color shades from white to light grey (Fig. 2). Chemosynthetic mats can rapidly colonize new substrates (e.g. seep sites, vents, wood, whale carcasses) within a few months (Alain et al., 2004; Girard et al., 2020; Guezennec et al., 1998; Kalenitchenko et al., 2016), as well as being rapidly eroded away or consumed by grazers when the chemical substrate is no longer present (Niemann et al., 2013; Seabrook et al., 2019; Sen et al., 2018b; Thurber et al., 2013). The appearance of the mats might therefore suggest that the fluid supply from underneath has been recently decreasing. It is known that cold seeps can be dynamic environments characterized by spatial and temporal variations in gas seepage distribution (Ferré et al., 2020; Greinert, 2008). A substantial variability in seepage activity as seen in water column backscatter anomalies was reported between 2018 and 2022 at this site (Ferré et al., 2024). Temporal fluctuations in methane fluxes could also explain the concave shape of the sulfate concentration profiles observed in our dataset and deeper SMTZs in some microbial mat microhabitats (Fig. S1). The profile shapes range from concave-up (PusC-04, PusC08?) to concave-down (PusC-07), which is a common indication of rapid increases and decreases in methane flux, respectively (Hensen et al., 2003; Kasten et al., 2003; Minami and Tatsumi, 2012). Strong bottom currents present in this area (Ferré et al., 2022, 2024) might also contribute to the observed reduced surface area of the single microbial mat patches, since mats are sensitive to physical disturbances (Cardoso et al., 2019; Noffke, 2010; Pan et al., 2019). Although the ROV samplings were conducted accurately, targeting the inner part of the mat patches, it is possible that the anomalous profiles reflect a mixed signal due to the sampling area of our coring devices (push corer = 50 cm2; blade corer = 250 cm2) being comparable to the size of the investigated microhabitats. Moreover, the ex-situ pore water extraction using 5 cm long rhizon filters inserted transversally into the core stratigraphy is also yielding bulk geochemical values. In-situ sensors such as the Raman probe (Zhang et al., 2010, 2011), are crucial for resolving challenges associated with cm-scale horizontal variations in geochemical gradients in microhabitats of small extent, such as those mapped in our study. Regarding the spatial distribution of microhabitats, the seep carbonate pavements in the CWC area act as a barrier to upward migrating sulfide-rich fluids, controlling the distribution of mats around the edges of carbonate crusts or in apparently carbonate-free seafloor areas (Fig. 2). Siboglinid tubeworms are common at Arctic cold seeps (Argentino et al., 2022; Sen et al., 2018a, b) and represent the dominant benthic community at nearby seeps in canyons on the lower continental slope off Lofoten–Vesterålen islands (Sen et al., 2019). The general lack of tubeworms in the investigated seepage site must be related to the sedimentary substrate (Bellec et al., 2024; Sen et al., 2019), i.e. hard carbonate pavements and/or coarse sediment (Fig. 2), since tubeworms require softer sediments to settle (Hilário et al., 2011; Southward et al., 2005).
Geochemical anomalies in δ13C of foraminifera in PusC08 appear below ∼ 18 cm, with remarkably negative isotopic values compared to the reference core. Those anomalies match the modern SMTZ as indicated by DIC data and the sharp drop in sulfate concentration right above (Fig. 4). Based on these observations, we ascribe the foraminiferal anomalies to precipitation of methane-derived carbonate as secondary overgrowth on the tests. Oxygen isotopes in PusC-08 show high δ18O values (up to 4.6 ‰) and similarly high values seem recorded at the bottom of the reference core BlaC-01 (Fig. 4A). Previous measurements on pure carbonate crusts from this site (Crémière et al., 2016; Sauer et al., 2017) indicated seep carbonate precipitation in isotopic equilibrium with seawater, with no influence from gas hydrates, at least during Holocene. Additional foraminiferal investigations on longer sediment cores are needed to interpret the oxygen trends and reconstruct the history of seepage.
5.2 Geochemical and genetic indications of methanotrophic biomass within the sediments
In this section we interpret the geochemical anomalies measured in the sediment of PusC-08 and discuss the putative origin of a white biofilm found at 10 cm in the same core (Fig. S2). Generally, the bulk organic matter composition of marine sediments includes a mixture of marine algae and vascular plants debris from land (Burdige, 2005; Emerson and Hedges, 1988; Meyers, 1994, 1997). The elemental and isotopic signatures of these two categories have been widely used for assessing the contribution of marine versus terrestrial organic inputs in paleoceanography (e.g. Kang et al., 2007; Knies and Martinez, 2009; Hasegawa et al., 2013; Knies, 2022). Marine-derived organic matter is commonly associated with δ13C values between −23 ‰ and − 16 ‰ (Emerson and Hedges, 1988; Meyers, 1994), δ15N values as high as 7 ‰ (Wada, 1980), and ratios between 4 and 10 (Meyers, 1994). Terrestrial organic matter from C3 plants which dominate the Arctic regions has lower δ13C values ‰ (Meyers, 1994; Naidu et al., 2000; Rachold and Hubberten, 1999; Ruttenberg and Goñi, 1997), δ15N values ∼ 0 ‰ (Kienast et al., 2005), and ratios >20 (Emerson and Hedges, 1988; Meyers, 1994). These ranges can vary regionally but they hold true for most marine settings, with minor adjustments of the end-member compositions (Argentino et al., 2023; Knies et al., 2007; Knies and Martinez, 2009). In PusC-08, δ13Corg reaches values as negative as −43.4 ‰, which cannot be explained by a simple mixture of marine and terrestrial organic matter. Similar values in bulk sediment were reported from cold seeps in the SW Barents Sea (−42 ‰, Argentino et al., 2023), offshore Pakistan (−42 ‰, Yoshinaga et al., 2014) and interpreted as indicative of significant contributions of methanotrophic biomass to the organic matter pool. In fact, methanotrophic microbes inhabiting cold seeps incorporate variable amounts of isotopically-fractionated carbon from methane (Kurth et al., 2019; Stock et al., 2025; Wegener et al., 2008), which is known to carry negative δ13C signals, generally ‰ (Judd and Hovland, 2007; Milkov and Etiope, 2018; Whiticar, 2020). Therefore, we confidently ascribe the isotopically-depleted δ13C values in PusC-08 between 5.5 and 11.5 cm to higher contents of AOM consortium biomass. The concomitant drop in values in that interval supports this interpretation, since bacterial organic matter has low ratios ∼ 5 (Madigan et al., 2017). Differently from other studies reporting a decrease in δ15N in AOM-impacted layers (Argentino et al., 2023; Hu et al., 2020), here we observe a slight increase (Fig. 4). This difference can be due to different nitrogen assimilation pathways by AOM-SRB consortia and/or different nitrogen substrates isotopic composition (Dekas et al., 2009, 2014; Gruber and Galloway, 2008). This AOM-impacted interval hosts a biofilm and is stratigraphically located above the modern SMTZ (Fig. 4). Macroscopic biofilms similar to the one found in our study have been reported by Briggs et al. (2011) from methane seeps at Hydrate Ridge (offshore of Oregon, USA), northern Cascadia Margin (offshore of Vancouver Island, Canada), and the Indian Ocean (offshore India), and by Gründger et al. (2019) from gas hydrate pingoes in the NW Barents Sea. In those studies, the biofilms consist of ANME-SRB consortia performing AOM (Briggs et al., 2011; Gründger et al., 2019), and the reported isotopic values for the pure biofilm masses yielded δ13C as negative as −43 ‰ (Briggs et al., 2011). We did not conduct carbon-nitrogen analyses on the biofilm material to directly compare with previous studies, but we found a similar value (−43.4 ‰) in bulk sediment from the same interval. What leads to the accumulation of macroscopic masses is the occurrence of pockets, cracks or fractures within the sediment, which provide high accommodation space (Briggs et al., 2011; Gründger et al., 2019). In PusC-08 we observed a network of microfractures in the core but no clear correlation with biofilm position was noticed during sediment slicing. The lack of indication of paleo-SMTZs in geochemical profiles of foraminifera (Fig. 4B) enable us to exclude that the biofilm could represent past conditions with higher methane fluxes but rather suggests ongoing activity. The stratigraphic position of the biofilm suggests that there is methane bypassing the SMTZ and being locally consumed within fractures (Briggs et al., 2011).
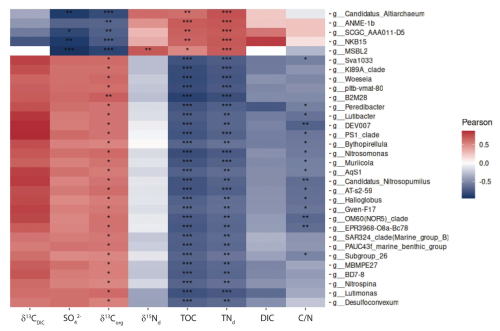
Figure 6Pearson correlation of environmental parameters and 16S rRNA gene-informed ASVs on genus level. Only genera with at least one high score “***” correlation were selected.
Correlations between biogeochemical parameters from pore waters and carbon-nitrogen geochemistry with 16S rRNA ASVs at the genus level reveal a clear division of the microbial community into two distinct groups, driven primarily by TOC, TNd, SO, and δ13Corg (Fig. 6). The first group, comprising archaeal taxa, is associated with organic-rich, nitrogen-rich, sulfate-depleted environments and plays a key role in methane oxidation and carbon fixation. In contrast, the second group, consisting largely of bacterial taxa, thrives in sulfate-rich, inorganic carbon-enriched conditions and is involved in sulfate reduction and sulfur cycling, highlighting the complementary roles of these groups in driving key biogeochemical processes. The first group includes the top 5 genera: Candidatus_Altiarchaeum, ANME-1b, SCGC_AAA011-D5, MSBL2, and NKB15. Among these, Candidatus_Altiarchaeum is known to act as a carbon dioxide sink, contributing to carbon fixation (Probst et al., 2014). Similarly, SCGC_AAA011-D5 belongs to the Nanoarchaeota archaeon, which is also adapted to anaerobic conditions. MSBL2 (classified as Methylocella) is a methanotroph, suggesting its involvement in methane oxidation. Together, these taxa likely contribute to carbon fixation, methane cycling, and organic matter degradation. The negative correlation with δ13Corg suggests that these taxa, particularly ANME-1b, use isotopically light organic matter, likely incorporating the strongly negative isotopic signature of methane-derived carbon into their biomass, further supporting their potential link to AOM. The second group is more diverse, with many taxa positively correlated with SO and δ13Corg, suggesting adaptation to sulfate-rich environments and involvement in sulfur cycling, likely including sulfate-reducing bacteria (SRB). Other genera showing strong inverse correlations with sulfate concentration in pore water and with δ13Corg correspond to sulfate-reducing Deltaproteobacteria which assimilates bicarbonate released by organoclastic sulfate reduction in marine sediments, i.e. MSBL2 (Vuillemin et al., 2022). The positive correlation with δ13C could indicate preference for isotopically heavier carbon sources, potentially linked to sulfur-based metabolic pathways. Other correlations relate to genera which are found in “normal” marine sediments unaffected by seepage. Future metagenomic analyses will be conducted on samples from PusC-08 for identifying the key microbial players and their ecological roles.
5.3 Geomorphological and biogeochemical factors influencing the distribution of CWCs at the Vesterålen site
Along the Norwegian coast, several coral fields have been mapped over the last decades by MAREANO (https://www.mareano.no, last access: 17 December 2025; Hovland, 2008), with some reef associations with hydrocarbon seeps. Among them, the Hola Trough became a case study to investigate the relationships between corals and seeps and monitor their response to environmental perturbations such as ocean acidification. Our study is part of a larger set of investigations conducted within the EMAN7 project. Here we did not focus the analyses on corals, but on the cold seep environment over which they superimpose. Still, we can discuss our results in light of recently-published studies from EMAN7 (Ferré et al., 2024; Sert et al., 2025), and previous work in the same area (e.g. Crémière et al., 2016; Hovland, 1990; Hovland and Thomsen, 1997; Sauer et al., 2015, 2017), to draw some lines of evidence about the geological and biogeochemical influence of seeps on CWCs. During ROV Dives conducted in 2022 (Ferré et al., 2022) microbial mats and MDACs were frequently observed in direct contact (either adjacent to or at their base) with coral mounds shown in Fig. 1b (Ferré et al., 2024), although in our orthomosaics and habitat maps covering the broader seepage environment (Fig. 2) corals are absent. Based on ROV imagery and the generated datasets, we found that CWCs in this area are mostly associated with chemosynthetic substrates (MDAC, microbial mats) and bubbling (Ferré et al., 2024). This is true also for the site to the north-east of the three mosaics, where hundreds of corals occur inside an intense gas emission area (Ferré et al., 2024). From a sediment perspective, we do not expect any difference in methane-related biogeochemical processes getting closer or further from CWCs, with AOM-associated sulfide fluxes sustaining chemosynthesis at the seafloor in spots where the MDAC “seal” is broken. Sub-bottom profiles crossing the two main mounds and reported in Ferré et al. (2024) revealed that the CWCs occur on top of topographic highs, likely related to glacial deposits (Buhl-Mortensen et al., 2012), which overly gas accumulations. Gas seeps identified outside of the profile coverage also show a correlation with small positive structures in the seafloor morphology. Our microbathymetric maps revealed a relatively flat morphology dominated by carbonate pavements. There is a strong topographic control for both gas migration in the shallow subsurface and coral occurrence. The presence of widespread MDAC deposits close to or at the seafloor explains the gas migration paths, which concentrate the gas toward topographic highs. Long-lasting seepage of deep-sourced thermogenic gas (Crémière et al., 2016; Sauer et al., 2015, 2017) resulted in the formation of widespread hard substrate, while the elevated structures shaped by glacial activity provided better exposure to strong bottom currents that supply food to the corals. At the CWC mounds, cold seeps influence the chemical and isotopic composition of dissolved organic carbon (Sert et al., 2025). Whether there is any seep-derived carbon uptake from corals remains unclear. Future results from ongoing coral transplantation experiments at this site will offer insights into this and other biochemical processes.
Methane seepage in the Hola area off the coast of Vesterålen (northern Norway) is known for its peculiar association with Desmophyllum pertusum cold water corals, but only recently it was possible to explore the distribution of seafloor ecosystems and to conduct microhabitat-specific samplings for biogeochemical investigations. Using ROV imagery collected in 2022 during expedition CAGE22-3, we generated three orthomosaics at millimetric resolution and habitat maps covering a total area of 1307 m2 in proximity of two coral mounds. A striking feature of this site is the presence of widespread MDAC pavements, partially exposed at the seafloor. White microbial mats forming small patches with an average area of 200 cm2 thrive along the edges of exposed carbonate crusts and on sedimented spots. We discussed the peculiar morphology and extent of mat microhabitats at this site, which are influenced by seep-related and environmental factors: widespread carbonate cap that channels rising fluids, spatial and temporal variations in methane flux, and strong bottom currents. We report the pore fluid composition of nine ROV-guided pushcores and blade cores from mats and background areas to compare their subsurface biogeochemistry and link it to seafloor habitats. Sulfate-methane transition zones as shallow as ∼ 5 cm and an increase in DIC carrying a 13C-depleted methane signature are measured beneath the mats, indicating high methane fluxes and intense AOM. Pore water profiles show concave-up and concave-down shapes, which reflect temporal variations in methane flux in agreement with variable seepage activity observed in cruises in the same area. Remarkably negative δ13C values (as low as −18.5 ‰) in foraminiferal tests were found in association with the modern SMTZ in one core, suggesting precipitation of MDAC as secondary overgrowth. Organic matter (TOC, δ13C, TNd, δ15N) and 16S rRNA gene analyses on this core provide evidence for the accumulation of methanotrophic biomass leading to bulk sediment δ13Corg values as negative as −43.4 ‰ and putatively attributed to ANME-1b anaerobic methane-oxidizing archaea. The same AOM-impacted interval hosts a macroscopic white biofilm located above the modern SMTZ that will be characterized in future studies.
Microbathymetry and orthomosaics from the EMAN7 Expedition 2022 are openly available on Dataverse.NO at https://doi.org/10.18710/1FCVAE (Argentino et al., 2026). Geochemistry data are available on Dataverse.NO at https://doi.org/10.18710/1FCVAE (Argentino et al., 2026). Raw sequencing DNA data have been submitted to the European Nucleotide Archive (ENA) and is available under accession PRJEB96327.
The supplement related to this article is available online at https://doi.org/10.5194/bg-23-3139-2026-supplement.
G.P. and C.A. conceived the study and conducted the sediment and pore fluid samplings onboard, C.A. wrote the initial manuscript with specific inputs from S.B., H.C.B and I.B. for the microbiological section, J.G.C. for the introduction on corals, and L.F. for image processing. C.A., L.F. and S.B. produced the figures and tables. All the authors contributed to the discussion and improvement of the paper since the early draft.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
We acknowledge the captain and crew onboard R/V Kronprins Hakon for their assistance during the expedition. We are grateful to Abidemi Akinselure and Matteus Lindgren (SIL-UiT) for sample preparation and IRMS analyses. We thank the anonymous reviewers and associate editor Mark Lever for constructive feedback.
This research was funded by EMAN7 project (Research Council of Norway grant No. 320100) and supported by AKMA project (Research Council of Norway grant No. 287869) and EXTREMES (UArctic UA 06/2024). IBA is supported by Swiss National Science Foundation (project number: 221959). CA is currently supported by Equinor ASA. Publication costs are covered by UiT's Publishing Fund.
This paper was edited by Mark Lever and reviewed by three anonymous referees.
Aalto, N. J., Schweitzer, H. D., Krsmanovic, S., Campbell, K., and Bernstein, H. C.: Diversity and Selection of Surface Marine Microbiomes in the Atlantic-Influenced Arctic, Front. Microbiol., 13, 892634, https://doi.org/10.3389/fmicb.2022.892634, 2022.
Alain, K., Zbinden, M., Le Bris, N., Lesongeur, F., Quérellou, J., Gaill, F., and Cambon-Bonavita, M.: Early steps in microbial colonization processes at deep-sea hydrothermal vents, Environ. Microbiol., 6, 227–241, https://doi.org/10.1111/j.1462-2920.2003.00557.x, 2004.
Argentino, C., Waghorn, K. A., Vadakkepuliyambatta, S., Polteau, S., Bünz, S., and Panieri, G.: Dynamic and history of methane seepage in the SW Barents Sea: new insights from Leirdjupet Fault Complex, Sci. Rep., 11, 4373, https://doi.org/10.1038/s41598-021-83542-0, 2021.
Argentino, C., Savini, A., and Panieri, G.: Integrating Fine-Scale Habitat Mapping and Pore Water Analysis in Cold Seep Research: A Case Study from the SW Barents Sea, in: World Atlas of Submarine Gas Hydrates in Continental Margins, Springer International Publishing, Cham, 505–514, https://doi.org/10.1007/978-3-030-81186-0_43, 2022.
Argentino, C., Wittig, C., Peckmann, J., and Panieri, G.: Nitrogen uptake by methanotrophic consortia in deep-water gas hydrate-bearing sediments, Chem. Geol., 636, 121638, https://doi.org/10.1016/j.chemgeo.2023.121638, 2023.
Argentino, C., Borrelli, C., Akinselure, A., Correa-Diaz, M., and Panieri, G.: Biogeochemical investigations of methane seepage at the ultraslow-spreading Arctic mid-ocean ridge: Svyatogor ridge, Fram Strait, Mar. Pet. Geol., 162, 106761, https://doi.org/10.1016/j.marpetgeo.2024.106761, 2024.
Argentino, C., Ferré, B., and Panieri, G.: Replication Data for: Seafloor chemosynthetic habitats and AOM-influenced sediment microbiome at a cold-water coral site off the Vesterålen coast, northern Norway, V1, DataverseNO [data set], https://doi.org/10.18710/1FCVAE, 2026.
Bagarinao, T.: Sulfide as an environmental factor and toxicant: tolerance and adaptations in aquatic organisms, Aquat. Toxicol., https://doi.org/10.1016/0166-445X(92)90015-F, 1992.
Barrenechea, I. A., Argentino, C., Cermakova, K., Holzmann, M., Pawlowski, J., and Panieri, G.: Sediment eDNA metabarcoding reveals the endemism in benthic foraminifera from Arctic methane cold seepages, ISME Commun., ycaf058, https://doi.org/10.1093/ismeco/ycaf058, 2025.
Beaumont, N. J., Austen, M. C., Atkins, J. P., Burdon, D., Degraer, S., Dentinho, T. P., Derous, S., Holm, P., Horton, T., Van Ierland, E., Marboe, A. H., Starkey, D. J., Townsend, M., and Zarzycki, T.: Identification, definition and quantification of goods and services provided by marine biodiversity: Implications for the ecosystem approach, Mar. Pollut. Bull., 54, 253–265, https://doi.org/10.1016/j.marpolbul.2006.12.003, 2007.
Becker, E. L., Cordes, E. E., Macko, S. A., and Fisher, C. R.: Importance of seep primary production to Lophelia pertusa and associated fauna in the Gulf of Mexico, Deep-Sea Res., 56, 786–800, https://doi.org/10.1016/j.dsr.2008.12.006, 2009.
Bellec, V. K., Chand, S., Knies, J., Bjarnadóttir, L. R., Lepland, A., Sen, A., and Thorsnes, T.: New cold seep sites on the continental slope southwest to Svalbard, Front. Earth Sci., 12, 1328357, https://doi.org/10.3389/feart.2024.1328357, 2024.
Benz, U. C., Hofmann, P., Willhauck, G., Lingenfelder, I., and Heynen, M.: Multi-resolution, object-oriented fuzzy analysis of remote sensing data for GIS-ready information, ISPRS J. Photogramm., 58, 239–258, https://doi.org/10.1016/j.isprsjprs.2003.10.002, 2004.
Biastoch, A., Treude, T., Rpke, L. H., Riebesell, U., Roth, C., Burwicz, E. B., Park, W., Latif, M., Böning, C. W., Madec, G., and Wallmann, K.: Rising Arctic Ocean temperatures cause gas hydrate destabilization and ocean acidification, Geophys. Res. Lett., 38, 1–6, https://doi.org/10.1029/2011GL047222, 2011.
Blaschke, T.: Object based image analysis for remote sensing, ISPRS J. Photogramm., 65, 2–16, https://doi.org/10.1016/j.isprsjprs.2009.06.004, 2010.
Blumenberg, M., Walliser, E.-O., Taviani, M., Seifert, R., and Reitner, J.: Authigenic carbonate formation and its impact on the biomarker inventory at hydrocarbon seeps – A case study from the Holocene Black Sea and the Plio-Pleistocene Northern Apennines (Italy), Mar. Petrol. Geol., 66, 532–541, https://doi.org/10.1016/j.marpetgeo.2015.05.013, 2015.
Bøe, R., Bellec, V. K., Dolan, M. F. J., Buhl-Mortensen, P., Rise, L., and Buhl-Mortensen, L.: Cold-water coral reefs in the Hola glacial trough off Vesterålen, North Norway, Geol. Soc. Lond. Mem., 46, 309–310, https://doi.org/10.1144/M46.8, 2016.
Boetius, A. and Wenzhöfer, F.: Seafloor oxygen consumption fuelled by methane from cold seeps, Nat. Geosci., 6, 725–734, https://doi.org/10.1038/ngeo1926, 2013.
Boetius, A., Ravenschlag, K., Schubert, C. J., Rickert, D., Widdel, F., Gieseke, A., Amann, R., Jørgensen, B. B., Witte, U., and Pfannkuche, O.: A marine microbial consortium apparently mediating anaerobic oxidation of methane, Nature, 407, 623–626, https://doi.org/10.1038/35036572, 2000.
Boretto, G. M., Sabino, M., Torricella, F., Ingrosso, G., Nogarotto, A., Panieri, G., Hefter, J., Mollenhauer, G., Langone, L., Giuliani, S., Tesi, T., and Capotondi, L.: Taphonomic signature of benthic foraminifera linked to methane release in the Barents Sea, Mar. Geol., 492, 107693, https://doi.org/10.1016/j.margeo.2025.107693, 2026.
Briggs, B. R., Pohlman, J. W., Torres, M., Riedel, M., Brodie, E. L., and Colwell, F. S.: Macroscopic Biofilms in Fracture-Dominated Sediment That Anaerobically Oxidize Methane, Appl. Environ. Microbiol., 77, 6780–6787, https://doi.org/10.1128/AEM.00288-11, 2011.
Buhl-Mortensen, L., Bøe, R., Dolan, M. F. J., Buhl-Mortensen, P., Thorsnes, T., Elvenes, S., and Hodnesdal, H.: Banks, Troughs, and Canyons on the Continental Margin off Lofoten, Vesterålen, and Troms, Norway, in: Seafloor Geomorphology as Benthic Habitat, Elsevier, 703–715, https://doi.org/10.1016/B978-0-12-385140-6.00051-7, 2012.
Burdige, D. J.: Burial of terrestrial organic matter in marine sediments: A re-assessment, Global Biogeochem. Cy., 19, https://doi.org/10.1029/2004GB002368, 2005.
Callahan, B. J., McMurdie, P. J., Rosen, M. J., Han, A. W., Johnson, A. J. A., and Holmes, S. P.: DADA2: High-resolution sample inference from Illumina amplicon data, Nat. Methods, 13, 581–583, https://doi.org/10.1038/nmeth.3869, 2016.
Cardoso, D. C., Cretoiu, M. S., Stal, L. J., and Bolhuis, H.: Seasonal development of a coastal microbial mat, Sci. Rep., 9, 9035, https://doi.org/10.1038/s41598-019-45490-8, 2019.
Chand, S., Rise, L., Bellec, V., Dolan, M., Bøe, R., Thorsnes, T., and Buhl-Mortensen, P.: Active Venting System Offshore Northern Norway, EOS T. Am. Geophys. Un., 89, 261–262, https://doi.org/10.1029/2008EO290001, 2008.
Crémière, A., Lepland, A., Chand, S., Sahy, D., Condon, D. J., Noble, S. R., Martma, T., Thorsnes, T., Sauer, S., and Brunstad, H.: Timescales of methane seepage on the Norwegian margin following collapse of the Scandinavian Ice Sheet, Nat. Commun., 7, 11509, https://doi.org/10.1038/ncomms11509, 2016.
Dekas, A. E., Poretsky, R. S., and Orphan, V. J.: Deep-Sea Archaea Fix and Share Nitrogen in Methane-Consuming Microbial Consortia, Science, 326, 422–426, https://doi.org/10.1126/science.1178223, 2009.
Dekas, A. E., Chadwick, G. L., Bowles, M. W., Joye, S. B., and Orphan, V. J.: Spatial distribution of nitrogen fixation in methane seep sediment and the role of the ANME archaea, Environ. Microbiol., 16, 3012–3029, https://doi.org/10.1111/1462-2920.12247, 2014.
Deng, Y., Chen, F., Li, N., Jin, M., Cao, J., Chen, H., Zhou, Y., Wu, C., Zhuang, C., Zhao, Y., and Cheng, S.: Cold-Water Corals in Gas Hydrate Drilling Cores from the South China Sea: Occurrences, Geochemical Characteristics and Their Relationship to Methane Seepages, Minerals, 9, 742, https://doi.org/10.3390/min9120742, 2019.
Egger, M., Riedinger, N., Mogollón, J. M., and Jørgensen, B. B.: Global diffusive fluxes of methane in marine sediments, Nat. Geosci., 11, 421–425, https://doi.org/10.1038/s41561-018-0122-8, 2018.
Emerson, S. and Hedges, J. I.: Processes controlling the organic carbon content of open ocean sediments, Paleoceanography, 3, 621–634, https://doi.org/10.1029/PA003i005p00621, 1988.
Eyre, B. D., Cyronak, T., Drupp, P., De Carlo, E. H., Sachs, J. P., and Andersson, A. J.: Coral reefs will transition to net dissolving before end of century, Science, 359, 908–911, https://doi.org/10.1126/science.aao1118, 2018.
Fallati, L., Saponari, L., Savini, A., Marchese, F., Corselli, C., and Galli, P.: Multi-Temporal UAV Data and Object-Based Image Analysis (OBIA) for Estimation of Substrate Changes in a Post-Bleaching Scenario on a Maldivian Reef, Remote Sens., 12, 2093, https://doi.org/10.3390/rs12132093, 2020.
Fallati, L., Panieri, G., Argentino, C., Varzi, A. G., Bünz, S., and Savini, A.: Characterizing Håkon Mosby Mud Volcano (Barents Sea) cold seep systems by combining ROV-based acoustic data and underwater photogrammetry, Front. Mar. Sci., 10, 1–15, https://doi.org/10.3389/fmars.2023.1269197, 2023.
Ferré, B., Jansson, P. G., Moser, M., Serov, P., Portnov, A., Graves, C. A., Panieri, G., Gründger, F., Berndt, C., Lehmann, M. F., and Niemann, H.: Reduced methane seepage from Arctic sediments during cold bottom-water conditions, Nat. Geosci., 13, 144–148, https://doi.org/10.1038/s41561-019-0515-3, 2020.
Ferré, B., Panieri, G., Argentino, C., Sert, M. F., Stetzler, M., Fallati, L., Dills, M. S., Petters, S., Rohan, T., Barreyre, T., Lien, M. E., Kutti, T., Corrales, J., Kjær, M., Ting, R., Bojesen, M. D., Walta, A., and Jensen, S.: CAGE22-3 Cruise Report: EMAN7 cruise, CAGE – Cent. Arct. Gas Hydrate Environ. Clim. Rep. Ser., 10, https://doi.org/10.7557/cage.6760, 2022.
Ferré, B., Barreyre, T., Bünz, S., Argentino, C., Corrales-Guerrero, J., Dølven, K. O., Stetzler, M., Fallati, L., Sert, M. F., Panieri, G., Rastrick, S., Kutti, T., and Moser, M.: Contrasting Methane Seepage Dynamics in the Hola Trough Offshore Norway: Insights From Two Different Summers, J. Geophys. Res.-Oceans, 129, e2024JC020949, https://doi.org/10.1029/2024JC020949, 2024.
Fischer, D., Sahling, H., Nöthen, K., Bohrmann, G., Zabel, M., and Kasten, S.: Interaction between hydrocarbon seepage, chemosynthetic communities, and bottom water redox at cold seeps of the Makran accretionary prism: insights from habitat-specific pore water sampling and modeling, Biogeosciences, 9, 2013–2031, https://doi.org/10.5194/bg-9-2013-2012, 2012.
Foley, N. S., Van Rensburg, T. M., and Armstrong, C. W.: The ecological and economic value of cold-water coral ecosystems, Ocean Coast. Manage., 53, 313–326, https://doi.org/10.1016/j.ocecoaman.2010.04.009, 2010.
Fosså, J. H., Mortensen, P. B., and Furevik, D. M.: The deep-water coral Lophelia pertusa in Norwegian waters: Distribution and fishery impacts, Hydrobiologia, 471, 1–12, https://doi.org/10.1023/A:1016504430684, 2002.
Foucher, J. P., Westbrook, G. K., Boetius, A., Ceramicola, S., Dupré, S., Mascle, J., Mienert, J., Pfannkuche, O., Pierre, C., and Praeg, D.: Cold Seep Ecosystems Structure and Drivers of Cold Seep Ecosystems, Oceanography, 22, 92–109, 2007.
Freiwald, A.: Cold-Water Coral Reefs, in: Encyclopedia of Modern Coral Reefs, edited by: Hopley, D., Springer Netherlands, Dordrecht, 225–229, https://doi.org/10.1007/978-90-481-2639-2_68, 2011.
Garcia-Tigreros, F., Leonte, M., Ruppel, C. D., Ruiz-Angulo, A., Joung, D. J., Young, B., and Kessler, J. D.: Estimating the Impact of Seep Methane Oxidation on Ocean pH and Dissolved Inorganic Radiocarbon Along the U.S. Mid-Atlantic Bight, J. Geophys. Res.-Biogeo., 126, e2019JG005621, https://doi.org/10.1029/2019JG005621, 2021.
Ghosh, A., Mazumdar, A., Sadique, Mohd., Peketi, A., Zatale, A., Sivan, K., Pillai, S. R., and Sparsh, A.: Chemosynthetic Organic Matter Reveals a Protracted Episode of Past Methane Seepage Activities at a Newly Discovered Cold Seep Site (Mannar Basin) Off the East Coast of India, Geophys. Res. Lett., 52, e2025GL116435, https://doi.org/10.1029/2025GL116435, 2025.
Gilbert, J. A., Jansson, J. K., and Knight, R.: The Earth Microbiome project: successes and aspirations, BMC Biol., 12, 69, https://doi.org/10.1186/s12915-014-0069-1, 2014.
Girard, F., Sarrazin, J., and Olu, K.: Impacts of an Eruption on Cold-Seep Microbial and Faunal Dynamics at a Mud Volcano, Front. Mar. Sci., 7, 241, https://doi.org/10.3389/fmars.2020.00241, 2020.
Greinert, J.: Monitoring temporal variability of bubble release at seeps: The hydroacoustic swath system GasQuant, J. Geophys. Res.-Oceans, 113, 2007JC004704, https://doi.org/10.1029/2007JC004704, 2008.
Gruber, N. and Galloway, J. N.: An Earth-system perspective of the global nitrogen cycle, Nature, 451, 293–296, https://doi.org/10.1038/nature06592, 2008.
Gründger, F., Carrier, V., Svenning, M. M., Panieri, G., Vonnahme, T. R., Klasek, S., and Niemann, H.: Methane-fuelled biofilms predominantly composed of methanotrophic ANME-1 in Arctic gas hydrate-related sediments, Sci. Rep., 9, 1–10, https://doi.org/10.1038/s41598-019-46209-5, 2019.
Guezennec, J., Ortega-Morales, O., Raguenes, G., and Geesey, G.: Bacterial colonization of artificial substrate in the vicinity of deep-sea hydrothermal vents, FEMS Microbiol. Ecol., 26, 89–99, https://doi.org/10.1111/j.1574-6941.1998.tb00495.x, 1998.
Hasegawa, H., Kita, I., Tsukamoto, S., Chiyonobu, S., and Kuwahara, Y.: Synchronous Changes of Nitrogen and Carbon Isotopic Ratios and Nannoplankton Assemblage in Marine Sediments off Peru at 250 ka: A Role of Phytoplankton in Primary Ocean Productivity, Open J. Geol., 03, 113–120, https://doi.org/10.4236/ojg.2013.32015, 2013.
Hauri, C., Irving, B., Hayes, D., Abdi, E., Kemme, J., Kinski, N., and McDonnell, A. M. P.: Expanding seawater carbon dioxide and methane measuring capabilities with a Seaglider, Ocean Sci., 20, 1403–1421, https://doi.org/10.5194/os-20-1403-2024, 2024.
Hay, G. J. and Castilla, G.: Geographic Object-Based Image Analysis (GEOBIA): A new name for a new discipline, in: Object-Based Image Analysis, edited by: Blaschke, T., Lang, S., and Hay, G. J., Springer Berlin Heidelberg, Berlin, Heidelberg, 75–89, https://doi.org/10.1007/978-3-540-77058-9_4, 2008.
Hensen, C., Zabel, M., Pfeifer, K., Schwenk, T., Kasten, S., Riedinger, N., Schulz, H. D., and Boetius, A.: Control of sulfate pore-water profiles by sedimentary events and the significance of anaerobic oxidation of methane for the burial of sulfur in marine sediments, Geochim. Cosmochim. Ac., 67, 2631–2647, https://doi.org/10.1016/S0016-7037(03)00199-6, 2003.
Hilário, A., Capa, M., Dahlgren, T. G., Halanych, K. M., Little, C. T. S., Thornhill, D. J., Verna, C., and Glover, A. G.: New Perspectives on the Ecology and Evolution of Siboglinid Tubeworms, PLoS ONE, 6, e16309, https://doi.org/10.1371/journal.pone.0016309, 2011.
Hilmi, N., Allemand, D., Dupont, S., Safa, A., Haraldsson, G., Nunes, P. A. L. D., Moore, C., Hattam, C., Reynaud, S., Hall-Spencer, J. M., Fine, M., Turley, C., Jeffree, R., Orr, J., Munday, P. L., and Cooley, S. R.: Towards improved socio-economic assessments of ocean acidification's impacts, Mar. Biol., 160, 1773–1787, https://doi.org/10.1007/s00227-012-2031-5, 2013.
Hossain, M. D. and Chen, D.: Segmentation for Object-Based Image Analysis (OBIA): A review of algorithms and challenges from remote sensing perspective, ISPRS J. Photogramm., 150, 115–134, https://doi.org/10.1016/j.isprsjprs.2019.02.009, 2019.
Hovland, M.: Do carbonate reefs form due to fluid seepage?, Terra Nova, 2, 8–18, https://doi.org/10.1111/j.1365-3121.1990.tb00031.x, 1990.
Hovland, M.: Deep-water Coral Reefs, Springer Netherlands, Dordrecht, https://doi.org/10.1007/978-1-4020-8460-7, 2008.
Hovland, M. and Thomsen, E.: Cold-water corals – Are they hydrocarbon seep related?, Mar. Geol., 137, 159–164, https://doi.org/10.1016/S0025-3227(96)00086-2, 1997.
Hu, Y., Feng, D., Peng, Y., Peckmann, J., Kasten, S., Wang, X., Liang, Q., Wang, H., and Chen, D.: A prominent isotopic fingerprint of nitrogen uptake by anaerobic methanotrophic archaea, Chem. Geol., 558, 119972, https://doi.org/10.1016/j.chemgeo.2020.119972, 2020.
Husebø, Å., Nøttestad, L., Fosså, J. H., Furevik, D. M., and Jørgensen, S. B.: Distribution and abundance of fish in deep-sea coral habitats, Hydrobiologia, 471, 91–99, https://doi.org/10.1023/A:1016549203368, 2002.
Joye, S. B.: The Geology and Biogeochemistry of Hydrocarbon Seeps, Annu. Rev. Earth Planet. Sc., 48, 205–231, https://doi.org/10.1146/annurev-earth-063016-020052, 2020.
Judd, A., Noble-James, T., Golding, N., Eggett, A., Diesing, M., Clare, D., Silburn, B., Duncan, G., Field, L., and Milodowski, A.: The Croker Carbonate Slabs: extensive methane-derived authigenic carbonate in the Irish Sea – nature, origin, longevity and environmental significance, Geo-Mar. Lett., 40, 423–438, https://doi.org/10.1007/s00367-019-00584-0, 2020.
Judd, A. G. and Hovland, M.: Seabed fluid flow: the impact of geology, Biology and the Marine Environment, 1–442, https://doi.org/10.1007/s00254-004-1086-0, 2007.
Kalenitchenko, D., Dupraz, M., Le Bris, N., Petetin, C., Rose, C., West, N. J., and Galand, P. E.: Ecological succession leads to chemosynthesis in mats colonizing wood in sea water, ISME J., 10, 2246–2258, https://doi.org/10.1038/ismej.2016.12, 2016.
Kang, H., Won, E., Shin, K., and Yoon, H. I.: Organic carbon and nitrogen composition in the sediment of the Kara Sea, Arctic Ocean during the Last Glacial Maximum to Holocene times, Geophys. Res. Lett., 34, 2007GL030068, https://doi.org/10.1029/2007GL030068, 2007.
Karaca, D., Hensen, C., and Wallmann, K.: Controls on authigenic carbonate precipitation at cold seeps along the convergent margin off Costa Rica, Geochem. Geophy. Geosy., 11, 1–19, https://doi.org/10.1029/2010GC003062, 2010.
Kasten, S., Zabel, M., Heuer, V., and Hensen, C.: Processes and Signals of Nonsteady-State Diagenesis in Deep-Sea Sediments and their Pore Waters, in: The South Atlantic in the Late Quaternary, Springer, Berlin, Heidelberg, https://doi.org/10.1007/978-3-642-18917-3_20, 2003.
Kienast, M., Higginson, M. J., Mollenhauer, G., Eglinton, T. I., Chen, M.-T., and Calvert, S. E.: On the sedimentological origin of down-core variations of bulk sedimentary nitrogen isotope ratios, Paleoceanography, 20, https://doi.org/10.1029/2004PA001081, 2005.
Kline, D. I., Teneva, L., Okamoto, D. K., Schneider, K., Caldeira, K., Miard, T., Chai, A., Marker, M., Dunbar, R. B., Mitchell, B. G., Dove, S., and Hoegh-Guldberg, O.: Living coral tissue slows skeletal dissolution related to ocean acidification, Nat. Ecol. Evol., 3, 1438–1444, https://doi.org/10.1038/s41559-019-0988-x, 2019.
Knies, J.: Nitrogen Isotope Evidence for Changing Arctic Ocean Ventilation Regimes During the Cenozoic, Geophys. Res. Lett., 49, 1–8, https://doi.org/10.1029/2022GL099512, 2022.
Knies, J. and Martinez, P.: Organic matter sedimentation in the western Barents Sea region: Terrestrial and marine contribution based on isotopic composition and organic nitrogen content, Nor. Geol. Tidsskr., 89, 79–89, 2009.
Knies, J., Brookes, S., and Schubert, C. J.: Re-assessing the nitrogen signal in continental margin sediments: New insights from the high northern latitudes, Earth Planet. Sc. Lett., 253, 471–484, https://doi.org/10.1016/j.epsl.2006.11.008, 2007.
Kurth, J. M., Smit, N. T., Berger, S., Schouten, S., Jetten, M. S. M., and Welte, C. U.: Anaerobic methanotrophic archaea of the ANME-2d clade feature lipid composition that differs from other ANME archaea, FEMS Microbiol. Ecol., 95, fiz082, https://doi.org/10.1093/femsec/fiz082, 2019.
Lapham, L. L., Lloyd, K. G., Fossing, H., Flury, S., Jensen, J. B., Alperin, M. J., Rehder, G., Holzhueter, W., Ferdelman, T., and Jørgensen, B. B.: Methane leakage through the sulfate–methane transition zone of the Baltic seabed, Nat. Geosci., 17, 1277–1283, https://doi.org/10.1038/s41561-024-01594-z, 2024.
Lee, D. H., Lee, Y. M., Kim, J. H., Jin, Y. K., Paull, C., Niemann, H., Kim, J. H., and Shin, K. H.: Discriminative biogeochemical signatures of methanotrophs in different chemosynthetic habitats at an active mud volcano in the Canadian Beaufort Sea, Sci. Rep., 9, 1–13, https://doi.org/10.1038/s41598-019-53950-4, 2019.
Levin, L. A.: Ecology of Cold Seep Sediments: Interactions of Fauna With Flow, Chemistry and Microbes, Oceanogr. Mar. Biol. Annu. Rev., 43, 1–46, https://doi.org/10.1201/9781420037449-3, 2005.
Levin, L. A., Baco, A. R., Bowden, D. A., Colaco, A., Cordes, E. E., Cunha, M. R., Demopoulos, A. W. J. J., Gobin, J., Grupe, B. M., Le, J., Metaxas, A., Netburn, A. N., Rouse, G. W., Thurber, A. R., Tunnicliffe, V., Van Dover, C. L., Vanreusel, A., and Watling, L.: Hydrothermal Vents and Methane Seeps: Rethinking the Sphere of Influence, Front. Mar. Sci., 3, 1–23, https://doi.org/10.3389/fmars.2016.00072, 2016.
Liebetrau, V., Eisenhauer, A., and Linke, P.: Cold seep carbonates and associated cold-water corals at the Hikurangi Margin, New Zealand: New insights into fluid pathways, growth structures and geochronology, Mar. Geol., 272, 307–318, https://doi.org/10.1016/j.margeo.2010.01.003, 2010.
Lim, A., Wheeler, A. J., Price, D. M., O'Reilly, L., Harris, K., and Conti, L.: Influence of benthic currents on cold-water coral habitats: a combined benthic monitoring and 3D photogrammetric investigation, Sci. Rep., 10, 19433, https://doi.org/10.1038/s41598-020-76446-y, 2020.
Liu, C., Cui, Y., Li, X., and Yao, M.: microeco: an R package for data mining in microbial community ecology, FEMS Microbiol. Ecol., 97, fiaa255, https://doi.org/10.1093/femsec/fiaa255, 2021.
Madigan, M. T., Bender, K. S., Buckley, D. H., Sattley, M. W., and Stahl, D. A.: Brock Biology of Microorganisms, Global Edition, Pearson Education Limited, ISBN 978-0134261928, 2017.
Meyers, P. A.: Preservation of elemental and isotopic source identification of sedimentary organic matter, Chem. Geol., 114, 289–302, https://doi.org/10.1016/0009-2541(94)90059-0, 1994.
Meyers, P. A.: Organic geochemical proxies of paleoceanographic, paleolimnologic, and paleoclimatic processes, Org. Geochem., 27, 213–250, https://doi.org/10.1016/S0146-6380(97)00049-1, 1997.
Milkov, A. V. and Etiope, G.: Revised genetic diagrams for natural gases based on a global dataset of > 20,000 samples, Org. Geochem., 125, 109–120, https://doi.org/10.1016/j.orggeochem.2018.09.002, 2018.
Millero, F. J.: Chemical Oceanography, CRC Press, https://doi.org/10.1201/9780429258718, 2005.
Minami, H. and Tatsumi, K.: Possible variation in methane flux caused by gas hydrate formation on the northeastern continental slope off Sakhalin, 525–534, https://doi.org/10.1007/s00367-012-0287-x, 2012.
Montes-Herrera, J. C., Hill, N., Cummings, V. J., Johnstone, G. J., Stark, J. S., and Lucieer, V.: Remote sensing of Antarctic polychaete reefs (Serpula narconensis): reproducible workflows for quantifying benthic structural complexity with action cameras, remotely operated vehicles and structure-from-motion photogrammetry, Remote Sens. Ecol. Conserv., 10, 72–90, https://doi.org/10.1002/rse2.358, 2024.
Naidu, A. S., Cooper, L. W., Finney, B. P., Macdonald, R. W., Alexander, C., and Semiletov, I. P.: Organic carbon isotope ratios (δ13C) of Arctic Amerasian Continental shelf sediments, Int. J. Earth Sci., 89, 522–532, https://doi.org/10.1007/s005310000121, 2000.
Niemann, H., Linke, P., Knittel, K., MacPherson, E., Boetius, A., Brückmann, W., Larvik, G., Wallmann, K., Schacht, U., Omoregie, E., Hilton, D., Brown, K., and Rehder, G.: Methane-Carbon Flow into the Benthic Food Web at Cold Seeps – A Case Study from the Costa Rica Subduction Zone, PLoS ONE, 8, e74894, https://doi.org/10.1371/journal.pone.0074894, 2013.
Noffke, N.: Geobiology: Microbial Mats in Sandy Deposits from the Archean Era to Today, Springer Berlin Heidelberg, Berlin, Heidelberg, https://doi.org/10.1007/978-3-642-12772-4, 2010.
Oksanen, J., Simpson, G. L., Blanchet, F. G., Kindt, R., Legendre, P., Minchin, P. R., O'Hara, R. B., Solymos, P., Stevens, M. H. H., Szoecs, E., Wagner, H., Barbour, M., Bedward, M., Bolker, B., Borcard, D., Borman, T., Carvalho, G., Chirico, M., De Caceres, M., Durand, S., Evangelista, H. B. A., FitzJohn, R., Friendly, M., Furneaux, B., Hannigan, G., Hill, M. O., Lahti, L., Martino, C., McGlinn, D., Ouellette, M.-H., Ribeiro Cunha, E., Smith, T., Stier, A., Ter Braak, C. J. F., and Weedon, J.: vegan: Community Ecology Package, https://doi.org/10.32614/CRAN.package.vegan, 2001.
Osman, E. O., Vohsen, S. A., Girard, F., Cruz, R., Glickman, O., Bullock, L. M., Anderson, K. E., Weinnig, A. M., Cordes, E. E., Fisher, C. R., and Baums, I. B.: Capacity of deep-sea corals to obtain nutrition from cold seeps aligned with microbiome reorganization, Glob. Change Biol., 29, 189–205, https://doi.org/10.1111/gcb.16447, 2023.
Ottesen, D., Dowdeswell, J. A., and Rise, L.: Submarine landforms and the reconstruction of fast-flowing ice streams within a large Quaternary ice sheet: The 2500-km-long Norwegian-Svalbard margin (57°–80° N), Geol. Soc. Am. Bull., 117, 1033, https://doi.org/10.1130/B25577.1, 2005.
Pan, J., Perillo, V. L., and Cuadrado, D. G.: Quantification of microbial mat response to physical disruption in siliciclastic sediments, Estuar. Coast. Shelf S., 230, 106434, https://doi.org/10.1016/j.ecss.2019.106434, 2019.
Panieri, G., Argentino, C., Ramalho, S. P., Vulcano, F., Savini, A., Fallati, L., Brekke, T., Galimberti, G., Riva, F., Balsa, J., Eilertsen, M. H., Stokke, R., Steen, I. H., Sahy, D., Kalenitchenko, D., Büenz, S., and Mattingsdal, R.: An Arctic natural oil seep investigated from space to the seafloor, Sci. Total Environ., 907, 167788, https://doi.org/10.1016/j.scitotenv.2023.167788, 2024.
Phrampus, B. J., Lee, T. R., and Wood, W. T.: A Global Probabilistic Prediction of Cold Seeps and Associated SEAfloor FLuid Expulsion Anomalies (SEAFLEAs), Geochem. Geophy. Geosy., 21, https://doi.org/10.1029/2019GC008747, 2020.
Pirlet, H., Wehrmann, L. M., Foubert, A., Brunner, B., Blamart, D., De Mol, L., Van Rooij, D., Dewanckele, J., Cnudde, V., Swennen, R., Duyck, P., and Henriet, J.: Unique authigenic mineral assemblages reveal different diagenetic histories in two neighbouring cold-water coral mounds on Pen Duick Escarpment, Gulf of Cadiz, Sedimentology, 59, 578–604, https://doi.org/10.1111/j.1365-3091.2011.01267.x, 2012.
Price, D. M., Robert, K., Callaway, A., Lo Lacono, C., Hall, R. A., and Huvenne, V. A. I.: Using 3D photogrammetry from ROV video to quantify cold-water coral reef structural complexity and investigate its influence on biodiversity and community assemblage, Coral Reefs, 38, 1007–1021, https://doi.org/10.1007/s00338-019-01827-3, 2019.
Probst, A. J., Weinmaier, T., Raymann, K., Perras, A., Emerson, J. B., Rattei, T., Wanner, G., Klingl, A., Berg, I. A., Yoshinaga, M., Viehweger, B., Hinrichs, K.-U., Thomas, B. C., Meck, S., Auerbach, A. K., Heise, M., Schintlmeister, A., Schmid, M., Wagner, M., Gribaldo, S., Banfield, J. F., and Moissl-Eichinger, C.: Biology of a widespread uncultivated archaeon that contributes to carbon fixation in the subsurface, Nat. Commun., 5, 5497, https://doi.org/10.1038/ncomms6497, 2014.
Quast, C., Pruesse, E., Yilmaz, P., Gerken, J., Schweer, T., Yarza, P., Peplies, J., and Glöckner, F. O.: The SILVA ribosomal RNA gene database project: improved data processing and web-based tools, Nucleic Acids Res., 41, D590–596, https://doi.org/10.1093/nar/gks1219, 2013.
Rachold, V. and Hubberten, H.-W.: Carbon Isotope Composition of Particulate Organic Material in East Siberian Rivers, in: Land-Ocean Systems in the Siberian Arctic, edited by: Kassens, H., Bauch, H. A., Dmitrenko, I. A., Eicken, H., Hubberten, H.-W., Melles, M., Thiede, J., and Timokhov, L. A., Springer Berlin Heidelberg, Berlin, Heidelberg, 223–238, https://doi.org/10.1007/978-3-642-60134-7_21, 1999.
R Core Team: R: A language and environment for statistical computing, R Foundation for Statistical Computing, Vienna, https://www.R-project.org/ (last access: 27 April 2026), 2021.
Riesch, R., Tobler, M., and Plath, M.: Hydrogen Sulfide-Toxic Habitats, in: Extremophile Fishes, edited by: Riesch, R., Tobler, M., and Plath, M., Springer International Publishing, Cham, 137–159, https://doi.org/10.1007/978-3-319-13362-1_7, 2015.
Rincón-Tomás, B., Duda, J.-P., Somoza, L., González, F. J., Schneider, D., Medialdea, T., Santofimia, E., López-Pamo, E., Madureira, P., Hoppert, M., and Reitner, J.: Cold-water corals and hydrocarbon-rich seepage in Pompeia Province (Gulf of Cádiz) – living on the edge, Biogeosciences, 16, 1607–1627, https://doi.org/10.5194/bg-16-1607-2019, 2019.
Roberts, J. M., Wheeler, A., Freiwald, A., and Cairns, S.: Cold-Water Corals: The Biology and Geology of Deep-Sea Coral Habitats, 1st edn., Cambridge University Press, https://doi.org/10.1017/CBO9780511581588, 2009.
Robeson, M. S., O'Rourke, D. R., Kaehler, B. D., Ziemski, M., Dillon, M. R., Foster, J. T., and Bokulich, N. A.: RESCRIPt: Reproducible sequence taxonomy reference database management, PLOS Comput. Biol., 17, e1009581, https://doi.org/10.1371/journal.pcbi.1009581, 2021.
Ruttenberg, K. C. and Goñi, M. A.: Phosphorus distribution, C : N : P ratios, and δ13Coc in arctic, temperate, and tropical coastal sediments: tools for characterizing bulk sedimentary organic matter, Mar. Geol., 139, 123–145, https://doi.org/10.1016/S0025-3227(96)00107-7, 1997.
Sahling, H., Rickert, D., Lee, R. W., Linke, P., and Suess, E.: Macrofaunal community structure and sulfide flux at gas hydrate deposits from the Cascadia convergent margin, NE Pacific, Mar. Ecol.-Prog. Ser., 231, 121–138, https://doi.org/10.3354/meps231121, 2002.
Sanna, G., Büscher, J. V., and Freiwald, A.: Cold-water coral framework architecture is selectively shaped by bottom current flow, Coral Reefs, 42, 483–495, https://doi.org/10.1007/s00338-023-02361-z, 2023.
Sauer, S., Knies, J., Lepland, A., Chand, S., Eichinger, F., and Schubert, C. J.: Hydrocarbon sources of cold seeps off the Vesterålen coast, northern Norway, Chem. Geol., 417, 371–382, https://doi.org/10.1016/j.chemgeo.2015.10.025, 2015.
Sauer, S., Crémière, A., Knies, J., Lepland, A., Sahy, D., Martma, T., Noble, S. R., Schönenberger, J., Klug, M., and Schubert, C. J.: U-Th chronology and formation controls of methane-derived authigenic carbonates from the Hola trough seep area, northern Norway, Chem. Geol., 470, 164–179, https://doi.org/10.1016/j.chemgeo.2017.09.004, 2017.
Schneider, A., Panieri, G., Lepland, A., Consolaro, C., Crémière, A., Forwick, M., Johnson, J. E., Plaza-Faverola, A., Sauer, S., and Knies, J.: Methane seepage at Vestnesa Ridge (NW Svalbard) since the Last Glacial Maximum, Quaternary Sci. Rev., 193, 98–117, https://doi.org/10.1016/j.quascirev.2018.06.006, 2018.
Schroeder, W. W.: Observations of Lophelia pertusa and the surficial geology at a deep-water site in the northeastern Gulf of Mexico, Hydrobiologia, 471, 29–33, https://doi.org/10.1023/A:1016580632501, 2002.
Seabrook, S., De Leo, F. C., and Thurber, A. R.: Flipping for Food: The Use of a Methane Seep by Tanner Crabs (Chionoecetes tanneri), Front. Mar. Sci., 6, 43, https://doi.org/10.3389/fmars.2019.00043, 2019.
Sen, A., Duperron, S., Hourdez, S., Piquet, B., Léger, N., Gebruk, A., Le Port, A. S., Svenning, M. M., and Andersen, A. C.: Cryptic frenulates are the dominant chemosymbiotrophic fauna at Arctic and high latitude Atlantic cold seeps, PLoS ONE, 13, https://doi.org/10.1371/journal.pone.0209273, 2018a.
Sen, A., Åström, E. K. L., Hong, W.-L., Portnov, A., Waage, M., Serov, P., Carroll, M. L., and Carroll, J.: Geophysical and geochemical controls on the megafaunal community of a high Arctic cold seep, Biogeosciences, 15, 4533–4559, https://doi.org/10.5194/bg-15-4533-2018, 2018b.
Sen, A., Himmler, T., Hong, W. L., Chitkara, C., Lee, R. W., Ferré, B., Lepland, A., and Knies, J.: Atypical biological features of a new cold seep site on the Lofoten-Vesterålen continental margin (northern Norway), Sci. Rep., 9, 1–14, https://doi.org/10.1038/s41598-018-38070-9, 2019.
Sert, M. F., Bernstein, H. C., Dølven, K. O., Petters, S., Kekäläinen, T., Jänis, J., Corrales-Guerrero, J., and Ferré, B.: Cold Seeps and Coral Reefs in Northern Norway: Carbon Cycling in Marine Ecosystems With Coexisting Features, J. Geophys. Res.-Biogeo., 130, e2024JG008475, https://doi.org/10.1029/2024JG008475, 2025.
Southward, E. C., Schulze, A., and Gardiner, S. L.: Pogonophora (Annelida): form and function, in: Morphology, Molecules, Evolution and Phylogeny in Polychaeta and Related Taxa, vol. 179, edited by: Bartolomaeus, T. and Purschke, G., Springer-Verlag, Berlin/Heidelberg, 227–251, https://doi.org/10.1007/1-4020-3240-4_13, 2005.
Stock, L., Wegener, G., Wang, Y., Zander, Y., and Elvert, M.: Marine Cold Seep ANME-2/SRB Consortia Produce Their Lipid Biomass From Inorganic Carbon, Environ. Microbiol., 27, e70213, https://doi.org/10.1111/1462-2920.70213, 2025.
Sundahl, H., Buhl-Mortensen, P., and Buhl-Mortensen, L.: Distribution and Suitable Habitat of the Cold-Water Corals Lophelia pertusa, Paragorgia arborea, and Primnoa resedaeformis on the Norwegian Continental Shelf, Front. Mar. Sci., 7, 213, https://doi.org/10.3389/fmars.2020.00213, 2020.
Szymczycha, B., Diak, M., Hong, W.-L., Böttcher, M. E., Lepland, A., Makuch, P., Ten Hietbrink, S., Saghravani, S. R., Sen, A., Winogradow, A., and Silberberger, M. J.: Submarine groundwater discharge and gas hydrate dissociation fuel organic matter formation in Arctic fjord sediments, Mar. Chem., 273, 104570, https://doi.org/10.1016/j.marchem.2025.104570, 2025.
Thurber, A. R., Levin, L. A., Rowden, A. A., Sommer, S., Linke, P., and Kröger, K.: Microbes, macrofauna, and methane: A novel seep community fueled by aerobic methanotrophy, Limnol. Oceanogr., 58, 1640–1656, https://doi.org/10.4319/lo.2013.58.5.1640, 2013.
Tobler, M., Passow, C. N., Greenway, R., Kelley, J. L., and Shaw, J. H.: The Evolutionary Ecology of Animals Inhabiting Hydrogen Sulfide–Rich Environments, Annu. Rev. Ecol. Evol. S., 47, 239–262, https://doi.org/10.1146/annurev-ecolsys-121415-032418, 2016.
Tseng, Y., Smrzka, D., Lin, S., Schröder-Ritzrau, A., Frank, N., and Bohrmann, G.: Authigenic Carbonate Precipitation at Yam Seep Controlled by Continuous Fracturing and Uplifting of Four-Way Closure Ridge Offshore SW Taiwan, Geochem. Geophy. Geosy., 24, https://doi.org/10.1029/2022GC010652, 2023.
Vuillemin, A., Coskun, Ö. K., and Orsi, W. D.: Microbial Activities and Selection from Surface Ocean to Subseafloor on the Namibian Continental Shelf, Appl. Environ. Microb., 88, e00216-22, https://doi.org/10.1128/aem.00216-22, 2022.
Wada, E.: Nitrogen isotope fractionation and its significance in biogeochemical processes occurring in marine environments, in: Isotope Marine Chemistry, edited by: Goldberg, E. D. and Horibe, Y., Uchida Rokakuho, Tokyo, 375–398, 1980.
Wegener, G., Niemann, H., Elvert, M., Hinrichs, K.-U., and Boetius, A.: Assimilation of methane and inorganic carbon by microbial communities mediating the anaerobic oxidation of methane, Environ. Microbiol., 10, 2287–2298, https://doi.org/10.1111/j.1462-2920.2008.01653.x, 2008.
Whiticar, M. J.: The Biogeochemical Methane Cycle, in: Hydrocarbons, Oils and Lipids: Diversity, Origin, Chemistry and Fate, Springer International Publishing, Cham, 1–78, https://doi.org/10.1007/978-3-319-54529-5_5-1, 2020.
Wisshak, M., Schönberg, C. H. L., Form, A., and Freiwald, A.: Ocean Acidification Accelerates Reef Bioerosion, PLoS ONE, 7, e45124, https://doi.org/10.1371/journal.pone.0045124, 2012.
Xu, A., Chen, Z., Qu, Y., Tian, Y., Shu, C., Zheng, X., Li, G., Yan, W., and Zhao, M.: Cold-water corals in a cold seep area on the northern continental slopes of the South China sea and their isotopic characteristics, Deep-Sea Res., 152, 103043, https://doi.org/10.1016/j.dsr.2019.05.003, 2019.
Yao, H., Niemann, H., and Panieri, G.: Multi-proxy approach to unravel methane emission history of an Arctic cold seep, Quaternary Sci. Rev., 244, 106490, https://doi.org/10.1016/j.quascirev.2020.106490, 2020.
Yilmaz, P., Parfrey, L. W., Yarza, P., Gerken, J., Pruesse, E., Quast, C., Schweer, T., Peplies, J., Ludwig, W., and Glöckner, F. O.: The SILVA and “All-species Living Tree Project (LTP)” taxonomic frameworks, Nucleic Acids Res., 42, D643–D648, https://doi.org/10.1093/nar/gkt1209, 2014.
Yoshinaga, M. Y., Holler, T., Goldhammer, T., Wegener, G., Pohlman, J. W., Brunner, B., Kuypers, M. M. M., Hinrichs, K., and Elvert, M.: Carbon isotope equilibration during sulphate-limited anaerobic oxidation of methane, Nat. Geosci., 7, https://doi.org/10.1038/NGEO2069, 2014.
Zhang, X., Walz, P. M., Kirkwood, W. J., Hester, K. C., Ussler, W., Peltzer, E. T., and Brewer, P. G.: Development and deployment of a deep-sea Raman probe for measurement of pore water geochemistry, Deep-Sea Res., 57, 297–306, https://doi.org/10.1016/j.dsr.2009.11.004, 2010.
Zhang, X., Kirkwood, W. J., Hester, K. C., Walz, P. M., Ussler, W., Peltzer, E. T., Shane, F., and Brewer, P. G.: In situ Raman probe for quantitative observation of sediment pore waters in the Deep Ocean – Development and applications, OCEANS 2011 IEEE – Spain, 1–8, https://doi.org/10.1109/Oceans-Spain.2011.6003421, 2011.




